Optimizing Modifier Concentration in Supercritical Fluid Extraction: A Strategic Guide for Enhanced Bioactive Compound Recovery
This article provides a comprehensive guide for researchers and drug development professionals on the strategic use of modifiers in Supercritical Fluid Extraction (SFE) to enhance the yield and selectivity of...
Optimizing Modifier Concentration in Supercritical Fluid Extraction: A Strategic Guide for Enhanced Bioactive Compound Recovery
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on the strategic use of modifiers in Supercritical Fluid Extraction (SFE) to enhance the yield and selectivity of bioactive compounds. It covers the foundational role of modifiers like ethanol and methanol in solubilizing polar molecules, details methodological approaches for optimization using statistical design, and addresses common troubleshooting challenges. Furthermore, it presents validation data comparing SFE with modifiers to conventional extraction techniques, highlighting advantages in extract purity, bioactivity, and environmental impact for pharmaceutical and biomedical applications.
The Role of Modifiers in SFE: Unlocking the Extraction of Polar Bioactives
Defining Modifiers and Cosolvents in a Supercritical CO2 System
In supercritical fluid extraction (SFE), supercritical carbon dioxide (SC-CO2) serves as an excellent solvent for non-polar compounds due to its low polarity [1]. However, its effectiveness diminishes significantly when extracting polar bioactive compounds. Modifiers, also called cosolvents, are small quantities of polar solvents added to SC-CO2 to overcome this limitation [1] [2]. These substances fundamentally alter the solvent properties of SC-CO2, enabling the extraction of a wider range of compounds and optimizing the efficiency of the extraction process within the context of thesis research on modifier concentration optimization.
The primary function of a modifier is to increase the solubility of polar analytes in SC-CO2 and to enhance desorption of matrix-bound compounds [1]. Ethanol, methanol, water, and acetone are commonly used, with ethanol often preferred for food and pharmaceutical applications due to its low toxicity [1] [3]. The modifier choice and concentration are critical parameters that directly impact extraction yield, selectivity, and the bioactive profile of the final extract [4].
Key Concepts and Definitions
What are Modifiers and Cosolvents?
In SFE terminology, modifiers and cosolvents are synonymous, referring to a secondary, typically polar, solvent added in small quantities to a primary supercritical fluid (SC-CO2) to enhance its solvating power [1]. While SC-CO2 is highly effective for lipophilic compounds, its application is limited for more polar molecules. Modifiers address this by:
- Increasing Solvent Polarity: Modifiers like ethanol and methanol increase the overall polarity of the supercritical phase, thereby improving the solubility of mid- to high-polarity compounds [1] [3].
- Improving Mass Transfer: They can interact with the sample matrix, swelling plant tissues or disrupting matrix-analyte interactions (e.g., hydrogen bonding), which facilitates the release of target compounds [1].
- Increasing Density: In some cases, modifiers can slightly increase the density of the supercritical phase, contributing to enhanced solvation power [2].
Mechanism of Action
The enhancement mechanism operates on multiple levels. A modifier like ethanol effectively increases the solubility of polar compounds by providing a more polar environment within the non-polar SC-CO2 [3]. Furthermore, it can act directly on the solid matrix, such as plant material, by swelling the cellular structures and breaking hydrogen bonds that bind target compounds to the matrix, thereby dramatically improving mass transfer and overall yield [1].
Troubleshooting Guide: Common Modifier-Related Issues
Problem: Low Extraction Yield of Polar Compounds
- Description: The target polar bioactive compounds (e.g., certain polyphenols, glycosides) are not being efficiently extracted, even when using a modifier.
- Potential Causes:
- Incorrect modifier type for the target compound polarity.
- Insufficient modifier concentration.
- Inefficient mixing of the modifier with SC-CO2 and the sample matrix.
- Solutions:
- Systematic Solvent Screening: Test a range of modifiers (e.g., ethanol, methanol, ethyl acetate) at 1-5% (v/v) to identify the most effective one for your specific analyte [3].
- Optimize Concentration: Use an experimental design (e.g., Response Surface Methodology) to find the optimal modifier percentage. Excessive amounts can co-extract undesirable impurities [4].
- Ensure Proper Mixing: If using a static modifier addition method, ensure an adequate equilibration time for the modifier to saturate the matrix before starting the dynamic flow.
Problem: Co-extraction of Unwanted Compounds
- Description: The extract is impure, containing high levels of chlorophyll, waxes, or other non-target components alongside the desired compound.
- Potential Causes:
- Modifier concentration is too high, leading to a non-selective extraction.
- The modifier's polarity profile is too broad.
- Solutions:
- Fine-tune Modifier Polarity: Use a binary mixture of modifiers (e.g., ethanol-water) for finer control over selectivity [3].
- Employ Fractional Separation: Couple the extraction with multiple separators in series. The extract is precipitated in stages by sequentially reducing pressure and/or temperature, allowing for the selective collection of different compound classes based on their solubility [1] [5].
Problem: System Blockage or Precipitation
- Description: Solids form within the tubing, valves, or separator, causing pressure drops and blockages.
- Potential Causes:
- The extracted compounds are precipitating due to an overly rapid pressure drop across the restrictor.
- The modifier is not properly mixed, leading to localized concentration gradients.
- Water in the sample or modifier is freezing upon depressurization.
- Solutions:
- Optimize Depressurization: Use a heated restrictor or back-pressure regulator to prevent the freezing of CO2 (dry ice) or the precipitation of solutes [6].
- Pre-dry Materials: Ensure the raw plant material has a low moisture content (typically 8-12%) to prevent water from entering the system and freezing [5].
- Clean Regularly: Perform monthly cleaning of pipes and valves to avoid residue buildup that can nucleate blockages [7].
Frequently Asked Questions (FAQs)
Q1: What is the most common modifier used in SFE for pharmaceutical research? Ethanol is frequently the modifier of choice for pharmaceutical and food-grade applications because it is generally recognized as safe (GRAS), non-toxic, and an effective solvent for a wide range of medium-polarity bioactive compounds [1] [3].
Q2: At what concentration are modifiers typically used? Modifier concentrations typically range from 1% to 15% (v/v) of the total solvent flow, with 5-10% being a common starting point for method development. The optimal concentration is highly dependent on the polarity of the target compound and must be determined experimentally [4] [5].
Q3: How is the modifier introduced into the SFE system? There are two primary methods:
- Static Addition: The modifier is directly mixed with the solid sample in the extraction vessel before the process begins.
- Dynamic Addition: The modifier is pumped and mixed with the SC-CO2 stream continuously throughout the extraction, allowing for more precise control [1].
Q4: Can water be used as a modifier in SC-CO2? Yes, water is used as a modifier, especially for highly polar compounds like sugars or certain glycosides. However, it has low solubility in SC-CO2, so it is often used in combination with a primary modifier like ethanol (e.g., as an ethanol-water mixture). Special care must be taken to prevent ice formation and corrosion [3].
Q5: How does modifier concentration affect the global yield and composition of an extract? Increasing modifier concentration generally increases the global extraction yield by enhancing the solubility of a wider range of compounds. However, it also changes the extract's composition. Higher concentrations can improve the yield of specific polar bioactivities but may also reduce selectivity by co-extracting more impurities. Optimization is required to find a balance [4].
Experimental Protocol: Optimizing Modifier Concentration
Objective
To determine the optimal type and concentration of modifier for the supercritical CO2 extraction of γ-oryzanol from rice bran, maximizing both yield and bioactivity [4].
Materials and Reagents
Table: Research Reagent Solutions and Essential Materials
| Item | Function in the Experiment |
|---|---|
| Rice Bran | The raw plant material containing the target bioactive compounds (γ-oryzanol, fatty acids) [4]. |
| Food-Grade CO₂ (≥99.9%) | The primary supercritical fluid solvent. High purity prevents contamination and system blockages [5]. |
| Anhydrous Ethanol | A GRAS-certified polar modifier used to enhance the solubility of γ-oryzanol in SC-CO₂ [4]. |
| n-Hexane | A conventional organic solvent used for benchmark extractions to compare yield and bioactivity [4]. |
Methodology
- Sample Preparation: Mill the rice bran to a particle size of 40-80 mesh and adjust the moisture content to 8-12% to ensure efficient mass transfer [5].
- Experimental Design: Employ a Central Composite Face-Centered Design (CCFD) to efficiently explore the experimental space. The independent variables are:
- Pressure (X₁): 200 - 500 bar
- Temperature (X₂): 40 - 80 °C
- Ethanol Modifier Concentration (X₃): 0 - 10% (v/v) [4].
- SFE Procedure:
- Load 20g of prepared rice bran mixed with glass beads into the extraction vessel.
- Dynamically pump CO₂ and the specified percentage of ethanol at a constant flow rate (e.g., 15 g/min).
- Maintain extraction for a fixed time (e.g., 3 hours).
- Collect the extract in the separator after depressurization [4].
- Analysis:
- Gravimetric Analysis: Weigh the extract to determine the global yield.
- Chemical Analysis: Use HPLC to quantify the concentration of γ-oryzanol in the extract (mg per gram of extract).
- Bioactivity Assay: Evaluate the cellular antioxidant activity and antiproliferative effect on colorectal cancer cells (e.g., Caco-2 cell line) [4].
Data Analysis and Optimization
- Fit the experimental yield and bioactivity data to a quadratic polynomial model using multiple regression.
- Generate response surface plots to visualize the relationship between variables (pressure, temperature, modifier %) and the responses (yield, γ-oryzanol concentration).
- Identify the optimal set of parameters that simultaneously maximize yield and bioactivity. For example, a study found optimal conditions of 500 bar and 62°C (modifier concentration was also a variable in the model) [4].
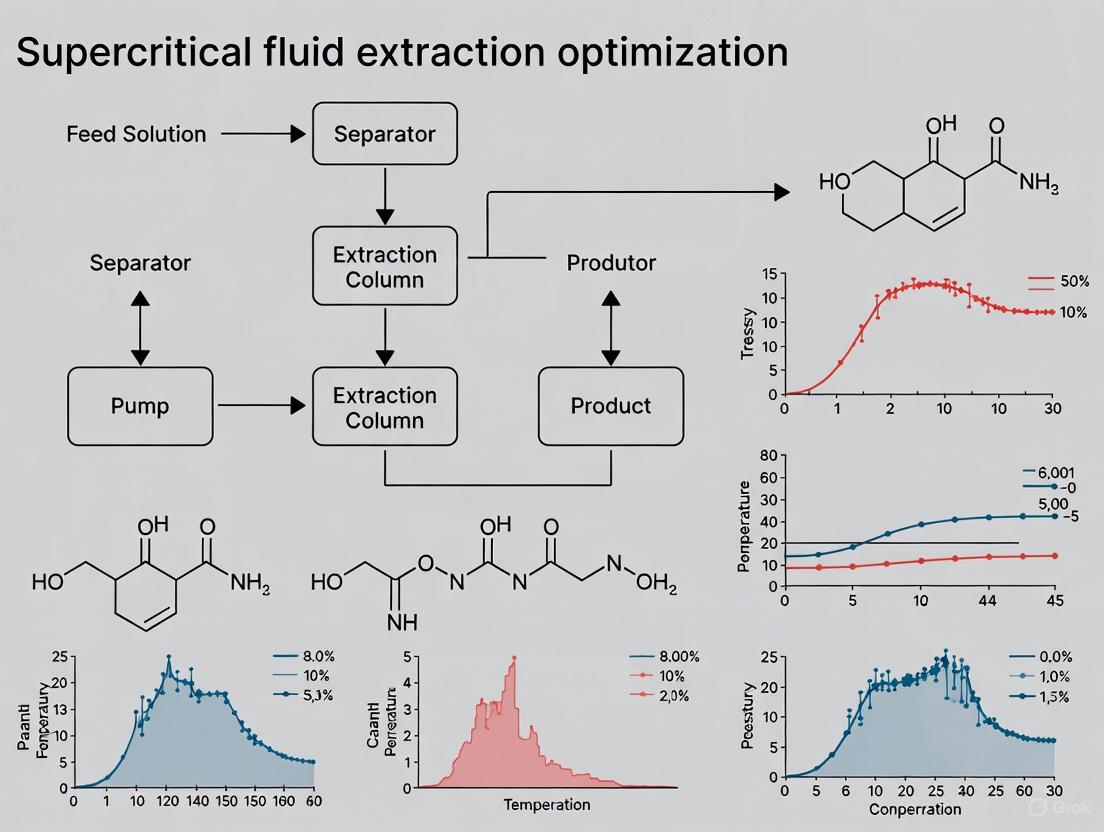
Workflow and Data Visualization
Modifier Optimization Workflow: This flowchart outlines the systematic approach to optimizing modifier use in SFE, from initial goal definition to final protocol validation.
Table: Example of Optimized SFE Conditions for Different Applications
| Application | Target Compound | Optimal Pressure | Optimal Temperature | Optimal Modifier | Key Reference Findings |
|---|---|---|---|---|---|
| Rice Bran | γ-Oryzanol & Fatty Acids | 500 bar | 62 °C | Ethanol (implied) | Achieved 17.3% mass yield; 36.6 mg γ-oryzanol/g extract; superior bioactivity vs. hexane extract [4]. |
| Waste Oil Drilling Mud | Hydrocarbons | 140 bar (~14 MPa) | 45 °C | Not Specified | Achieved 55.46% extraction efficiency; optimized via RSM and kinetic modeling [8]. |
| General Polar Compounds | Various Bioactives | 180 - 300 bar | 40 - 60 °C | Ethanol (5-10%) | Modifier significantly increases yield of polar compounds; required for many phytochemicals [1] [3]. |
Frequently Asked Questions (FAQs)
Q1: What is the primary function of a modifier (co-solvent) in Supercritical Fluid Extraction (SFE)? The primary function of a modifier is to significantly enhance the solubility and alter the selectivity of supercritical CO2 by increasing its polarity. Pure supercritical CO2 is excellent for extracting non-polar compounds (such as oils and fats) but is a poor solvent for more polar molecules like most polyphenols and flavonoids [9]. A modifier like ethanol increases the polarity of the supercritical fluid, thereby improving its ability to interact with, desorb, and dissolve these mid- to high-polarity target analytes from the solid matrix [10].
