Supercritical Fluid vs. Conventional Solvent Extraction: A Comprehensive Yield and Efficiency Study for Pharmaceutical Development
This article provides a critical comparative analysis of Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction (CSE) methods, focusing on yield, bioactive compound preservation, and scalability for pharmaceutical and nutraceutical...
Supercritical Fluid vs. Conventional Solvent Extraction: A Comprehensive Yield and Efficiency Study for Pharmaceutical Development
Abstract
This article provides a critical comparative analysis of Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction (CSE) methods, focusing on yield, bioactive compound preservation, and scalability for pharmaceutical and nutraceutical applications. We explore the foundational principles of both techniques, detail methodological parameters and industrial applications, and offer troubleshooting and optimization strategies. By synthesizing validation data and recent case studies, this review serves as a strategic guide for researchers and drug development professionals in selecting and optimizing extraction protocols to maximize the therapeutic potential of natural products, aligning with the growing demand for green and sustainable technologies.
Understanding the Core Principles: How SFE and Conventional Extraction Work
Supercritical Fluid Extraction (SFE) represents a sophisticated separation technology that utilizes solvents under specific temperature and pressure conditions that exceed their critical point, creating a unique supercritical state [1]. In this state, the fluid exhibits hybrid properties of both liquids and gases, possessing the density and solvating power of a liquid combined with the high diffusivity and low viscosity of a gas [2]. This combination enables supercritical fluids to penetrate solid matrices more effectively than liquid solvents, facilitating efficient extraction of target compounds [1]. Among the various solvents available, carbon dioxide (CO₂) has emerged as the predominant choice for SFE applications, particularly in sensitive industries such as pharmaceuticals, food, and nutraceuticals, due to its non-toxic, non-flammable, and environmentally benign characteristics [3] [4].
The critical point of a substance defines the temperature and pressure above which distinct liquid and gas phases no longer exist. For CO₂, this critical temperature is 31.1°C and critical pressure is 7.38 MPa (approximately 74 bar) [3] [5]. Beyond these parameters, CO₂ transforms into a supercritical state (SC-CO₂), making it an ideal solvent for extracting heat-sensitive compounds because of its mild critical temperature [4]. The versatility of SFE technology allows its application to range from small-scale analytical sample preparation to large-scale industrial processing for obtaining valuable products like essential oils or removing unwanted components such as caffeine [5].
Fundamental Principles of SFE
The Supercritical State
The operational principle of SFE centers on achieving and utilizing the supercritical state of a solvent. When a substance is heated above its critical temperature and pressurized beyond its critical pressure, it enters a supercritical phase where the typical physical distinctions between liquid and gas disappear [2]. This transition yields a fluid with remarkable properties ideally suited for extraction processes. The density of a supercritical fluid approximates that of a liquid, granting it comparable solvating power, while its diffusivity is significantly higher and its viscosity substantially lower than liquid solvents [1] [2]. These combined characteristics enable supercritical fluids to effectively dissolve materials and rapidly diffuse into solid matrices, accessing pores typically inaccessible to liquids [1].
Tunable Solvation Power
A defining advantage of supercritical fluids, particularly SC-CO₂, is their tunable solvation power. The density of SC-CO₂, which directly correlates with its dissolving capacity, can be precisely controlled through adjustments in pressure and temperature [3]. This tunability enables operators to selectively extract target compounds by manipulating system parameters. For instance, lower pressures may suffice for extracting volatile oils, while higher pressures are required for lipids or phospholipids [5]. This selective extraction minimizes co-extraction of unwanted compounds, reducing downstream purification requirements and preserving the integrity of heat-sensitive bioactive compounds [3]. The table below summarizes how pressure adjustments influence the extraction of different compound classes.
Table: Influence of Pressure on Compound Extraction in SC-CO₂
| Pressure Range | Compound Class | Extraction Efficiency | Applications |
|---|---|---|---|
| Low Pressure (~100 bar) | Volatile Oils, Terpenes | High | Essential Oils, Flavors [5] |
| Medium Pressure (200-300 bar) | Lipids, Triglycerides | Medium to High | Seed Oils, Nutraceuticals [1] |
| High Pressure (>300 bar) | Phospholipids, Polar Compounds | High (often with modifier) | Specialty Lipids, Polar Bioactives [5] |
The SFE Process Workflow
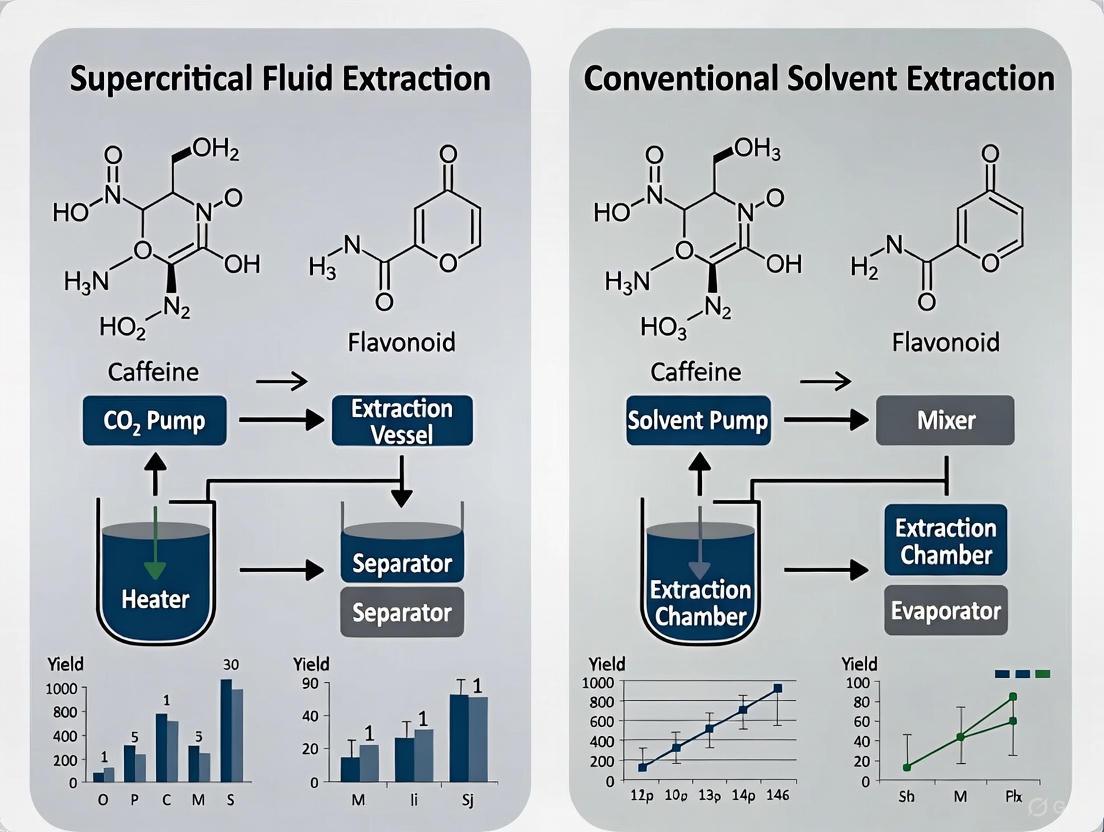
A typical SFE system comprises several key components: a pump for the CO₂, a pressure cell (extraction vessel) to contain the sample, a mechanism for maintaining system pressure, and a collection vessel [5]. The process begins with cooling and pressurizing CO₂ into a liquid state for efficient pumping. The liquid CO₂ then passes through a heating zone where it reaches supercritical conditions before entering the extraction vessel containing the raw material [5]. Within the vessel, SC-CO₂ permeates the matrix, dissolving the target compounds. This solution then flows into a separator where reduced pressure causes the CO₂ to lose its solvating power, precipitating the extracted material [5]. The CO₂ gas can then be recycled in a closed-loop system or vented, leaving behind a high-purity, solvent-free extract [3] [2].
Figure 1: SFE System Process Flow. The diagram illustrates the closed-loop pathway of CO₂ from liquid to supercritical state and back, highlighting the solvent recovery and recycle process [5] [2].
Carbon Dioxide as the Supercritical Solvent of Choice
Critical Properties and Advantages
Carbon dioxide's predominance in SFE applications stems from its favorable physical and chemical properties. With a critical temperature of 31.1°C and a critical pressure of 7.38 MPa, CO₂ can reach the supercritical state under relatively mild and economically achievable conditions [3] [5]. This low critical temperature is particularly advantageous for processing thermolabile compounds found in pharmaceuticals, essential oils, and food products, as it prevents thermal degradation during extraction [3] [4]. Furthermore, CO₂ is non-toxic, non-flammable, chemically inert, and readily available in high purity at relatively low cost [4]. Its gaseous state at ambient conditions enables easy separation from the extract through simple depressurization, eliminating the energy-intensive evaporation steps required with organic solvents [3].
Environmental and Safety Benefits
The environmental profile of SC-CO₂ extraction presents significant advantages over conventional solvent-based methods. CO₂ is not classified as a Volatile Organic Compound (VOC) and its use in closed-loop SFE systems minimizes greenhouse gas emissions [2]. Unlike hydrocarbon solvents, SC-CO₂ leaves no toxic residues in the final product, a critical consideration for pharmaceutical, food, and cosmetic applications [3]. The closed-loop operation of industrial SFE systems allows for recycling and reuse of CO₂, dramatically reducing solvent consumption and waste generation compared to traditional methods that require disposal of spent solvents [3] [6]. This aligns with the principles of green chemistry and meets increasingly stringent regulatory standards across multiple industries [6].
Modifiers for Enhanced Polarity
A limitation of pure SC-CO₂ is its inherently non-polar character, which restricts its effectiveness for extracting highly polar compounds [5] [7]. To overcome this, small quantities of polar modifiers (co-solvents) such as ethanol, methanol, or water are added to the CO₂ stream to enhance its solvating power for polar molecules [5] [8]. Ethanol is frequently preferred for food and pharmaceutical applications due to its safety profile and regulatory acceptance [8] [7]. The incorporation of modifiers like ethanol significantly expands the range of extractable compounds, enabling efficient recovery of polyphenols, flavonoids, and other polar bioactive substances without compromising the environmental benefits of the process [8] [7]. Research on hemp seed oil extraction demonstrated that adding 10% ethanol to SC-CO₂ significantly increased the yield of phenolic compounds and tocopherols while maintaining favorable oil quality parameters [7].
SFE vs. Conventional Solvent Extraction: A Comparative Analysis
Operational and Performance Comparison
When objectively comparing SFE to conventional solvent extraction methods like Soxhlet, maceration, or reflux extraction, distinct differences emerge in operational parameters, performance characteristics, and final product quality. The following table summarizes key comparative factors based on experimental studies and technical data.
Table: Comprehensive Comparison of SFE vs. Conventional Solvent Extraction
| Characteristic | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction |
|---|---|---|
| Solvent Type | Primarily CO₂ (non-toxic, non-flammable) [3] | Organic solvents (hexane, ethanol, methanol, chloroform) [3] [8] |
| Selectivity | High (Tunable via pressure & temperature) [3] [5] | Low to Moderate (Fixed by solvent choice) [3] |
| Extraction Speed | Fast (10-60 minutes) due to high diffusivity [5] | Slow (several hours to days) limited by liquid mass transfer [3] |
| Operating Temperature | Low (e.g., 31-60°C), suitable for heat-sensitive compounds [3] [4] | Often high (e.g., 60-100°C), risk of thermal degradation [3] |
| Solvent Residue | None (CO₂ evaporates) [3] | Potential for toxic residues requiring post-processing [3] |
| Environmental Impact | Low (Closed-loop, recyclable solvent) [3] [2] | High (Hazardous waste disposal needed) [3] |
| Product Purity | High, with selective fractionation possible [5] | May contain co-extracted impurities and solvent residues [3] |
| Capital Cost | High initial investment [3] | Lower initial investment [3] |
| Operational Cost | Lower solvent costs, energy for compression [3] | Ongoing solvent purchase and waste disposal costs [3] |
Experimental Yield and Recovery Data
Comparative studies across various plant materials consistently demonstrate the efficacy of SFE for recovering valuable bioactive compounds. The following table compiles experimental yield data from recent research investigations.
Table: Experimental Yield Comparisons Between SFE and Conventional Methods
| Plant Material | Target Compound | SFE Conditions | SFE Yield/Recovery | Conventional Method & Yield/Recovery | Citation |
|---|---|---|---|---|---|
| Nepeta crispa | Essential Oil | 25 MPa, 40°C, 3.5% EtOH | 1.812% yield | Hydrodistillation: Lower yield (exact value N/S) | [4] |
| Syzygium campanulatum | Secondary Metabolites (Flavanones, Chalcone) | 30 MPa, 40°C, 5% EtOH | 25.5–84.9% recovery | Soxhlet (n-hexane:methanol): 0.92–66.00% recovery | [8] |
| Hemp Seed | Oil | 20 MPa, 50°C, 244 min | 28.83 g/100g yield | Cold Pressing: ~60-80% oil recovery from seeds | [7] |
| Hemp Seed | Phenolic Compounds | 20 MPa, 50°C, 10% EtOH | 294.15 GAE mg/kg | Not specified in source | [7] |
Detailed Experimental Protocols
Protocol 1: SFE of Nepeta crispa Essential Oil
This protocol optimized the extraction of essential oils from Nepeta crispa using Response Surface Methodology (RSM) [4]:
- Raw Material Preparation: Fresh plant specimens were shade-dried at ambient temperature for one week, ground mechanically, and sieved to a particle size of 0.2 mm [4].
- SFE Equipment: A lab-scale SFE system equipped with a CO₂ pump, modifier pump, automated back-pressure regulator, and oven serving as the extraction chamber was employed [4].
- Extraction Parameters: The study investigated pressures (15-25 MPa), temperatures (40-60°C), and ethanol co-solvent percentages (0.5-3.5%) in a Box-Behnken experimental design [4].
- Optimal Conditions: Maximum yield of 1.812% was achieved at 25 MPa, 40°C, with 3.5% ethanol modifier [4].
- Analysis: GC-Mass analysis identified 1,8-cineol and nepetalactone as primary constituents. The SFE extract demonstrated superior antioxidant and antibacterial activity compared to hydrodistillation [4].
Protocol 2: SFE of Bioactive Compounds from Syzygium campanulatum
This comparative study evaluated SFE against three conventional techniques for recovering specific secondary metabolites [8]:
- Raw Material Preparation: Dried leaves were ground to fine powder using an electric grinder. Particle sizes of 300, 500, and 700 μm were tested for SFE [8].
- SFE Parameters: Extractions were performed at pressures of 10, 20, and 30 MPa, temperatures of 40, 60, and 80°C, with food-grade ethanol as modifier (5% of CO₂) at a flow rate of 4 mL/min for two hours [8].
- Conventional Extractions: Twenty extracts were produced using ten different solvents with maceration, soxhletion, and reflux techniques [8].
- Results: SFE with ethanol modifier produced significantly higher recovery (25.5-84.9%) of target flavanones, chalcone, and triterpenoids compared to the best conventional method (n-hexane:methanol soxhletion at 0.92-66.00%) [8].
Essential Research Reagent Solutions for SFE
Successful implementation of SFE requires specific reagents and materials tailored to the extraction objectives. The following table details key research reagent solutions essential for SFE experiments.
Table: Essential Research Reagent Solutions for SFE Experiments
| Reagent/Material | Specifications | Function in SFE | Application Notes |
|---|---|---|---|
| Carbon Dioxide (CO₂) | High purity (SFE or food grade), ≥99.9% [8] | Primary supercritical solvent | Low critical point (31.1°C, 74 bar); inert, non-toxic [5] |
| Ethanol (Modifier) | Food grade or chemical codex [8] | Polar co-solvent to increase solubility of polar compounds | Typically 1-10% of CO₂ flow; enhances phenolic compound recovery [7] |
| Methanol (Modifier) | HPLC grade [8] | Polar co-solvent for analytical applications | More effective for polar compounds than ethanol but toxic for food/pharma [8] |
| Sample Matrix | Controlled particle size (e.g., 0.2-0.7 mm) [4] [8] | Source of target compounds | Smaller particles increase surface area; moisture content affects yield [1] |
| Solid Support | Inert matrix like glass beads | Packing material for small samples | Ensures even CO₂ flow distribution through extraction vessel |
Supercritical Fluid Extraction using carbon dioxide represents a technologically advanced, environmentally sustainable, and highly efficient alternative to conventional solvent extraction methods. The tunable solvation power of SC-CO₂ through precise pressure and temperature control enables selective extraction that preserves the integrity of heat-sensitive bioactive compounds [3] [5]. While the initial capital investment for SFE equipment exceeds that of traditional extraction systems, the long-term benefits of superior product purity, elimination of solvent residues, reduced environmental impact, and lower operational costs present a compelling value proposition [3] [6].
Experimental evidence consistently demonstrates that SFE can achieve comparable or superior extraction yields while recovering compounds with enhanced biological activity due to the gentle processing conditions [4] [8]. The growing global demand for SFE equipment across pharmaceutical, food, and cosmetic industries underscores its commercial viability and technological maturity [6]. As regulatory pressures against hazardous solvents intensify and consumer preference for clean-label products grows, SFE with CO₂ is positioned to become the extraction technology of choice for high-value, sensitive applications where product quality, safety, and environmental sustainability are paramount.
Within the broader research context of supercritical fluid extraction versus conventional solvent extraction yield studies, understanding established conventional methods is a fundamental prerequisite for accurate comparative analysis [9] [10]. Conventional techniques such as maceration, Soxhlet, and reflux extraction have served as the benchmark for decades in laboratories and industries focused on isolating natural products for drug development [11] [12]. Despite the emergence of greener and more efficient technologies, these methods remain widely used due to their operational simplicity and well-understood principles [13]. This guide provides an objective comparison of these three conventional techniques, detailing their methodologies, performance, and applications to furnish researchers and scientists with a clear baseline for evaluating advanced extraction systems.
Principles and Methodologies
This section outlines the core principles and standard experimental protocols for each extraction technique, providing a foundation for their comparison.
Maceration
Maceration is a simple, cold extraction technique that involves soaking a solid plant matrix in a solvent for an extended period [11].
- Principle: The process relies on the passive diffusion of soluble compounds from the plant material into the surrounding solvent [12]. The driving force is the concentration gradient between the interior of the plant cell and the external solvent [11].
- Experimental Protocol: The standard protocol involves using a finely ground solid sample (e.g., 1 g of lyophilized plant powder) combined with a selected solvent (e.g., ethanol, water, acetone) at a typical material-to-liquid ratio of 1:30 (g/mL) [14]. The mixture is kept in a sealed container at room temperature with periodic stirring for a duration ranging from several hours to days. The supernatant is subsequently separated from the solid residue via filtration or centrifugation (e.g., 10,000×g for 10 min) and concentrated, often using a rotary evaporator [14].
Soxhlet Extraction
Soxhlet extraction is a continuous, automated method that uses repeated cycles of solvent percolation to achieve exhaustive extraction [15] [16].
- Principle: The apparatus uses the principles of solvent reflux and siphoning [9]. The system continuously delivers fresh, warm solvent to the solid sample, maintaining a high concentration gradient and thereby improving mass transfer and extraction efficiency compared to simple maceration [9] [15].
- Experimental Protocol: A dried, ground sample is placed in a porous cellulose thimble, which is then loaded into the main chamber of the Soxhlet apparatus [16]. A suitable solvent (e.g., hexane, petroleum ether, ethanol) is added to a round-bottom flask and heated. The solvent vaporizes, is condensed back into liquid, and drips onto the sample. Once the liquid in the extraction chamber reaches the siphon's apex, it automatically empties back into the flask, carrying the extracted compounds with it. This cycle repeats automatically for many hours, typically between 4 and 24 [16]. After extraction, the solvent in the flask is evaporated to recover the final extract [15].
Reflux Extraction
Reflux extraction employs a condenser attached to a boiling flask to prevent solvent loss during heated extraction [9].
- Principle: This method involves boiling a mixture of solid sample and solvent while continuously condensing the vapor and returning it to the reaction flask [9] [11]. This allows for the use of temperatures above the solvent's atmospheric boiling point without losing solvent, which can increase the solubility of target compounds and accelerate the extraction rate [9].
- Experimental Protocol: A known mass of solid sample is combined with solvent in a round-bottom flask. A reflux condenser is attached vertically to the flask. The mixture is heated to a gentle boil, and the extraction proceeds for a set time, often 1 to 2 hours. The condensed solvent continuously drips back into the flask. After cooling, the extract is filtered or centrifuged to remove particulate matter, and the solvent is evaporated to concentrate the product [11].
The following diagram illustrates the operational workflow and logical relationship between the steps of the Soxhlet extraction cycle, the most complex of the three conventional methods.
Comparative Performance Analysis
A critical comparison of these techniques reveals significant differences in their efficiency, operational requirements, and suitability for different types of compounds. Quantitative data from research studies highlight these performance variations.
Table 1: Comparative Analysis of Conventional Extraction Techniques
| Feature | Maceration | Soxhlet Extraction | Reflux Extraction |
|---|---|---|---|
| Principle | Passive diffusion & soaking [11] | Continuous solvent recycling & percolation [9] [15] | Heated solvent circulation with condensation [9] |
| Temperature | Room temperature [11] | Boiling point of the solvent [15] | Boiling point of the solvent [9] |
| Extraction Time | Long (several hours to days) [11] | Long (4 to 24 hours) [16] | Moderate (1 to 2 hours typical) [11] |
| Solvent Consumption | Large volume [11] | Moderate volume, but recycled [16] | Moderate volume, conserved by reflux [9] |
| Efficiency | Low to moderate [11] [12] | High (exhaustive) [15] [16] | High [9] |
| Suitability | Thermolabile compounds [11] | Stable, non-volatile compounds; ideal for lipids [15] [16] | Volatile and heat-stable compounds [9] |
| Advantages | Simple, low-cost equipment, ideal for heat-sensitive compounds [11] [16] | Continuous process, no filtration required, high reproducibility [9] [16] | Faster than maceration/Soxhlet, prevents solvent loss [9] |
| Disadvantages | Time-consuming, low efficiency, high solvent use [11] | Long time, high heat degrades thermolabile compounds [15] [16] | Unsuitable for thermolabile compounds [9] |
Table 2: Experimental Yield and Bioactivity Data from Comparative Studies
| Study Extract / Compound | Method (Solvent) | Key Performance Metric & Yield | Comparative Bioactivity (e.g., IC₅₀) |
|---|---|---|---|
| Furanochromones from Ammi visnaga [17] | Maceration / Reflux (Water) | Furanochromones: 5.95% (w/w) | Cytotoxicity (MCF-7): Weak (IC₅₀ >90 µg/mL) |
| Maceration (95% Ethanol) | Furanochromones: 8.23% (w/w) | Cytotoxicity (MCF-7): Moderate | |
| Soxhlet (95% Ethanol) | Furanochromones: 6.59% (w/w) | Cytotoxicity (MCF-7): Moderate | |
| SCFE (CO₂ + MeOH) | Furanochromones: 30.10% (w/w) | Cytotoxicity (MCF-7): Strong (IC₅₀ 12.54-17.53 µg/mL) | |
| Phytochemicals from Matthiola ovatifolia [14] | Maceration (Ethanol) | Total Phenolics: Lower yield | Not Specified |
| Microwave-Assisted (Ethanol) | Total Phenolics: 69.6 mg GAE/g (Highest) | Antioxidant & Cytotoxic activity: Highest | |
| Phenolic Compounds (General) [13] | Maceration / Soxhlet | Total Phenolic Content: 48.6 - 71 mg GAE/g | Varies by compound stability |
| Microwave-Assisted Extraction | Total Phenolic Content: Up to 227.63 mg GAE/g | Generally better preserved |
The Scientist's Toolkit: Essential Research Reagent Solutions
The selection of an appropriate solvent is a critical parameter that directly influences the yield, selectivity, and safety of the extraction process. The following table details common solvents used in conventional extraction methods.
Table 3: Key Solvents and Reagents in Conventional Extraction
| Reagent / Solvent | Primary Function in Extraction | Key Considerations for Researchers |
|---|---|---|
| Ethanol | Universal solvent for polar and non-polar substances [9] [11]. | GRAS (Generally Recognized as Safe) status; commonly used for nutraceutical and pharmaceutical extracts [17]. |
| n-Hexane | Selective extraction of non-polar compounds (e.g., lipids, oils) [9]. | Industry standard for oil extraction; highly flammable and has significant environmental impact [9]. |
| Petroleum Ether | Extraction of non-polar lipids and volatile compounds [9]. | Low boiling point; highly flammable; used in maceration and Soxhlet [9]. |
| Water | Extraction of high-polarity compounds (phenolics, sugars) [11]. | Safe, cheap, and green; but can co-extract impurities and requires high energy for removal [17]. |
| Methanol | Efficient extraction of a wide range of phenolic compounds and alkaloids [11]. | More efficient than ethanol for many phytochemicals but is toxic, limiting its use for food/pharma products [11]. |
| Ethyl Acetate | Extraction of medium-polarity compounds like flavonoids and coumarins [9]. | Offers selective separation due to intermediate polarity. |
| Carbon Dioxide (SCF) | Green solvent for supercritical fluid extraction (SFE) [10]. | Non-toxic, non-flammable; tunable solvent power by adjusting P/T; requires high-pressure equipment [10] [3]. |
Maceration, Soxhlet, and reflux extraction form the foundational toolkit for isolating bioactive compounds from natural products. While Soxhlet offers exhaustive extraction and reflux provides a faster heated process, both involve prolonged thermal stress that can degrade thermolabile bioactives [9] [15] [16]. Maceration, while gentle, is inefficient and time-consuming [11]. As demonstrated in the comparative data, these limitations of conventional methods have catalyzed the development of advanced techniques like Supercritical Fluid Extraction (SFE), which can achieve superior yields of heat-sensitive compounds without toxic solvent residues [10] [17] [3]. For researchers in drug development, the choice of an extraction method is a critical determinant of the yield, bioactivity, and safety profile of the final extract, guiding the path from natural material to therapeutic candidate.
The core distinction between Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction (CSE) lies in the fundamental state and tunability of the solvent. SFE utilizes a supercritical fluid, most commonly carbon dioxide (CO₂), which exists at a temperature and pressure above its critical point, exhibiting unique hybrid properties of both gases and liquids [3] [18]. In contrast, CSE relies on conventional organic solvents in their liquid state, such as ethanol, hexane, or methanol, whose solvating power is largely fixed and not easily adjustable [9] [3]. This fundamental difference in solvent nature dictates all subsequent variations in mass transfer, selectivity, and the overall mechanism for isolating compounds from a solid matrix, forming the basis for their performance differences in yield, purity, and environmental impact [19] [20].
Comparative Analysis: SFE vs. CSE
The following tables summarize the key differences in solvent properties, mass transfer characteristics, and overall process attributes between the two extraction techniques.
Table 1: Comparison of Solvent Properties and Mass Transfer Characteristics
| Feature | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction (CSE) |
|---|---|---|
| Solvent State | Supercritical fluid (e.g., CO₂) [18] | Liquid (e.g., Ethanol, Hexane, Methanol) [9] |
| Solvent Properties | Tunable density and solvating power; Low viscosity; High diffusivity [3] [18] | Fixed solubility; Higher viscosity; Lower diffusivity [3] |
| Mass Transfer Mechanism | Combination of convection and enhanced diffusion due to gas-like properties [19] | Primarily diffusion-limited, governed by concentration gradients [9] |
| Driving Force | Pressure and temperature controlling solvent density [19] [18] | Concentration gradient between solid matrix and bulk solvent [9] |
| Selectivity | High and adjustable by modulating pressure (P) and temperature (T) [3] | Low to moderate, primarily dependent on solvent choice [3] |
Table 2: Comparison of Process and Performance Attributes
| Attribute | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction (CSE) |
|---|---|---|
| Operational Parameters | High pressure (e.g., >7.38 MPa for CO₂), Moderate temperature (e.g., 40-60°C) [18] | Atmospheric pressure, Often higher temperatures (e.g., Soxhlet reflux) [9] |
| Extraction Speed | Fast due to high diffusion rates and low viscosity [3] | Slow, limited by liquid mass transfer [3] |
| Product Integrity | High; preserves thermolabile compounds (e.g., antioxidants, volatile oils) [3] | Low to moderate; risk of thermal degradation for sensitive compounds [9] [3] |
| Residual Solvent | Virtually none; CO₂ reverts to gas upon depressurization [3] | Risk of toxic solvent residues, requiring post-processing [9] [3] |
| Environmental Impact | Low; closed-loop CO₂ recycling, no toxic waste [3] | High; requires disposal of large volumes of hazardous solvents [9] [3] |
Experimental Data and Yield Comparisons
Empirical studies across various plant matrices consistently highlight the performance differences between SFE and CSE, particularly in selectivity and the preservation of bioactive compounds.
Table 3: Comparative Experimental Yields from Recent Studies
| Plant Material | Target Compound | SFE Yield & Conditions | CSE Yield & Conditions | Key Finding | Source |
|---|---|---|---|---|---|
| Ammi visnaga (Khella) | Khellin & Visnagin (Furanochromones) | N/A (More selective purified extract) | N/A | SFE produced a more selective extract with better cytotoxic activity. | [21] |
| Ruta graveolens | Polyphenolics (e.g., Gallic acid) | Gallic acid: 1380 µg/g (SC-CO₂) | Gallic acid: Lower than SFE | SFE yielded higher concentrations for most quantified phenolic compounds. | [22] |
| Ruta graveolens | -- | IC₅₀ (DPPH): 5.81 µg/mL (SC-CO₂) | IC₅₀ (DPPH): 7.86 µg/mL (Methanol Soxhlet) | SFE extract demonstrated superior antioxidant activity. | [22] |
| Matthiola ovatifolia | Total Phenolics & Flavonoids | N/A | Max TPC: 69.6 mg GAE/g (MAE, Ethanol) | Advanced techniques like MAE (a modern method) can optimize CSE yields. | [14] |
| General | Thermolabile compounds | High integrity of extracts | Risk of degradation | SFE is superior for heat-sensitive compounds due to low-temperature operation. | [3] |
Detailed Experimental Protocol: SFE vs. Soxhlet for Polyphenol Recovery
The following methodology, derived from a comparative study on Ruta graveolens, outlines a standardized protocol for evaluating the two techniques [22].
1. Plant Material Preparation:
- The aerial parts of the plant are dried and finely ground into a homogeneous powder to increase the surface area for solvent contact.
- The powder is often sieved to achieve a consistent particle size, which ensures uniform extraction kinetics.
2. Supercritical Fluid Extraction (SFE):
- Equipment: A high-pressure SFE system comprising a CO₂ pump, an extraction vessel, a pressure control valve, and a separator.
- Solvent: Food-grade carbon dioxide (CO₂).
- Procedure:
- The plant powder is loaded into the extraction vessel.
- CO₂ is pressurized above its critical point (typically >7.38 MPa) and heated above its critical temperature (typically >31°C). A common operating condition is 30°C and 50 bar as a starting point [22].
- A co-solvent (e.g., methanol, ethanol) may be added in small percentages (e.g., 5-15%) to enhance the solubility of more polar compounds like polyphenols [18].
- The supercritical CO₂ percolates through the plant matrix, dissolving the target compounds.
- The solution is then passed into a separator where pressure is reduced, causing CO₂ to revert to a gas and leaving the extracted compounds behind.
- The extract is collected, and the CO₂ can be recycled in a closed-loop system [22] [3].
3. Conventional Solvent Extraction (Soxhlet):
- Equipment: Standard Soxhlet apparatus, rotary evaporator.
- Solvent: Methanol, ethanol, or hexane, depending on target compound polarity.
- Procedure:
- A defined mass of plant powder is placed in a thimble within the Soxhlet extractor.
- The chosen solvent is heated to reflux in a bottom flask. The solvent vapor condenses and drips onto the plant material, leaching out the soluble components.
- When the liquid in the thimble reaches a siphon level, it flows back to the bottom flask, enriching the solvent with the extract over multiple cycles.
- The process continues for a set number of cycles or time (e.g., 10 hours) [22].
- The solvent is finally removed under reduced pressure using a rotary evaporator to obtain the crude extract [9].
4. Analysis:
- The resulting extracts from both methods are analyzed using techniques like High-Performance Liquid Chromatography (HPLC) for quantifying specific bioactive compounds (e.g., gallic acid, catechin) [22].
- Bioactivities, such as antioxidant capacity (e.g., DPPH radical scavenging assay) and antimicrobial potency, are measured and compared [22].
Mechanisms and Workflows
The underlying mechanisms of SFE and CSE are fundamentally different, which directly impacts their efficiency and selectivity.
Mechanism of Supercritical Fluid Extraction
SFE leverages the unique properties of a supercritical fluid. The mechanism involves three consecutive steps, enhanced by the fluid's physical properties [19] [18]:
- Convective Transport: The supercritical CO₂, with its low viscosity and high diffusivity, easily penetrates the porous structure of the plant matrix.
- Solubilization: The solute (target compound) dissolves into the supercritical fluid. Its solvating power, which is proportional to its density, can be finely tuned by adjusting the system's pressure and temperature. This allows for selective extraction.
- Counter-Diffusion: The solute diffuses from the matrix into the bulk supercritical CO₂ stream, a process accelerated by the fluid's high diffusion coefficient.
SFE Mechanism: Pressure-Driven Mass Transfer
Mechanism of Conventional Solvent Extraction
CSE is primarily governed by Fickian diffusion and is characterized by several sequential and often slower steps [9]:
- Solvent Penetration: The liquid solvent soaks into the plant material.
- Solvation: The solvent interacts with and dissolves the target compounds located within the cells.
- Internal Diffusion: The dissolved solutes slowly diffuse through the saturated plant matrix to the particle surface. This step is often rate-limiting due to the high viscosity of liquid solvents.
- External Transport: The solutes are convected away by the bulk solvent flow.
CSE Mechanism: Diffusion-Limited Process
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of SFE and CSE requires specific reagents and equipment. The table below details essential items for setting up and performing these extractions in a research context.
Table 4: Key Research Reagent Solutions for Extraction Studies
| Item | Function/Description | Relevance |
|---|---|---|
| Supercritical CO₂ (Food Grade) | Primary solvent for SFE; inert, non-flammable, and tunable. | Essential for SFE process. Its purity is critical for reproducible results. [3] [18] |
| Cosolvents (e.g., Ethanol, Methanol) | Added in small quantities (1-15%) to modify the polarity of SC-CO₂, enabling extraction of more polar molecules like polyphenols. | Crucial for expanding the range of compounds extractable via SFE. [22] [18] |
| Organic Solvents (e.g., Hexane, Ethanol, Methanol) | Primary extraction media in CSE. Choice depends on target compound polarity (e.g., ethanol for phenolics, hexane for oils). | Core reagents for all conventional methods like Soxhlet, maceration, etc. [14] [9] |
| High-Pressure Extraction Vessel | Constructed from stainless steel to withstand high pressures; contains the plant biomass during SFE. | Critical and safety-critical component of any SFE system. [3] |
| Soxhlet Extractor Apparatus | Classic glassware for continuous CSE, using solvent reflux and siphoning. | Standard, low-cost equipment for benchmark conventional extractions. [9] [22] |
| Rotary Evaporator | Used to concentrate the extract solution by removing the bulk solvent under reduced pressure and moderate heat. | Essential post-processing step for CSE to obtain a concentrated crude extract. [9] [22] |
| Inert Solid Matrix (e.g., Diatomaceous Earth) | Often mixed with wet plant samples to prevent clumping and improve solvent flow during extraction. | Aids in achieving consistent and efficient extraction from various sample types. |
The Critical Impact of Extraction Choice on Phytochemical Integrity and Final Product Bioactivity
The selection of an extraction technique is a fundamental decision in natural product research, directly determining the yield, profile, and biological activity of the resulting phytochemical compounds. With the growing application of plant-derived extracts in pharmaceuticals, nutraceuticals, and functional foods, understanding the trade-offs between conventional solvent extraction and advanced methods like Supercritical Fluid Extraction (SFE) has become critical for researchers and drug development professionals. This guide provides an objective comparison based on recent scientific investigations, highlighting how extraction choice impacts final product quality and therapeutic potential.
SFE, particularly using supercritical carbon dioxide (SC-CO₂), has emerged as a green and sustainable alternative to conventional methods. It leverages the unique properties of fluids above their critical point (for CO₂: temperature > 31.1 °C and pressure > 73.8 bar), which exhibit gas-like diffusivity and liquid-like solvating power [23]. This review synthesizes experimental data comparing these methodologies, focusing on their influence on phytochemical integrity and bioactivity—a key consideration for developing efficacious therapeutic agents.
Comparative Analysis of Extraction Performance
Extraction Yield and Phytochemical Composition
The performance of conventional solvent extraction and SFE varies significantly across different plant matrices and target compounds. The tables below summarize key experimental findings from recent studies.
Table 1: Comparison of Extract Yields and Key Compound Recovery
| Plant Material | Extraction Method | Solvent/ Conditions | Total Extract Yield (%) | Target Compound Yield/Content | Reference |
|---|---|---|---|---|---|
| Ammi visnaga (Khella) fruits | Conventional | 30% Ethanol | 15.44% | Furanochromones: 6.59% w/w | [17] |
| Conventional | 95% Ethanol | - | Furanochromones: 8.23% w/w | [17] | |
| SFE | CO₂ + 5% MeOH | 4.50% | Furanochromones: 30.1% w/w | [17] | |
| Boehmeria rugulosa wood | Conventional | 100% Water | 31.97% | - | [24] |
| Conventional | 70% Ethanol | 32.59% | Total Phenols: 229.3 mg GAE/g | [24] | |
| Matthiola ovatifolia aerial parts | MAE (Conventional) | Ethanol | - | Total Phenolics: 69.6 mg GAE/g | [25] |
Table 2: Bioactivity Comparison of Extracts from Different Methods
| Bioactivity Assay | Plant Material | SFE Extract Result | Conventional Solvent Extract Result | Reference |
|---|---|---|---|---|
| Cytotoxicity (IC₅₀) vs. MCF-7 & Hep G2 | Ammi visnaga | Strong (12.54 - 17.53 µg/mL) | Moderate to Weak (20 - >90 µg/mL) | [17] |
| Antioxidant Activity | Matthiola ovatifolia | - | Highest in MAE Ethanolic extract | [25] |
| Antibacterial Activity | Boehmeria rugulosa | - | Highest in 70% Aqueous Ethanol extract | [24] |
The data reveals a critical distinction: while conventional solvents often produce a higher crude extract yield, SFE can generate a more concentrated and selective extract for specific bioactive compounds. For instance, in Ammi visnaga, SFE yielded a far purer fraction of furanochromones (30.1%) despite a lower overall mass output [17]. This selectivity directly translated to superior cytotoxic activity against cancer cell lines. The composition of extracts is also notably different; HPLC analysis showed that conventional extracts contained other coumarins and coloring pigments absent in the cleaner SFE chromatogram [17].
Operational Advantages and Environmental Impact
Advanced techniques like SFE offer significant operational and sustainability benefits over conventional methods.
Table 3: Comparison of Operational and Environmental Parameters
| Parameter | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction |
|---|---|---|
| Solvent Consumption | Reduces solvent use by 80-90% [10] | High solvent consumption |
| Energy Consumption | 30-50% lower than traditional methods [10] | High energy demand for solvent removal |
| Extract Purity | ~95% purity achievable [10] | Typically 70-80% purity [10] |
| Process Temperature | Low (e.g., ~31°C for SC-CO₂), preserves heat-labile compounds [10] [23] | Often high, risking degradation of thermosensitive phytochemicals [26] [27] |
| Solvent Residues | Non-toxic, easily removed solvents (e.g., CO₂) [28] [23] | Potential for toxic solvent residues in extract |
| Selectivity | Highly tunable by adjusting pressure, temperature, and co-solvents [28] | Limited selectivity, largely dependent on solvent polarity |
The tunable solubility of SFE is a key advantage. By manipulating pressure and temperature, operators can fine-tune the density and solvating power of the supercritical fluid, allowing for selective targeting of desired compounds [28]. Furthermore, SFE operates at lower temperatures, which is crucial for preserving the integrity of heat-sensitive compounds like flavonoids and polyphenols, whose degradation in conventional Soxhlet extraction has been documented [26] [27].
Detailed Experimental Protocols
To ensure reproducibility and provide a clear basis for comparison, detailed methodologies from key cited studies are outlined below.
Protocol for Supercritical Fluid Extraction ofAmmi visnagaFuranochromones
This protocol is adapted from the study that demonstrated high selectivity for khellin and visnagin [17].
- Raw Material Preparation: Ammi visnaga fruits were dried and ground to a homogeneous particle size.
- Extraction Equipment: A commercial supercritical fluid extraction system was used.
- Supercritical Fluid: Carbon dioxide (CO₂) with 5% methanol as a co-solvent.
- Procedure:
- The ground plant material was loaded into the high-pressure extraction vessel.
- The system was pressurized and heated to the desired supercritical conditions (specific temperature and pressure were optimized but not detailed in the abstract).
- The supercritical CO₂ with co-solvent was passed through the plant matrix at a controlled flow rate.
- The dissolved extract was separated from the CO₂ in a downstream separator by reducing the pressure, causing the compounds to precipitate.
- The extract was collected as a white, slightly fatty solid with a strong characteristic odor.
- Analysis: The extracted furanochromones (khellin and visnagin) were quantified using High-Performance Liquid Chromatography (HPLC).
Protocol for Conventional Solvent Extraction ofMatthiola ovatifoliaPhytochemicals
This protocol for Microwave-Assisted Extraction (MAE) yielded the highest levels of bioactive compounds in its study [25].
- Raw Material Preparation: Aerial parts of Matthiola ovatifolia were dried and powdered.
- Extraction Equipment: A laboratory-scale microwave-assisted extraction system was used.
- Solvent: Ethanol was identified as the most effective solvent.
- Procedure:
- A defined mass of powdered plant material was mixed with ethanol in a sealed microwave-transparent vessel.
- The mixture was subjected to microwave irradiation under controlled power, temperature, and time parameters to heat the solution rapidly.
- After irradiation, the vessel was cooled, and the mixture was filtered to separate the plant residue (marc) from the liquid extract.
- The solvent was removed from the filtrate under reduced pressure using a rotary evaporator to obtain a concentrated dry extract.
- Analysis: The extract was analyzed spectrophotometrically for total phenolics, flavonoids, tannins, alkaloids, and saponins, and tested for various biological activities.
Visualizing the SFE Process and Its Impact
The following diagrams illustrate the core principles of SFE and the logical relationship between extraction choice and phytochemical integrity.
The Scientist's Toolkit: Key Research Reagent Solutions
This section details essential materials and reagents used in the featured extraction experiments, providing a practical resource for laboratory setup.
Table 4: Essential Reagents and Materials for Extraction Studies
| Item Name | Function/Application | Example from Research |
|---|---|---|
| Supercritical CO₂ System | Core equipment for SFE; provides pressurized and heated CO₂. | Used for selective extraction of furanochromones from Ammi visnaga [17]. |
| Microwave-Assisted Extraction (MAE) System | Equipment for rapid, solvent-based extraction using microwave energy. | Used to achieve highest phytochemical yield from Matthiola ovatifolia [25]. |
| Carbon Dioxide (Food Grade) | Primary solvent for SFE; non-toxic, non-flammable, and recyclable. | The supercritical fluid of choice in most SFE studies [10] [28] [17]. |
| Co-solvents (e.g., Ethanol, Methanol) | Modifies the polarity of SC-CO₂ to enhance extraction of polar compounds. | 5% Methanol used to boost furanochromone yield in SFE [17]. Ethanol is common and GRAS. |
| HPLC-MS System | For precise identification and quantification of individual phytochemicals in complex extracts. | Used to profile volatile compounds in Boehmeria rugulosa [24] and Schinus molle [29]. |
| Spectrophotometer | For spectrophotometric quantification of total phytochemical groups (e.g., phenolics, flavonoids). | Used to measure total phenols and flavonoids in Matthiola ovatifolia [25] and Boehmeria rugulosa [24]. |
The choice between conventional solvent extraction and supercritical fluid extraction presents a clear trade-off: maximizing crude yield versus optimizing target compound purity and bioactivity. Conventional methods, while sometimes yielding more raw extract mass, often result in diluted, less selective mixtures where thermosensitive bioactives may be compromised.
In contrast, SFE emerges as a superior technique for generating potent, high-purity extracts. Its tunable selectivity, low-temperature operation, and minimal environmental footprint make it particularly suited for pharmaceutical and high-value nutraceutical applications where bioactivity and safety are paramount. The experimental data confirms that SFE can produce extracts with significantly enhanced therapeutic potency, as evidenced by stronger cytotoxic effects, despite lower overall crude yields. For researchers prioritizing the isolation of intact, highly active phytochemical principles, SFE represents a critical technology advancing modern natural product-based drug development.
Methodology in Practice: Parameters, Protocols, and Industrial Applications
Supercritical Fluid Extraction (SFE) has emerged as a green and sustainable technique for isolating high-value bioactive compounds from natural sources, offering a superior alternative to conventional solvent extraction methods. This method leverages the unique properties of supercritical fluids, particularly supercritical carbon dioxide (SC-CO₂), which exhibits physicochemical properties intermediate between those of liquids and gases. SC-CO₂ possesses the diffusivity and viscosity of a gas while maintaining the density and solvating power of a liquid, enabling enhanced selectivity, efficiency, and environmental sustainability of the extraction process [23]. The critical point of CO₂ is achieved at mild conditions (31.1 °C and 73.8 bar), making it particularly suitable for heat-sensitive compounds and providing significant advantages over traditional organic solvents which often involve toxicity, higher energy consumption, and environmental pollution [23].