Q2: How does modifier concentration affect extraction yield and selectivity? Modifier concentration has a direct and often non-linear effect on both yield and selectivity. Increasing the modifier concentration enhances the fluid's polarity, which typically increases the yield of polar compounds up to an optimal point [10]. For instance, in the extraction of flavonoids from hops, the yield increased as ethanol concentration rose to 80%, but beyond this point, the yield dropped, likely because the fluid properties were no longer optimal [10]. Selectivity is controlled because different compound classes have varying polarities; by fine-tuning the modifier concentration, you can tailor the fluid's polarity to be more selective for your target compounds over unwanted matrix interferences [11].
Q3: What are the most common modifiers used in SFE, and why? Ethanol is the most prevalent modifier, especially in food, nutraceutical, and pharmaceutical applications. It is effective, environmentally benign, and has a Generally Recognized As Safe (GRAS) status [10] [12]. Water is also used, sometimes in combination with ethanol, for creating highly polar conditions in techniques like subcritical water extraction [9]. Methanol is effective but is less preferred due to toxicity concerns, restricting its use in products for human consumption [11].
Q4: I am not getting the expected yield of my target compound. Could the modifier concentration be a factor? Yes, an incorrect modifier concentration is a very common factor in low yields. If the concentration is too low, the supercritical CO2 may not have sufficient polarity to efficiently solubilize your target analyte. Conversely, a very high modifier concentration can alter the critical parameters of the fluid mixture excessively and potentially reduce the solubility of your target compound [10]. You should perform a systematic optimization, for example using a central composite design, to determine the optimal modifier percentage for your specific raw material and target compound [13].
Troubleshooting Guides
Problem: Low Extraction Yield of Target Polar Compound
- Potential Cause 1: Insufficient modifier concentration.
- Potential Cause 2: The modifier is not appropriate for the polarity of your target compound.
- Solution: Re-evaluate the chemical nature of your analyte. For very polar compounds (e.g., sugars, glycosides), a mixture of ethanol and water might be necessary instead of pure ethanol [9].
Problem: Poor Extract Selectivity (Co-extraction of Unwanted Compounds)
- Potential Cause: The modifier concentration is too high, making the fluid excessively powerful and non-selective.
- Solution: Reduce the modifier concentration to make the supercritical CO2 more selective. Alternatively, employ a sequential extraction strategy: start with a lower modifier concentration to extract your target compounds, then increase the concentration or change modifiers in a second step to remove more polar interferences [9] [11].
Problem: Inconsistent Extraction Results Between Batches
- Potential Cause: Inaccurate or inconsistent delivery of the modifier into the CO2 stream.
- Solution: Calibrate the pump responsible for delivering the co-solvent. Ensure that the modifier is thoroughly mixed with the CO2 before it enters the extraction vessel. Prepare modifier-CO2 mixtures in a dedicated vessel beforehand if your system allows, to ensure homogeneity [9].
Table 1: Optimized Modifier Conditions for Bioactive Compound Extraction
| Source Material | Target Compound(s) | Optimal Modifier & Concentration | Key Optimized Parameters (P, T) | Extraction Yield / Result |
|---|---|---|---|---|
| Waste Hops [10] | Flavonoids (Xanthohumol) | 80% Ethanol | 25 MPa, 50 °C | 7.8 mg/g |
| Coccomyxa onubensis (Microalga) [14] | Lutein, Phenols, Antioxidants | 50% Ethanol (v/v) | 40 MPa, 70 °C | 66.98% Lutein Recovery |
| Ammodaucus leucotrichus Fruits [12] | Phenolic Compounds | 15% Ethanol | 30 MPa, 70 °C | Optimized for TPC and Anti-Alzheimer activity |
| Grapefruit [13] | Lycopene | 5% Ethanol | 30.5 MPa, 70 °C | Optimized yield via RSM (R² = 0.9885) |
Table 2: Common Modifiers and Their Applications
| Modifier | Polarity | Common Applications | Advantages | Safety & Regulatory Notes |
|---|---|---|---|---|
| Ethanol | Medium-High | Polyphenols, Flavonoids, Carotenoids, Lutein [9] [14] [13] | GRAS status, low toxicity, biodegradable [11] | Preferred for food and pharmaceutical applications. |
| Water | Very High | Polar antioxidants, sugars, phenolics (often as subcritical water) [9] | Non-toxic, inexpensive, can change dielectric constant with temperature. | Can hydrolyze thermally labile compounds at high T [9]. |
| Methanol | High | Various polar analytes (analytical scale) | Strong modifier, good for challenging separations. | Toxic; avoided in consumable product extraction. |
Detailed Experimental Protocols
Protocol 1: Optimizing Modifier Concentration Using a Single-Factor Approach
This protocol is ideal for an initial, straightforward investigation into the effect of modifier concentration.
- Sample Preparation: Pulverize your plant material (e.g., hops, grapefruit peel) to a consistent particle size (e.g., ≤ 0.42 mm or 40-mesh) [10] [13]. For moisture-sensitive compounds, lyophilize the sample before grinding.
- Fixed Parameters: Set your SFE system to constant values for pressure, temperature, and CO2 flow rate based on literature for similar materials. For example, 25 MPa, 50°C, and a constant flow rate [10].
- Variable Parameter (Modifier): Perform a series of extractions where you vary only the concentration of your modifier (e.g., ethanol). A typical range could be 0%, 5%, 10%, 15%, and 20% (v/v or w/w).
- Extraction & Analysis: Run each extraction for a fixed time (e.g., 90 minutes). Collect the extract, and quantitatively analyze it for your target compound(s) using a validated method like HPLC-MS [10] or SFC [13].
- Data Interpretation: Plot the yield of your target compound against the modifier concentration to identify the optimal value.
Protocol 2: Systematic Optimization Using Response Surface Methodology (RSM)
For a more efficient and robust optimization that can model interactions between parameters.
- Experimental Design: Select an appropriate design such as a Box-Behnken Design (BBD) or Central Composite Design (CCD) [12] [14] [13]. These designs typically involve 3-5 levels for each factor.
- Key Factors: The most common factors to investigate are:
- A: Extraction Pressure (e.g., 25, 40, 55 MPa)
- B: Extraction Temperature (e.g., 30, 50, 70 °C)
- C: Modifier (Ethanol) Concentration (e.g., 0, 25, 50% v/v) [14]
- Execution: Conduct the experiments in the randomized order prescribed by the design. The response variable(s) could be global yield, total phenolic content (TPC), or the specific yield of your target molecule.
- Modeling & Optimization: Use statistical software to fit the data to a second-order polynomial model. The software will then generate response surfaces and identify the precise combination of pressure, temperature, and modifier concentration that provides the maximum response [13].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for SFE with Modifiers
| Item | Function / Role in the Experiment | Example from Literature |
|---|---|---|
| Carbon Dioxide (CO₂) | The primary supercritical fluid solvent. Chosen for its mild critical point, non-toxicity, and low cost [9] [15]. | Purity > 99.5% is standard for extraction [13]. |
| Ethanol (Absolute) | The most common GRAS modifier to increase the polarity of scCO₂ for extracting medium-polarity bioactive compounds [9] [10]. | Used at concentrations from 5% to over 80% depending on the target compound's polarity [10] [12] [13]. |
| Water (HPLC Grade) | Used as a co-modifier with ethanol or alone in subcritical water extraction for highly polar compounds [9]. | Applied for the extraction of polyphenols from winery by-products [9]. |
| Analytical Standards | Pure reference compounds for quantifying the target analytes in the extract via chromatographic methods. | e.g., Xanthohumol for hops [10], Lutein for microalgae [14], Lycopene for grapefruit [13]. |
| Solid-Phase Extraction (SPE) Sorbents | For post-extraction clean-up of the extract to remove co-extracted impurities and enhance purity [16]. | Select a sorbent phase (e.g., C18, silica) based on the chemistry of the target analyte. |
Mechanistic Pathways of Modifier Action
The following diagram illustrates the core mechanisms through which a modifier enhances solubility and selectivity in SFE.
Troubleshooting Guides and FAQs
This section addresses common challenges researchers face when using modifiers in Supercritical Fluid Extraction (SFE).
FAQ 1: Why is a co-solvent necessary in supercritical CO2 extraction, and when should I use one?
Supercritical CO2 is inherently non-polar, with dissolving power similar to hexane, making it excellent for non-polar compounds like lipids but poor for polar molecules [17]. A co-solvent, or modifier, is necessary to increase the solvating power of the supercritical fluid for polar compounds [17] [18]. You should employ a co-solvent when your target analytes are polar, such as certain polyphenols, water-soluble vitamins, or specific glycosides [18] [19]. The addition of even a small quantity of a polar co-solvent like ethanol or methanol can significantly enhance the solubilization of these molecules [17].
FAQ 2: My extraction yield for polar compounds is low even with a modifier. What parameters should I check?
Low yields can be attributed to several factors. First, verify the concentration of your co-solvent; it may be insufficient for your specific analyte-matrix combination [18]. Second, review the pressure and temperature settings. While higher pressure generally increases solubility, the effect of temperature is more complex; near the critical point, increased temperature can decrease fluid density and dissolving power, whereas at higher pressures, it typically enhances solubility [20]. Finally, ensure your system is configured for efficient co-solvent introduction, maintaining a constant percentage in the vessel throughout the dynamic extraction phase [17].
FAQ 3: How do I maintain a fixed percentage of co-solvent, like ethanol, in the extraction vessel during dynamic flow?
Maintaining a fixed co-solvent percentage requires calculating the replacement rate. The amount of co-solvent in the system should be based on the vessel volume [17]. For example, for a 100 ml vessel requiring 5% ethanol, you would initially add 5 ml of co-solvent. During dynamic flow, you must replace the co-solvent being flushed out by the CO2. Using a mass flow meter to gauge the CO2 flow rate, you would set your co-solvent pump to add back a volume of ethanol equal to 5% of the CO2 volume flowing out of the vessel [17].
FAQ 4: What are the critical safety and regulatory considerations when selecting a modifier for pharmaceutical applications?
For pharmaceutical applications, regulatory compliance is paramount. Ethanol is generally preferred as it is non-toxic, considered safe (GRAS), and is approved by regulatory bodies like the FDA for use in pharmaceuticals [17] [21]. It leaves no toxic residues, simplifying validation and meeting pharmacopeia standards (e.g., USP, EP) [21]. Methanol, while an effective modifier for polar compounds, is toxic and not suitable for extractions intended for human consumption or therapeutic use [18] [19]. Its use should be restricted to analytical-scale preparations. Water is safe and can assist in swelling the plant matrix to improve recovery, but its low solubility in supercritical CO2 can present technical challenges [18].
FAQ 5: I am experiencing blockages in my restrictor or back-pressure regulator. What is the cause and how can I prevent this?
Blockages are often caused by the adiabatic expansion of CO2, which results in significant cooling (the Joule-Thomson effect) [20]. This can cause extracted water or other compounds to freeze in the restrictor. To prevent this, the restrictor or back-pressure regulator must be heated [20]. For small-scale systems, an electrically heated restrictor or even a hairdryer can be used. For larger systems, ensure that the separator is also adequately heated to compensate for the temperature drop during expansion [20].
Experimental Protocols and Data Presentation
This section provides a detailed methodology for a representative experiment and summarizes quantitative data in a structured format.
Detailed Experimental Protocol: Optimizing γ-Oryzanol Extraction from Rice Bran
The following protocol is adapted from a study optimizing the supercritical fluid extraction of γ-oryzanol from rice bran, demonstrating the application of a co-solvent and a structured experimental design [4].
- 1. Objective: To identify optimal SFE conditions (temperature, pressure, co-solvent) for the selective extraction of γ-oryzanol and fatty acids from rice bran, maximizing yield and bioactivity [4].
- 2. Materials and Equipment:
- Raw Material: Rice bran (e.g., Japônica variety). Pre-treat by drying and milling to a specific particle size [4].
- Solvents: Supercritical CO2 (99.95% purity); Food-grade ethanol (co-solvent) [4].
- Equipment: SFE system (e.g., 500 mL extraction cell), CO2 pump with chiller, co-solvent pump, pressure vessels, separators, back-pressure regulator, and heating ovens [4] [17].
- 3. Experimental Design:
- A Central Composite Face-Centered Design (CCF) is recommended to optimize multiple parameters efficiently [4].
- Independent Variables:
- Temperature: 40°C to 80°C
- Pressure: 200 bar to 500 bar
- Co-solvent (Ethanol) Concentration: 0-10% (v/v)
- Dependent Variables (Responses): Global yield (%), γ-oryzanol concentration (mg/g extract), fatty acid concentration (mg/g extract) [4].
- 4. Procedure:
- Sample Preparation: Load 20g of rice bran mixed with glass beads (to reduce dead volume) into the extraction vessel [4].
- System Pressurization: Pressurize the vessel with CO2 to the desired pressure using an automated back-pressure regulator. The liquid CO2 is pumped at a rate of 15 g/min [4].
- Co-solvent Addition: Use a co-solvent pump to introduce the predetermined percentage of ethanol into the system. This can be done by pre-doping the vessel or by simultaneously pumping CO2 and ethanol at a set ratio [17].
- Static Extraction (Optional): Close the outlet valve and allow the mixture to equilibrate for a set time (e.g., 15-30 minutes) to enable solvent penetration [1].
- Dynamic Extraction: Open the outlet valve and perform continuous extraction for a fixed time (e.g., 3 hours). The CO2/co-solvent mixture passes through the matrix, dissolving the target compounds, and is expanded into a fraction collector [4].
- Separation and Collection: The extract is collected in an ice-cooled separator where the pressure reduction causes the compounds to precipitate. The CO2 is vented or recycled [20].
- Analysis: Weigh the extract to determine mass yield. Analyze the composition using HPLC or GC-MS to quantify γ-oryzanol and fatty acids [4].
The workflow for this experiment is outlined below.
The table below consolidates data on the performance of different modifiers in various SFE applications, highlighting their impact on yield and selectivity.
Table 1: Performance of Common Modifiers in Supercritical Fluid Extraction
| Modifier | Typical Concentration Range | Target Compounds / Application | Reported Impact on Yield / Performance | Key Advantages & Limitations |
|---|---|---|---|---|
| Ethanol | 5 - 15% (v/v) [18] | Polar compounds (polyphenols, flavonoids), cannabinoids, chiral separations [18] [21] [22]. | Increased recovery of polar mycotoxins (97-100%) at 80°C [19]. In rice bran SFE, led to extracts with superior anticancer activity (EC~50~ = 0.9 mg/mL) vs. hexane extract [4]. | GRAS, non-toxic, pharma-approved. Ideal for food and drug applications. Less polar than methanol, so may be less effective for some highly polar analytes [17] [21]. |
| Methanol | 1 - 10% (v/v) [19] | Highly polar molecules (e.g., mycotoxins, specific glycosides) in analytical chemistry [19]. | Proven highly effective as a modifier for the extraction of polar macrocyclic lactone mycotoxins from food matrices [19]. | Highly effective for polar compounds. Toxic. Not suitable for extractions intended for human consumption; primarily for analytical scale [18] [19]. |
| Water | Low percentages (often with ethanol) [18] | Used to swell the plant matrix; can enhance extraction of water-soluble compounds [18]. | – | |
| Data on pure water as a modifier in scCO2 is less commonly quantified in isolation. | Safe, non-toxic. Limited solubility in supercritical CO2, can cause ice blockages in restrictors if not properly managed [18] [20]. |
The Scientist's Toolkit: Essential Research Reagent Solutions
This table details key reagents and materials essential for conducting SFE experiments with modifiers.
Table 2: Essential Research Reagents and Materials for SFE with Modifiers
| Item | Function / Purpose | Technical Specifications & Considerations |
|---|---|---|
| Supercritical CO2 | Primary extraction solvent. | High purity (99.95%), supplied in cylinders. Requires a chiller to maintain liquid state before pumping [17] [1]. |
| Pharmaceutical-Grade Ethanol | GRAS co-solvent for polar compound extraction. | Anhydrous or high-purity (e.g., 95-99%) to avoid water introduction. Preferred for pharmaceutical and nutraceutical applications [17] [21]. |
| HPLC-Grade Methanol | Potent polar co-solvent for analytical applications. | High purity. Use is restricted due to toxicity; not for extractions intended for human consumption [18] [19]. |
| Co-solvent Pump | Precisely delivers modifier into the SFE system. | Typically an HPLC-type pump capable of handling the system pressure. Must be calibrated to maintain a fixed ratio with CO2 flow [17]. |
| Back-Pressure Regulator | Maintains system pressure above the critical point. | Can be mechanical or electronic. Requires heating to prevent freezing and blockages during depressurization [17] [20]. |
| Extraction Vessels | Contain the solid sample during extraction. | ASME-certified pressure vessels ranging from 5 mL to 20+ L. Material (e.g., stainless steel) must be compatible with CO2 and modifiers [17]. |
The decision-making process for selecting an appropriate modifier is summarized in the following diagram.
In supercritical fluid extraction (SFE), carbon dioxide (SC-CO2) is the most widely used solvent due to its low critical temperature (31°C), low critical pressure (74 bar), low toxicity, and low cost [15] [1]. However, a significant limitation of pure SC-CO2 is its non-polar nature, which makes it a poor solvent for polar organic compounds [23] [1]. The introduction of polar modifiers, also called co-solvents, such as ethanol, methanol, or water, is a critical strategy to overcome this limitation. These modifiers interact with the supercritical CO2, fundamentally altering its polarity and solvent strength, thereby dramatically expanding its application range to include polar bioactive compounds like flavonoids and polyphenols [23] [10] [1]. Understanding these interaction effects is essential for optimizing SFE processes, particularly in pharmaceutical and natural product research where the extraction of polar molecules is often required. This guide addresses the most frequent questions and troubleshooting challenges researchers face when working with modifiers.
Frequently Asked Questions (FAQs)
1. How does a polar modifier actually enhance the solvent power of SC-CO2? A polar modifier enhances SC-CO2 solvent power through two primary mechanisms. First, it directly increases the polarity of the supercritical fluid mixture, thereby improving the solubility of polar target analytes. This occurs because the modifier alters the overall solubility parameter of the mixed fluid [23]. The solubility parameter (δ) for the SC-CO2/modifier mixture is calculated as δmix = φCO2δCO2 + φmδm, where φ is the volume fraction and δm is the solubility parameter of the modifier [23]. Second, the modifier can interact with the sample matrix itself, promoting faster desorption of analytes from active sites, covering matrix active sites to prevent re-adsorption, and improving the accessibility of the fluid to the matrix [23] [24].
2. Do modifiers change the density of SC-CO2, and how does this affect extraction? While modifiers directly alter the polarity of the fluid, their effect on density is more complex and is intertwined with the effects of temperature and pressure. The density of a supercritical fluid is a key determinant of its solvating power and is highly responsive to changes in pressure and temperature [23] [1]. The addition of a modifier can influence the critical parameters of the mixture. The primary role of a modifier is not to increase density, but to introduce polar interactions (e.g., hydrogen bonding) that pure CO2 lacks. However, the overall extraction efficiency is a result of the combined effect of the fluid's density and its altered chemical nature due to the modifier [10].
3. What is the typical range for modifier concentration, and what are the trade-offs? Modifier concentrations typically range from 1% to 30% (by volume or mass) of the total solvent flow, though most applications use 5% to 15% [23] [25] [10]. The optimal concentration involves a trade-off:
- Low Concentration (<5%): May provide insufficient polarity enhancement for very polar compounds.
- High Concentration (>15-30%): Can excessively alter the critical parameters of the mixture, potentially moving the fluid out of its supercritical state and reducing the beneficial transport properties (e.g., high diffusivity, low viscosity) that make SFE attractive [10]. It can also make the final solvent removal more difficult if food-grade or low-toxicity solvents like ethanol are not a requirement.
4. Which modifier should I choose for my specific application? The choice of modifier depends on the polarity of your target compound and the requirements for your final product (e.g., food-grade). The table below summarizes common modifiers and their applications.
Table 1: Common Modifiers and Their Typical Applications in SFE
| Modifier | Polarity | Common Applications | Notes |
|---|---|---|---|
| Methanol | High | High-efficiency extraction of polar templates; analytical applications [23] | Can be too toxic for food/pharma products. |
| Ethanol | Moderate-High | Flavonoids, polar lipids, food-grade and pharmaceutical extractions [25] [10] [4] | Preferred for its low toxicity and GRAS status. |
| Water | High | Polar compounds; often used in mixed modifiers [23] [25] | Can cause pore collapse in some matrices; requires careful control [23]. |
| Formic/Acetic Acid | High | Acidic compounds; can protonate basic analytes to improve solubility [23] | Useful for specific pH-controlled extractions. |
Troubleshooting Guides
Problem: Low Extraction Yield for Polar Compounds
Possible Causes and Solutions:
- Insufficient Modifier Polarity or Concentration:
- Solution: Increase the concentration of your modifier in a stepwise manner (e.g., 5%, 10%, 15%). If using ethanol is ineffective, consider a more polar modifier like methanol for analytical applications, or a mixed ethanol-water modifier [25] [10].
- Experimental Protocol: Perform a series of extractions on your matrix where pressure and temperature are held constant, but the modifier percentage is varied. Analyze the yield to determine the optimal concentration.
- Suboptimal Solubility Parameters:
- Solution: Adjust the pressure and temperature to tune the density and solubility parameter of the SC-CO2/modifier mixture. Higher pressures generally increase density and solvent strength [23] [10].
- Experimental Protocol: Use a statistical experimental design (e.g., Box-Behnken or Central Composite Design) to simultaneously optimize pressure, temperature, and modifier concentration. The response can be the yield of your target compound [25] [4] [8].
Problem: Poor Selectivity or Co-extraction of Undesired Compounds
Possible Causes and Solutions:
- Excessively Strong Solvation Power:
- Solution: Reduce the modifier concentration or use a less polar modifier. Alternatively, implement a fractional extraction strategy by starting with pure CO2 at lower pressures to extract non-polar compounds (e.g., lipids), then introduce the modifier at higher pressures to extract the polar target compounds [20].
- Experimental Protocol: Conduct a stepwise extraction. First, use pure SC-CO2 at 100-200 bar. Then, sequentially increase pressure and add modifier, collecting extracts separately at each step for analysis [4].
- Fluid Density Too High:
- Solution: For selectivity, slightly reducing the pressure can decrease the fluid density, making the solvent less powerful and more selective. The effect of temperature is complex and should be experimentally verified [20].
Problem: Inconsistent Results or Extraction Efficiency
Possible Causes and Solutions:
- Improper Mixing of Modifier with SC-CO2:
- Solution: Ensure your SFE system is properly configured for modifier addition. The modifier should be mixed thoroughly with the CO2 before it enters the extraction vessel. Using a dedicated co-solvent pump is recommended for consistent and precise mixing.
- Experimental Protocol: Visually inspect the mixing point if possible. Systematically check the flow rates of both CO2 and the modifier pump for stability.
- Inadequate Extraction Time in Solubility-Limited Regime:
- Solution: If the extraction is limited by the solubility of the analyte in the fluid (rather than diffusion inside the particle), increasing the flow rate or the static extraction time may be necessary [20].
- Experimental Protocol: Generate an extraction curve (yield vs. time) for your specific matrix. If the curve is linear, the process is solubility-limited, and higher flow rates are needed. If it is a steep curve that quickly plateaus, the process is diffusion-limited [20].
Experimental Protocols & Data
Example Protocol: Optimizing Flavonoid Extraction from Plant Material
This protocol is adapted from research on extracting polar flavonoids from various plant matrices [25] [10].
- Sample Preparation: Dry and mill the plant material to a consistent particle size (e.g., 40-mesh sieve, Dp ≤ 0.42 mm) to ensure uniform extraction [10].
- Extraction Setup: Load the extraction vessel with the sample mixed with an inert material like glass beads to reduce dead volume [4].
- Initial Conditions: Set the extractor to a moderate pressure and temperature (e.g., 20 MPa, 50°C) with a CO2 flow rate of 15 g/min [4].
- Modifier Introduction: Introduce a food-grade modifier like ethanol or an ethanol-water mixture (e.g., 80:20) at a concentration of 10-20% of the total solvent flow [25] [10].
- Dynamic Extraction: Perform dynamic extraction for a predetermined time (e.g., 30-90 minutes) [25] [10].
- Collection: Depressurize the fluid stream into a collection vessel containing a solvent like ethanol or methanol to trap the extracted analytes [20].
- Analysis: Quantify the yield of your target compounds using a suitable analytical method such as UHPLC-DAD or HPLC-MS [25] [10].
Quantitative Data on Modifier Performance
The following table summarizes experimental data from the literature demonstrating the effect of different modifiers on extraction efficiency.
Table 2: Effect of Modifiers on Extraction Efficiency in Various Applications
| Source Material | Target Compound | Optimal Modifier / Conditions | Extraction Efficiency / Yield | Reference |
|---|---|---|---|---|
| Hexagonal Mesoporous Silica (HMS) | Dodecylamine (template) | Methanol, Formic Acid, Acetic Acid, Water (at 65°C, 15.0 MPa) | >85% amine removal | [23] |
| Waste Hops (SC-CO2 extracted) | Flavonoids (Xanthohumol) | 80% Ethanol, 50°C, 25 MPa, 50% solvent-to-material ratio | 7.8 mg/g | [10] |
| Robinia pseudoacacia Heartwood | Flavonoids (Dihydrorobinetin & Robinetin) | CO2:EtOH:H2O (80:16:4), 80°C, 10 MPa | 49.2 mg/g of dried wood | [25] |
| Rice Bran | γ-Oryzanol & Fatty Acids | Pure CO2, 62°C, 50 MPa | 36.6 mg γ-oryzanol / g extract | [4] |
Visualizing Modifier Interactions and Experimental Workflows
Diagram: How a Modifier Alters SC-CO2 Polarity
The following diagram illustrates the mechanism by which a polar modifier, such as ethanol, enhances the extraction of a polar target molecule (e.g., a flavonoid) from a solid plant matrix.
Diagram: SFE Experimental Workflow with Modifier
This flowchart outlines the key stages of a supercritical fluid extraction process that includes the use of a modifier.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for SFE with Modifiers
| Item | Function / Purpose | Example Specifications / Notes |
|---|---|---|
| Carbon Dioxide (CO2) | Primary supercritical solvent. | High purity grade (e.g., 99.95%). Supplied as a liquid in a cylinder with a dip tube [10] [4]. |
| Polar Modifiers | Enhance polarity and solvation power of SC-CO2. | Ethanol: Food-grade, preferred for nutraceuticals. Methanol: High efficiency for analytical work. Water: Used in mixed modifiers [23] [25] [10]. |
| Collection Solvent | Traps the analyte after depressurization. | A solvent like methanol or ethanol in which the target analytes are highly soluble [20]. |
| Inert Packing Material | Reduces dead volume in the extraction vessel. | Glass beads (e.g., 2-3 mm diameter) [4]. |
| Standard Reference Materials | For quantification and method validation. | Pure analytical standards of the target compounds (e.g., xanthohumol, γ-oryzanol, robinetin) [10] [4]. |
Methodological Strategies for Modifier Optimization and Application
In supercritical fluid extraction (SFE), particularly when using supercritical CO₂ (scCO₂), the addition of polar modifiers (co-solvents) is often essential for efficient extraction of moderately polar to polar bioactive compounds. scCO₂ alone, with solvating power similar to hexane, is excellent for non-polar compounds but has limited effectiveness for polar molecules. Modifiers like ethanol, methanol, or water enhance the solvating power of scCO₂ for a wider range of compounds. The method of introducing these modifiers significantly impacts extraction efficiency, reproducibility, and process control. The two primary methods for introducing modifiers are Direct Mixing (also called static or co-solvent doping) and In-Line Addition (also called dynamic addition). This guide details both methods within the context of thesis research focused on optimizing modifier concentration.