The efficiency of SFE is governed by three primary parameters: temperature, pressure, and the use of co-solvents. These parameters directly influence the solvating power of the supercritical fluid by affecting its density and mass transfer properties. Pressure and temperature adjustments can tune the density of SC-CO₂, thereby controlling its solubility potential, while co-solvents are added to modify the polarity of the supercritical fluid and enhance the extraction of more polar compounds [23]. Understanding the interplay of these parameters is crucial for optimizing extraction yields, selectivity, and the bioactivity of the final extract. This guide provides a comprehensive, data-driven comparison of how these parameters influence SFE performance across various applications, offering researchers detailed experimental protocols and quantitative comparisons with conventional extraction methods.
The Fundamental Role of Temperature and Pressure
Temperature and pressure are the most critical operational parameters in SFE, as they directly dictate the density and, consequently, the solvating power of the supercritical fluid. The relationship between these parameters is complex, with a phenomenon known as the "crossover pressure" identified, where solubility does not depend linearly on pressure or temperature alone, but rather on the fluid density [23]. In general, increasing pressure at constant temperature increases the fluid density, which enhances solute solubility. Conversely, the effect of temperature is more dualistic: at constant pressure, increasing temperature can decrease fluid density (reducing solvating power) but simultaneously increase the vapor pressure of the solute (enhancing solubility) [23].
The optimization of these parameters is therefore system-specific. For instance, in the recovery of hydrocarbons from waste oil-based drilling mud, the optimal conditions were identified as 14 MPa (140 bar) pressure and 45 °C temperature, yielding an extraction efficiency of 55.46% [30]. In contrast, for extracting γ-oryzanol-rich extracts from rice bran, a much higher pressure of 500 bar combined with 62 °C temperature was optimal, resulting in a mass yield of 17.3% and a γ-oryzanol concentration of 36.6 mg per gram of extract [31]. This higher pressure significantly increases the density of SC-CO₂, facilitating the extraction of less volatile and higher molecular weight compounds.
The extraction of bioactive compounds from Peruvian Schinus molle leaves was studied across a range of temperatures (35, 45, 55 °C) and pressures (150, 250, 350 bar). The highest yield was obtained at 150 bar and 45 °C, demonstrating that maximum solubility is not always achieved at the highest pressures and that different compound classes have distinct optimal points [29]. The following workflow outlines the typical procedure for optimizing temperature and pressure in an SFE process:
Table 1: Impact of Temperature and Pressure on SFE Yield in Various Applications
| Source Material | Optimal Pressure | Optimal Temperature | Resulting Yield/Concentration |
|---|---|---|---|
| Waste Oil-Based Mud [30] | 140 bar | 45 °C | 55.46% Extraction Efficiency |
| Rice Bran [31] | 500 bar | 62 °C | 17.3% mass yield, 36.6 mg/g γ-oryzanol |
| Schinus molle Leaves [29] | 150 bar | 45 °C | Highest extract yield |
| Ammi visnaga Fruits [17] | Not Specified | Not Specified | 30.1% Furanochromones |
Co-solvent Effects and Selectivity Enhancement
While SC-CO₂ is excellent for extracting non-polar compounds, its ability to dissolve polar molecules is limited. This constraint is overcome through the use of co-solvents or entrainers, which are typically small quantities of polar solvents such as ethanol, methanol, or water added to the SC-CO₂ to modify its polarity and improve the solubility of polar solutes [23]. Co-solvents function by specific molecular interactions between the co-solvent and solute, such as hydrogen bonding, which significantly enhance the solubility of polar compounds without substantially increasing the operational pressure requirements.
A compelling example of co-solvent efficacy comes from the extraction of furanochromones from Ammi visnaga (Khella) fruits. In this study, SFE with CO₂ containing 5% methanol as a co-solvent produced an extract with a furanochromone content of 30.1%, which was substantially higher than that achieved with conventional solvents like 95% ethanol (8.23%) or boiling water (5.95%) [17]. Furthermore, the SCFE extract demonstrated superior cytotoxic activity against cancer cell lines (IC₅₀ < 20 µg/mL) compared to conventional extracts, highlighting that co-solvent-enhanced SFE not only improves yield and selectivity but can also enhance the bioactivity of the final product [17].
The selectivity enabled by co-solvents is another significant advantage. HPLC analysis of the Ammi visnaga extracts revealed that conventional solvent extracts contained numerous other coumarins and coloring pigments not present in the SFE extract, which was selectively enriched with the target furanochromones (khellin and visnagin) and had a white, slightly fatty appearance with a strong characteristic odor [17]. This purity can reduce downstream purification costs and is particularly valuable in pharmaceutical applications.
Comparative Analysis: SFE vs. Conventional Solvent Extraction
When objectively comparing SFE to conventional solvent extraction (CSE), the advantages of SFE extend beyond its green credentials to include tangible benefits in selectivity, extract purity, and bioactivity. While total mass yield from CSE may sometimes be higher due to the non-selective dissolution of a wider range of compounds, SFE often provides superior performance in the targeted recovery of specific high-value bioactive components.
The comparison between SFE and n-hexane extraction of rice bran is illustrative. While both methods showed similar global yields (SFE: 17.3% vs. n-hexane: 18.0%) and fatty acid concentrations, SFE demonstrated higher selectivity and yield for the target bioactive γ-oryzanol (6.3 mg/g of rice bran for SFE vs. 3.3 mg/g for hexane) [31]. Moreover, the SFE extract exhibited enhanced bioactivity, showing a lower EC₅₀ (0.9 mg/mL) in inhibiting colorectal cancer cell growth compared to the hexane extract (EC₅₀ = 1.15 mg/mL) [31].
Similarly, for Ammi visnaga fruits, although 30% ethanol extraction gave the highest total extract yield (15.44%), SFE with co-solvent yielded a much more concentrated furanochromone extract (30.1% vs. 6.59% for 30% ethanol) [17]. This underscores a key distinction: CSE often maximizes bulk mass, while SFE can optimize the concentration and potency of desired actives. The following table provides a direct comparison of these two case studies.
Table 2: SFE vs. Conventional Solvent Extraction: Direct Experimental Comparisons
| Parameter | Rice Bran Extraction [31] | Ammi visnaga Fruit Extraction [17] |
|---|---|---|
| SFE Yield | 17.3% mass yield | 4.50% mass yield |
| Conventional Solvent Yield | 18.0% (n-hexane) | 15.44% (30% Ethanol) |
| Target Compound | γ-Oryzanol | Furanochromones (Khellin & Visnagin) |
| Target Compound Yield in SFE | 6.3 mg/g rice bran | 30.1% in extract |
| Target Compound Yield in Conventional | 3.3 mg/g rice bran (n-hexane) | 6.59% in extract (30% Ethanol) |
| Bioactivity Advantage | SFE extract more effective at inhibiting cancer cell growth (EC₅₀ = 0.9 mg/mL) | SFE extract showed strong cytotoxic activity (IC₅₀ < 20 µg/mL) |
Advanced Modeling and Optimization Techniques
Modern SFE optimization has moved beyond one-factor-at-a-time approaches to sophisticated statistical design and machine learning (ML) models that capture complex non-linear interactions between process parameters. Response Surface Methodology (RSM) is widely employed; for example, a Box-Behnken design was used to optimize the SFE of hydrocarbons from waste oil-based mud, establishing a quadratic polynomial regression model with an excellent R² value of 0.98229 [30].
More recently, ensemble machine learning frameworks have demonstrated remarkable predictive accuracy for drug solubility in SC-CO₂. One study combined Extreme Gradient Boosting (XGBoost), Light Gradient Boosting (LightGBM), and CatBoost regression, optimized with bio-inspired algorithms, achieving a predictive accuracy of R² = 0.9920 [32]. Another study found that the XGBoost model alone could achieve an R² of 0.9984 and an RMSE of 0.0605 in predicting drug solubility, with 97.68% of data points falling within the model's applicability domain [33]. These ML models utilize input parameters such as temperature, pressure, drug molecular weight, melting point, critical properties, and acentric factor to provide highly accurate solubility predictions, thereby reducing the need for extensive experimental trials.
The following diagram illustrates the integrated approach of combining traditional experimental design with modern machine learning for SFE optimization:
Experimental Protocols and Methodologies
- Experimental Design: Box-Behnken Design (BBD) with three factors (pressure, temperature, time).
- Optimal Parameters: 140 bar pressure, 45 °C temperature, 60 min extraction time.
- Material Preparation: Waste oil-based mud was used as received without further treatment.
- Extraction Process: Supercritical CO₂ was passed through the sample in a high-pressure extraction vessel.
- Analysis: Extraction efficiency was calculated based on recovered hydrocarbon fractions. A kinetic model was formulated to elucidate the mass transfer mechanism.
- Model Validation: The regression model was validated with experimental data, achieving R² = 0.98229.
- Material Preparation: Leaves were dried at room temperature, vacuum-packed, stored at -20 °C, then ground using an immersion blender.
- Experimental Design: 3² factorial design testing three temperature levels (35, 45, 55 °C) and three pressure levels (150, 250, 350 bar).
- Extraction Process: Supercritical CO₂ extractions were performed with a flow rate of 3.28 g CO₂/min.
- Modeling: Overall extraction curves (OEC) were modeled using Spline, logistic, and Esquível models.
- Analysis: Extracts were analyzed by GC-MS, identifying 26 compounds primarily sesquiterpenoids (57.17%), sesquiterpenes (24.50%), and triterpenoids (10.48%).
- Co-solvent Application: Supercritical CO₂ with 5% methanol as co-solvent.
- Comparison Extractions: Conventional extractions with boiling water, 30% ethanol, 95% ethanol, and acetone.
- Analysis: HPLC analysis for khellin and visnagin content.
- Bioactivity Testing: Cytotoxic activity evaluated using sulforhodamine B assay on breast cancer (MCF-7) and hepatocellular carcinoma (Hep G2) cell lines.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for SFE Experimentation
| Reagent/Material | Function in SFE | Application Example |
|---|---|---|
| Supercritical CO₂ | Primary extraction fluid | Universal solvent for non-polar compounds [23] |
| Methanol | Polar co-solvent | Enhanced extraction of furanochromones from Ammi visnaga [17] |
| Ethanol | Green polar co-solvent | Commonly used for extraction of polar bioactive compounds [23] |
| Rice Bran | Model substrate for nutraceuticals | γ-Oryzanol and fatty acid extraction [31] |
| Schinus molle Leaves | Botanical source of terpenoids | Extraction of sesquiterpenoids and triterpenoids [29] |
| Ammi visnaga Fruits | Source of furanochromones | Selective extraction of khellin and visnagin [17] |
| Waste Oil-Based Mud | Industrial waste material | Hydrocarbon recovery and environmental remediation [30] |
The optimization of Supercritical Fluid Extraction requires a nuanced understanding of the complex interplay between temperature, pressure, and co-solvent effects. Through strategic manipulation of these parameters, SFE can achieve superior selectivity, yield, and bioactivity for target compounds compared to conventional extraction methods. The integration of advanced modeling approaches, particularly machine learning, with traditional experimental design provides a powerful framework for accelerating SFE process development. As the pharmaceutical, nutraceutical, and food industries continue to prioritize green and sustainable technologies, SFE stands out as a versatile and efficient extraction platform capable of producing high-value extracts with enhanced functional properties. The experimental data and protocols presented in this guide provide researchers with a comprehensive foundation for designing and optimizing SFE processes tailored to their specific applications.
The extraction of bioactive compounds from plant materials is a foundational step in natural product research, pharmaceutical development, and the creation of functional foods [34]. Among the various techniques available, conventional solvent extraction remains widely used due to its simplicity, low initial equipment costs, and minimal technical requirements [35]. The efficacy of these conventional methods hinges critically on the optimization of three fundamental parameters: solvent polarity, extraction time, and process temperature [36] [37]. These parameters interact in complex ways to influence both the yield and quality of the extracted phytochemicals, creating a series of trade-offs that researchers must carefully balance [26]. Within the broader context of comparing supercritical fluid extraction (SFE) with conventional solvent extraction, understanding these parameter relationships is essential for making informed methodological choices and for appreciating the advantages and limitations of each approach [10]. This guide provides a comprehensive comparison of conventional extraction parameters, supported by experimental data and analysis of their impacts on final extract yield, composition, and bioactivity.
Fundamental Principles of Conventional Extraction
Conventional extraction methods, including maceration and Soxhlet extraction, operate on the principle of using liquid solvents to dissolve and transport bioactive compounds from plant matrices into solution [13]. The process involves several mass transfer stages: penetration of the solvent into the plant material, dissolution of target compounds, and diffusion of these compounds to the bulk solvent [20]. The efficiency of this process is governed by multiple interdependent factors, with solvent polarity, temperature, and time representing the most significant controllable parameters.
Solvent polarity determines which classes of compounds will be effectively dissolved based on the "like dissolves like" principle. Polar solvents such as water, ethanol, and methanol are effective for extracting hydrophilic compounds like phenolics and flavonoids, while non-polar solvents like hexane are better suited for lipophilic compounds [26] [37]. Temperature influences solubility, diffusion rates, and solvent viscosity, with higher temperatures generally increasing extraction efficiency but potentially degrading thermolabile compounds [20]. Extraction time must be sufficient to allow for adequate compound recovery but prolonged exposure may lead to oxidative degradation [35]. The interrelationship between these parameters creates a complex optimization landscape that researchers must navigate to maximize target compound recovery while maintaining bioactivity.
Comparative Analysis of Extraction Parameters
Solvent Polarity and Composition
Solvent selection is arguably the most critical parameter in conventional extraction, directly determining which compound classes will be preferentially extracted. The dielectric constant of the solvent serves as a reliable indicator of its polarity and can guide selection based on target compound properties [26].
Table 1: Effect of Solvent Polarity on Phytochemical Recovery
| Solvent System | Total Phenolic Content (TPC) | Total Flavonoid Content (TFC) | Preferred Compound Classes | Optimal Applications |
|---|---|---|---|---|
| Water (polar) | Low to moderate [37] | Low [37] | Polar glycosides, polysaccharides, tannins | Food-grade extracts, safety-critical applications |
| Ethanol (polar) | High [35] | High [36] | Flavonoids, phenolic acids, saponins | Nutraceuticals, tinctures, food applications |
| Methanol (polar) | Very high [37] | Moderate to high [14] | Alkaloids, flavonoids, phenolics | Analytical applications, research settings |
| Acetone (mid-polarity) | Moderate [14] | Moderate [14] | Medium polarity flavonoids, some terpenoids | Selective extraction of intermediate polarity compounds |
| Ethanol-Water Mixtures | High to very high [35] [36] | High [36] | Broad spectrum of phenolics and flavonoids | Most applications requiring balanced polarity |
Experimental evidence consistently demonstrates that binary solvent systems often outperform pure solvents. In olive leaf extraction, ≥75% aqueous methanol provided the highest total phenolic content, while ≥75% ethanol maximized flavonoid recovery and antioxidant capacity measured by ABTS assay [37]. Similarly, optimization studies on potato peels determined that 80% aqueous ethanol represented the optimal balance for phenolic compound extraction [35]. The mechanism behind this enhancement involves the modified dielectric constant of the mixture, which improves the solubility of a wider range of compound polarities and enhances cell wall swelling for better compound release [26].
Temperature Optimization and Trade-offs
Temperature exerts a dual effect on extraction efficiency, enhancing compound recovery while potentially compromising stability. Elevated temperatures reduce solvent viscosity and surface tension while increasing diffusion rates and solute solubility, collectively improving mass transfer [20].
Table 2: Temperature Effects on Extraction Efficiency and Compound Stability
| Temperature Range | Extraction Efficiency | Impact on Compound Stability | Recommended Applications |
|---|---|---|---|
| Room Temperature (25-30°C) | Low to moderate, extended time required | Minimal degradation, ideal for thermolabile compounds | Maceration of heat-sensitive flavonoids, aromatic compounds |
| Moderate (40-60°C) | Good efficiency with reasonable time | Some volatile loss, minimal phenolic degradation | Most phenolic compounds, saponins, some alkaloids |
| High (>60°C) | Very high efficiency, rapid extraction | Significant risk of thermolabile compound degradation | Stable compounds, cellulose-bound metabolites |
Quantitative studies demonstrate these trade-offs clearly. Research on Pyrus spinosa fruit extraction showed that increasing temperature from 20°C to 80°C enhanced total polyphenol content by up to 345% and antioxidant capacity by 2356% [36]. Similarly, olive leaf extractions at temperatures >50°C increased phenolic yields up to fivefold compared to room temperature extraction [37]. However, these impressive yield increases may not translate directly to bioactivity preservation, as high temperatures can degrade heat-sensitive flavonoids and alter phenolic profiles [26]. The optimal temperature must therefore be determined based on the stability of the target compounds, with thermolabile substances requiring more conservative temperature profiles.
Time Parameter Optimization
Extraction time represents a balance between process efficiency and practical considerations. Insufficient time prevents equilibrium establishment, while excessive duration offers diminishing returns and risks compound degradation [35].
Conventional extraction typically requires longer timeframes than modern assisted techniques. Optimization studies for potato peel phenolics determined 150 minutes as the ideal duration [35], while Pyrus spinosa fruit extraction achieved optimal results within 60-120 minutes depending on temperature and solvent combination [36]. Evidence suggests that most phenolic compounds are recovered within ≤1 hour under optimized conditions, with extended times providing minimal additional benefit [37]. The interaction between time and temperature is particularly important, as higher temperatures can significantly reduce the time required to reach extraction equilibrium through enhanced diffusion rates [20].
Experimental Protocols for Parameter Optimization
Standardized Maceration Protocol
The following protocol provides a standardized approach for optimizing conventional extraction parameters, adapted from methodologies used in potato peel and Pyrus spinosa research [35] [36]:
Sample Preparation: Fresh plant material should be washed, dried (40-50°C convection oven or freeze-drying), and ground to a particle size of 200-500 μm. Proper drying preserves bioactive compounds while facilitating grinding.
Experimental Design: Implement a Response Surface Methodology (RSM) approach, such as Box-Behnken design, to efficiently explore parameter interactions. Key factors include ethanol concentration (0-100%), temperature (20-80°C), and time (30-180 minutes).
Extraction Procedure: Use a consistent solid-to-liquid ratio of 1:20 to 1:30 (g:mL). Conduct extraction with continuous agitation (500 rpm) using a magnetic stirrer. Protect light-sensitive compounds by performing extractions in amber vessels or dark conditions.
Sample Recovery: Centrifuge mixtures at 3000-4000 × g for 10 minutes. Collect supernatant and store at -40°C until analysis to prevent degradation.
Analysis: Quantify total phenolic content using Folin-Ciocalteu method, total flavonoids via aluminum chloride method, and antioxidant capacity through DPPH and FRAP assays.
Parameter Interaction Studies
To effectively capture parameter interactions, researchers should employ a systematic approach:
- Solvent Gradients: Test ethanol-water mixtures at 20% intervals (0%, 20%, 40%, 60%, 80%, 100%) while holding time and temperature constant.
- Temperature Profiles: Evaluate extraction efficiency at 30°C, 45°C, 60°C, and 75°C using the optimal solvent system.
- Time Course Analysis: Monitor compound recovery at 30, 60, 120, and 180 minutes to identify the point of diminishing returns.
This comprehensive approach enables the construction of predictive models that can identify optimal parameter combinations for specific plant materials and target compounds.
Comparative Performance: Conventional vs. Modern Techniques
When evaluating conventional extraction within the broader context of extraction technologies, distinct advantages and limitations emerge, particularly in comparison with supercritical fluid extraction (SFE).
Table 3: Conventional vs. Supercritical Fluid Extraction Comparative Performance
| Parameter | Conventional Solvent Extraction | Supercritical Fluid Extraction (SFE) |
|---|---|---|
| Solvent Consumption | High (typical solvent-to-feed ratio >20) [13] | Reduced by 80-90% [10] |
| Extraction Time | Long (30-180 minutes) [35] | Short (often <60 minutes) [10] |
| Temperature | Wide range (room temperature to >60°C) [36] | Moderate (typically 31-60°C) [10] |
| Extract Purity | Moderate (70-80%) [10] | High (~95%) [10] |
| Energy Requirements | Moderate to high | 30-50% lower than conventional methods [10] |
| Capital Cost | Low | High |
| Operating Cost | Moderate (primarily solvent replacement) | Moderate to high |
| Environmental Impact | Higher (solvent waste, energy use) | Lower (green solvents, reduced energy) |
| Thermolabile Compound Preservation | Variable (depends on temperature selection) | Excellent (low-temperature operation) |
The economic and environmental trade-offs between these techniques are significant. While SFE offers substantial reductions in solvent consumption (80-90%) and energy use (30-50%) [10], the high capital investment remains a barrier to implementation. Conventional methods provide accessibility and technical simplicity but generate greater environmental impact through solvent waste and higher energy requirements [26]. The selection between these approaches ultimately depends on application-specific requirements, including extract quality needs, production scale, regulatory considerations, and available infrastructure.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Research Reagents for Extraction Optimization Studies
| Reagent/Equipment | Function/Application | Technical Considerations |
|---|---|---|
| Ethanol (Food/Pharma Grade) | Primary extraction solvent, generally recognized as safe (GRAS) | Optimal at 60-80% aqueous concentration for phenolic compounds [35] |
| Folin-Ciocalteu Reagent | Quantification of total phenolic content | Reacts with phenolic hydroxyl groups; use gallic acid for calibration curve [35] |
| Aluminum Chloride | Flavonoid content determination | Forms acid-stable complexes with flavonoid C-4 keto group [35] |
| DPPH (2,2-Diphenyl-1-picrylhydrazyl) | Free radical scavenging antioxidant assay | Measures hydrogen-donating capacity; results as IC50 or TEAC [35] |
| FRAP Reagent | Ferric reducing antioxidant power assay | Evaluates electron-transfer mechanism of antioxidants [36] |
| Rotary Evaporator | Solvent removal and extract concentration | Use temperature <40°C to prevent thermal degradation [14] |
| Lyophilizer | Sample drying and preservation | Maintains compound stability better than thermal drying methods [36] |
Workflow Visualization
Extraction Parameter Optimization Workflow: This diagram illustrates the systematic approach to optimizing conventional extraction parameters, highlighting the critical decision points and parameter trade-offs that researchers must navigate throughout the process.
The optimization of conventional extraction parameters presents a complex interplay of competing priorities. Solvent polarity fundamentally determines extraction selectivity, with ethanol-water mixtures (60-80% ethanol) generally providing the optimal balance for phenolic compounds [35] [36]. Temperature control represents a careful compromise between extraction efficiency and compound stability, with moderate temperatures (40-60°C) offering the best balance for most applications [20] [37]. Extraction time should be sufficient to reach equilibrium while avoiding unnecessary prolongation that risks degradation [35]. When compared with supercritical fluid extraction, conventional methods demonstrate higher solvent consumption and environmental impact but remain accessible and cost-effective for many applications [10] [13]. The selection of extraction methodology ultimately depends on the specific research or production context, including target compounds, quality requirements, economic constraints, and sustainability considerations. Through systematic parameter optimization and understanding of these fundamental trade-offs, researchers can maximize the potential of conventional extraction techniques while making informed decisions about when advanced alternatives like SFE may be warranted.