FAQs & Troubleshooting Guides
Basic Principles and Definitions
Q1: What is a modifier in SFE and why is it used? A modifier, or co-solvent, is a small amount of a polar solvent added to the main supercritical fluid (typically CO₂) to increase its solvating power for polar compounds. Neat scCO₂ has dissolving properties similar to hexane, making it ideal for non-polar materials. The addition of even a small quantity of a co-solvent like ethanol or methanol enhances its ability to dissolve much more polar molecules [17] [26]. This is crucial for efficiently extracting a broad spectrum of bioactive compounds from plant and other biomass materials.
Q2: What are the most common modifiers used in SFE? The most common modifiers are ethanol, methanol, and water. Ethanol is often preferred for food and pharmaceutical applications because it is inexpensive, available in high purity, FDA-approved, and generally regarded as safe (GRAS) [17] [26].
Method-Specific Questions
Direct Mixing (Static Addition)
Q3: What is the Direct Mixing method? In the Direct Mixing method, the calculated volume of co-solvent is added directly to the extraction vessel containing the sample before the vessel is sealed and pressurized with CO₂ [17]. The modifier is statically mixed with the sample matrix at the start of the extraction process.
Q4: When should I choose the Direct Mixing method? This method is often used for smaller-scale extractions, when the sample has a high affinity for the modifier, or for simpler system setups. It is a straightforward approach that does not require an additional pump for the modifier.
Q5: What is a key limitation of the Direct Mixing method? The main limitation is the inability to maintain a precise, constant modifier concentration throughout the entire dynamic extraction phase. As fresh CO₂ flows through the vessel, it continuously dilutes and removes the pre-loaded modifier, leading to a decreasing modifier concentration over time [17]. This can result in inconsistent extraction efficiency, especially for longer extraction runs.
In-Line Addition (Dynamic Addition)
Q6: What is the In-Line Addition method? In the In-Line Addition method, the modifier is pumped directly into the CO₂ stream using a dedicated pump (e.g., an HPLC-type pump) continuously and dynamically throughout the extraction process. The CO₂ and modifier are mixed in a line before entering the extraction vessel [17].
Q7: What are the primary advantages of the In-Line Addition method? The key advantage is the ability to maintain a precise and constant modifier-to-CO₂ ratio throughout the entire dynamic extraction process. This leads to superior reproducibility, better process control, and is essential for method scalability and optimization studies [17].
Q8: My In-Line Addition system seems to have fluctuating pressure. What could be wrong? Ensure that the CO₂ pump and the co-solvent pump are both actuated simultaneously with the restrictor valve open. The pump speeds are designed to maintain the system's pressure set point by adjusting to the flow set by the restrictor. Think of the restrictor as a back-pressure regulator; as you adjust it, the pumps will respond to maintain the desired pressure [17] [26].
Concentration and Optimization
Q9: How do I calculate and maintain the modifier concentration in the vessel for the In-Line method? Concentration is based on the volumetric flow rates of CO₂ and the modifier. To maintain a fixed percentage (e.g., 5%) of ethanol during dynamic flow, you must replace the ethanol flushed out by the CO₂. Using a mass flow meter for CO₂, calculate 5% of the CO₂ volume flowing out of the vessel and set the co-solvent pump to add that equivalent volume of ethanol back into the system [17].
Q10: What are typical optimal modifier concentrations reported in recent research? Optimal concentrations are highly dependent on the target compound and plant matrix. Recent studies show a range of effective concentrations:
- 10% ethanol was optimal for enhancing phenolic compound recovery in Hemp Seed Oil, significantly increasing yield and bioactive content [27].
- 15% ethanol was identified as optimal for the supercritical fluid extraction of bioactive compounds from Ammodaucus leucotrichus fruits [12].
- Ethanol volumes as a modifier have been used in a range from 150 μL (spiked directly onto the sample) to much higher volumes for different applications [28].
Table 1: Exemplary Modifier Concentrations from Recent SFE Studies
| Source Material | Target Compound(s) | Optimal Modifier & Concentration | Key Outcome |
|---|---|---|---|
| Hemp Seed [27] | Phenolic compounds, Tocopherols | 10% Ethanol | Increased oil yield, TPC, and oxidative stability. |
| Ammodaucus leucotrichus Fruits [12] | Total Phenolic Content, Anti-Alzheimer activity | 15% Ethanol | Identified as part of optimal conditions (300 bar, 70°C). |
| Angelica sinensis [29] | Coniferyl ferulate | Ethyl Acetate (60 mL volume) | Selected as the most effective modifier for this specific compound. |
| Flixweed Seed [28] | Essential Oil | Methanol (150 μL volume) | Used in the optimized method for highest yield. |
Troubleshooting Common Problems
Q11: I am getting low yields of my target polar compound even with a modifier. What should I check?
- Verify the modifier type: Ensure the polarity of your modifier matches your target compound. For very polar compounds, you may need to test different modifiers (e.g., ethanol vs. methanol vs. water mixtures).
- Check the introduction method: If using Direct Mixing, the modifier concentration may be dropping too rapidly. Switch to In-Line Addition to maintain a constant concentration.
- Confirm system temperature: Ensure you are using a pre-heater for the CO₂-modifier fluid. Without a pre-heater, especially at high flow rates, the system may not hold temperature accurately, affecting solubility and efficiency [17] [26].
Q12: Why is a pre-heater recommended for the fluid stream? Regardless of vessel size, heating efficiency through vessel band heaters alone is limited. A liquid CO₂ pre-heater is used to regulate the temperature of the carbon dioxide and co-solvent before they reach the main sample vessel. This is critical for maintaining accurate and reproducible extraction temperatures, particularly during dynamic flow, and is highly recommended for all efficient extraction work [17] [26].
Experimental Protocols for Method Comparison
Protocol 1: Direct Mixing (Static) Method
This protocol is adapted from methods used in the optimization of SFE for essential oils from Flixweed seed [28].
- Sample Preparation: Grind the plant material (e.g., Flixweed seed) to a consistent particle size (e.g., 0.5 mm mean diameter). Load a known mass (e.g., 0.5 g) mixed with inert glass beads into the extraction vessel.
- Modifier Addition: Spike the calculated volume of modifier (e.g., 150 μL of methanol) directly onto the sample in the extraction vessel.
- System Assembly: Attach the extraction vessel to the SFE system.
- Pressurization and Extraction: Pump liquid CO₂ into the vessel to pressurize it to the desired set point. The pump will shut off if no flow out of the vessel occurs (static period). For dynamic extraction, open the restrictor valve. The pump will actuate to maintain pressure as the mixture of CO₂, modifier, and dissolved analytes flows out of the vessel for collection.
- Collection: Collect the extract from the restrictor outlet in a collection vial containing a solvent like n-hexane, placed in an ice bath to improve recovery.
Protocol 2: In-Line Addition (Dynamic) Method
This protocol is based on technical descriptions of SFE systems and the optimization of phenolic extraction from hemp seed [17] [27].
- System Setup: Ensure the SFE system is equipped with a dedicated co-solvent pump (e.g., an HPLC-type pump). The CO₂ line and co-solvent line should meet at a mixing point before entering the pre-heater and extraction vessel.
- Sample Loading: Load the prepared biomass into the extraction vessel as in Protocol 1.
- Parameter Setting: Set the desired temperature, pressure, CO₂ flow rate (e.g., 15 g/min [4] or 0.25 kg/h [27]), and the modifier flow rate to achieve the target concentration (e.g., 10% ethanol [27]).
- Dynamic Extraction: Actuate the CO₂ pump and the co-solvent pump simultaneously with the restrictor valve open. The pumps will work together to maintain the system pressure and the precise modifier-to-CO₂ ratio throughout the entire extraction time (e.g., 3 hours [4]).
- Collection and Analysis: Collect the extract in a suitable container. The extract can then be analyzed gravimetrically for yield and using techniques like GC-MS or HPLC-MS for chemical profiling [12] [27].
Workflow and Logical Diagrams
The diagram below illustrates the logical decision-making process for selecting and implementing co-solvent introduction methods in SFE.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for SFE Modifier Optimization
| Item | Function/Application | Key Considerations |
|---|---|---|
| Carbon Dioxide (CO₂) | The primary supercritical fluid solvent. | Must be high purity (e.g., 99.95% [4] or 99.99% [28]). GRAS status makes it safe for food/pharma [30] [17]. |
| Ethanol | A polar co-solvent, preferred for food, cosmetic, and pharmaceutical applications. | GRAS status [17]. Effective for increasing solubility of phenolic compounds [27]. |
| Methanol | A polar co-solvent. | Stronger solvent strength than ethanol, but more toxic. Often used in analytical-scale extractions [28]. |
| Water | A polar co-solvent. | Used to modify the polarity of scCO₂, sometimes in combination with other solvents. |
| Ethyl Acetate | An organic co-solvent. | Can be selected for specific applications where it shows higher extraction efficiency for target compounds, such as coniferyl ferulate [29]. |
| Diatomaceous Earth | A solid support for samples in some extraction protocols. | Used to create a better interface for extraction and can help prevent channeling [31]. |
In supercritical fluid extraction (SFE), achieving optimal conditions is crucial for maximizing yield, purity, and efficiency of bioactive compounds from natural sources. The "black box" nature of SFE, where numerous factors interact in complex ways, makes systematic optimization essential [32]. Response Surface Methodology (RSM) provides a collection of mathematical and statistical techniques for experimental optimization where the functional form of the objective function is unknown [33]. Within RSM, designs like Central Composite Design (CCD) and Box-Behnken Design (BBD) are specifically employed to efficiently navigate the factor space, model quadratic relationships, and locate optimal process parameters [34] [35]. This technical guide addresses common challenges researchers face when applying these designs to optimize parameters such as modifier concentration in SFE processes.
Frequently Asked Questions (FAQs)
Q1: What is the fundamental difference between screening and optimization designs, and when should I use each?
- Screening Designs are used in the initial phase of experimentation to identify the most influential factors from a large set of potential variables. They are efficient for examining qualitative, quantitative, and mixer-related factors simultaneously with fewer runs. Common screening designs include Full Factorial, Fractional Factorial, and Plackett-Burman designs [32].
- Optimization Designs are employed after key factors are identified to determine their optimal settings. These designs can model curvature and interaction effects to find a precise optimum. CCD and BBD are the most common optimization designs for SFE processes [32].
- When to Use: Always start with a screening design when dealing with many factors (e.g., >5) to avoid unnecessary experimentation. Proceed with an optimization design like CCD or BBD once you have narrowed down the critical few factors (typically 3-5) [32].
Q2: How do I choose between Central Composite Design (CCD) and Box-Behnken Design (BBD)?
Your choice depends on the experimental constraints and the region of interest you wish to explore. The table below summarizes the key differences:
Table: Comparison of Central Composite Design (CCD) and Box-Behnken Design (BBD)
| Feature | Central Composite Design (CCD) | Box-Behnken Design (BBD) |
|---|---|---|
| Basic Structure | Combines a factorial/fractional factorial design with axial ("star") points and center points [34]. | An incomplete factorial design formed by combining two-level factorial designs with incomplete block designs [35]. |
| Factor Levels | Typically requires 5 levels for each factor (for a circumscribed CCD) [34]. | Requires only 3 levels for each factor [35]. |
| Region of Exploration | Explores a larger area, including extreme conditions (especially the Circumscribed type) [34]. | Explores a spherical region within the factor space, never including the corner (extreme) points [35]. |
| Runs/Experiments | Number of runs = 2^(k) + 2k + cp (for a full factorial core), where k is factors and cp is center points [34]. | Generally fewer runs than CCD for 3-6 factors; the number of runs increases as shown in the BBD table [35]. |
| Best Use Case | When you need to fit a precise quadratic model and are interested in exploring a wider process space, including extremes. | When you want to avoid extreme factor combinations due to practical or safety constraints, or when working within a smaller, spherical region of interest [35]. |
Q3: What are the key steps in implementing an RSM-based optimization strategy for SFE?
A standard RSM algorithm involves the following steps [33]:
- Fit a First-Order Model: Conduct a designed experiment (e.g., a factorial design) in a small sub-region and fit a linear model to the data.
- Follow the Path of Steepest Descent: Use the linear model to determine the direction that improves the response most rapidly. Conduct experiments along this path until the response no longer improves.
- Fit a Second-Order Model: Once curvature is detected or near the optimum, a more elaborate experiment (like a CCD or BBD) is conducted to fit a quadratic model.
- Canonical Analysis: Locate the stationary point (maximum, minimum, or saddle point) of the fitted response surface and characterize the response.
Q4: In the context of SFE, what specific parameters do CCD and BBD typically optimize?
These designs are used to model and optimize critical SFE parameters, including:
- Pressure and Temperature, which directly affect the density and solvating power of the supercritical fluid [28] [12].
- Modifier (Co-solvent) Concentration, which enhances the solubility of target compounds (e.g., using methanol or ethanol) [28] [12].
- Extraction Time (static and dynamic), which influences the process kinetics and overall yield [28].
- CO₂ Flow Rate, though this is sometimes fixed in some experimental setups.
Troubleshooting Guides
Problem 1: Poor Model Fit or Low Predictive Power
Symptoms: Low R² (coefficient of determination) value, large difference between R² and adjusted R², poor performance of validation experiments.
Possible Causes and Solutions:
- Cause: Important factors or interactions were omitted from the model.
- Solution: If you started directly with an optimization design, go back and perform a screening design to ensure all significant factors are included [32].
- Cause: The experimental region is too large, and the true response surface is highly complex and non-quadratic.
- Solution: Reduce the region of interest and re-run the experimental design in a smaller area where a quadratic model might be more appropriate.
- Cause: Insufficient replication leading to a poor estimate of pure error.
Problem 2: Inability to Reach a Clear Optimum
Symptoms: The optimization algorithm suggests moving to the edge of the experimental region, or the canonical analysis reveals a saddle point instead of a maximum/minimum.
Possible Causes and Solutions:
- Cause: The true optimum lies outside the current experimental region.
- Solution: Expand the experimental region and add new axial points (in the case of CCD) or conduct a new design centered on the current best point. A standard RSM approach involves iterating steps of linear modeling and steepest descent until curvature is detected [33].
- Cause: The path of steepest descent is prone to zigzagging, slowing convergence.
- Solution: Consider advanced RSM algorithms that use gradient deflection methods (e.g., conjugate gradient methods). These methods utilize information from previous iterations to build knowledge of the curvature, leading to faster and more stable convergence [33].
Problem 3: Practical Difficulties with Factor Levels in the Design
Symptoms: The design suggests running experiments at factor levels that are practically impossible, unsafe, or too costly (e.g., extremely high pressure or modifier volume).
Possible Causes and Solutions:
- Cause: Using a Circumscribed CCD (CCC) which places star points outside the factorial cube.
- Solution: Use a Face-Centered CCD (CCF), where the star points are placed at the center of each face of the factorial space (α = ±1). This requires only 3 levels per factor and keeps all experiments within the original factor range [34]. Alternatively, switch to a Box-Behnken Design, which inherently avoids corner points and works within a spherical region [35].
Key Research Reagent Solutions for SFE Optimization
This table details essential materials and reagents commonly used in SFE experiments, particularly those focused on optimizing modifier concentration for bioactive compound extraction.
Table: Essential Research Reagents and Materials for SFE Experiments
| Reagent/Material | Function in SFE Experiment | Example from Literature |
|---|---|---|
| Supercritical CO₂ | Primary extraction solvent. It is non-toxic, non-flammable, cost-efficient, and has tunable solvating power by changing pressure and temperature [28]. | Used as the main solvent for extracting essential oils and fatty acids from Flixweed seed [28] and bioactive molecules from Ammodaucus leucotrichus seeds [12]. |
| Methanol | Polar modifier (co-solvent) added to supercritical CO₂ to enhance the solubility of medium-polarity to polar compounds. | 150 μL was used as a modifier to optimize the extraction yield from Descurainia Sophia L. seed [28]. |
| Ethanol | A "green" and GRAS (Generally Recognized As Safe) polar modifier. Often preferred for extractions intended for food or pharmaceutical applications. | Optimized at 15% as a co-solvent for the SFE of bioactive compounds from Ammodaucus leucotrichus fruits [12]. |
| n-Hexane | Organic solvent used to collect the extract exiting the SFE system. It traps the analytes as the CO₂ expands through the restrictor. | Used as a collection solvent for Flixweed seed extracts in a 3-mL volumetric flask placed in an ice bath [28]. |
| Plant Material (e.g., Seeds) | The sample matrix containing the target bioactive compounds. Particle size and moisture content are critical parameters. | Descurainia Sophia L. seeds were ground to a mean diameter of 0.5 mm [28]. Ammodaucus leucotrichus fruits were also used [12]. |
Experimental Protocol: Optimizing Modifier Concentration using a Box-Behnken Design
This protocol outlines the specific methodology for applying a BBD to optimize modifier concentration, based on a published study [12].
1. Experimental Setup and Design:
- Apparatus: Use a laboratory-scale SFE system equipped with a CO₂ pump, a co-solvent modifier pump, a pressurized extraction vessel, temperature control, and a back-pressure regulator.
- Sample Preparation: Clean and dry the plant material (e.g., Ammodaucus leucotrichus seeds). Grind to a homogeneous particle size (e.g., 0.5 mm mean diameter) to ensure consistent extraction [28].
- Factor Selection: Based on prior screening, select three critical factors for optimization. For example:
- A: Pressure (e.g., 200 - 400 bar)
- B: Temperature (e.g., 40 - 70 °C)
- C: Modifier (Ethanol) Concentration (e.g., 5 - 15 % v/v)
- Design Generation: Use statistical software to generate a 3-factor BBD. This design will typically require 15 experiments, including 12 non-center points and 3 center points [35] [12].
2. Execution and Analysis:
- Run Experiments: Perform the extractions in a randomized order to minimize the effects of uncontrolled variables. Keep constant other parameters like CO₂ flow rate, extraction time (static and dynamic), and sample load.
- Response Measurement: For each run, gravimetrically determine the extraction yield. Further analyze the extracts for Total Phenolic Content (TPC) using the Folin-Ciocalteu method and antioxidant activity (e.g., DPPH assay) [12].
- Model Fitting and Optimization: Input the response data into the software to fit a second-order quadratic model. Perform ANOVA to check the model's significance and lack-of-fit. Use the model's desirability function to identify the optimal combination of pressure, temperature, and modifier concentration that maximizes your desired responses [12].
Workflow and Relationship Diagrams
Diagram 1: Overall Workflow for RSM-Based SFE Optimization
Diagram 2: The Role of a Modifier in SFE
Technical FAQs: Supercritical Fluid Extraction with Methanol Modifiers
How does methanol volume affect the yield of Acetylcholinesterase (AChE) inhibitors in SFE?
Methanol acts as a polar co-solvent (entrainer) in supercritical CO₂, significantly altering the solvent's polarity and its ability to extract target compounds [36]. The volume used directly impacts yield and selectivity.
- Low Methanol Volume (<5% of total solvent): Results in poor yield of polar AChE inhibitors (e.g., coumarin derivatives, huperzine A analogues) because the supercritical CO₂ lacks sufficient polarity to dissolve these compounds effectively [36].
- Optimal Methanol Volume (5-15%): Strikes a balance, maximizing the solubility of most AChE inhibitors without excessive co-extraction of unwanted polar impurities. The exact percentage depends on the specific inhibitor's polarity [37] [36].
- High Methanol Volume (>15%): Can increase yield but reduces selectivity. It may lead to co-extraction of undesirable compounds like chlorophylls, sugars, or tannins, which complicate subsequent purification and can potentially foul the SFE system [36].
What are the common symptoms of suboptimal methanol volume, and how are they resolved?
Researchers can diagnose issues by observing the extract's characteristics and system behavior. The table below outlines common symptoms and corrective actions.
Table: Troubleshooting Guide for Methanol Volume in SFE of AChE Inhibitors
| Observed Symptom | Potential Cause | Recommended Corrective Action |
|---|---|---|
| Low extraction yield of target AChE inhibitors | Methanol volume too low; fluid polarity insufficient [36]. | Systematically increase methanol volume by 2-5% increments while monitoring yield. |
| High yield but low inhibitory activity in bioassays | Methanol volume too high; excessive co-extraction of inactive polar compounds [36]. | Reduce methanol volume; implement a pressure/temperature gradient to fractionate extracts. |
| Inconsistent yield between batches | Fluctuations in co-solvent pumping or improper mixing with SC-CO₂ [36]. | Calibrate co-solvent pump; ensure use of a static extraction step for equilibration. |
| Precipitation of extract in tubing or separator | Rapid pressure drop causing "shock" precipitation. | Optimize pressure reduction rate across separators; consider a multi-stage separation setup. |
What is the systematic protocol for optimizing methanol volume?
A robust optimization protocol is crucial for method development.
- Preliminary Scoping: Conduct initial extractions with a broad range of methanol volumes (e.g., 0%, 5%, 10%, 15%, 20%) while keeping other parameters (pressure, temperature, flow rate) constant [36].
- Yield Analysis: Quantify the total mass of the crude extract obtained at each methanol volume.
- Bioactivity Screening: Analyze the bioactivity of each extract using a standard AChE inhibition assay (e.g., Ellman's method) [38]. The goal is to identify the fraction with the highest specific activity (inhibition per unit mass of extract), not just the highest total yield.
- Chemical Profiling: Use analytical techniques like TLC or HPLC to compare the chemical profiles of the most active extracts. This helps confirm that the target inhibitors are being selectively enriched [39].
- Fine-Tuning: Narrow the volume range around the most promising value from the initial scoping (e.g., testing 7%, 9%, 11%) to pinpoint the optimum.
How do other SFE parameters interact with methanol volume?
Methanol volume does not operate in isolation. Its effect is intertwined with core SFE parameters [36]:
- Pressure and Temperature: These parameters control the density and solvent power of SC-CO₂. A higher pressure can sometimes compensate for slightly lower methanol volume by increasing density, and vice-versa. The vapor pressure of target solutes also increases with temperature.
- CO₂ Flow Rate: A very high flow rate may not allow sufficient time for the methanol-modified CO₂ to penetrate the matrix and solubilize the targets, reducing effective extraction even at an optimal volume.
- Raw Material Preparation: Finer particle sizes and controlled moisture content create more surface area and improve contact between the modified fluid and the plant matrix, making methanol volume optimization more effective [36].
Experimental Protocol: Optimizing Methanol Modifier Concentration
The following diagram illustrates the logical workflow for the optimization experiment.
Detailed Methodology
Aim: To determine the optimal volume percentage of methanol modifier in supercritical CO₂ for the maximum recovery of acetylcholinesterase inhibitors from a selected plant matrix.
Materials and Equipment
- Supercritical Fluid Extractor system (with co-solvent pump) [36]
- Liquid CO₂ supply source
- Anhydrous methanol (HPLC grade)
- Plant material (e.g., dried, milled, and sieved Galanthus leaves or other relevant source)
- Standard AChE inhibition assay kit (e.g., electric eel AChE, acetylthiocholine iodide, DTNB) [38]
Procedure
- Material Preparation: Prepare the plant matrix by freeze-drying, grinding, and sieving to a uniform particle size (e.g., 0.5-1.0 mm). Record the moisture content [37].
- System Setup: Load a fixed mass (e.g., 10g) of prepared material into the extraction vessel. Set the base SFE parameters based on literature or preliminary tests. A suggested starting point is:
- Pressure: 250 bar
- Temperature: 50°C
- CO₂ Flow Rate: 10 g/min
- Extraction Time: 120 min (including a 15-minute static period) [36]
- Scoping Experiment: Perform a series of extractions where the only variable is the methanol volume percentage. A recommended design is outlined in the table below.
- Extract Collection: Collect the total extract from each run in a pre-weighed collection vial. Flush the system with pure CO₂ between runs to prevent cross-contamination. Weigh the vials to determine the total extraction yield.
- Bioactivity Testing: Prepare standardized solutions of each extract. Evaluate the AChE inhibitory activity of each solution using the Ellman's assay or a similar validated method [38]. Include a positive control (e.g., galantamine or donepezil) and a negative control.
- Data Analysis: Calculate the IC₅₀ values or percentage inhibition at a fixed concentration for each extract. Plot the total yield and bioactivity against the methanol volume to identify the optimum.
Table: Experimental Design for Scoping Methanol Volume
| Experiment Run | Static Methanol Volume (%) | Dynamic Methanol Volume (%) | Pressure (bar) | Temperature (°C) |
|---|---|---|---|---|
| 1 | 0 | 0 | 250 | 50 |
| 2 | 5 | 5 | 250 | 50 |
| 3 | 10 | 10 | 250 | 50 |
| 4 | 15 | 15 | 250 | 50 |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table: Key Reagents and Materials for SFE of AChE Inhibitors
| Item Name | Function / Rationale | Technical Notes |
|---|---|---|
| Supercritical CO₂ | Primary solvent; non-toxic, tunable solvent power. | Critical point: 31.1°C, 73.8 bar. Food-grade purity (99.9%) is recommended [36]. |
| Methanol (HPLC Grade) | Polar co-solvent (modifier); enhances extraction of polar AChE inhibitors. | Anhydrous methanol prevents ice formation in the system. Typically used at 5-15% (v/v) [36]. |
| Acetylcholinesterase | Target enzyme for bioactivity screening. | Electric eel AChE is commonly used. Aliquots should be stored at -20°C [38]. |
| Acetylthiocholine Iodide | Substrate for the Ellman's assay. | Decomposed by AChE to thiocholine, which reacts with DTNB [38]. |
| DTNB (Ellman's Reagent) | Chromogenic agent; produces yellow anion for spectrophotometric detection. | 5,5'-dithiobis-(2-nitrobenzoic acid). Measure absorbance at 412 nm [38]. |
| Reference Inhibitors | Positive controls for validating the bioassay (e.g., Donepezil, Galantamine). | Ensure potency and purity. Used to calculate relative inhibition potency of extracts [39]. |
Troubleshooting Guide: Common Experimental Challenges
Problem 1: Emulsion Formation During Extraction or Post-Processing
- Possible Cause: High concentration of surfactant-like compounds (e.g., phospholipids, free fatty acids) in the rice bran matrix [31].
- Solution: Gently swirl the extraction vessel instead of using vigorous shaking to reduce agitation. If an emulsion forms, disrupt it by centrifugation or by adding brine (salt water) to increase the ionic strength of the aqueous layer and force phase separation [31].
Problem 2: Low Extraction Yield of γ-Oryzanol
- Possible Cause: Inefficient solvent strength of pure CO₂ for target compounds [18] [17].
- Solution: Incorporate a polar co-solvent like ethanol to enhance the solubilizing power of supercritical CO₂ for polar molecules. Ensure the ethanol is added at the correct ratio (e.g., 10% w/w) [40] [17].
Problem 3: Inconsistent Yield Between Experimental Runs
- Possible Cause: Inaccurate maintenance of co-solvent concentration during the dynamic flow phase of Supercritical Fluid Extraction (SFE) [17].
- Solution: Actively replace the co-solvent flushed out during dynamic extraction. Use a mass flow meter to measure CO₂ outflow and set the co-solvent pump to add ethanol at a rate equivalent to 5% of this volume to maintain a constant level in the vessel [17].
Problem 4: Clogging in the Restrictor Valve or Back Pressure Regulator
- Possible Cause: Water or other extracted material freezing due to adiabatic expansion of CO₂, which causes significant cooling [20].