The choice of extraction technique is a critical determinant of the yield, purity, and bioactivity of compounds isolated from natural sources. Within pharmaceutical and nutraceutical development, the polarity of target molecules presents a fundamental challenge. This guide objectively compares the performance of Supercritical Fluid Extraction (SFE), predominantly using supercritical CO₂ (SC-CO₂), and conventional liquid solvent extraction (LSE) for recovering lipophilic and polar bioactive compounds. The core thesis is that SFE offers a green, selective, and highly efficient platform for lipophilic compounds, while the effectiveness for polar molecules is heavily dependent on the strategic use of co-solvents and optimized protocols. This comparison is framed within the broader context of yield optimization studies, providing researchers with data-driven insights for method selection.
Core Principles and Comparative Mechanisms
Supercritical Fluid Extraction (SFE)
SFE utilizes a fluid, typically carbon dioxide, above its critical temperature (31.1 °C) and critical pressure (73.8 bar) [23]. In this supercritical state, the fluid exhibits unique properties: it possesses the diffusivity and low viscosity of a gas, allowing for deep penetration into solid matrices, combined with the density and solvating power of a liquid [3] [38]. The most significant advantage of SC-CO₂ is its tunable solvent power. By precisely controlling pressure and temperature, operators can manipulate the fluid's density, thereby adjusting its selectivity for different compounds [3]. This enables targeted extractions that are difficult to achieve with conventional solvents. Furthermore, upon depressurization, CO₂ reverts to a gas, separating cleanly from the extract and yielding a solvent-free product [3].
Conventional Liquid Solvent Extraction (LSE)
Conventional LSE relies on organic solvents (e.g., hexane, ethanol, methanol) or water to dissolve desired compounds from a solid matrix based on the principle of "like dissolves like" [9]. These methods, including maceration, percolation, and Soxhlet extraction, are often performed at elevated temperatures and atmospheric pressure. The selectivity is primarily fixed by the choice of solvent, with limited scope for fine-tuning during the process itself [3]. A major drawback is the potential for thermal degradation of heat-sensitive compounds and the challenging, energy-intensive removal of solvent residues from the final extract, which can pose regulatory and safety concerns [3] [39].
Performance Data: A Quantitative Comparison
The following tables summarize key experimental data from published studies, highlighting the performance differences between SFE and LSE across various metrics and specific applications.
Table 1: Overall Process Performance and Economic Comparison
| Metric | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction (LSE) | Source |
|---|---|---|---|
| Solvent Consumption | Reduces solvent use by 80-90% [10] | High solvent consumption | [10] |
| Energy Consumption | 30-50% lower than traditional methods [10] | High energy for heating and solvent removal | [10] |
| Extract Purity | ~95% purity, no toxic solvent residues [10] | 70-80% purity, risk of toxic solvent residues [10] | [3] [10] |
| Typical Solvent | Non-toxic, recyclable CO₂ [3] | Various organic liquids (e.g., Hexane, Ethanol) [3] | [3] |
| Operational Temperature | Low (e.g., 40-60°C), protects heat-sensitive compounds [3] [7] | Often requires high heat, causing degradation [3] | [3] [7] |
| Environmental Impact | Low; recyclable solvent, closed-loop system [3] | High; hazardous waste disposal necessary [3] | [3] |
Table 2: Comparative Extraction Efficiency for Specific Bioactives
| Target Compound / Source | SFE Performance (Conditions) | Conventional LSE Performance (Solvent) | Key Finding | Source |
|---|---|---|---|---|
| Furanochromones (Ammi visnaga) | Yield: 135.0 mg, Purity: 30.1% w/w [17] | Best LSE (95% Ethanol): Yield: 111.1 mg, Purity: 8.23% w/w [17] | SFE yielded a purer extract with superior cytotoxic activity [17] | [17] |
| Phenolic Compounds (Hemp Seed Oil) | Total Phenols: 294.15 GAE mg/kg (with 10% ethanol) [7] | Not specified in source | Ethanol as co-solvent significantly enhanced polar compound recovery [7] | [7] |
| Polar Compounds (Pomegranate Pericarp) | Total Target Compounds: 19.59 mg/g (40°C, 20% EtOH:H₂O) [40] | Traditional maceration takes days, large solvent use [40] | Optimized SFE with polar modifier efficiently recovered ellagic acid & punicalagins [40] | [40] |
| Lipid Bioactives (General) | High selectivity for lipophilic compounds, concentrated target compounds [39] | Effective but non-selective; co-extracts pigments & impurities [17] | SFE's tunability allows for selective extraction of lipophilics [39] [17] | [39] [17] |
Detailed Experimental Protocols
Protocol 1: SFE of Furanochromones fromAmmi visnagaL. Fruits
This protocol is derived from a study that directly compared SFE with conventional solvents for extracting khellin and visnagin [17].
- Objective: To selectively extract furanochromones (khellin and visnagin) from Ammi visnaga fruits.
- SFE Setup and Conditions:
- Solvent: Supercritical CO₂ with 5% methanol as a co-solvent.
- Extraction Vessel: Packed with pre-treated, powdered plant material.
- Pressure and Temperature: Maintained above the critical point of CO₂ (exact parameters optimized by the researcher).
- Process: The SC-CO₂ flows through the vessel, dissolving the target compounds.
- Separation: The extract-laden fluid is passed through a separator where pressure is reduced, causing CO₂ to revert to gas and the extract to precipitate.
- Collection: The dry, solvent-free extract is collected. The CO₂ gas can be recycled in a closed-loop system [17].
- Comparison with LSE: The same plant material was extracted in parallel using boiling water, 30% ethanol, 95% ethanol, and acetone using standard maceration or reflux techniques, followed by solvent evaporation [17].
- Key Outcome: The SFE extract was white and of high purity (30.1% furanochromones), while the LSE extracts were brown and contained lower concentrations of target compounds (5.95-8.23%) due to co-extraction of other materials [17].
Protocol 2: Optimized SFE of Polar Phenolics fromLabisia pumila
This protocol exemplifies the use of Response Surface Methodology (RSM) to optimize SFE for polar compounds [41].
- Objective: To maximize the yield of phenolic acids (gallic acid, methyl gallate, caffeic acid) from Labisia pumila leaves.
- Optimization Design:
- Method: Response Surface Methodology (RSM) using a Box-Behnken Design.
- Parameters Optimized: Pressure, temperature, percentage of ethanol in co-solvent, and total co-solvent concentration.
- Optimal SFE Conditions:
- Pressure: 283 bar
- Temperature: 32 °C
- Co-solvent: 16% (v/v) of a 78% (v/v) ethanol-water mixture.
- Flow Rate & Time: 4 mL/min for 240 minutes (after 30 min static time) [41].
- Key Outcome: Under these optimized conditions, the extraction yielded 14.05% (g/g) of extract containing 1.27% gallic acid, 0.44% methyl gallate, and 1.38% caffeic acid, demonstrating the successful application of modified SFE for polar phenolics [41].
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for SFE and LSE Experiments
| Item | Function in Extraction | Example Application / Note |
|---|---|---|
| Supercritical CO₂ | Primary solvent in SFE; non-toxic, non-flammable, tunable. | Food-grade, commercial-grade (99.9% purity) is typically used [41]. |
| Ethanol (Food/Pharma Grade) | Green polar co-solvent for SFE; primary solvent for polar compounds in LSE. | Used to modify polarity of SC-CO₂ (e.g., 5-20%) [7] [41]. Common LSE solvent [17]. |
| Water (HPLC Grade) | Additive in co-solvent mixture to further increase polarity for SFE; solvent in LSE. | Used in hydro-ethanol mixtures for SFE of highly polar compounds (e.g., punicalagins) [40]. |
| Methanol (HPLC Grade) | Polar co-solvent for SFE; solvent for analytical standard preparation. | Sometimes used as a co-solvent for analytical-scale SFE [17]. |
| Hexane or Petroleum Ether | Non-polar organic solvent for LSE of lipids and lipophilic compounds. | Effective for oils but poses residue and toxicity concerns [3] [9]. |
| High-Pressure Extraction Vessel | Contains the solid sample and withstands operational pressures for SFE. | Constructed from stainless steel; core component of any SFE system [3]. |
| Back-Pressure Regulator | Maintains system pressure above the critical point of CO₂ during SFE. | Critical for maintaining the supercritical state [41]. |
| Response Surface Methodology (RSM) | Statistical tool for optimizing multiple SFE parameters efficiently. | Used to model and optimize pressure, temperature, and co-solvent [7] [41]. |
Analysis of Compound-Dependent Recovery Efficiency
The relationship between compound polarity and extraction method efficiency is the cornerstone of selectivity. The schematic above visualizes this critical decision-making pathway.
Lipophilic Compounds: SFE with pure SC-CO₂ is the unequivocal champion for non-polar molecules. Its gas-like diffusivity allows superior penetration, and the solvent power is perfectly matched to oils, fats, and essential oils. The result is high-purity extracts without the contamination risks associated with hydrocarbon solvents like hexane [3] [39]. For these compounds, SFE outperforms LSE in both purity and environmental safety.
Polar Compounds: For highly polar molecules like certain phenolic acids and sugars, traditional hydro-ethanolic LSE remains a straightforward and effective, though less green, option [9] [17]. Its effectiveness is based on the inherent polarity of the solvents. The primary challenge with LSE is the subsequent need for solvent removal, which can be energy-intensive and may leave behind regulated residues [3] [10].
The Middle Ground: The most significant advancement in SFE is its ability to bridge this polarity gap. By adding a polar co-solvent (or modifier) like ethanol, the polarity of SC-CO₂ can be dramatically increased. This transforms SFE from a solely lipophilic-targeting method into a versatile platform capable of efficiently extracting medium-polarity compounds, as demonstrated with phenolic compounds in hemp seed and Labisia pumila [7] [41]. While this requires optimization (e.g., using RSM), it offers a cleaner alternative to LSE for a much wider range of bioactive molecules.
The empirical data and experimental protocols presented confirm that the recovery of targeted compounds is highly dependent on the synergy between the extraction technique and the physicochemical properties of the solute. SFE using SC-CO₂ is the superior choice for lipophilic compounds, providing unmatched selectivity, purity, and environmental credentials. For polar compounds, conventional solvent extraction remains effective, but SFE modified with co-solvents presents a powerful, green alternative that avoids toxic solvent residues, provided process parameters are meticulously optimized.
Future research will continue to enhance the scope of SFE through improved co-solvent systems, hybridization with other green techniques (e.g., ultrasound-assisted SFE), and advanced process control. For researchers in drug development, the decision matrix is clear: prioritize SFE for lipophilics and invest in co-solvent optimization for polar targets where solvent-free extracts and superior purity are critical to the development pipeline.
The transition from laboratory research to full-scale manufacturing presents a critical challenge for scientists and engineers in the pharmaceutical and nutraceutical industries. The choice between Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction (CSE) methods significantly impacts final product quality, process sustainability, and economic feasibility. SFE, particularly using supercritical carbon dioxide (SC-CO₂), has emerged as a transformative technology delivering unparalleled purity and process efficiency compared to traditional solvent-based methods [42]. This comprehensive guide objectively compares the performance of these competing extraction technologies, providing experimental data and scale-up methodologies to inform strategic decision-making for research and development professionals engaged in natural product extraction.
Fundamental Principles
Conventional Solvent Extraction (CSE) encompasses traditional methods like maceration, percolation, and Soxhlet extraction that utilize organic solvents (e.g., ethanol, acetone, hexane) to dissolve and separate target compounds from plant matrices. These methods often involve high temperatures, extended processing times, and significant solvent volumes, which can degrade thermolabile compounds and create environmental concerns [10].
Supercritical Fluid Extraction (SFE) employs substances at temperatures and pressures above their critical point, where they exhibit properties between gases and liquids. Supercritical CO₂ (SC-CO₂), with a critical temperature of 31.1°C and critical pressure of 73.8 bar, is particularly advantageous for extracting thermosensitive compounds. Its tunable solvation power, achieved by modulating pressure and temperature, enables selective extraction while minimizing environmental impact [43].
Market Adoption and Growth Trajectory
The global supercritical fluid extraction chemicals market, valued at USD 2.9 billion in 2024, is projected to grow at a CAGR of 10.8% from 2025 to 2034, reaching USD 7.9 billion by 2034 [43]. This robust growth is fueled by several factors:
- Stringent regulatory requirements in the pharmaceutical industry demanding high-purity extracts with minimal solvent residues
- Consumer preference for clean-label products and natural ingredients
- Environmental regulations restricting organic solvent use and promoting green chemistry principles
- Technological advancements improving SFE efficiency, scalability, and cost-effectiveness
The pharmaceutical sector constitutes the largest end-use segment, accounting for 39.8% of the market share in 2024, followed by food & beverage and cosmetics & personal care industries [43].
Table 1: Global Market Overview for Supercritical Fluid Extraction Chemicals
| Market Aspect | 2024 Status | 2034 Projection | CAGR (2025-2034) |
|---|---|---|---|
| Market Value | USD 2.9 billion | USD 7.9 billion | 10.8% |
| Pharmaceutical Share | 39.8% | Increasing | - |
| SC-CO₂ Segment Value | USD 2 billion | Significant growth | - |
| North America Growth | Leader | Sustained growth | 10.9% |
| Europe Growth | Strong | Accelerated growth | 10.7% |
| Asia-Pacific Growth | Emerging | Rapid expansion | 10.9% |
Comprehensive Performance Comparison
Extraction Efficiency and Yield
Direct comparative studies reveal significant differences in extraction performance between SFE and CSE methods. In a study examining khella (Ammi visnaga L.) furanochromones, conventional solvent extraction with 95% ethanol produced the highest crude extract yield (15.44%), while SFE yielded substantially less crude extract (4.50%) [17]. However, when analyzing target bioactive compounds, SFE demonstrated superior performance, yielding 30.1% furanochromones compared to 8.23% for 95% ethanol and 6.59% for 30% ethanol [17]. This highlights a key distinction: CSE often extracts more total material but with lower specificity, while SFE provides more selective extraction of target compounds.
The enhanced selectivity of SFE was further demonstrated through HPLC analysis, which showed that conventional solvent extracts contained additional compounds not present in SFE extracts, resulting in darker colors and potential impurities [17]. This purification during extraction represents a significant advantage for pharmaceutical applications where compound specificity is crucial.
Table 2: Yield Comparison Between SFE and CSE for Khella (Ammi visnaga L.) Furanochromones
| Extraction Method | Solvent/Parameters | Total Extract Yield (%) | Furanochromones Yield (%) | Purity Assessment |
|---|---|---|---|---|
| SFE | SC-CO₂ + 5% methanol | 4.50 | 30.10 | High (clean chromatogram) |
| CSE | 95% Ethanol | 15.44 | 8.23 | Moderate (additional compounds) |
| CSE | 30% Ethanol | 15.44 | 6.59 | Moderate (additional compounds) |
| CSE | Acetone | 10.90 | 7.52 | Moderate (additional compounds) |
| CSE | Boiling Water | 13.79 | 5.95 | Low (additional compounds) |
Bioactivity Preservation
The extraction technique significantly influences the bioactivity of the final extract, with implications for pharmaceutical efficacy. In cytotoxicity evaluations against breast cancer (MCF-7) and hepatocellular carcinoma (Hep G2) cell lines, SFE extracts of Ammi visnaga and standard furanochromones (khellin and visnagin) demonstrated strong cytotoxic activity (IC₅₀ ranging between 12.54 ± 0.57 and 17.53 ± 1.03 µg/mL) [17]. In contrast, ethanol and acetone extracts showed only moderate cytotoxic activity (IC₅₀ 20-90 µg/mL), while aqueous extracts displayed weak activity (IC₅₀ > 90 µg/mL) [17]. This enhanced bioactivity of SFE extracts underscores the technology's ability to preserve compound integrity and pharmacological potency.
Similar trends were observed in studies on Matthiola ovatifolia Boiss., where extraction method significantly influenced phytochemical composition and associated biological activities, including antioxidant, antibacterial, and anti-inflammatory properties [14].
Environmental and Economic Considerations
From a sustainability perspective, SFE offers substantial advantages over conventional methods. Comparative analyses demonstrate that SFE can reduce solvent usage by 80-90% and lower energy consumption by 30-50% compared to traditional extraction methods [10]. The primary factor in energy reduction is SFE's ability to operate at moderate temperatures (often near ambient temperature), avoiding the prolonged heating required by many CSE methods.
Economic assessments of SFE processes for Phyllanthus niruri extraction have demonstrated encouraging values for return on investment (ROI) and net present value (NPV) across various scale-up capacities [44]. While SFE requires higher initial capital investment, its operational costs become competitive at scale due to reduced solvent consumption, shorter extraction times, and lower purification costs. A detailed profitability analysis revealed that industrial SFE could reduce costs by up to 89% compared to Soxhlet extraction when implemented at commercial scale [10].
Scale-up Methodologies and Experimental Protocols
Scale-up Criteria and Validation
Successful transition from laboratory to production scale requires systematic scale-up approaches. For SFE, the solvent-to-feed ratio (S/F) has been validated as a reliable scale-up criterion across multiple plant matrices. In a scale-up study of SFE for clove and sugarcane residue, maintaining constant S/F ratio enabled successful 15-fold scale-up from a 290 mL extraction vessel to a 5.15 L system [45]. The pilot-scale yields were slightly higher than laboratory-scale, demonstrating the robustness of this scaling parameter.
Similar success was reported for Phyllanthus niruri extraction, where S/F ratio scale-up validation experiments using 0.5 kg feed mass showed nearly identical extraction yields and overall extraction curves to predictions, with errors of just 5.13% and 14.2% for the two operating conditions tested [44]. This approach facilitates predictable technology transfer from benchtop to production.
Experimental Protocols
Supercritical Fluid Extraction Protocol
Objective: Extract bioactive compounds from plant materials using supercritical CO₂ with co-solvent modification.
Materials and Equipment:
- Supercritical fluid extraction system with co-solvent capability
- CO₂ supply (food/pharmaceutical grade)
- Co-solvents (ethanol, methanol, water - HPLC grade)
- Plant material (dried, ground to 0.3-0.5 mm particle size)
Methodology:
- Sample Preparation: Plant material is dried to 8-10% moisture content and ground to appropriate particle size (0.3-0.5 mm for lab-scale, 0.5-3.0 mm for pilot-scale to prevent channelling) [44].
- Extraction Vessel Loading: Pack extraction vessel with plant material, ensuring uniform density to avoid channeling.
- System Pressurization: Pressurize system to target pressure (200-400 bar) and temperature (40-80°C) based on target compounds.
- Static Extraction: Maintain conditions for 30-60 minutes to allow saturation.
- Dynamic Extraction: Initiate CO₂ flow (1.5-3.0 mL/min for lab-scale) with co-solvent (5-15% v/v) for 2-4 hours.
- Fraction Collection: Collect extracts in separators at reduced pressure.
- Solvent Removal: Remove residual co-solvent under reduced pressure at 40°C.
Critical Parameters:
- Pressure and temperature (control solvent density and selectivity)
- Co-solvent type and concentration (enhance polarity range)
- Flow rate and extraction time (impact yield and throughput)
- Particle size and packing density (influence mass transfer)
Conventional Solvent Extraction Protocol
Objective: Extract bioactive compounds using traditional solvent extraction methods.
Materials and Equipment:
- Solvent extraction system (reflux, Soxhlet, or maceration apparatus)
- Organic solvents (ethanol, acetone, hexane - analytical grade)
- Plant material (dried, ground to 0.5-1.0 mm particle size)
Methodology:
- Sample Preparation: Plant material is dried and ground to 0.5-1.0 mm particle size [46].
- Solvent Addition: Mix plant material with solvent at specified solid-to-liquid ratio (typically 1:10 to 1:30).
- Extraction: Conduct extraction with stirring (300 rpm) at specified temperature (25-70°C) for 1-6 hours.
- Filtration: Separate spent plant material from extract by filtration.
- Concentration: Remove solvent under reduced pressure at 40°C.
- Drying: Lyophilize or vacuum-dry to obtain solid extract.
Critical Parameters:
- Solvent type and concentration (determine extraction selectivity)
- Temperature and time (impact yield and compound stability)
- Solid-to-liquid ratio (influences mass transfer driving force)
- Number of extraction stages (affects completeness of extraction)
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful development and scale-up of extraction processes requires carefully selected materials and reagents. The following table outlines key solutions and their functions in SFE and CSE processes.
Table 3: Essential Research Reagents and Materials for Extraction Studies
| Category | Specific Items | Function/Application | Technical Considerations |
|---|---|---|---|
| Supercritical Fluids | Carbon dioxide (≥99.7%) | Primary extraction solvent | Critical point: 31.1°C, 73.8 bar; GRAS status |
| Co-solvents/Modifiers | Ethanol, Methanol, Water | Enhance polarity range for SFE | Typically 5-15% (v/v); ethanol preferred for pharmaceutical applications |
| Organic Solvents | Ethanol, Acetone, Hexane | Extraction solvents for CSE | Varying polarities for selective extraction; residue concerns |
| Plant Material Prep | GM 200 knife mill (Retsch) | Particle size reduction | Optimal size: 0.3-0.5mm (lab), 0.5-3.0mm (production) |
| Analytical Standards | Gallic acid, Quercetin, Catechin | Quantification reference compounds | HPLC/UV-Vis calibration; purity ≥95% |
| Antioxidant Assays | DPPH, FRAP, ABTS reagents | Bioactivity assessment | Free radical scavenging capacity measurement |
| Cell Culture | MCF-7, Hep-G2 cell lines | Cytotoxicity evaluation | Cancer models for bioactivity screening |
Technological Advancements and Future Outlook
Emerging Innovations
The SFE technology landscape is evolving rapidly, with several advancements enhancing its pharmaceutical and nutraceutical applications:
Hybrid Extraction Systems: Combining SFE with other technologies like ultrasound or microwave assistance improves extraction efficiency and reduces processing time [47]. Simultaneous ultrasound-assisted SFE enhances cell wall disruption, increasing compound accessibility.
Process Intensification: Continuous flow innovations have accelerated cycle times and reduced energy consumption, enabling higher throughput without sacrificing extract quality [42]. Industry 4.0 capabilities allow remote optimization of extraction parameters, reducing downtime and facilitating rapid scale-up.
AI-Enabled Optimization: Incorporating artificial intelligence and machine learning tools refines solvent tuning and predictive maintenance, further optimizing yield and reducing operational risk [42] [47]. These technologies enable real-time monitoring and advanced algorithms enhancing reproducibility.
Sequential Extraction Approaches: Implementing SFE as an initial defatting step followed by polarized solvent extraction maximizes recovery of diverse bioactive compounds from single biomass sources [10].
Regulatory and Economic Trajectory
Increasing regulatory pressure on organic solvent usage is driving SFE adoption across regulated industries. Government agencies and international bodies now favor methodologies that minimize hazardous waste, leading to incentives for companies adopting solvent-free processing [42]. This regulatory momentum has catalyzed capital deployment into research and pilot programs, expanding the technology's reach.
From an economic perspective, while SFE requires substantial initial investment (a significant barrier for smaller operators), total cost of ownership becomes favorable at commercial scale due to reduced solvent consumption, waste disposal costs, and purification requirements [10] [43]. Collaborative leasing and service-based models are emerging to improve accessibility for smaller operators [42].
The transition from laboratory to production scale requires careful consideration of technical, economic, and regulatory factors. SFE technology offers distinct advantages for pharmaceutical and nutraceutical applications where compound purity, bioactivity preservation, and residual solvent concerns are paramount. While CSE methods remain relevant for certain applications, particularly in research settings or for established processes, the industry trajectory clearly favors supercritical extraction for high-value bioactive compounds.
Successful scale-up necessitates systematic approaches, with the solvent-to-feed ratio (S/F) emerging as a reliable criterion for predictable technology transfer. As innovations in process intensification, hybrid systems, and AI-enabled optimization continue to advance, SFE is positioned to become increasingly accessible and economically viable across the product development lifecycle. Research and development professionals should consider these comparative insights when designing extraction strategies for new therapeutic compounds or nutraceutical ingredients.
Overcoming Challenges: Optimization Strategies for Maximum Yield and Purity
Supercritical Fluid Extraction (SFE), particularly using carbon dioxide (SC-CO₂), has emerged as a green alternative to conventional solvent extraction in pharmaceutical and nutraceutical research. The technique leverages the unique properties of supercritical fluids—gas-like diffusivity and liquid-like density—to achieve efficient extraction of bioactive compounds from natural sources [23]. Supercritical CO₂ operates under mild critical conditions (31.1°C, 73.8 bar), making it suitable for heat-sensitive compounds and eliminating toxic solvent residues—a significant advantage for drug development [3] [23]. The global SFE market, valued at USD 2.9 billion in 2024 and projected to reach USD 7.9 billion by 2034, reflects its growing adoption across research sectors [43].
However, two significant limitations impede broader implementation: substantial initial capital investment and challenges in extracting polar molecules due to the non-polar nature of CO₂ [43] [23]. This guide objectively examines these limitations against conventional methods, providing experimental data and protocols to inform research decision-making within the broader context of extraction efficiency and compound selectivity studies.
The High Initial Cost of SFE: Investment versus Operational Economics
Capital Investment and Operational Cost Analysis
The initial investment for SFE equipment substantially exceeds that for conventional extraction setups. Industrial-scale SFE systems represent a significant capital outlay, with costs influenced by pressure rating, automation features, and scale. The market for supercritical CO₂ extraction equipment is segmented by vessel capacity, with systems over 200L commanding the highest prices but offering economies of scale for industrial production [48]. This high entry cost presents a notable barrier for smaller research laboratories and startups [43].
However, a comprehensive cost analysis reveals a different long-term economic profile. The operational expenditure (OPEX) of SFE is frequently lower than conventional methods due to reduced solvent consumption and waste disposal costs. SFE utilizes carbon dioxide in a closed-loop system where approximately 90% of the CO₂ is recycled, dramatically reducing both raw material costs and environmental footprint [3] [43]. In contrast, traditional solvent extraction generates substantial hazardous waste, with hexane classified as a hazardous air pollutant requiring costly disposal and containment measures [49].
Table 1: Cost Structure Comparison: SFE vs. Conventional Solvent Extraction
| Cost Factor | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction |
|---|---|---|
| Initial Equipment Investment | High ($79M market 2025, projected growth 7.7% CAGR) [48] | Significantly lower for basic setups |
| Solvent Costs | Low (CO₂ is inexpensive and 90% recyclable) [43] | Continuous expense (hexane, ethanol, methanol) |
| Waste Disposal Costs | Minimal (closed-loop system) [3] | High (hazardous solvent disposal required) [49] |
| Energy Consumption | Variable (high-pressure pumping) | High (prolonged heating for solvent removal) [49] |
| Process Time | Faster extraction cycles [3] | Slower (limited by liquid mass transfer) [3] |
| Regulatory Compliance Costs | Lower (GRAS solvent) [43] | Higher (strict solvent residue limits) [43] |
Experimental Evidence: Economic Assessment Protocols
Research institutions evaluating SFE implementation should conduct lifecycle cost analyses using standardized protocols. The methodology below outlines a framework for economic assessment:
Protocol: Five-Year Cost-Benefit Analysis Model
- Equipment Costing: Document capital investment for comparable capacity systems, including installation and commissioning.
- Solvent Consumption Tracking: For SFE, measure initial CO₂ charge and monthly makeup amounts. For solvent extraction, record purchase volumes of hexane, ethanol, or other solvents.
- Waste Management Accounting: Quantify costs for solvent disposal according to hazardous waste regulations for conventional methods [49].
- Energy Monitoring: Install power meters to record electricity consumption for both extraction and post-processing (e.g., solvent evaporation).
- Labor Cost Assessment: Track operator time requirements for both processes, noting SFE's advantage of minimal post-processing for solvent removal [3].
- Output Quality Valuation: Apply premium valuation for SFE extracts with no solvent residues, particularly important for pharmaceutical applications [43].
Experimental studies consistently demonstrate that despite higher initial investment, SFE achieves lower operating costs over a 5-year period, particularly for high-value extracts where solvent-free status commands premium pricing [3]. One equipment manufacturer reports that SFE can reduce harmful solvent waste by nearly 90% compared to traditional methods, significantly impacting operational economics and sustainability metrics [43].
Extraction of Polar Compounds: Technical Challenges and Solutions
The Polarity Limitation and Selectivity Trade-Offs
The fundamental challenge in SFE lies in the non-polar nature of supercritical CO₂, which exhibits solubility characteristics similar to pentane or hexane [23]. This property makes it ideal for extracting non-polar compounds (lipids, essential oils, terpenes) but inefficient for more polar bioactive molecules such as polyphenols, flavonoids, and glycosides—compounds of significant pharmaceutical interest for their antioxidant and therapeutic properties [23].
This limitation, however, can be transformed into an advantage for selective extraction. Research on Ammi visnaga L. furanochromones demonstrates that SFE yielded a lower overall mass (4.50%) compared to conventional 30% ethanol extraction (15.44%), but produced a significantly more concentrated active fraction—30.1% furanochromones in SFE extract versus 6.59% in ethanol extract [17]. This enhanced selectivity reduces downstream purification requirements, particularly valuable in pharmaceutical isolation workflows.
Table 2: Extraction Efficiency Comparison for Bioactive Compounds
| Extraction Method | Plant Material | Target Compound | Yield | Purity/Concentration | Reference |
|---|---|---|---|---|---|
| SFE with 5% MeOH | Ammi visnaga fruits | Furanochromones | 4.50% (extract) | 30.1% of extract [17] | [17] |
| 30% Ethanol | Ammi visnaga fruits | Furanochromones | 15.44% (extract) | 6.59% of extract [17] | [17] |
| SFE (Optimized) | Rosmarinus officinalis L. | Polyphenols | 75-115 mg GAE/g | Superior antioxidant activity (IC₅₀: 0.14-11.7 μg/mL) [50] | [50] |
| Hydrodistillation | Rosmarinus officinalis L. | Polyphenols | Lower than SFE | Reduced antioxidant activity [50] | [50] |
| SFE-CO₂ | Various plant seeds | Phytosterols | Variable by species | Enhanced total phytosterol content [49] | [49] |
| Hexane Extraction | Various plant seeds | Phytosterols | Higher oil yield | Lower phytosterol concentration [49] | [49] |
Experimental Protocols for Polar Compound Extraction
Co-solvent Modification Methodology
The most effective approach to overcome polarity limitations involves adding polar co-solvents (typically 1-15% by volume) to SC-CO₂. Ethanol and methanol are most common, with ethanol preferred for food and pharmaceutical applications due to its GRAS (Generally Recognized As Safe) status [23].
Protocol: Co-solvent Assisted SFE for Polar Compounds
- Matrix Preparation: Grind plant material to 250-500μm particle size. For Rosmarinus officinalis L. studies, leaves were dried and ground to improve mass transfer [50].
- Extraction Vessel Loading: Pack biomass evenly into extraction vessel to prevent channeling.
- Co-solvent Addition: Introduce food-grade ethanol (5-10% of total solvent volume) via co-solvent pump directly into CO₂ stream.
- Parameter Optimization: For rosemary antioxidants, optimal conditions were 35-80°C temperature and 10-50 MPa pressure [51]. Specific optimization required for different plant matrices.
- Fraction Collection: Separate extracts at different pressure/temperature conditions to fractionate compounds by polarity.
- Analysis: Analyze extracts for target polar compounds using HPLC and assess antioxidant activity via DPPH or ORAC assays [50].
Experimental results demonstrate the efficacy of this approach. SFE with ethanol as co-solvent yielded rosemary extracts enriched with polyphenols (75-115 mg GAE/g) and flavonoids (16-19 mg QE/g), demonstrating potent radical scavenging activity (IC₅₀: 0.14-11.7 μg/mL) that surpassed conventional extraction results [50].
Hybrid Extraction Workflows
For complex matrices containing both polar and non-polar bioactive compounds, researchers can implement sequential or hybrid extraction workflows:
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for SFE Research
| Research Reagent/Material | Function/Application | Research Considerations |
|---|---|---|
| Supercritical CO₂ (≥99.9%) | Primary extraction fluid | GRAS status; non-flammable; recyclable in closed-loop systems [23] |
| Food-Grade Ethanol | Polar co-solvent for phenolic compounds | Preferred for pharmaceutical/food applications; typically 5-15% by volume [50] [23] |
| Pharmaceutical-Grade Methanol | Co-solvent for analytical applications | Higher polarity modifier; restricted in pharmaceutical products [17] |
| In-Line Adsorbents (Silica, Alumina) | Selective fractionation during extraction | Placed post-extraction vessel to trap specific compound classes [3] |
| High-Pressure Vessels (Stainless Steel) | Contain biomass during extraction | Safety-critical; rated for ≥50 MPa; corrosion-resistant [3] |
| Back-Pressure Regulators | Maintain system pressure; control separation | Precision components for fractionation at different pressures [3] |
Addressing the dual limitations of high initial cost and polar compound extraction requires a nuanced approach to SFE implementation in research settings. The economic challenge is primarily front-loaded, with lifecycle cost analysis generally favoring SFE over 3-5 year horizons, especially for high-value extracts where solvent-free status provides regulatory and marketing advantages [3] [43].
The technical limitation of polar compound extraction has been effectively addressed through co-solvent modification and hybrid approaches, transforming SFE's selectivity into a advantage for obtaining purified fractions with enhanced bioactivity, as demonstrated in the cytotoxicity research on Ammi visnaga extracts [17]. Future directions include continued optimization of co-solvent systems, AI-enabled process optimization to reduce energy consumption, and development of more cost-effective equipment designs to lower barriers to adoption [43] [6].
For research institutions and drug development professionals, strategic implementation may begin with pilot-scale systems for method development and process optimization, with subsequent scale-up based on specific application requirements. The compelling advantages of solvent-free extracts for pharmaceutical applications, combined with advancing SFE technology, position this green extraction method as an increasingly viable and essential tool in natural product research.
Conventional Solvent Extraction (CSE) methods, such as maceration, Soxhlet, and reflux extraction, have long been standard for isolating bioactive compounds from plant materials. However, these techniques present significant drawbacks that impact their efficiency, safety, and environmental footprint. The core limitations include the use of large volumes of toxic organic solvents (e.g., n-hexane, petroleum ether), extended extraction times ranging from hours to days, and the application of high temperatures that degrade thermolabile phytochemicals [9] [26]. These disadvantages are particularly problematic for pharmaceutical and nutraceutical applications where solvent residues pose safety concerns, and the degradation of active compounds diminishes therapeutic value [4] [26].
In response to these challenges, Supercritical Fluid Extraction (SFE) has emerged as a sustainable and efficient alternative. By utilizing supercritical carbon dioxide (SC-CO₂) as a green solvent, SFE addresses the fundamental limitations of CSE while offering enhanced selectivity and preservation of bioactive compounds [4] [20]. This guide provides an objective comparison of these technologies, supported by experimental data and protocols, to inform researchers and drug development professionals in selecting optimal extraction methodologies.
Supercritical Fluid Extraction: Mechanism and Advantages
Fundamental Principles of SFE
Supercritical Fluid Extraction employs solvents at temperatures and pressures above their critical points, where they exhibit unique properties intermediate between gases and liquids. Carbon dioxide (CO₂) is the most widely used supercritical fluid due to its accessible critical point (31.1°C, 73.8 bar), non-toxicity, non-flammability, and low cost [4] [20]. In its supercritical state, CO₂ possesses gas-like diffusivity and viscosity, enabling deep penetration into plant matrices, coupled with liquid-like density, providing superior solvating power [52].
The SFE process operates in a closed system where CO₂ is pressurized and heated to supercritical conditions, passed through a vessel containing the plant material, and then decompressed to separate the extract. The dissolved compounds precipitate out, and the CO₂ can be recycled back into the system [20] [53]. This cyclic process eliminates solvent residue concerns and minimizes waste generation.
Key Advantages Over Conventional Methods
Elimination of Toxic Solvents: SC-CO₂ is GRAS (Generally Recognized as Safe) for food and pharmaceutical applications, replacing hazardous solvents like n-hexane and chloroform [4] [9]. Post-extraction, CO₂ reverts to gas, leaving no toxic residues [7].
Prevention of Thermal Degradation: Low operational temperatures (typically 31-60°C) preserve heat-sensitive compounds such as flavonoids, antioxidants, and volatile aromatics that degrade in conventional extraction [4] [26].
Reduced Extraction Time: Enhanced diffusivity and mass transfer rates significantly shorten extraction times compared to maceration or Soxhlet methods [4] [53].
Tunable Selectivity: By modulating pressure and temperature, operators can adjust solvent density and polarity to selectively target specific compound classes [52] [7]. The addition of polar co-solvents like ethanol further enhances extraction efficiency for phenolic compounds [7].
Comparative Experimental Data: SFE vs. CSE
Extraction Yield and Efficiency
Table 1: Comparative Extraction Yields of SFE and CSE Methods
| Plant Material | Target Compound | SFE Yield | CSE Yield | Experimental Conditions | Citation |
|---|---|---|---|---|---|
| Nepeta crispa | Essential oil | 1.812% | Lower than SFE (HD method) | SFE: 25 MPa, 40°C, 3.5% co-solvent | [4] |
| Schinus molle Leaves | Bioactive compounds | 1.23-2.31% (varies with parameters) | N/A | SFE: 150-350 bar, 35-55°C | [29] |
| Hemp Seed | Oil and phenolics | 30.13% (with ethanol co-solvent) | 60-80% (screw pressing) | SFE: 20 MPa, 50°C, 10% ethanol | [7] |
Bioactive Compound Recovery and Quality
Table 2: Bioactivity Comparison of SFE vs. CSE Extracts
| Plant Material | Extraction Method | Bioactive Content | Bioactivity Results | Citation |
|---|---|---|---|---|
| Nepeta crispa | SFE (SC-CO₂) | 1,8-cineol and nepetalactone | Superior antioxidant and antibacterial activity vs. hydrodistillation | [4] |
| Nepeta crispa | Hydrodistillation (CSE) | Same major compounds | Lower bioactivity compared to SFE | [4] |
| Hemp Seed | SFE (SC-CO₂ + 10% ethanol) | Total phenolics: 294.15 GAE mg/kg; Total tocopherols: 484.38 mg/kg | Significantly enhanced phenolic and tocopherol content vs. conventional | [7] |
| Various Plants | Modern Green Methods | Higher retention of flavonoids and polyphenols | Improved antioxidant and anti-inflammatory activities vs. conventional | [26] |
Experimental Protocols for SFE Optimization
Standard SFE Protocol for Plant Materials
The following protocol, adapted from multiple studies, outlines the general procedure for supercritical fluid extraction of bioactive compounds from plant matrices [4] [29] [7]:
Sample Preparation: Plant materials should be air-dried at ambient temperature, protected from direct sunlight. Grind the dried material using a mechanical grinder and sieve to achieve uniform particle size (typically 0.2-0.5 mm). Maintain moisture content below 10% to prevent ice formation during extraction.
Extraction Setup: Load the prepared plant material into the extraction vessel. For laboratory-scale systems, typical loadings range from 5-50 g. Ensure even packing to prevent channeling, which reduces extraction efficiency.
Parameter Optimization: Based on the target compounds, set the operational parameters:
- Pressure: 150-350 bar (15-35 MPa) for most applications
- Temperature: 35-60°C, balancing solubility and compound stability
- CO₂ Flow Rate: 0.25-3.28 g/min, optimized for the specific matrix
- Co-solvent Addition: For polar compounds, add 2.5-10% ethanol as a modifier
Extraction Cycle: Conduct extraction in dynamic mode, with the supercritical CO₂ continuously flowing through the plant matrix. Typical extraction times range from 120-300 minutes, depending on the material and scale.
Separation and Collection: Depressurize the CO₂-extract mixture through a separation vessel maintained at lower pressure (50-60 bar) and temperature (15-25°C) to precipitate the extract. Collect the extract in a suitable container and store under inert atmosphere if necessary.
Response Surface Methodology for SFE Optimization
Recent studies employ Response Surface Methodology (RSM) with Box-Behnken or Central Composite Designs to optimize SFE parameters [4] [7]. This statistical approach systematically evaluates the effects of multiple parameters and their interactions on extraction yield and quality:
Experimental Design: Select three key independent variables (pressure, temperature, and co-solvent percentage) at three coded levels (-1, 0, +1).
Model Fitting: Conduct experiments according to the design matrix and fit the data to a second-order polynomial model to understand linear, quadratic, and interactive effects.
Validation: Verify the model adequacy using ANOVA with p-values and R² statistics, then validate predicted optimum conditions through confirmatory experiments.
This methodology was successfully applied to Nepeta crispa extraction, identifying optimal conditions of 25 MPa, 40°C, and 3.5% co-solvent with a maximum yield of 1.812% [4].
Visualization of SFE Process and Advantages
SFE System Workflow
Comparative Advantages: SFE vs. CSE
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Reagents and Equipment for SFE Research
| Item | Function/Application | Specifications/Alternatives |
|---|---|---|
| Supercritical CO₂ | Primary extraction solvent | Food-grade, 99.95% purity, critical point: 31.1°C, 73.8 bar |
| Ethanol (Co-solvent) | Modifier for polar compounds | HPLC grade, 95-99% purity, GRAS status |
| Plant Materials | Extraction matrix | Air-dried, ground (0.2-0.5 mm), low moisture content (<10%) |
| SFE System | Extraction equipment | Includes CO₂ tank, pump, extraction vessel, heating unit, separator |
| Analytical Standards | Compound identification | Reference compounds for GC-MS/HPLC quantification |
| Antioxidant Assay Kits | Bioactivity assessment | FRAP, DPPH, ORAC assays for antioxidant capacity |
| Antimicrobial Test Materials | Bioactivity assessment | Bacterial strains, culture media, dilution buffers |
The comparative analysis presented in this guide demonstrates that Supercritical Fluid Extraction effectively addresses the primary limitations of Conventional Solvent Extraction. Through the replacement of toxic solvents with supercritical CO₂, reduction of extraction time, and operation at mild temperatures that preserve thermolabile compounds, SFE represents a superior approach for pharmaceutical and nutraceutical applications.
Experimental evidence confirms that SFE not only matches but often exceeds the extraction efficiency of conventional methods while producing extracts with enhanced bioactivity profiles. The tunability of SFE parameters enables precise targeting of specific compound classes, offering researchers unparalleled control over extract composition.
For drug development professionals seeking to maximize therapeutic potential while adhering to green chemistry principles, SFE presents a viable, efficient, and sustainable alternative to conventional extraction methodologies. Future advancements in SFE modeling, scale-up protocols, and hybrid approaches will further solidify its position as the extraction method of choice for high-value bioactive compounds.
The pursuit of efficient, sustainable, and high-yield extraction methods represents a critical focus in modern phytochemistry and pharmaceutical development. For decades, conventional extraction techniques such as Soxhlet extraction, maceration, and hydrodistillation have dominated industrial and laboratory practices despite significant limitations including prolonged processing times, excessive solvent consumption, and thermal degradation of bioactive compounds [26] [9]. The emergence of green extraction technologies has revolutionized this landscape, with supercritical fluid extraction (SFE) establishing itself as a particularly promising method. SFE, typically using supercritical CO₂ (SC-CO₂), offers exceptional selectivity, minimal solvent residue, and preservation of thermolabile compounds [50] [54] [20]. However, no single extraction technology is universally superior, as each presents distinct advantages for specific compound classes and matrix types [55] [9].
Recent research has increasingly focused on hybrid extraction strategies that combine the strengths of multiple technologies to achieve synergistic effects. The integration of SFE with microwave-assisted extraction (MAE) and ultrasound-assisted extraction (UAE) represents a particularly promising frontier. These hybrid approaches leverage the unique mechanisms of each technology: SFE provides superior selectivity and green solvent properties, UAE enhances cell wall disruption through acoustic cavitation, and MAE enables rapid intracellular heating via dielectric mechanisms [56] [57] [58]. This comprehensive analysis compares the performance of individual and integrated extraction technologies, providing experimental data and methodologies to guide researchers in optimizing extraction protocols for pharmaceutical and nutraceutical applications.
Fundamental Principles of Individual Extraction Technologies
Supercritical Fluid Extraction (SFE)
SFE utilizes solvents maintained at temperatures and pressures above their critical points, resulting in unique physico-chemical properties including gas-like diffusivity and viscosity combined with liquid-like density [54] [20]. Carbon dioxide (SC-CO₂) is the most widely used supercritical fluid due to its low critical parameters (31.1°C, 73.8 bar), non-toxicity, and environmental acceptability [50] [54]. The extraction efficiency of SFE is influenced by multiple parameters including pressure, temperature, cosolvent addition, and flow rates, which collectively determine solvent density and solvating power [50] [55]. The tunable solvation power of supercritical fluids enables selective extraction of target compounds by precisely controlling pressure and temperature conditions [52] [20]. SFE has demonstrated exceptional performance in extracting lipophilic compounds, antioxidants, and essential oils while preserving their bioactivity and structural integrity [50] [54].