- Solution: Electrically heat the restrictor or back pressure regulator to prevent freezing and subsequent blockages [20].
Frequently Asked Questions (FAQs)
Q1: Why is ethanol the preferred co-solvent for extracting γ-oryzanol from rice bran? Ethanol is preferred because it is a safe, food-grade solvent with a lower carbon footprint and global warming potential compared to alternatives like methanol [40]. It effectively increases the polarity of supercritical CO₂, enhancing the solubility of γ-oryzanol, which is a moderately polar compound [18] [40]. Experimental optimizations have identified 10 w/w% ethanol as a highly effective concentration for maximizing γ-oryzanol yield from defatted rice bran [40].
Q2: How does the addition of a co-solvent like ethanol enhance the SFE process? Supercritical CO₂ by itself has dissolving power similar to hexane, making it excellent for non-polar compounds but less effective for polar molecules [18] [17]. The addition of a small quantity of a polar co-solvent like ethanol enhances the solubilizing power of the supercritical fluid, making it possible to extract much more polar compounds efficiently [18] [17]. Co-solvents can also reduce the required process temperature and pressure, thereby improving overall extraction efficiency [18].
Q3: What is the most critical parameter to control when using a co-solvent in SFE? Maintaining a consistent and precise concentration of the co-solvent throughout the dynamic extraction phase is crucial [17]. The initial doping of the vessel must be followed by a continuous, metered addition of co-solvent during CO₂ flow to replace what is being flushed out, ensuring the solvent strength remains constant for reproducible results [17].
Q4: My system pressure is limited to 250 bar. Can I still achieve a high γ-oryzanol yield? Yes. While some studies use very high pressures (e.g., 500 bar [41]), research has shown that excellent yields can be obtained at lower pressures by optimizing other parameters. One study achieved an optimal γ-oryzanol yield of 36,000 mg/kg at a pressure of 23.9 MPa (approx. 239 bar) by using 10% ethanol as a co-solvent and optimizing temperature and CO₂ flow rate [40].
Experimental Protocols & Data
Detailed Methodology for SFE of γ-Oryzanol with Ethanol Modifier
Apparatus Setup:
- A supercritical CO₂ extraction system equipped with a high-pressure pump, a pressure cell (extraction vessel), a means of maintaining pressure (e.g., a back pressure regulator), a co-solvent addition pump, a fluid pre-heater, and a collection vessel is required [20].
- A pre-heater for the CO₂ and co-solvent stream before it enters the main extraction vessel is highly recommended for accurate and reproducible temperature control, especially at high flow rates [17].
Sample Preparation:
- Obtain rice bran (e.g., Japônica variety [41]) and store at -20°C to prevent enzymatic degradation until use [40].
- The raw bran can be used directly, or defatted bran can be employed as a starting material for concentrating γ-oryzanol [40].
- The moisture content of the bran should be analyzed and standardized (e.g., ~8.34 w/w% [40]).
Extraction Procedure:
- Load the Vessel: Place an appropriate mass of rice bran (e.g., 20g [41]) into the extraction vessel. Mixing with an inert material like glass beads can reduce dead volume [41].
- Initial Doping (Optional but recommended): For a 100 ml vessel, if a 5% co-solvent level is desired, add 5 ml of ethanol to the vessel before sealing [17].
- Pressurize and Heat: Pressurize the system with liquid CO₂ and heat the extraction vessel to the target temperature.
- Dynamic Extraction with Co-solvent:
- Collection: The extract, containing γ-oryzanol and other lipids, is collected in a vessel downstream after the pressure is reduced, causing the CO₂ to lose its solvating power [20].
- Analysis: The collected extract is analyzed for γ-oryzanol content using validated analytical methods, such as HPLC.
Table 1: Optimized SFE Parameters for γ-Oryzanol Recovery from Rice Bran
| Parameter | Study A: Defatted Rice Bran [40] | Study B: Rice Bran [41] | Function/Rationale |
|---|---|---|---|
| Pressure | 23.9 MPa (239 bar) | 500 bar | Higher pressure increases solvent density and dissolving power [20]. |
| Temperature | 48.9 °C | 62 °C | Effect is complex; higher T can increase solute vapor pressure but reduce solvent density [20]. |
| CO₂ Flow Rate | 29.8 g/min | 15 g/min | Balances extraction speed and solvent consumption [20]. |
| Ethanol (Co-solvent) Concentration | 10 w/w% | Not specified | Enhances solubility of polar γ-oryzanol in non-polar CO₂ [18]. |
| Reported γ-Oryzanol Yield | 36,000 mg/kg | 36.6 mg/g of extract | Demonstrates process efficacy under different conditions and material states. |
Table 2: Key Research Reagent Solutions
| Reagent/Equipment | Specification / Function | Safety & Environmental Notes |
|---|---|---|
| Carbon Dioxide (CO₂) | Primary supercritical solvent. Liquid CO₂ is pumped, then heated to supercritical state (Tc > 31°C, Pc > 74 bar) [20]. | GRAS (Generally Recognized as Safe), non-flammable, non-toxic. Leaves no residual solvent [18] [42]. |
| Ethanol (Co-solvent) | Polar modifier. Increases solubility of γ-oryzanol and other polar bioactives in supercritical CO₂ [18] [40]. | Food-grade, preferable due to lower carbon footprint and global warming potential vs. methanol [40]. |
| Rice Bran | Raw material from milling. Source of γ-oryzanol, fatty acids, and other bioactive compounds [41] [40]. | Should be stored at -20°C to avoid rancidity and enzymatic degradation [40]. |
| n-Hexane | Conventional solvent for benchmark extractions (e.g., Soxhlet) [41]. | Flammable, toxic, poses environmental and residual solvent concerns [41] [18]. |
Workflow Visualization
SFE Experimental Workflow
Troubleshooting Logic Flow
Artificial Neural Networks (ANN) Coupled with Genetic Algorithms (GA)
Frequently Asked Questions (FAQs)
FAQ 1: Why should we use an ANN-GA approach over traditional statistical methods like RSM for optimizing SFE processes?
ANN-GA is often superior for modeling the complex, non-linear relationships typical in Supercritical Fluid Extraction (SFE). While Response Surface Methodology (RSM) fits a predefined polynomial equation to the data, ANN is a non-parametric model that learns directly from data without requiring a pre-specified kinetic model, allowing it to capture more complex patterns [43] [44]. A direct comparison in the optimization of Ferulago angulata SFE showed the ANN model (R² = 0.9971) had a significantly better fit than the RSM model (R² = 0.9645) [45]. Furthermore, the GA efficiently navigates complex multi-variable spaces to find a global optimum, often leading to more precise and robust optimal conditions than RSM [46] [47].
FAQ 2: We are getting a good fit during training, but the ANN model's predictions are poor. What might be wrong?
This is a classic sign of overfitting, where the model learns the training data too well, including its noise, but fails to generalize to new data. To address this:
- Data Division: Ensure your experimental data is properly divided into three sets: a training set (~80%) for model learning, a validation set (~10%) to tune hyperparameters and detect overfitting, and a testing set (~10%) for a final, unbiased evaluation of model performance [46].
- Dataset Size: Use an adequate number of experimental data points. The network should be trained on a dataset that is substantially larger than the number of connection weights within the ANN to ensure proper generalization [44].
- Early Stopping: Halt the training process when the error on the validation set begins to increase, even if the training error continues to decrease [46].
FAQ 3: How do we determine the optimal architecture for the ANN (e.g., number of hidden layers and neurons)?
There is no universal rule; the optimal architecture is problem-dependent and found empirically. A common and effective approach is to:
- Start with a simple network featuring a single hidden layer.
- Systematically vary the number of neurons in that hidden layer (e.g., from 1 to 20).
- Train each configuration multiple times to account for random weight initializations.
- Select the architecture that yields the lowest error on the validation set [44] [46]. For instance, an ANN with a [5-6-1] architecture (5 inputs, 6 hidden neurons, 1 output) was optimal for SFE of Argemone mexicana seeds [44], while other studies found success with different configurations through similar testing.
FAQ 4: The GA is not converging to a satisfactory solution. How can we improve its performance?
- Adjust GA Parameters: Tune key parameters such as the population size, crossover rate, and mutation rate. A larger population increases diversity but is computationally more expensive.
- Check the Fitness Function: Ensure your fitness function (e.g., maximization of extraction yield) is correctly defined and that the ANN model providing the predictions is highly accurate.
- Hybrid Approach: Consider using the results from an RSM optimization as an initial guess or within the initial population for the GA, which can help guide the search and accelerate convergence.
Troubleshooting Guides
Issue 1: Poor Predictive Performance of the ANN Model
| Symptom | Possible Cause | Solution |
|---|---|---|
| High error on both training and test data (Underfitting). | The network architecture is too simple to capture the underlying trends. | Increase the number of neurons in the hidden layer or add a second hidden layer [44] [46]. |
| Low error on training data but high error on test data (Overfitting). | The model has memorized the training data noise. | 1) Implement early stopping using a validation set [46]. 2) Increase the size of the experimental dataset. 3) Use a simpler network with fewer neurons. |
| Inconsistent model performance across training sessions. | The random initialization of weights leads to different local minima. | Train the network several times with different initial random weights and select the best-performing model, or use a more robust training algorithm [44]. |
Issue 2: Failure to Achieve High Extraction Yield at ANN-GA Predicted Optimum
| Symptom | Possible Cause | Solution |
|---|---|---|
| Experimental validation yield is significantly lower than the predicted value. | The model may be extrapolating beyond the range of the experimental data used to train it. | Ensure the GA-found optimum lies within the experimental boundaries of the training data for all parameters (e.g., pressure, temperature, modifier concentration). |
| Yield is low and the model fit is good. | There may be a physical or chemical constraint in the SFE system not captured by the model (e.g., channeling, particle agglomeration). | Verify the physical setup of the SFE extractor, ensure homogeneous packing of the raw material, and check for technical issues like clogged lines. |
| The optimal modifier concentration predicted is at the extreme of the tested range. | The true optimum may lie outside the experimental design space. | Consider expanding the upper or lower limit of the modifier concentration in a new experimental design to explore a wider range. |
Performance Comparison: ANN-GA vs. RSM
The following table summarizes quantitative comparisons between ANN-GA and RSM as reported in various extraction optimization studies.
Table 1: Comparative performance of RSM and ANN-GA in extraction optimization.
| Source Material | Extraction Method | Response Variable | Model Performance (R²) | Key Finding | Reference |
|---|---|---|---|---|---|
| RSM | ANN-GA | ||||
| Pouteria lucuma Seed | Supercritical CO₂ | Oil Yield | 0.9891 | 0.9999 | ANN-GA provided a greater proportion of the total variability, facilitating better parameter identification [43] [48]. |
| Orange Paprika | Accelerated Solvent | Zeaxanthin Yield | N/A | N/A | The relative error under optimal conditions was 10.46% for RSM vs. 2.18% for ANN-GA, showing superior accuracy for ANN-GA [49]. |
| Phylloporia ribis Mushroom | Soxhlet | Antioxidant Activity | N/A | N/A | Extracts from ANN-GA optimized conditions showed superior antioxidant activity and higher phenolic content compared to RSM [46]. |
| Ferulago angulata | Supercritical CO₂ | Essential Oil Yield | 0.9645 | 0.9971 | ANN model was more accurate than RSM, confirmed by lower MSE and AARD values [45]. |
Experimental Protocol: A Representative Workflow
The following is a generalized detailed methodology for developing and applying an ANN-GA model to optimize modifier concentration in SFE, based on established protocols [43] [44] [46].
1. Experimental Design and Data Generation:
- Input Variables: Design experiments considering key SFE parameters. A typical Box-Behnken Design (BBD) or Central Composite Design (CCD) is suitable. Variables often include:
- Pressure (e.g., 100 - 300 bar)
- Temperature (e.g., 40 - 60 °C)
- Modifier (Co-solvent) Concentration (e.g., 0 - 10% of CO₂ flow rate)
- CO₂ Flow Rate
- Extraction Time
- Response Variable: Conduct the SFE experiments and measure your target response (e.g., cumulative extraction yield of the target compound, antioxidant activity).
- Data Division: Randomly split the resulting experimental data into three subsets: Training (70-80%), Validation (10-15%), and Testing (10-15%).
2. Artificial Neural Network (ANN) Modeling:
- Network Structure: Use a feed-forward, multi-layer perceptron (MLP) network. The input layer has neurons corresponding to each input variable, and the output layer has neurons for each response.
- Hidden Layer Configuration: Determine the optimal number of hidden layers and neurons empirically. A single hidden layer is often sufficient. The hyperbolic tangent sigmoid (
tansig) or logistic sigmoid are common activation functions for the hidden layer, while a linear (purelin) function is used for the output layer [44] [46]. - Training: Train the network using the training dataset. The Levenberg-Marquardt (LM) backpropagation algorithm is widely used for its fast convergence [44] [46]. The training process should be monitored using the validation set to prevent overfitting.
- Model Evaluation: Evaluate the final model's performance on the unseen testing set using statistical metrics:
- Coefficient of Determination (R²)
- Mean Square Error (MSE)
- Mean Absolute Percentage Error (MAPE) or Absolute Average Relative Deviation (AARD)
3. Optimization with Genetic Algorithm (GA):
- Fitness Function: The trained and validated ANN model serves as the fitness function for the GA. The objective is to find the input variable combination that maximizes (or minimizes) the ANN's predicted output.
- GA Execution: Run the GA with the following typical steps:
- Initialization: Generate an initial population of random candidate solutions (sets of extraction parameters).
- Selection: Evaluate candidates using the ANN fitness function and select the best-performing ones for reproduction.
- Crossover and Mutation: Create a new generation of candidates by combining (crossover) and randomly altering (mutation) the parameters of the selected parents.
- Iteration: Repeat the selection and reproduction steps over many generations until a convergence criterion is met (e.g., a maximum number of generations or no improvement in fitness).
4. Validation:
- Conduct a new SFE experiment using the optimal parameters predicted by the ANN-GA framework.