Microwave-Assisted Extraction (MAE)
MAE employs electromagnetic radiation to generate heat within plant matrices through two primary mechanisms: ionic conduction and dipole rotation [56] [57]. This internal heating significantly enhances mass transfer rates by rapidly elevating intracellular pressure, resulting in cell wall rupture and release of bioactive compounds. MAE offers substantial advantages over conventional methods, including reduced extraction times, lower solvent consumption, and improved extraction yields [56] [58]. A comparative study on stevia leaves demonstrated that MAE outperformed UAE, yielding 8.07%, 11.34%, and 5.82% higher total phenolic content (TPC), total flavonoid content (TFC), and antioxidant activity (AA), respectively, with 58.33% less extraction time [56]. The efficiency of MAE is governed by parameters such as microwave power, irradiation time, temperature, and solvent characteristics, which collectively influence the dielectric heating and mass transfer processes [56] [57].
Ultrasound-Assisted Extraction (UAE)
UAE utilizes high-frequency sound waves (typically 20-100 kHz) to generate acoustic cavitation in solvent systems [56] [57]. The formation, growth, and implosive collapse of microbubbles near cell walls create extreme local temperatures and pressures, resulting in cell disruption and enhanced solvent penetration. This mechanism significantly improves mass transfer while operating at relatively low temperatures, making UAE particularly suitable for heat-sensitive compounds [26] [57]. UAE has demonstrated superior efficiency compared to conventional methods, with citrus peel studies showing enhanced extraction yields and reduced processing times [57] [58]. The effectiveness of UAE depends on multiple factors including ultrasound frequency, power density, extraction temperature, and duration, which collectively influence cavitation intensity and distribution [56] [58].
Table 1: Fundamental Mechanisms and Advantages of Advanced Extraction Technologies
| Technique | Fundamental Mechanism | Key Operational Parameters | Primary Advantages |
|---|---|---|---|
| SFE | Tunable solvation power of supercritical fluids | Pressure, temperature, CO₂ flow rate, cosolvents | Selective extraction, minimal solvent residue, preserves thermolabile compounds |
| MAE | Dielectric heating through microwave radiation | Microwave power, irradiation time, temperature, solvent dielectric constant | Rapid extraction, reduced solvent consumption, enhanced yield for polar compounds |
| UAE | Acoustic cavitation disrupting cell walls | Ultrasound frequency, power density, extraction time, temperature | Low-temperature operation, efficient cell disruption, suitable for heat-sensitive compounds |
Quantitative Performance Comparison of Extraction Techniques
Yield and Efficiency Metrics
Direct comparative studies provide valuable insights into the performance characteristics of individual extraction technologies. In rosemary antioxidant extraction, SFE demonstrated superior performance compared to conventional methods, yielding polyphenols (75-115 mg GAE/g) and flavonoids (16-19 mg QE/g) with exceptional radical scavenging activity (IC₅₀: 0.14-11.7 μg/mL) [50]. The selectivity of SFE was further evidenced in carnostic acid recovery, with Density Functional Theory (DFT) and ADMET analyses confirming its pharmaceutical potential [50]. However, technology performance is highly matrix-dependent, as evidenced by a pinewood sawdust study where accelerated solvent extraction (ASE) achieved 4.2% lipophilic compound yield compared to 2.5% for SFE under optimal conditions for each method [55].
The time efficiency of advanced technologies represents a significant advantage over conventional methods. In orange essential oil extraction, MAE and UAE achieved yields of 11.5% in just 30 minutes, compared to 10.5% yield obtained through hydrodistillation requiring 180 minutes [57]. This 83% reduction in processing time substantially decreases energy consumption while potentially reducing thermal degradation. Similarly, SC-CO₂ extraction of lipids and carotenoids from Rhodotorula toruloides yeast demonstrated not only higher yields (332.09 ± 27.32 μg/g dry weight versus 19.9 ± 2.74 μg/g for conventional acetone extraction) but also superior preservation of compound integrity, particularly for unsaturated lipids and oxygen-sensitive carotenoids like torularhodin [54].
Bioactive Compound Preservation
The preservation of bioactivity and structural integrity of extracted compounds is a critical metric for pharmaceutical applications. Modern extraction technologies significantly outperform conventional methods in maintaining compound stability and functionality. SFE has demonstrated exceptional performance in preserving carnostic acid and other antioxidant compounds from rosemary, with confirmed bioactivity through computational and pharmacokinetic studies [50]. The gentle processing conditions of SFE (moderate temperatures and oxygen-free environment) prevent degradation of sensitive molecules, making it particularly valuable for pharmaceutical applications where compound integrity directly influences therapeutic efficacy [50] [54].
MAE and UAE also demonstrate superior bioactive preservation compared to conventional methods, though through different mechanisms. MAE's rapid heating minimizes thermal exposure, while UAE's low-temperature operation maintains stability of thermolabile compounds. In stevia extraction studies, MAE-produced extracts showed significantly higher antioxidant activity compared to UAE, indicating better preservation of redox-active compounds [56]. The combination of these technologies with SFE in hybrid approaches creates synergistic effects that further enhance bioactive compound preservation while improving extraction efficiency.
Table 2: Quantitative Performance Comparison of Extraction Technologies for Various Matrices
| Source Material | Extraction Technique | Optimal Conditions | Key Performance Metrics | Reference |
|---|---|---|---|---|
| Rosemary | SFE | Varied temperature, pressure, and cosolvent | Polyphenols: 75-115 mg GAE/g; Flavonoids: 16-19 mg QE/g; IC₅₀: 0.14-11.7 μg/mL | [50] |
| Pinewood Sawdust | ASE | 160°C, 12.5 min, 1 static cycle | Lipophilic compound yield: 4.2% | [55] |
| Pinewood Sawdust | SFE | 50°C, 300 bar, CO₂ 3.2 ml/min, cosolvent 2 ml/min | Lipophilic compound yield: 2.5% | [55] |
| Orange Peel | MAE/UAE | 30 min extraction time | Essential oil yield: 11.5% | [57] |
| Orange Peel | Hydrodistillation | 180 min extraction time | Essential oil yield: 10.5% | [57] |
| R. toruloides Yeast | SC-CO₂ | Supercritical conditions | Total carotenoids: 332.09 ± 27.32 μg/g DW | [54] |
| R. toruloides Yeast | Conventional acetone | Solvent extraction | Total carotenoids: 19.9 ± 2.74 μg/g DW | [54] |
| Stevia Leaves | MAE | 5.15 min, 284.05 W, 53.10% ethanol, 53.89°C | Higher TPC, TFC, and AA than UAE with 58.33% less time | [56] |
Hybridization Strategies: Experimental Protocols and Workflows
Sequential Integration Methodologies
The strategic combination of extraction technologies creates synergistic effects that enhance overall efficiency and yield. One effective approach involves sequential integration of non-thermal pre-treatments (UAE or MAE) followed by SFE. This methodology leverages the cell-disrupting capabilities of UAE or MAE to increase solvent accessibility, subsequently enhancing the efficiency of SFE [57] [58]. In citrus lemon peel processing, a hybrid MAE-UAE approach demonstrated significant improvements, with optimized conditions (microwave power of 516.74 W for 101.86 seconds followed by UAE at 40°C for 21.033 minutes) yielding 2283.72 mg GAE/100 g TPC and 987.58 mg QE/100 g TFC [58]. This integrated approach reduced energy consumption by 23.42% compared to UAE alone while simultaneously lowering CO₂ emissions [58].
The experimental workflow for hybrid extraction typically begins with sample preparation (drying, grinding, and sieving) to achieve optimal particle size, followed by a pre-treatment step using either MAE or UAE to disrupt cellular structures. The pre-treated material then undergoes SFE under carefully optimized parameters (pressure, temperature, cosolvent addition, and flow rate) [57] [58]. Response Surface Methodology (RSM) and Artificial Neural Networks coupled with Genetic Algorithms (ANN-GA) have proven valuable for modeling complex parameter interactions and identifying optimal conditions in these multi-step processes [56] [58].
Simultaneous Hybridization Approaches
While sequential integration represents the most common hybridization strategy, recent technological advances have enabled simultaneous combination of extraction mechanisms. Although technically challenging, these approaches offer potentially greater synergistic effects by applying multiple energy forms concurrently. The development of specialized extraction vessels capable of withstanding both supercritical conditions while permitting ultrasound transmission or microwave irradiation is essential for such applications [57]. Though current literature primarily focuses on sequential approaches, ongoing research in reactor design suggests simultaneous hybridization represents a promising future direction for extraction technology enhancement.
Comparative Analysis of Hybrid Versus Conventional Approaches
Efficiency and Yield Enhancements
Hybrid extraction techniques demonstrate measurable improvements across multiple performance metrics compared to individual technologies. The MAE-UAE hybrid approach for citrus lemon peel not only enhanced bioactive compound yields but also reduced total processing time and solvent consumption [58]. Similarly, the integration of UAE as a pre-treatment for SFE in orange peel processing created micro-fractures in the plant matrix, significantly improving SC-CO₂ penetration and essential oil recovery [57]. These synergistic effects are particularly pronounced for matrices with robust cellular structures or when targeting intracellular compounds that are difficult to access through single-mechanism extraction.
The efficiency gains from hybridization extend beyond simple yield improvements to encompass compound selectivity and purity. In SFE of rosemary, the addition of cosolvents like ethanol enhanced polyphenol recovery while maintaining the superior selectivity of the supercritical process [50]. When combined with mechanical pre-treatments like UAE, this approach enables targeted recovery of specific compound classes with pharmaceutical value, as demonstrated by the enhanced carnostic acid extraction with confirmed antioxidant activity through DFT studies [50].
Sustainability and Economic Considerations
From an environmental perspective, hybrid techniques offer significant advantages through reduced energy consumption, decreased solvent utilization, and shorter processing times. The MAE-UAE hybrid for citrus lemon peel achieved 23.42% reduction in energy consumption compared to standalone UAE [58]. Similarly, SFE-based hybrid processes eliminate or substantially reduce the need for organic solvents, minimizing environmental contamination and reducing downstream purification requirements [50] [54] [20]. These environmental benefits align with the principles of green chemistry and sustainable processing, increasingly important considerations in pharmaceutical development.
The economic viability of hybrid techniques depends on balancing enhanced yields against increased capital investment and operational complexity. While SFE equipment requires substantial initial investment, its combination with lower-cost technologies like UAE can improve overall cost-effectiveness. Similarly, the reduction in processing time achieved through MAE integration decreases operational costs while increasing throughput [56] [57] [58]. Life cycle assessment studies indicate that the environmental benefits of hybrid approaches, including reduced carbon emissions and solvent waste, further enhance their economic appeal through reduced waste management requirements and alignment with sustainable manufacturing priorities.
Table 3: Sustainability Metrics of Advanced Extraction Technologies
| Extraction Technique | Energy Consumption | Solvent Usage | Processing Time | Environmental Impact |
|---|---|---|---|---|
| Conventional Solvent Extraction | High | High | Long (hours) | High solvent waste, potential contamination |
| SFE | Moderate | Low (solvent-free) | Moderate | Low, CO₂ is recyclable and non-toxic |
| MAE | Low | Moderate | Short (minutes) | Moderate, reduced energy footprint |
| UAE | Low | Moderate | Short (minutes) | Moderate, reduced energy footprint |
| Hybrid MAE-UAE | 23.42% lower than UAE alone | Moderate | Very short | Lower CO₂ emissions, reduced energy use |
| SFE with Pre-treatment | Moderate | Low | Moderate | Very low, enhanced efficiency with green solvent |
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of hybrid extraction methodologies requires specific reagents, solvents, and analytical tools. The selection of appropriate materials directly influences extraction efficiency, compound stability, and analytical accuracy.
Table 4: Essential Research Reagent Solutions for Hybrid Extraction Studies
| Reagent/Equipment | Function in Hybrid Extraction | Application Examples | Considerations |
|---|---|---|---|
| Supercritical CO₂ | Primary solvent for SFE | Extraction of lipophilic compounds, antioxidants, essential oils | High purity (99.9%), with cosolvent modifiers for polarity adjustment |
| Food-Grade Ethanol | Cosolvent for SFE, solvent for MAE/UAE | Polyphenol and flavonoid extraction, polarity modification in SFE | GRAS status, optimal concentration 50-70% for bioactive compounds |
| Analytical Standards | Quantification and identification | Gallic acid (TPC), quercetin (TFC), carnostic acid | Certified reference materials for calibration curves |
| Antioxidant Assay Kits | Bioactivity assessment | DPPH, FRAP, ORAC assays | Standardized protocols for comparative bioactivity analysis |
| UHPLC-HRMS | Compound separation and identification | Carotenoid profiling, phenolic compound characterization | High-resolution mass detection for structural elucidation |
| Response Surface Methodology Software | Experimental design and optimization | Design-Expert, MATLAB for RSM and ANN-GA modeling | Multi-parameter optimization for hybrid process conditions |
Hybrid extraction techniques integrating SFE with MAE and UAE represent a significant advancement in bioactive compound recovery, offering enhanced efficiency, improved yields, and superior sustainability profiles compared to individual technologies. The synergistic combination of these methods leverages their complementary mechanisms: MAE and UAE provide effective cell disruption, while SFE enables selective and gentle extraction of target compounds [57] [58]. The experimental data and protocols presented in this analysis provide researchers with evidence-based guidance for implementing these advanced extraction strategies.
Future developments in hybrid extraction technology will likely focus on several key areas: (1) advanced reactor designs enabling simultaneous application of multiple extraction mechanisms; (2) integration of real-time monitoring and process control using PAT (Process Analytical Technology) principles; (3) expanded application to novel biomass sources including microalgae, agricultural byproducts, and marine resources [54] [20]; and (4) enhanced sustainability through solvent recycling and energy optimization. As the pharmaceutical and nutraceutical industries continue to prioritize both efficiency and environmental responsibility, hybrid extraction methodologies offer a promising pathway to meet these dual objectives while maintaining the highest standards of product quality and bioactivity.
In the pursuit of efficient and sustainable industrial processes, advanced process control techniques are paramount. Within extraction technologies, particularly supercritical fluid extraction (SFE), optimizing process parameters—such as pressure, temperature, and solvent composition—is crucial for maximizing yield, purity, and selectivity while minimizing environmental impact and operational costs [59]. This guide objectively compares two powerful optimization methodologies: Response Surface Methodology (RSM), a well-established statistical approach, and Artificial Intelligence (AI) techniques, primarily Artificial Neural Networks (ANN), often coupled with optimization algorithms like Genetic Algorithms (GA). The comparative performance of these methods is evaluated within the context of SFE, a green alternative to conventional solvent extraction, providing researchers and scientists with data-driven insights for method selection.
Performance Comparison: RSM vs. AI-based Models
Direct comparisons from recent studies reveal nuanced performance differences between RSM and AI-based models like ANN and ANN-GA. The following table summarizes quantitative findings from various optimization studies in the field of bioactive compound extraction.
Table 1: Comparative Performance of RSM and AI Models in Extraction Optimization
| Source Material | Optimization Method | Key Performance Metrics | Reference |
|---|---|---|---|
| Terminalia chebula Pulp | RSMANN | R²: 0.9987R²: 0.9973 | [60] |
| Stevia rebaudiana Leaves (MAE) | RSMANN-GA | R²: 0.8893 - 0.9533R²: 0.9985, MSE: 0.7029 | [56] |
| Stevia rebaudiana Leaves (UAE) | RSMANN-GA | R²: 0.9177 - 0.9326R²: 0.9981, MSE: 0.8362 | [56] |
| Phylloporia ribis Mushroom | RSMANN-GA | Lower Antioxidant ActivityHigher Antioxidant Activity & Phenolic Content | [61] |
| Pouteria lucuma Seed Oil | RSMANN-GA | R²: 0.9891R²: 0.9999 | [59] |
| Diclofenac Potassium Removal | RSMANN | Strong CorrelationBest Predictive Accuracy | [62] |
Key Performance Insights
- Predictive Accuracy: ANN-based models, particularly when hybridized with genetic algorithms (ANN-GA), consistently demonstrate superior predictive accuracy over RSM, as indicated by higher R² values and lower mean squared error (MSE) in studies on stevia, lucuma seed oil, and pharmaceutical wastewater treatment [59] [62] [56]. This is attributed to ANN's superior capability in modeling complex, non-linear relationships between process parameters and responses [56].
- Experimental Outcome Optimization: In applications where the goal is not just to model but to find the absolute best outcome, ANN-GA often finds a more optimal solution. For instance, Phylloporia ribis extracts optimized via ANN-GA exhibited greater antioxidant activity and higher concentrations of key phenolic compounds compared to RSM-optimized samples [61].
- Model Robustness and Simplicity: While ANN-GA can show superior performance, RSM models are also highly effective and can sometimes be sufficient, offering the advantage of being less computationally complex and providing explicit polynomial equations that describe the interaction of factors [60]. The choice between methods may depend on the specific process complexity and the required precision [60].
Experimental Protocols & Methodologies
Core Protocol for Supercritical Fluid Extraction (SFE)
The following generalized protocol, synthesized from multiple studies, outlines the standard procedure for SFE used in generating data for RSM and ANN modeling [59] [41] [63].
- Sample Preparation: Plant material (e.g., leaves, seeds) is washed, dried, and ground into a fine powder. A uniform particle size is achieved by sieving (e.g., 0.5-0.8 mm) [59] [41]. The moisture content is typically reduced to a constant level (e.g., 5-6% w/w) [41].
- SFE System Setup: The SFE system typically includes a CO₂ pump, a co-solvent pump (if used), an extraction vessel housed in an oven, a back-pressure regulator, and a sample collection vessel [41].
- Extraction Run: A predetermined mass of sample (e.g., 5-1000 g, depending on vessel size) is loaded into the extraction vessel [41] [63]. The system is pressurized and heated to the desired conditions. Extraction often occurs in two modes:
- Extract Collection and Analysis: The extract is collected, and the yield is determined gravimetrically. The extract is then analyzed using techniques like HPLC (for specific compounds like CBD or phenolics), GC-MS (for oils and volatiles), or spectrophotometric assays (for total phenolic content and antioxidant activity) [61] [59] [63].
Optimization Workflow: RSM vs. ANN-GA
The fundamental difference between the two approaches lies in how the experimental data is modeled and used for optimization. The workflow for both is illustrated below.
Response Surface Methodology (RSM) Protocol
- Experimental Design: A structured design (e.g., Central Composite Design (CCD) or Box-Behnken Design (BBD)) is employed to define the experimental runs. This design efficiently explores the influence of multiple input variables (e.g., pressure, temperature, time) at different levels on the response(s) (e.g., yield, purity) [59] [63] [64].
- Model Development and Analysis: A second-order polynomial regression model is fitted to the experimental data. The model's significance and adequacy are evaluated using Analysis of Variance (ANOVA) and the coefficient of determination (R²) [61] [60].
- Optimization: The fitted model is used to generate response surfaces. The optimal conditions are identified either through numerical techniques or a desirability function that simultaneously optimizes multiple responses [60].
Artificial Neural Network with Genetic Algorithm (ANN-GA) Protocol
- Network Architecture and Training: An ANN model (e.g., Multilayer Perceptron (MLP)) is designed with input neurons (process parameters), hidden layers, and output neurons (responses). The dataset is divided into training, validation, and testing subsets. The network is trained using algorithms like Levenberg-Marquardt to learn the complex, non-linear relationships in the data [65] [61] [56].
- Model Validation: The trained ANN model's predictive accuracy is validated using statistical metrics like Mean Squared Error (MSE) and Mean Absolute Percentage Error (MAPE) on the test dataset [61] [56].
- Optimization with GA: The validated ANN model serves as the fitness function for a Genetic Algorithm. The GA, mimicking natural selection, evolves a population of potential solutions (parameter sets) to find the combination that maximizes or minimizes the predicted response, effectively performing a global search for the optimum [61] [56].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for SFE Optimization Studies
| Item | Function / Application | Specific Examples from Research |
|---|---|---|
| Carbon Dioxide (CO₂) | Primary supercritical solvent; non-toxic, tunable solvation power. | Commercial-grade liquefied CO₂ (99.9%) [41] [63]. |
| Co-solvents (Modifiers) | Enhance polarity of SC-CO₂, improving extraction of polar compounds. | Ethanol, methanol, ethyl acetate [41] [64]. Ethanol-water mixtures are common [61] [41]. |
| Analytical Standards | Identification and quantification of target compounds via chromatography. | Gallic acid, methyl gallate, caffeic acid [41]. Cannabidiol (CBD) standard [63]. Fatty Acid Methyl Ester (FAME) mix [59]. |
| Antioxidant Assay Reagents | Quantify total antioxidant capacity of extracts. | DPPH (2,2-diphenyl-1-picrylhydrazyl), ABTS, Folin-Ciocalteu reagent (for Total Phenolic Content) [59] [56] [60]. |
| Chromatography Solvents | Mobile and stationary phases for HPLC and GC-MS analysis. | HPLC-grade acetonitrile, methanol, n-hexane [41] [64]. |
Both RSM and AI-based models like ANN-GA are powerful tools for optimizing SFE processes. The choice between them hinges on the project's specific goals. RSM provides a robust, computationally less intensive method that yields interpretable polynomial models, making it an excellent choice for understanding factor interactions and for processes where high predictability is sufficient [60]. In contrast, ANN-GA offers superior predictive accuracy and is better suited for navigating highly complex, non-linear systems to find a global optimum, often resulting in marginally better experimental outcomes [61] [59] [56]. For researchers pushing the boundaries of yield and bioactivity in extractable compounds, ANN-GA represents a cutting-edge approach, while RSM remains a highly reliable and accessible workhorse for most optimization challenges.
Data-Driven Comparison: Validating Yield, Bioactivity, and Economic Viability
The selection of an optimal extraction technique is a critical determinant of success in natural product research and drug development. The choice between Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction (CSE) influences not only the yield but also the bioactivity, purity, and environmental footprint of the final extract. This guide provides an objective, data-driven comparison of these two methodologies, presenting recent experimental evidence to help researchers and scientists make informed decisions tailored to their specific project goals—whether prioritizing maximum crude yield, target compound concentration, or bioactivity.
The table below summarizes the core findings from recent head-to-head comparisons, highlighting how the optimal extraction method can vary significantly depending on the desired outcome.
Table 1: Summary of Recent Comparative Studies (2018-2025)
| Plant Material (Source) | Target Compound/Extract | Best Total Crude Yield (Method) | Best Target Compound Yield/Concentration (Method) | Key Performance Notes |
|---|---|---|---|---|
| Khella (Ammi visnaga) Fruits [66] [17] | Furanochromones (Khellin, Visnagin) | 15.44% (30% Ethanol, CSE) | 30.1% Concentration in extract (SCFE with 5% MeOH) | SCFE extract showed superior, selective concentration of furanochromones and significantly stronger cytotoxic activity ( [66] [17]). |
| Manjakani (Quercus infectoria) Gall [67] | Phenolic Compounds & Total Extract | 8.01% (SCFE-CO₂ with 1% MeOH) | ~75% Total Phenolic Acids (Both SCFE & CSE) | SCFE achieved nearly double the crude yield. Composition was similar, but CSE with methanol produced a toxic extract, unlike SCFE ( [67]). |
| Chamaecyparis formosensis Leaves [68] | Essential Oil & Chemical Profile | Dominated by α-pinene (83.4%) (HD) | Dominated by kaur-16-ene (51.1%) (SCFE) | Extraction method drastically altered the chemical profile, leading to differences in subsequent antifungal efficacy ( [68]). |
| Chamaecyparis obtusa var. formosana Leaves [68] | Essential Oil & Chemical Profile | Dominated by sabinene (36.2%) (HD) | Dominated by totarol (50.9%) (SCFE) | The antifungal activity against specific fungi was highest for the oil extracted via SFE, demonstrating method-dependent bioactivity ( [68]). |
Detailed Experimental Data and Protocols
This section details the methodologies and quantitative results from the key studies presented in the summary, providing a deeper understanding of the experimental groundwork.
Case Study: Ammi visnaga (Khella) Furanochromones
This 2021 study provides a direct comparison of SFE and various conventional solvents for extracting bioactive furanochromones (khellin and visnagin) [66] [17].
Table 2: Detailed Yield Data for Ammi visnaga Extractions
| Extraction Method | Solvent / Conditions | Total Crude Extract Yield (% w/w) | Total Furanochromones in Extract (g) | Furanochromones Concentration (% w/w of Extract) |
|---|---|---|---|---|
| Conventional (CSE) | Boiling Water | Not Reported | 82.11 ± 2.1 | 5.95% |
| Conventional (CSE) | Acetone | Not Reported | 103.21 ± 1.5 | 7.49% |
| Conventional (CSE) | 95% Ethanol | Not Reported | 111.10 ± 2.4 | 8.23% |
| Conventional (CSE) | 30% Ethanol | 15.44% | 101.74 ± 1.9 | 6.59% |
| Supercritical (SFE) | CO₂ + 5% MeOH, 50°C, 20 MPa | 4.50% | 135.00 ± 3.22 | 30.10% |
Experimental Protocol [66] [17]:
- Plant Material Preparation: Fruits of Ammi visnaga L. were powdered.
- Conventional Solvent Extraction (CSE): Used solvents (water, acetone, 30% and 95% ethanol) in a solvent-to-feed ratio of 10:1 (100 ml solvent per 10 g plant material). Extraction was conducted at 50°C for 8 hours. The extract was concentrated and dried.
- Supercritical Fluid Extraction (SFE): Conducted using supercritical CO₂ with 5% methanol as a co-solvent. Conditions were set at 50°C and 20 MPa (200 bar) for 60 minutes.
- Analysis: Extract yields were determined gravimetrically. Khellin and visnagin content was quantified using HPLC.
Key Findings: While 30% ethanol produced the highest mass of crude extract, SFE was vastly superior in selectivity, producing an extract with a much higher concentration (30.1%) of the target furanochromones. This translated directly to enhanced bioactivity, with the SFE extract demonstrating strong cytotoxic activity (IC₅₀ < 20 µg/mL) against breast cancer (MCF-7) and hepatocellular carcinoma (HepG2) cell lines, outperforming the conventional extracts [66] [17].
Case Study: Quercus infectoria (Manjakani) Gall Phenolics
A 2025 conference proceeding compared techniques for extracting phenolic compounds from Quercus infectoria galls [67].
Experimental Protocol [67]:
- Conventional Extraction: 10 g of plant material was extracted with 100 ml of methanol at 50°C for 8 hours.
- Supercritical Fluid Extraction: 10 g of the same material was processed with SFE-CO₂ at 50°C and 20 MPa for 60 minutes, using methanol as a 1% (w/w) co-solvent.
- Analysis: Yields were measured gravimetrically. Phenolic acid composition (tannic, gallic, ellagic, and quinic acid) was determined by HPLC. Toxicity was assessed using an MTT assay.
Key Findings: SFE with a co-solvent produced a higher crude yield (8.01%) compared to CSE with methanol (4.19%). Both methods yielded extracts with similar compositions of phenolic acids (e.g., ~67% tannic acid). A critical finding was on safety: the CSE extract was found to be toxic, whereas the SFE extract was non-toxic, underscoring SFE's potential to produce cleaner, safer extracts by avoiding toxic solvent residues [67].
The Scientist's Toolkit: Key Research Reagents and Materials
The following table outlines essential materials and their functions for designing SFE and CSE experiments, based on the reviewed methodologies.
Table 3: Essential Reagents and Materials for Extraction Studies
| Item | Function / Relevance in Extraction | Example from Studies |
|---|---|---|
| Carbon Dioxide (CO₂) | The most common supercritical fluid due to its mild critical point (31.1°C, 72.8 bar), non-toxicity, and low cost. Serves as the primary solvent in SFE. | Used as the primary SFE solvent in all cited studies [66] [67] [68]. |
| Co-solvents (Modifiers) | Added in small amounts (1-10%) to scCO₂ to polarity and improve the extraction efficiency of medium-polarity compounds (e.g., phenolics, furanochromones). | Methanol was used at 5% to extract furanochromones from Ammi visnaga [66] and at 1% for phenolics from Quercus infectoria [67]. |
| Organic Solvents (CSE) | Used in conventional extraction. Selectivity and yield are determined by the solvent's polarity (e.g., ethanol, acetone, methanol, hexane). | Water, ethanol (30% and 95%), and acetone were used for Ammi visnaga [66]. Methanol and hexane are common for essential oils and phenolics [67] [68]. |
| High-Pressure Extraction Vessel | A core component of SFE systems, designed to withstand high pressures (e.g., >200 bar) and allow flow-through of the supercritical fluid. | Implied in all SFE studies. Vessel size can be scaled from analytical (e.g., 10 ml [69]) to commercial scale [70]. |
| HPLC-MS/GC-MS System | For the qualitative and quantitative analysis of extract composition, crucial for comparing the selectivity and purity of different extraction methods. | HPLC was used to quantify khellin and visnagin [66] [17]. GC-MS was used to profile essential oil components from Chamaecyparis [68]. |
Decision Workflow and Technical Considerations
The following diagram maps the logical decision process for selecting and optimizing an extraction method based on research objectives and compound properties.
Critical Analysis and Research Implications
The empirical data demonstrates that the "best" extraction method is inherently defined by the research objective.
For Maximum Crude Yield: Conventional solvent extraction, particularly with aqueous ethanol, often provides the highest mass of crude extract, making it suitable for initial biomass assessments or when a broad spectrum of constituents is desired [66] [17].
For Selective Concentration and Bioactivity: SFE consistently produces extracts with higher purity and concentration of target compounds, as evidenced by the 30.1% furanochromone content in Ammi visnaga [66]. This enhanced purity directly translates to superior biological efficacy, a critical factor in drug discovery.
For Reproducibility and Safety: The tunable nature of SFE (by adjusting pressure, temperature, and co-solvents) allows for a highly controlled and reproducible process [71]. Furthermore, the avoidance of toxic organic solvents can result in safer extracts, as shown in the Quercus infectoria study [67].
For Chemical Profile and Novel Discovery: The choice of extraction technique can dramatically alter the chemical profile of an extract, as seen in the Chamaecyparis study, where different methods yielded oils dominated by entirely different major compounds [68]. This suggests that relying on a single extraction method may overlook significant portions of a plant's chemical repertoire.
The escalating demand for natural bioactive compounds in the pharmaceutical, nutraceutical, and cosmeceutical industries has intensified the focus on optimizing extraction technologies. Within research on supercritical fluid extraction versus conventional solvent extraction yield studies, a critical question emerges: how do these methods compare in isolating compounds with definitive antioxidant, antimicrobial, and cytotoxic activities? This guide provides a systematic comparison of extraction techniques based on quantitative bioactivity benchmarks, offering researchers a data-driven framework for method selection. The efficiency of an extraction method is no longer measured solely by yield, but by the biological potency of the resulting extracts, which is influenced by the method's ability to preserve delicate compound structures and functionalities.
Comparative Bioactivity Performance of Extraction Methods
The choice of extraction method significantly impacts the type, concentration, and biological activity of the recovered compounds. The following tables summarize key performance metrics across different techniques and source materials.
Table 1: Antioxidant Activity and Phenolic Content of Extracts from Various Matrices
| Source Material | Extraction Method | Key Solvent | Total Phenolic Content (mg GAE/g) | Antioxidant Activity (IC50/Other Metric) | Citation |
|---|---|---|---|---|---|
| Schinus terebinthifolius Leaves | Conventional (CE) | 50% Ethanol | 668.56 ± 11.52 | ABTS: 12,645.50 ± 60.31 µmol TE/g DM | [72] |
| Schinus terebinthifolius Leaves | Accelerated Solvent (ASE) | 50% Ethanol | 373.86 ± 8.04 | ABTS: 7,114.33 ± 77.91 µmol TE/g DM | [72] |
| Schinus terebinthifolius Leaves | Pulsed Electric Field (PEF) | 50% Ethanol | 293.43 ± 11.52 | ABTS: 5,864.33 ± 56.60 µmol TE/g DM | [72] |
| Pleurotus ostreatus Mushroom | Conventional | Chloroform | Flavonoids: 104.83 ± 29.46 mg/100g | Best overall IC50 across multiple assays | [73] |
| Pleurotus ostreatus Mushroom | Conventional | Methanol | Phenols: 8.87 ± 0.06 mg/100g | DPPH: 87.67% at 500 µg/mL | [73] |
| Matthiola ovatifolia Aerial Parts | Microwave-Assisted (MAE) | Ethanol | 69.6 ± 0.3 | Exhibited the highest antioxidant activity among tested methods | [14] |
Table 2: Antimicrobial and Cytotoxic Performance of Various Extracts and Formulations
| Active Agent / Source | Extraction/Formulation Method | Test Organism / Cell Line | Key Efficacy Metric | Citation |
|---|---|---|---|---|
| Bee Pollen | Ultrasound-Assisted (UAE) & Supercritical Fluid (SFE) | - | Optimized recovery of phenolic compounds and Vitamin C | [74] |
| Silver Nanoparticles (from I. viscosa) | Green Synthesis | Saos-2 & MCF-7 cells | Significant dose-dependent cytotoxicity | [75] |
| Ag-Based Metal-Organic Frameworks | Chemical Synthesis | E. coli & S. aureus | MIC: 5-15 ppm & 10-20 ppm, respectively | [76] |
| Essential Oils/Citrus Extract | Gelled Emulsion (GE) | B. subtilis (on bread) | Extended shelf-life, delayed proliferation to 36 days | [77] |
| Matthiola ovatifolia Aerial Parts | Microwave-Assisted (MAE) | Hep-G2 & MCF-7 cells | Highest cytotoxic activity among tested methods | [14] |
Detailed Experimental Protocols for Key Assays
To ensure the reproducibility of bioactivity benchmarking, researchers must adhere to standardized experimental protocols. Below are detailed methodologies for the most common assays used to generate the data in this guide.
Phytochemical Quantification Assays
Total Phenolic Content (TPC) by Folin-Ciocalteu Method
- Principle: Measures the reduction of phosphomolybdate-phosphotungstate reagent by phenolic compounds to form a blue chromophore.
- Procedure:
- Combine 0.5 mL of extract (e.g., 300 µg/mL concentration) with 2.5 mL of 10% Folin-Ciocalteu reagent.
- After 5 minutes, add 2 mL of sodium carbonate (7.5% w/v).
- Incubate the mixture in the dark for 30-60 minutes at room temperature.
- Measure the absorbance at 765 nm against a blank.
- Express results as mg Gallic Acid Equivalents (GAE) per g of dry weight using a gallic acid standard curve [14] [73].
Total Flavonoid Content (TFC)
- Procedure:
- Mix 1 mL of extract with 0.3 mL of NaNO₂ (5%).
- After 5 minutes, add 0.3 mL of AlCl₃ (10%).
- At 6 minutes, add 2 mL of NaOH (1 M).
- Dilute the solution immediately with 2.4 mL of ethanol and vortex.
- Measure absorbance at 510 nm. Results are expressed as mg Quercetin Equivalents (QE) per g dry weight [14].
Antioxidant Activity Assays
DPPH (2,2-diphenyl-1-picrylhydrazyl) Radical Scavenging Assay
- Principle: Antioxidants donate a hydrogen atom to the stable purple DPPH radical, decolorizing it to yellow. The extent of discoloration indicates free radical scavenging capacity.
- Procedure:
- Prepare a 0.1 mM DPPH solution in methanol.
- Add 1 mL of this solution to 1 mL of the extract at various concentrations.
- Vortex the mixture and incubate in the dark for 30 minutes.
- Measure the absorbance at 517 nm against a methanol blank.
- Calculate the percentage inhibition using the formula:
% Inhibition = [(A_control - A_sample) / A_control] × 100[73].
Oxygen Radical Absorbance Capacity (ORAC) Assay
- Principle: Measures the ability of antioxidants to protect a fluorescent probe (e.g., fluorescein) from peroxyl radical attack generated by AAPH.
- Procedure:
- In a microplate, mix 150 µL of fluorescein and 25 µL of extract or Trolox standard.
- Initiate the reaction by adding 25 µL of AAPH solution.
- Immediately place the plate in a fluorescence reader and monitor the decay of fluorescence every minute for 1-2 hours (excitation: 485 nm, emission: 535 nm).
- Calculate the area under the curve (AUC) and express results as µM Trolox Equivalents [72].
Cytotoxicity Assays
MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) Assay
- Principle: Living cells reduce the yellow tetrazolium salt MTT to purple formazan crystals. The amount of formazan produced is proportional to the number of viable cells.
- Procedure:
- Seed cancer cell lines (e.g., MCF-7, Saos-2) in a 96-well plate and incubate for 24 hours.
- Treat cells with a range of concentrations of the test extract and incubate for 24-72 hours.
- Add MTT solution to each well and incubate for 2-4 hours to allow formazan crystal formation.
- Carefully remove the medium, dissolve the formazan crystals in DMSO, and measure the absorbance at 570 nm.
- Calculate cell viability as a percentage relative to the untreated control and determine the IC₅₀ value [75].
Mechanistic Pathways of Bioactivity
Understanding the biological activity of extracts requires insight into their mechanisms of action at the cellular and molecular levels. The following diagrams illustrate key pathways.
Antioxidant Defense Pathway Activation
Plant extracts rich in polyphenols can activate the body's endogenous antioxidant defense system, a crucial mechanism for mitigating oxidative stress.
Multimodal Antibacterial Mechanisms
Advanced antibacterial agents like Metal-Organic Frameworks (MOFs) and silver nanoparticles exert their effects through several simultaneous mechanisms.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful bioactivity evaluation relies on a suite of specialized reagents and instruments. The following table catalogues essential items for setting up these experiments.
Table 3: Key Reagents and Solutions for Bioactivity Research
| Reagent/Solution | Primary Function | Application Example |
|---|---|---|
| Folin-Ciocalteu Reagent | Quantification of total phenolic content | Reacts with phenolic compounds to form a blue complex for spectrophotometric measurement [14]. |
| DPPH (2,2-diphenyl-1-picrylhydrazyl) | Assessment of free radical scavenging activity | Stable radical used to evaluate antioxidant potential via color change [73]. |
| MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) | Determination of cell viability and cytotoxicity | Reduced by metabolically active cells to a purple formazan product [75]. |
| Agar & Microbial Culture Media (e.g., Mueller-Hinton) | Support microbial growth for antimicrobial assays | Provides a solid or liquid medium for cultivating test bacteria and fungi in disc diffusion or MIC tests [14] [77]. |
| Silver Nitrate (AgNO₃) | Precursor for green synthesis of silver nanoparticles | Source of Ag⁺ ions, reduced by phytochemicals in plant extracts to form AgNPs [75]. |
| Essential Oils (e.g., Oregano, Cinnamon) | Natural antimicrobial agents | Used in encapsulation studies to create active formulations for food preservation and biomedicine [77]. |
| MOF Precursors (Metal salts, Organic linkers) | Synthesis of Metal-Organic Frameworks | Building blocks for constructing porous MOF structures with controlled release of antibacterial metal ions [76]. |
The extraction of bioactive compounds from natural products is a foundational step in pharmaceutical, nutraceutical, and fragrance development. For decades, Conventional Solvent Extraction (CSE) methods, such as maceration and Soxhlet extraction, have been the industry standard. However, the environmental footprint of these processes, characterized by high energy consumption and substantial use of often toxic organic solvents, has come under increased scrutiny [78]. In response, Supercritical Fluid Extraction (SFE), particularly using carbon dioxide (CO₂), has emerged as a technology frequently described as a "green solvent" alternative, promising to reduce ecological impacts [79] [80].
This guide provides an objective comparison of the environmental performance of SFE and CSE, grounded in Life Cycle Assessment (LCA) methodology. LCA offers a systematic, cradle-to-grave framework for quantifying the environmental impacts of products or processes, making it the gold standard for such evaluations [79]. For researchers and drug development professionals, understanding these nuances is critical for making informed, sustainable choices in process development that align with growing regulatory and consumer pressures for environmentally responsible practices [81].
Quantitative Environmental Impact Comparison
Life Cycle Assessment studies provide quantitative data that is essential for an objective comparison. The following tables summarize key environmental impact metrics and the factors that influence them, based on recent LCA reviews and case studies.
Table 1: LCA Impact Comparison for Extraction Technologies
| Technology | Global Warming Potential (GWP) | Primary Environmental Hotspot | Comparative Performance vs. CSE | Key Influencing Factors |
|---|---|---|---|---|
| Supercritical Fluid Extraction (SFE) | 0.2 - 153 kg CO₂eq/kg*e.g., 71.42 kg CO₂eq/g (Apple Pomace) [82] | Energy consumption (especially electricity for compression and heating) [79] [80] | Mixed results: 27 out of 70 reviewed LCA studies reported lower impacts than CSE; 18 reported higher impacts [79]. | Electricity mix, extraction scale, solvent recycling rate, feed concentration [79]. |
| Subcritical Water Extraction (SWE) | 6.20 kg CO₂eq/g (Apple Pomace) [82] | Energy consumption [79] | Can be significantly lower than SFE for the same feedstock [82]. | Temperature, pressure, processing time. |
| Conventional Solvent Extraction (CSE) | Highly variable; typically high due to solvent production and waste management [78] | Solvent production and waste disposal [78] | Often higher in toxicity and waste generation categories; performance is solvent-dependent [78]. | Solvent type, volume, and recyclability [78]. |
Note: The wide range for SFE GWP is application-dependent (e.g., gasification vs. extraction) and scale-dependent [79].
Table 2: Summary of Non-Greenhouse Gas Environmental Metrics
| Impact Category | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction (CSE) |
|---|---|---|
| Solvent Consumption | Lower; CO₂ is typically recycled within the system. Reduces use of conventional organic solvents [79]. | High; requires large volumes of organic solvents (e.g., hexane, petroleum ether) with potential for fugitive emissions [78]. |
| Toxicity & Safety | Generally safer; CO₂ is non-flammable and non-toxic. The final product is free from toxic solvent residues [78]. | Higher hazard potential; uses toxic, flammable solvents, risking worker safety and requiring stringent purification steps [78]. |
| Waste Generation | Lower; CO₂ vents as a gas, leaving minimal liquid waste [78]. | Higher; generates significant hazardous waste streams requiring treatment and disposal [78]. |
Detailed Experimental Protocols from Cited Studies
To contextualize the LCA data, it is vital to understand the underlying experimental methodologies. The following protocols are summarized from recent, relevant studies.
Protocol 1: Supercritical vs. Subcritical Extraction of Apple Pomace
This 2025 LCA study directly compared the carbon footprint of SFE and Subcritical Water Extraction (SWE) for obtaining bioactive compounds from apple pomace [82].
- Objective: To assess the carbon footprint (cradle-to-gate) of extracting 1 gram of bioactive compounds from apple pomace using SC-CO₂ and SWE.
- Software & Database: The analysis was performed using SimaPro software and the ecoinvent database.
- System Boundary: The LCA included all processes from raw material acquisition (e.g., CO₂ production, electricity generation, water use) to the final extraction step at the factory gate.
- Life Cycle Impact Assessment (LCIA): The primary impact category reported was Global Warming Potential (GWP), measured in kg of CO₂ equivalent (kg CO₂eq) per functional unit (1 g of bioactive compounds).
- Key Finding: The SFE process resulted in 71.42 kg CO₂eq per gram of extract, while SWE resulted in 6.20 kg CO₂eq, highlighting that the "green" label for SFE is not universally applicable and is highly dependent on process energy intensity [82].
Protocol 2: Microwave-Assisted vs. Conventional Solvent Extraction of Phytochemicals
A 2025 study on Matthiola ovatifolia provides a benchmark for the efficiency and environmental advantages of modern techniques over CSE [14].
- Plant Material: Aerial parts of Matthiola ovatifolia were collected, dried, lyophilized, and ground into a fine powder.
- Extraction Techniques Compared:
- Analysis: The extracts were analyzed for Total Phenolic Content (TPC), Total Flavonoid Content (TFC), and antioxidant activity.
- Key Finding: MAE using ethanol as the solvent yielded the highest concentrations of all phytochemical classes and the strongest biological activities. Crucially, MAE offers environmental benefits through reduced processing time and lower solvent consumption compared to CSE [14].
Protocol 3: Ultrasound-Assisted vs. Solid-Liquid Extraction of Hawthorn Leaves
A 2025 study optimized the extraction of bioactive compounds from Crataegus almaatensis leaves, comparing Ultrasound-Assisted Extraction (UAE) to conventional Solid-Liquid Extraction (SLE) [46].
- Experimental Design: A four-factor face-centered central composite design (FC-CCD) was used to optimize temperature, time, ethanol concentration, and solid-to-liquid ratio (or ultrasound power for UAE).
- SLE Protocol: Maceration on a magnetic stirrer at 300 rpm [46].
- UAE Protocol: Using an ultrasonic processor (UP 400S) at 24 kHz and 100 W power [46].
- Key Finding: The optimized UAE method achieved a 16% higher Total Phenolic Content (TPC) while using 40% (v/v) ethanol compared to the 75% (v/v) ethanol required for SLE. This demonstrates UAE's dual advantage of improved efficiency and reduced reliance on organic solvents, lowering its environmental impact [46].
Workflow and Technology Selection Diagram
The following diagram illustrates the general workflows for SFE and a typical CSE process (Soxhlet extraction), highlighting key stages where environmental impacts are accrued. This aligns with the experimental protocols and LCA findings detailed above.
Diagram Title: SFE and CSE Process Workflows
The Scientist's Toolkit: Research Reagent Solutions
This table details key materials and reagents used in the extraction processes discussed, with a focus on their function and role in environmental impact.