- Compare the experimentally obtained response value with the model's prediction to validate the model's accuracy and reliability.
Workflow and Architecture Visualization
ANN-GA Optimization Workflow
The diagram below illustrates the integrated steps for optimizing extraction processes using ANN and GA.
Typical Feed-Forward ANN Architecture
This diagram represents a typical ANN architecture used for modeling SFE processes, showing the flow of information from inputs to output.
Research Reagent Solutions
The following table lists key materials and reagents commonly used in SFE research coupled with ANN-GA modeling.
Table 2: Essential research reagents and materials for SFE-ANN-GA studies.
| Reagent/Material | Function in SFE and ANN-GA Research | Example from Literature |
|---|---|---|
| Supercritical CO₂ | Primary solvent for extraction; non-toxic, tunable solvation power. | Used as the main solvent in SFE of Pouteria lucuma seed oil [43] and Argemone mexicana seeds [44]. |
| Co-solvents/Modifiers (e.g., Ethanol, Methanol) | Added to SC-CO₂ to increase polarity and improve extraction yield of target compounds. Ethanol is preferred for food/pharma as a GRAS solvent. | Ethanol was used as a co-solvent in the SFE of Argemone mexicana seeds [44] and for extracting fucoxanthin [50]. |
| Analytical Standards (e.g., FAME Mix, Zeaxanthin) | Used for identification and quantification of extracted compounds via chromatography (GC, UPLC). Crucial for generating accurate response variable data. | Fatty acid methyl ester (FAME) standards were used to analyze lucuma seed oil [43]. Zeaxanthin standard was used for UPLC calibration [49]. |
| Antioxidant Assay Kits (e.g., ABTS, DPPH, TAS/TOS) | Used to measure the biological activity (antioxidant capacity) of extracts as a response variable for optimization. | TAS (Total Antioxidant Status) was used as the key response for optimizing Phylloporia ribis and Mentha longifolia extraction [46] [47]. |
| Sample Preparation Materials (e.g., Diatomaceous Earth) | Used as an inert dispersant to mix with the sample for improved solvent contact and flow in pressurized extraction cells. | Mixed with paprika powder in Accelerated Solvent Extraction cells [49]. |
Troubleshooting Modifier Use: From Emulsions to Degradation
Balancing Modifier Concentration to Prevent Matrix Swelling
Troubleshooting Guide: Matrix Swelling in SFE
Problem Description
Researchers often encounter matrix swelling during Supercritical Fluid Extraction (SFE) when using liquid modifiers like ethanol or methanol. This phenomenon occurs when the co-solvent interacts with the plant matrix, causing it to expand and potentially leading to reduced extraction yields, system clogging, and increased mass transfer resistance.
Root Causes
Matrix swelling primarily occurs when:
- High concentrations of polar modifiers (typically >10% ethanol) penetrate the plant cellular structure
- Moisture content in raw materials interacts with hydrophilic co-solvents
- Extended static extraction times allow excessive solvent-matrix interaction
- Specific plant matrices with high polysaccharide content are more susceptible
FAQ: Managing Modifier Concentration
Q1: What is the safe concentration range for ethanol to prevent matrix swelling? For most plant matrices, ethanol concentration should be maintained between 1-10% of the total CO₂ flow rate. Concentrations above 15% significantly increase swelling risk in susceptible materials [12] [51]. The optimal concentration depends on your specific biomass type and should be determined through preliminary testing.
Q2: How does matrix swelling affect my extraction efficiency? Matrix swelling creates several operational challenges:
- Reduced permeability of the packed bed, limiting solvent access to target compounds
- Increased mass transfer resistance as diffusion paths lengthen
- Potential channeling where CO₂ bypasses sections of the swollen matrix
- Increased pressure drops across the extraction vessel
- Possible system clogging requiring premature shutdowns [51]
Q3: Which plant materials are most prone to swelling effects? Materials with high polysaccharide, cellulose, or starch content demonstrate greater swelling propensity. This includes:
- Cereal brans (rice, wheat, oat bran)
- Roots and tubers
- Woody barks and plant stems
- Seeds with high mucilage content [41]
Q4: What experimental signs indicate matrix swelling is occurring? Monitor for these warning signs during SFE runs:
- Unexplained pressure increases across the extraction vessel
- Erratic CO₂ flow rates despite constant pump settings
- Sudden decreases in extraction yield after initial high recovery
- Visible compaction or changes in the matrix upon vessel emptying [51]
Experimental Protocol: Modifier Optimization
Objective
Determine the optimal modifier concentration that maximizes extraction yield while preventing matrix swelling.
Materials and Equipment
Table: Essential Research Reagent Solutions
| Item | Function | Technical Specifications |
|---|---|---|
| Supercritical CO₂ | Primary extraction fluid | Food-grade, 99.95% purity [41] |
| Anhydrous Ethanol | Polar co-solvent | HPLC grade, ≥99.9% purity [12] |
| Raw Plant Material | Extraction matrix | Particle size: 0.3-1.0 mm [51] |
| SFE System | Extraction apparatus | Equipped with co-solvent pump [41] |
| Analytical Balance | Yield quantification | Precision: ±0.0001 g |
Methodology
Step 1: Raw Material Preparation
- Reduce particle size to 0.3-1.0 mm using an appropriate mill
- Adjust moisture content to 5-10% by controlled drying if necessary
- Store prepared material in desiccators until use [51]
Step 2: Experimental Design
- Employ a Response Surface Methodology (RSM) with a Box-Behnken or Central Composite Design
- Test ethanol concentrations at 3-5 levels (e.g., 2%, 5%, 8%, 11%, 14%)
- Maintain other parameters constant: pressure (200-300 bar), temperature (40-60°C), CO₂ flow rate (1-2 L/min) [12] [41]
Step 3: Extraction Procedure
- Pre-mix CO₂ and ethanol at target concentration using the co-solvent pump
- Begin with 10-15 minute static extraction to allow solvent penetration
- Switch to dynamic extraction for 2-3 hours at predetermined flow rate
- Collect extract in separators with pressure reduction to 50-60 bar
- Weigh extracts and analyze for target compounds [41]
Step 4: Swelling Assessment
- Measure bed volume before and after extraction
- Calculate swelling index = (Final volume - Initial volume) / Initial volume × 100
- Document any operational issues (pressure spikes, flow restrictions)
Step 5: Data Analysis
- Correlate extraction yield with modifier concentration and swelling index
- Identify optimal concentration that balances yield with acceptable swelling (<5%)
Technical Data Reference
Table: Modifier Concentration Effects on Different Matrices
| Plant Material | Optimal Ethanol Concentration | Swelling Threshold | Yield Impact |
|---|---|---|---|
| Rice Bran [41] | 5-7% | >10% | 17.3% mass yield at optimal |
| Ammodaucus leucotrichus Seeds [12] | 15% | Not reported | Higher phenolic content |
| General Plant Material [51] | 1-10% | >15% | Varies by compound |
Workflow Visualization
Avoiding Co-extraction of Undesirable Compounds and Impurities
In the optimization of modifier concentration for Supercritical Fluid Extraction (SFE), a primary challenge is achieving selective recovery of target analytes while minimizing the co-extraction of undesirable compounds. These impurities—such as chlorophyll, waxes, fats, and heavy metals—can compromise extract purity, complicate downstream processing, and interfere with analytical results. This guide provides targeted troubleshooting strategies to enhance selectivity through precise control of modifier use, helping researchers and drug development professionals obtain cleaner, more reliable extracts.
Troubleshooting FAQs
FAQ 1: How can I prevent the co-extraction of chlorophyll and other pigments when extracting from green plant material?
- Issue: Chlorophyll, a common pigment, is often co-extracted with target bioactive compounds, giving the extract a green hue and potentially acting as a photosensitizer that promotes oxidation [27].
- Solution & Protocol: The key is to use pure supercritical CO₂ without a polar modifier for an initial defatting/decolorizing step, as chlorophyll has low solubility in pure CO₂.
- Experimental Protocol:
- Prepare the biomass: Commingle dried, ground plant material.
- Initial extraction with pure CO₂: Load the biomass into the extraction vessel. Perform SFE using pure CO₂ at moderate pressure and temperature (e.g., 250-300 bar, 40-50 °C) for a predetermined time. This step will remove non-polar lipids and some pigments without extracting significant chlorophyll [27].
- Elute target compounds with modified CO₂: Following the initial step, introduce a polar modifier like ethanol to the CO₂ stream. The optimal modifier concentration is compound-specific. For phenolic compounds in Labisia pumila, a concentration of 16% (v/v) of a 78% ethanol-water mixture was effective [52]. Adjust pressure and temperature to maximize the solubility of your target molecules.
- Collect separate fractions: Collect the extract from the initial pure CO₂ run and the subsequent modified CO₂ run separately. Analyze the second fraction for target compounds and chlorophyll content.
- Experimental Protocol:
FAQ 2: My extract is waxy or greasy at room temperature. How can I reduce the co-extraction of lipids and waxes?
- Issue: Neutral lipids and long-chain waxes can precipitate in extraction lines or analytical instruments, causing blockages and interfering with analysis.
- Solution & Protocol: Implement a fractional separation strategy by manipulating pressure and temperature across multiple separators in series. A pressure reduction cascade causes compounds to precipitate based on their decreasing solubility in CO₂.
- Experimental Protocol:
- Set up a multi-separator system: Configure your SFE system with at least two separators plumbed in series.
- Optimize separator conditions:
- Separator 1 (for impurities): Set to a high pressure and temperature (e.g., 100-150 bar, 50-60 °C). Under these conditions, lipids and waxes remain soluble in CO₂, while heavier target compounds may precipitate [41] [53].
- Separator 2 (for target compounds): Set to a lower pressure and near-ambiant temperature (e.g., 50-80 bar, 20-25 °C). The rapid pressure drop here will cause the target compounds to precipitate for collection.
- Extract and collect: Perform the dynamic extraction. Waxes and lipids will be retained in the CO₂ stream through the first separator and collected in the second, while the target compounds are isolated in the first. Monitor and adjust conditions based on your specific extract composition.
- Experimental Protocol:
FAQ 3: The selectivity for my target polar compound (e.g., a phenolic acid) is low. How can I optimize modifier concentration to improve it?
- Issue: Low yields of polar target compounds due to the limited solubility of polar molecules in pure supercritical CO₂.
- Solution & Protocol: Systematically optimize the type and concentration of a polar co-solvent to increase the polarity of the supercritical fluid without extracting highly polar impurities.
- Experimental Protocol:
- Select a co-solvent: Food-grade ethanol is a common, safe, and effective choice for polar compounds [27] [52].
- Design an experiment: Use a Response Surface Methodology (RSM) design, such as a Central Composite or Box-Behnken design, to model the effect of variables [41] [27] [52].
- Define variables and responses: The key variables are modifier concentration, pressure, and temperature. The responses are extraction yield of the target compound and purity (e.g., measured by HPLC or via the concentration of a known impurity).
- Execute and model: Run the extractions as per the experimental design. Fit the data to a quadratic model to find the optimal modifier concentration that maximizes target yield while minimizing co-extraction. For example, a study on hemp seed oil found that 10% ethanol maximized phenolic content without negatively affecting the oil's fatty acid profile [27].
- Experimental Protocol:
FAQ 4: How can I avoid extracting heavy metals from my plant or soil samples?
- Issue: Inorganic heavy metal ions are not soluble in supercritical CO₂, with or without standard organic modifiers.
- Solution & Protocol: SFE is inherently selective for organic compounds. The primary risk of metal contamination comes from the extraction system itself or from particulate matter in the sample.
- Experimental Protocol:
- Use high-purity reagents and equipment: Ensure CO₂ and modifiers are of high purity. Use extraction vessels and tubing made of high-quality stainless steel to prevent leaching.
- Pre-clean the sample: For complex matrices like soil, a pre-extraction wash with an appropriate aqueous solution (e.g., a chelating agent) can be performed, followed by thorough drying before SFE. Note that this is a separate, non-SFE step.
- Filter the extract: After SFE, pass the collected extract through a micro-filter (e.g., 0.45 µm) to remove any suspended particulate matter that may contain metals.
- System maintenance: Regularly clean and passivate the entire SFE system to prevent corrosion and metal contamination.
- Experimental Protocol:
Optimized Experimental Parameters for Selective Extraction
The table below summarizes optimized SFE parameters from recent research for recovering target compounds while minimizing co-extraction of specific impurities.
| Target Compound | Source Material | Key Impurity to Avoid | Optimized Modifier | Optimal Conditions (P, T, Modifier Conc.) | Key Outcome / Selectivity |
|---|---|---|---|---|---|
| γ-Oryzanol & Fatty Acids [41] | Rice Bran | Non-target lipids | Pure CO₂ (initial step) | 500 bar, 62 °C | Higher selectivity for γ-oryzanol vs. conventional hexane extraction. |
| Phenolic Acids [52] | Labisia pumila Leaves | Unspecified polar impurities | Ethanol-Water (78% v/v) | 283 bar, 32 °C, 16% (v/v) co-solvent | High yield of gallic acid, methyl gallate, and caffeic acid. |
| Phenolic Compounds [27] | Hemp Seeds | Chlorophyll, Lipids | Ethanol (10% as co-solvent) | 200 bar, 50 °C, 10% Ethanol | Increased phenolics & tocopherols without affecting fatty acid profile. |
| Cannabinoids [54] | Industrial Hemp | Acidic cannabinoid forms | Varies with target | 300-550 bar, 60 °C | Selective extraction of neutral (CBD, THC) vs. acidic (CBDA, THCA) forms. |
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent / Material | Function in SFE | Application Note |
|---|---|---|
| Supercritical CO₂ | Primary solvent; tunable density controls solubility [53]. | Must be high purity. Critical point: 31.1°C, 73.8 bar [53]. |
| Ethanol | Polar co-solvent (modifier); increases solubility of mid-to-high polarity targets [27] [52]. | GRAS (Generally Recognized as Safe) status ideal for food/pharma. |
| Water-Ethanol Mixtures | Modifier for highly polar compounds (e.g., phenolic acids, glycosides) [52]. | Allows fine-tuning of polarity. A 78% ethanol-water mix was optimal for some phenolics [52]. |
| Stainless Steel Vessels & Frits | Contain sample and withstand high pressure; frits filter particulates. | Ensure compatibility with solvents; clean regularly to prevent carryover. |
| Glass Beads | Packing material to reduce dead volume in the extraction vessel [41]. | Improves solvent flow dynamics and extraction efficiency. |
Workflow Diagram for Impurity Avoidance
The following diagram illustrates a logical decision-making workflow to prevent the co-extraction of common impurities.