Table 3: Essential Reagents and Materials for Extraction Research
| Reagent/Material | Function in Extraction | Environmental & Safety Considerations |
|---|---|---|
| Carbon Dioxide (CO₂) | The supercritical solvent in SFE. In its supercritical state, it has liquid-like density and gas-like diffusivity, enabling efficient penetration of plant matrixes [78]. | Non-flammable, non-toxic. Considered green, but energy required for compression is the primary environmental hotspot [79] [82]. |
| Ethanol | A polar solvent used in CSE, MAE, and UAE for extracting phenolics, flavonoids, and alkaloids [14] [46]. | Less toxic than hexane and biodegradable. Its environmental impact is tied to production energy and volume used [78]. |
| n-Hexane | A non-polar organic solvent conventionally used for extracting oils and lipids [78]. | Highly flammable, neurotoxic, and a hazardous air pollutant. Its production and disposal pose significant environmental and health risks [78]. |
| Water | A green, polar solvent. Used in subcritical water extraction (SWE) where its properties change at high T&P to extract less polar compounds [82]. | Non-toxic, safe, and abundant. The most environmentally benign solvent, though energy is needed for heating in SWE [82]. |
| Plant Material | The raw feedstock containing target bioactive compounds. Properties (e.g., particle size, moisture) affect extraction efficiency [14] [46]. | A renewable resource. Using agricultural by-products (e.g., apple pomace) enhances the sustainability profile of the extraction process [82]. |
The declaration of SFE as an unambiguously "greener" alternative to CSE is an oversimplification. The evidence from LCA studies reveals a more complex picture: while SFE eliminates the problem of toxic solvent residues and can reduce waste generation, its environmental impact is often dominated by high energy demands, leading to a GWP that can exceed that of CSE in some applications [79] [82]. Conversely, CSE's primary environmental drawbacks are linked to solvent-related impacts, including production, emission, and waste handling [78].
The choice between SFE and CSE should be guided by a project-specific evaluation. Researchers must consider the energy source (e.g., a grid with high renewable penetration significantly improves SFE's GWP), the scale of operation, the possibility of solvent recycling, and the nature of the target compound and feedstock [79]. Advanced techniques like MAE and UAE present compelling alternatives, often bridging the gap by offering reduced processing times and solvent use with lower capital and energy costs than SFE [14] [46]. Ultimately, a holistic, life-cycle perspective is indispensable for making truly sustainable decisions in scientific research and drug development.
The selection of an extraction method is a critical decision in pharmaceutical development, directly influencing the quality, safety, and economic viability of the final product. This guide provides an objective comparison between Supercritical Fluid Extraction (SFE) and Conventional Solvent Extraction methods, focusing on their performance in balancing the critical triad of purity, solvent residues, and production cost. As regulatory pressures intensify and the demand for clean-label pharmaceuticals grows, understanding the technical and economic trade-offs between these technologies is essential for researchers and drug development professionals. The analysis is framed within a broader thesis on extraction yield studies, providing both comparative data and detailed experimental protocols to inform method selection.
Fundamental Principles
Conventional Solvent Extraction relies on organic solvents such as ethanol, acetone, hexane, or their aqueous mixtures to solubilize and separate target compounds from raw materials. Techniques include maceration, percolation, reflux, and Soxhlet extraction, which often involve high temperatures and prolonged processing times, potentially degrading thermolabile bioactives [78].
Supercritical Fluid Extraction (SFE) utilizes fluids, typically carbon dioxide (CO₂), above their critical temperature and pressure (31.1°C and 73.8 bar for CO₂). In this supercritical state, the fluid exhibits gas-like diffusivity and liquid-like density, enabling deep penetration into matrices and efficient solubilization of target compounds. The solvating power is tunable by adjusting pressure and temperature, allowing for selective extraction. Co-solvents like ethanol can be added in small quantities to modify polarity [10] [43].
Quantitative Performance Comparison
The table below summarizes key performance metrics from industrial and research studies, highlighting the trade-offs between these technologies.
Table 1: Comprehensive Performance Comparison: SFE vs. Conventional Extraction
| Performance Metric | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction |
|---|---|---|
| Typical Extract Purity | Up to ~95% [10] | Typically 70-80% [10] |
| Solvent Consumption | Reduces solvent use by 80-90% [10] | High solvent volumes required |
| Residual Solvent in Product | Negligible with supercritical CO₂ [43] | Significant, requires post-processing removal [78] |
| Energy Consumption | 30-50% lower than conventional methods [10] | High due to heating and solvent recovery |
| Operational Temperature | Low (e.g., near 31°C for CO₂), preserves thermolabile compounds [10] | Often high (e.g., reflux temperatures), risks thermal degradation [78] |
| Selectivity | Highly tunable for selective compound isolation [10] [66] | Lower selectivity, co-extraction of impurities common [66] |
| Extraction Time | Generally faster due to high diffusivity [42] | Can be time-consuming (hours to days) [78] |
| Process "Greenness" | High; uses non-toxic, recyclable CO₂ [43] | Lower; often involves hazardous solvents [78] |
Case Study: Extraction of Khella (Ammi visnaga L.) Furanochromones
A direct comparative study on extracting khellin and visnagin from Khella fruits provides illustrative experimental data on yield versus purity.
Table 2: Experimental Yield and Purity Data from Khella Fruit Extraction [66]
| Extraction Method | Total Extract Yield (% w/w) | Total Furanochromone Content in Extract (%) |
|---|---|---|
| 30% Ethanol (Conventional) | 15.44% | Not Specified (Lower than SFE) |
| Acetone (Conventional) | 9.60% | Not Specified (Lower than SFE) |
| Supercritical Fluid Extraction | 4.50% | 30.1% |
Interpretation: While conventional 30% ethanol extraction produced a higher gross yield, SFE resulted in a significantly more concentrated extract of the target furanochromones. This demonstrates a key strength of SFE: its selectivity leads to a purer product with fewer impurities, reducing downstream purification costs [66].
Detailed Experimental Protocols
Protocol for Supercritical Fluid Extraction
This protocol outlines a method for extracting bioactive compounds from plant-based materials, adaptable for laboratory-scale systems.
3.1.1 Research Reagent Solutions & Essential Materials
Table 3: Key Materials for SFE and Conventional Extraction Protocols
| Item | Function/Description | Example Specification |
|---|---|---|
| Supercritical CO₂ Supply | Primary extraction fluid. | Food or pharmaceutical grade, 99.99% purity. |
| Co-solvent (e.g., Ethanol) | Modifies polarity to enhance solubility of polar compounds. | Pharmaceutical grade, 96% or absolute. |
| Raw Material (e.g., Plant Matrix) | Source of target bioactive compounds. | Dried, milled to 250-500 µm particle size. |
| On-line Modifier Pump | Precisely delivers co-solvent into the supercritical CO₂ stream. | High-pressure HPLC pump. |
| Extraction Vessel | Holds the raw material under high pressure. | Stainless steel, rated for > 400 bar. |
| Back-Pressure Regulator | Maintains and controls system pressure. | Automated, electronically controlled. |
| Collection Vessel | Collects the extract after depressurization. | Glass, cooled (e.g., 4°C). |
3.1.2 Step-by-Step Workflow
- Raw Material Pretreatment: The plant material is dried and ground to a uniform particle size (e.g., 250-500 µm) to increase surface area and disrupt cell walls, facilitating solvent penetration [10].
- System Preparation: The extraction vessel is loaded with the prepared biomass. The system is sealed, and a leak test is performed. The co-solvent reservoir is filled if required.
- Extraction Parameter Setup: Key parameters are set based on the target compounds:
- Dynamic Extraction: The system is brought to the set temperature and pressure. Supercritical CO₂ (with or without co-solvent) is passed continuously through the biomass bed for a predetermined time (e.g., 60-180 minutes).
- Fraction Collection: The solute-laden supercritical fluid passes through a back-pressure regulator, where depressurization causes the CO₂ to revert to gas, precipitating the extract into the collection vessel.
- Extract Processing: The collected extract is weighed and analyzed. Further steps may include solvent removal for co-solvent extracts or formulation.
Protocol for Conventional Soxhlet Extraction
Soxhlet extraction serves as a benchmark conventional method for solid samples.
3.2.1 Step-by-Step Workflow
- Sample Preparation: The dried, ground sample is placed in a cellulose or thimble.
- Apparatus Assembly: The thimble is placed in the extraction chamber of the Soxhlet apparatus. A distillation flask is filled with a suitable solvent (e.g., ethanol, hexane) and attached below. A condenser is secured atop the apparatus.
- Heating and Extraction: The solvent is heated to reflux. The solvent vapor rises, condenses in the condenser, and drips onto the sample in the thimble.
- Siphoning: When the solvent in the extraction chamber reaches a certain level, the siphon arm automatically empties the solvent, along with the dissolved solutes, back into the distillation flask. This cycle repeats automatically for 6-24 hours.
- Solvent Removal: After extraction, the solvent in the distillation flask is evaporated under reduced pressure using a rotary evaporator to concentrate the crude extract.
- Extract Analysis: The concentrated extract is analyzed for yield and composition [78].
Economic and Regulatory Analysis
Cost Structure Breakdown
The economic feasibility of SFE versus conventional methods involves a trade-off between capital expenditure (CAPEX) and operational expenditure (OPEX).
Table 4: Comparative Economic Analysis of Extraction Methods
| Cost Factor | Supercritical Fluid Extraction (SFE) | Conventional Solvent Extraction |
|---|---|---|
| Capital Cost (CAPEX) | High initial investment for high-pressure equipment [43] | Lower initial investment for atmospheric/low-pressure systems |
| Operational Cost (OPEX) | Lower solvent costs (CO₂ is cheap and recyclable); up to 89% cost reduction over Soxhlet reported [10]. Energy costs significant for compression [43]. | High solvent procurement and disposal costs. High energy costs for heating and distillation. |
| Solvent Recovery/Disposal | Minimal cost; CO₂ is gaseous at ambient conditions. | Significant cost for distillation and hazardous waste handling. |
| Downstream Processing | Lower cost due to higher initial purity and lack of solvent residues [10] [66]. | Higher cost for purification and solvent residue removal to meet regulatory standards. |
| Process Scalability | Well-established for some industries (e.g., decaffeination); scalability challenges persist for novel compounds [10]. | Highly scalable with established engineering principles. |
Regulatory and Safety Considerations
Regulatory drivers are increasingly favoring SFE technology due to its superior safety and environmental profile.
- Solvent Residues: Regulatory bodies like the FDA and EMA strictly limit residual solvents in pharmaceuticals (ICH Q3C guidelines). SFE using CO₂, which is "Generally Recognized As Safe" (GRAS), eliminates concerns over toxic solvent residues, a significant advantage over methods using hexane or chlorinated solvents [43].
- Environmental Impact: SFE reduces solvent use by 80-90% and cuts energy use by 30-50% compared to traditional methods, aligning with green chemistry principles and corporate sustainability goals [10].
- Process Safety: CO₂ is non-flammable and non-toxic, improving operational safety compared to flammable organic solvents [43].
The choice between Supercritical Fluid Extraction and Conventional Solvent Extraction is multifaceted. SFE offers a compelling profile of high selectivity, superior extract purity, negligible solvent residues, and a greener lifecycle, which directly addresses stringent regulatory and consumer demands. However, this comes with a high initial capital investment and persistent challenges in scalability for some applications. In contrast, conventional methods, while more straightforward to implement and scale, incur higher operational costs for solvent and waste management and present greater challenges in achieving high purity and meeting residual solvent standards.
Future developments in the SFE field are focused on improving its economic and operational competitiveness. Key research areas include:
- Process Intensification: Developing continuous extraction systems and optimizing parameters with AI to improve throughput and reduce costs [43] [42].
- Hybrid and Sequential Extraction: Using SFE as an initial step to remove oils, followed by other green methods to recover polar compounds, maximizes the value extracted from biomass [10].
- Circular Economy Applications: Leveraging SFE for waste valorization—extracting bioactives from food, agricultural, and pharmaceutical by-products—presents a growing opportunity for sustainable resource use [10] [43].
For researchers and drug developers, the decision must be based on a holistic analysis of the target compound, desired purity, regulatory constraints, and total cost of ownership. SFE is increasingly the technology of choice for high-value, thermolabile pharmaceuticals where purity and safety are paramount.
Conclusion
The comparative analysis unequivocally demonstrates that Supercritical Fluid Extraction offers a superior, sustainable alternative to conventional methods for many pharmaceutical applications, particularly in efficiently extracting lipophilic compounds with high purity and enhanced bioactivity. While SFE excels in selectivity, speed, and environmental profile, conventional methods remain relevant for specific polar compounds and lower-budget operations. The future of extraction lies not in a single technology, but in the intelligent, integrated application of hybrid methods (e.g., SFE-UAE, SFE-MAE) and the adoption of AI-driven optimization. For biomedical research, this evolution promises more reproducible, potent, and clinically viable natural product extracts, directly supporting the development of next-generation therapeutics and nutraceuticals. Future efforts should focus on standardizing these advanced protocols and further exploring their application in valorizing agri-food waste for a circular economy in drug development.
References
- 1. Carbon Dioxide Supercritical Extraction - an overview [https://www.sciencedirect.com/topics/engineering/carbon-dioxide-supercritical-extraction]
- 2. Principles of the supercritical extraction process [https://www.buffaloextracts.com/knowledge/what-are-the-principles-of-the-supercritical-extraction-process-explore-this-guide-to-learn/]
- 3. SFE vs. Traditional Solvent Extraction: Key Differences & ... [https://www.toptionlab.com/what-is-the-main-difference-between-sfe-and-traditional-solvent-extraction]
- 4. Supercritical CO2 green solvent extraction of Nepeta crispa [https://www.sciencedirect.com/science/article/abs/pii/S0896844624002869]
- 5. Supercritical fluid extraction [https://en.wikipedia.org/wiki/Supercritical_fluid_extraction]
- 6. Global Market Growth for Supercritical Fluid Extraction [https://www.buffaloextracts.com/knowledge/global-demand-for-supercritical-fluid-extraction-equipment-market-growth-and-opportunities/]
- 7. Optimizing ethanol-modified supercritical CO₂ extraction ... [https://www.nature.com/articles/s41598-025-91441-x]
- 8. A comparative study of conventional and supercritical fluid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5018615/]
- 9. Comparative analysis of extraction technologies for plant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11911331/]
- 10. Valorization of bioactive compounds from food by-products ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814625015286]
- 11. Techniques for extraction and isolation of natural products [https://pmc.ncbi.nlm.nih.gov/articles/PMC5905184/]
- 12. Techniques for extraction and isolation of natural products [https://cmjournal.biomedcentral.com/articles/10.1186/s13020-018-0177-x]
- 13. Recent advances and comparisons of conventional and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7550548/]
- 14. Comparative analysis of solvent and advanced extraction ... [https://www.nature.com/articles/s41598-025-23565-z]
- 15. Soxhlet Extraction - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/soxhlet-extraction]
- 16. ≫ Soxhlet Extraction: The Method, Equipment, and Its ... [https://pobel.com/en/blog/guides/soxhlet-extraction-the-method-equipment-and-its-essential-applications-in-the-laboratory]
- 17. Assessment of Conventional Solvent Extraction vs. ... [https://www.mdpi.com/1420-3049/26/5/1290]
- 18. Supercritical Fluid Extraction - an overview [https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/supercritical-fluid-extraction]
- 19. On the mass transfer of supercritical fluids, specifically ... [https://www.sciencedirect.com/science/article/pii/S1385894724040087]
- 20. New Trends in Supercritical Fluid Technology and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10254675/]
- 21. [PDF] Assessment of Conventional .... Solvent Extraction vs [https://www.semanticscholar.org/paper/Assessment-of-Conventional-Solvent-Extraction-vs.-Khalil-Bishr/05373a71ca21b603db59c0c9cb838ad92c3f22e6]
- 22. evaluation of Comparative CO2 and... :: BioResources supercritical [https://bioresources.cnr.ncsu.edu/resources/comparative-evaluation-of-supercritical-co2-and-methanol-extraction-of-ruta-graveolens-polyphenolic-compounds-in-vitro-characterization-of-antioxidant-and-antimicrobial-potentials/]
- 23. Supercritical Fluid Extraction: A Green and Sustainable ... [https://www.ajgreenchem.com/article_221328.html]
- 24. Effects of different solvents on phytochemical constituents ... [https://www.nature.com/articles/s41598-025-14506-x]
- 25. Comparative analysis of solvent and advanced extraction ... [https://pubmed.ncbi.nlm.nih.gov/41238571/]
- 26. Impact of extraction techniques on phytochemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12343529/]
- 27. Impact of extraction techniques on phytochemical ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1615338/full]
- 28. Supercritical CO2 technology for biomass extraction: Review [https://www.sciencedirect.com/science/article/pii/S0926669025008945]
- 29. Supercritical Fluid Extraction of Peruvian Schinus molle ... [https://www.mdpi.com/2227-9717/13/7/2191]
- 30. Optimization of supercritical fluid extraction from waste oil ... [https://ui.adsabs.harvard.edu/abs/2025JEST...2215921M/abstract]
- 31. Optimization of Supercritical Fluid Extraction for the ... [https://pubmed.ncbi.nlm.nih.gov/40002392/]
- 32. High-Fidelity Prediction of Drug Solubility in Supercritical ... [https://www.sciencedirect.com/science/article/pii/S0928098725003197]
- 33. Predicting drug solubility in supercritical carbon dioxide ... [https://www.nature.com/articles/s41598-025-24006-7]
- 34. A review of modern and conventional extraction techniques ... [https://www.sciencedirect.com/science/article/pii/S2468227623000443]
- 35. Optimization of Conventional Extraction Parameters for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9311538/]
- 36. Optimization of Extraction Parameters to Enhance the ... [https://www.mdpi.com/2306-5710/10/3/56]
- 37. Exploring the Influence of Extraction Methods, Solvents ... [https://www.mdpi.com/2297-8739/12/9/236]
- 38. Recent Advances in Supercritical Fluid Extraction of Natural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7504334/]
- 39. Lipid Extracts Obtained by Supercritical Fluid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9024703/]
- 40. Integrated Supercritical Fluid Extraction and Pre-Formulation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10745611/]
- 41. Parameter Effects and Optimisation in Supercritical Fluid ... [https://www.mdpi.com/2297-8739/9/12/385]
- 42. Supercritical Fluid Extraction System Market Size 2025-2032 [https://www.360iresearch.com/library/intelligence/supercritical-fluid-extraction-system]
- 43. Supercritical Fluid Extraction Chemicals Market Size [https://www.gminsights.com/industry-analysis/supercritical-fluid-extraction-chemicals-market]
- 44. Scale-up approach for supercritical fluid extraction with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8338961/]
- 45. Scale-up study of supercritical fluid extraction process for ... [https://www.sciencedirect.com/science/article/pii/S0896844610003992]
- 46. PMC Search Update - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12382680/]
- 47. Sustainable Optimization of Extraction Techniques for ... [https://chemrxiv.org/engage/chemrxiv/article-details/67ffcad3927d1c2e663e09bd]
- 48. Supercritical CO2 Extraction Equipment Future-Proof ... [https://www.marketreportanalytics.com/reports/supercritical-co2-extraction-equipment-21772]
- 49. Supercritical CO2 Extraction vs. Hexane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11644741/]
- 50. Advances in Supercritical Fluid Extraction, DFT, and ... [https://www.sciencedirect.com/science/article/pii/S2590123025034772]
- 51. Effectiveness of sustainable oil extraction techniques [https://www.sciencedirect.com/science/article/pii/S2666154324005830]
- 52. Supercritical Fluid Extraction—A Sustainable and Selective ... [https://www.mdpi.com/2076-3417/15/11/5914]
- 53. Modeling of supercritical fluid extraction bed: A critical review [https://www.sciencedirect.com/science/article/abs/pii/S0263876223002137]
- 54. Supercritical carbon dioxide extraction of lipids and ... [https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-025-02632-7]
- 55. A comparative study of supercritical fluid extraction and ... [https://www.sciencedirect.com/science/article/abs/pii/S2352554122000122]
- 56. Comparative evaluation and optimization of microwave and ... [https://pubs.rsc.org/en/content/articlehtml/2025/fb/d5fb00329f]
- 57. A Review on Microwave and Ultrasound-Assisted ... [https://link.springer.com/article/10.1007/s11947-025-03882-x]
- 58. Optimization of hybrid green extraction techniques for ... [https://www.sciencedirect.com/science/article/pii/S0023643825009570]
- 59. Optimization of extraction in supercritical fluids in obtaining ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2024.1491479/full]
- 60. Comparison of response surface methodology (RSM) and ... [https://www.sciencedirect.com/science/article/abs/pii/S0926669021005331]
- 61. Artificial intelligence-assisted optimization of extraction ... [https://www.nature.com/articles/s41598-025-25130-0]
- 62. Comparative analysis of the response surface ... [https://www.sciencedirect.com/science/article/pii/S2666086525000220]
- 63. Optimization of Supercritical Extraction of Cannabidiol ... [https://www.mdpi.com/2227-9717/12/11/2498]
- 64. Optimization of supercritical fluid extraction of essential oils ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4486533/]
- 65. Artificial Intelligence Modeling, RSM regression, and ... [https://www.sciencedirect.com/science/article/abs/pii/S0009250925012382]
- 66. Assessment of Conventional . Solvent ... Extraction vs Supercritical [https://pubmed.ncbi.nlm.nih.gov/33673560/]
- 67. Comparative study of yield, composition, reproducibility and toxicity ... [https://scholar.unair.ac.id/en/publications/comparative-study-of-yield-composition-reproducibility-and-toxici/]
- 68. evaluation of hydrodistillation, Comparative ... supercritical fluid [https://bioresources.cnr.ncsu.edu/resources/comparative-evaluation-of-hydrodistillation-supercritical-fluid-extraction-and-organic-solvent-extraction-on-leaf-essential-oils-of-chamaecyparis-formosensis-and-c-obtusa-var-formosana-and-their-p/]
- 69. Supercritical Fluid Extraction Method Development for ... [https://pubmed.ncbi.nlm.nih.gov/8953489/]
- 70. Solvent Supercritical Fluid Technologies to Extract ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6152233/]
- 71. Review Experimental design of supercritical fluid extraction [https://www.sciencedirect.com/science/article/abs/pii/S0260877413005128]
- 72. Comparative Study on Antioxidant Potential of Schinus ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12430624/]
- 73. Comparative antioxidant activity and phytochemical ... [https://www.nature.com/articles/s41598-024-54201-x]
- 74. Comparison of conventional and supercritical isolation ... [https://www.sciencedirect.com/science/article/pii/S0896844625000865]
- 75. Comparative Cytotoxic Effects of Silver Nanoparticles ... [https://link.springer.com/article/10.1007/s44411-025-00233-2]
- 76. Recent advances in metal–organic frameworks for ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra02955d?page=search]
- 77. Comparison of nanoemulsion, gelled emulsion, and ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429224001500]
- 78. Comparative analysis of extraction technologies for plant ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2025.1536590/full]
- 79. Environmental impacts of supercritical fluids processes [https://www.sciencedirect.com/science/article/pii/S0195925525003038]
- 80. Environmental impacts of supercritical fluids processes [https://hal.science/hal-05228336v1]
- 81. 80+ Sustainability Statistics for 2025: Ultimate List [https://www.arbor.eco/blog/sustainability-statistics]
- 82. Subcritical or supercritical? A comparative life cycle ... [https://www.sciencedirect.com/science/article/pii/S2666789425000571]