SFE Impurity Avoidance Workflow: This chart provides a step-by-step decision path for selecting the appropriate technique based on the specific impurity of concern.
Preventing Analyte Degradation and Ensuring Process Stability
Troubleshooting Guides
Issue 1: Formation of Emulsions During Extraction
Problem: Emulsions are forming during the liquid-liquid extraction step, preventing clean phase separation and potentially leading to analyte loss.
Causes:
- Sample contains high amounts of surfactant-like compounds (e.g., phospholipids, free fatty acids, triglycerides, proteins) [31].
- Overly vigorous shaking of the separation funnel [31].
Solutions:
- Prevention: Gently swirl the separatory funnel instead of shaking it vigorously to maintain surface area for extraction while reducing agitation [31].
- Disruption: If an emulsion forms, try these techniques:
- Salting Out: Add brine or salt water to increase the ionic strength of the aqueous layer, forcing the surfactant-like molecules to separate into one phase or the other [31].
- Filtration: Pass the emulsion through a glass wool plug or a highly silanized phase separation filter paper to isolate the desired layer [31].
- Centrifugation: Use centrifugation to isolate the emulsion material in the residue [31].
- Solvent Adjustment: Add a small amount of a different organic solvent to adjust the solvent properties and break the emulsion [31].
- Alternative Method: For samples prone to emulsions, use Supported Liquid Extraction (SLE). SLE involves applying the pH-adjusted aqueous sample to a solid support (e.g., diatomaceous earth), which creates an interface for extraction and prevents emulsion formation [31].
Issue 2: Thermal Degradation of Analytes
Problem: Target analytes are degrading during the extraction or subsequent decarboxylation process, leading to reduced yields and inaccurate profiles.
Causes:
- Excessively high temperatures or prolonged reaction times during decarboxylation [54].
- Exposure to high temperatures in conventional extraction methods like steam distillation [42].
Solutions:
- Optimize Decarboxylation: The optimal conditions for decarboxylation are compound-specific. For instance, for CBD, optimal conditions may be 140°C for 10 minutes. Higher temperatures and times can deplete neutral cannabinoids [54].
- Use Supercritical Fluid Extraction (SFE): SFE operates at relatively low temperatures, preserving thermolabile compounds often degraded by high-temperature methods [42]. Supercritical CO₂ is particularly suitable due to its low critical temperature (31.1°C) [42].
- Process Control: Implement control strategies for the SFE process. This can involve decomposing the process into subsystems (e.g., extractor and stripper units) with decentralized controllers to maintain stable pressure and temperature, thus ensuring consistent extraction conditions and preventing localized degradation [55].
Issue 3: Poor or Inconsistent Extraction Yield
Problem: The yield of the target analyte is low or varies significantly between experiments.
Causes:
- Suboptimal pressure and temperature conditions for the supercritical fluid [54] [41] [56].
- Lack of a co-solvent for more polar compounds [57].
- Inefficient extraction time [56].
Solutions:
- Optimize SFE Parameters: Systematically optimize pressure, temperature, and extraction time. The optimal conditions are matrix- and analyte-dependent. The tables below summarize optimal parameters from various studies.
- Use a Co-solvent: Adding a small percentage of a polar co-solvent like ethanol can significantly increase the yield of target compounds by modifying the polarity of supercritical CO₂ [57].
- Employ Experimental Design: Use statistical methods like Response Surface Methodology (RSM) with a Central Composite Face-Centered Design to efficiently find the optimal combination of extraction variables with the fewest possible trials [41] [56].
Table 1: Optimal SFE Conditions for Various Matrices and Analytes
| Plant Material | Target Compound(s) | Optimal Pressure | Optimal Temperature | Optimal Time | Key Findings | Source |
|---|---|---|---|---|---|---|
| Industrial Hemp | Cannabinoids (CBD, THC, etc.) | 300 - 550 bar | 60 °C | Not Specified | Higher temperatures and reaction times depleted neutral cannabinoids. | [54] |
| Rice Bran | γ-Oryzanol, Fatty Acids | 500 bar | 62 °C | 3 hours | Achieved a balance between yield and bioactive concentration; higher selectivity for γ-oryzanol vs. hexane. | [41] |
| Coreopsis tinctoria Nutt. | Oleoresin | 27.5 MPa (275 bar) | 45 °C | 3 hours | Yield was up to 3.163%; SFE resulted in more abundant components than steam distillation. | [56] |
| Filamentous Fungus | Azaphilone Pigments | Not Specified | Not Specified | Not Specified | SFE with CO₂/ethanol yielded twice as high as ethyl acetate maceration. | [57] |
Table 2: Example Experimental Design for SFE Optimization of Rice Bran Extracts [41]
| Experiment No. | Temperature (°C) | Pressure (bar) | Temperature (Coded) | Pressure (Coded) |
|---|---|---|---|---|
| 1 | 40 | 200 | -1 | -1 |
| 2 | 80 | 200 | +1 | -1 |
| 3 | 40 | 500 | -1 | +1 |
| 4 | 80 | 500 | +1 | +1 |
| 5 | 40 | 350 | -1 | 0 |
| 6 | 80 | 350 | +1 | 0 |
| 7 | 60 | 200 | 0 | -1 |
| 8 | 60 | 500 | 0 | +1 |
| 9 | 60 | 350 | 0 | 0 |
Frequently Asked Questions (FAQs)
Q1: What are the key advantages of SFE over traditional extraction methods in preventing degradation?
SFE offers several key advantages for ensuring analyte stability:
- Low-Temperature Operation: SFE uses CO₂ at relatively low temperatures (e.g., 30-80°C), which preserves thermolabile compounds that would be degraded by high-temperature methods like steam distillation [42].
- Inert Environment: The process occurs in an inert CO₂ atmosphere, minimizing oxidative degradation [42].
- No Toxic Solvents: CO₂ is non-toxic and easily removed, leaving no harmful residues that could interact with or degrade analytes [42] [56].
- Selective Extraction: The solvent power of supercritical CO₂ can be tuned by adjusting pressure and temperature, allowing for selective extraction of target compounds and leaving unwanted impurities behind [42].
Q2: How do I optimize the supercritical CO₂ extraction process in my research?
A robust optimization protocol involves the following steps:
- Define Variables and Ranges: Select key parameters like pressure, temperature, and extraction time. Choose ranges based on literature review; for example, pressures of 100-500 bar and temperatures of 40-80°C are common starting points [42] [41].
- Select an Experimental Design: Use a statistical design like a Central Composite Face-Centered Design (CCFC) or a Box-Behnken Design. This allows you to test the interactions between variables with a minimal number of experiments [41] [56].
- Perform Extractions: Conduct extractions according to the experimental design matrix.
- Analyze Responses: Measure your response variables, such as extraction yield or concentration of specific bioactive compounds.
- Model and Predict: Use statistical software to fit a model (e.g., a quadratic polynomial) to your data and identify the optimal conditions that maximize your desired response [56].
Q3: What is the role of decarboxylation in cannabinoid extraction, and how can it be controlled?
- Role: Cannabinoids like CBD and THC are naturally present in the acid form (CBDA, THCA) in the plant. Decarboxylation is a process that uses heat to remove a carboxyl group from these acid forms, converting them into the active neutral forms (CBD, THC) [54].
- Control: The decarboxylation process must be carefully optimized, as the conditions vary for different components. For example, one study found optimal conditions for CBD were 140°C for 10 minutes. Excessive temperature or time can lead to the significant depletion of these neutral cannabinoids [54].
Experimental Protocols
1. Objective: To optimize the supercritical CO₂ extraction of γ-oryzanol and fatty acids from rice bran. 2. Materials and Equipment:
- Raw Material: Rice bran.
- SFE System: A supercritical fluid extraction system (e.g., Thar Technology, SFE-500F-2-C50) with a 500 mL extraction cell and two separators.
- Solvent: Food-grade CO₂ (99.95% purity). 3. Methodology:
- Preparation: Place 20 g of rice bran mixed with glass beads into the extraction vessel.
- Pressurization and Heating: Pressurize the vessel with CO₂ to the target pressure (e.g., 200-500 bar) and heat to the target temperature (e.g., 40-80°C).
- Extraction: Maintain a constant CO₂ flow rate (e.g., 15 g/min) for a fixed duration (e.g., 3 hours) in continuous mode.
- Collection: The extract is collected in a fraction collector after CO₂ expansion and depressurization.
- Analysis: Weigh the extract to determine yield and analyze it for γ-oryzanol and fatty acid content using techniques like HPLC and GC-MS.
1. Objective: To find the optimal SFE conditions (time, pressure, temperature) for oleoresin yield from Coreopsis tinctoria Nutt. 2. Experimental Design:
- A Box-Behnken Design with three factors (time, pressure, temperature) at three levels (-1, 0, +1) is used.
- Variables and Levels:
- Time (X₁): 1 h (-1), 2 h (0), 3 h (+1)
- Pressure (X₂): 20 MPa (-1), 25 MPa (0), 30 MPa (+1)
- Temperature (X₃): 35°C (-1), 45°C (0), 55°C (+1) 3. Analysis:
- The yield (Y) from each experiment is fitted to a quadratic polynomial model:
Y = β₀ + β₁X₁ + β₂X₂ + β₃X₃ + β₁₁X₁² + β₂₂X₂² + β₃₃X₃² + β₁₂X₁X₂ + β₁₃X₁X₃ + β₂₃X₂X₃ - The model's significance and regression coefficient (R²) are evaluated to identify the optimal point.
Process Optimization Workflow
The following diagram illustrates the logical workflow for developing and troubleshooting a stable and efficient SFE process.
Experimental Design Logic
This diagram outlines the specific logic and decision points within the experimental design and optimization phase.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for SFE Optimization
| Item | Function / Purpose | Example / Note |
|---|---|---|
| Supercritical CO₂ | Primary extraction solvent. Its solvent power is tunable with pressure and temperature. | Use high-purity CO₂ (99.95%). Food-grade is often suitable [41]. |
| Co-solvents (Modifiers) | Enhance the solubility of polar compounds by modifying the polarity of supercritical CO₂. | Ethanol is a common, green, and safe choice [57]. |
| Brine (NaCl solution) | Used in "salting out" to break emulsions in liquid-liquid extraction by increasing aqueous phase ionic strength [31]. | A saturated solution is often effective. |
| Phase Separation Filter Paper | A highly silanized paper used to physically separate organic and aqueous phases when emulsions form [31]. | Allows one phase to pass through while retaining the other. |
| Reference Standards | Critical for calibrating analytical instruments and quantifying target analytes in the extract. | Pure analytical standards of the target compounds (e.g., CBD, γ-oryzanol). |
| Solid Support for SLE | Provides a high-surface-area interface for emulsion-free extraction in Supported Liquid Extraction [31]. | Diatomaceous earth is commonly used. |
Addressing Challenges in Scale-up and Industrial Transfer
FAQs: Troubleshooting Scale-Up in Supercritical Fluid Extraction
This technical support guide addresses common challenges researchers and scientists face when scaling up Supercritical Fluid Extraction (SFE) processes, with a specific focus on managing modifier concentrations.
Pump Cavitation and Inefficient Operation During Extended Runs
- Question: During scale-up to a continuous process, our CO₂ pump begins to cavitate and operate inefficiently, failing to maintain system pressure. What is the cause and solution?
- Answer: Pump cavitation often occurs when liquid CO₂ flashes to gas in the pump head due to heat buildup from compression and piston friction. This is a significant risk in scaled-up operations with longer run times.
- Cause: The heat of compression causes the liquid CO₂ to vaporize, preventing the pump from efficiently moving the fluid.
- Solution: Use a chiller or recirculator to cool the pump head to approximately -5°C. This counteracts the heat and ensures only liquid CO₂ is pumped, eliminating cavitation. While helium headspace CO₂ tanks (at ~1500 psi) can also prevent this, they are significantly more expensive, making a chiller a more cost-effective long-term solution for industrial-scale operations [17] [26].
Maintaining Target Modifier Concentration in a Dynamic Flow System
- Question: How can we maintain a fixed percentage of a co-solvent (modifier), like ethanol, in the extraction vessel during dynamic flow in a large-scale system?
- Answer: Precise control is achieved by matching the co-solvent introduction rate to the volume of CO₂ flowing through the system.
- Methodology: Calculate the co-solvent volume needed as a percentage of the vessel's total volume. During dynamic extraction, use a mass flow meter to measure the CO₂ outflow. The co-solvent pump should then be set to deliver a volume of modifier equal to the chosen percentage of the CO₂ flow volume. This continuously replaces the co-solvent being flushed out, maintaining a constant concentration throughout the extraction [17].
Inconsistent Temperature Control at High Flow Rates
- Question: Why does the temperature in our large-scale extraction vessel fluctuate and become difficult to control accurately during high-flow-rate processing?
- Answer: Larger vessels have a small surface area relative to their volume, which limits the efficiency of external band heaters. At high flow rates, the incoming fluid can cause temperature instabilities.
- Solution: Install a fluid pre-heater to regulate the temperature of the CO₂ and co-solvent stream before it enters the main sample vessel. This ensures the fluid reaches the desired temperature and protects the main vessel's heaters from being overwhelmed, leading to more efficient and reproducible extractions [17] [26].
Achieving Selectivity for Polar Compounds with CO₂
- Question: Supercritical CO₂ is excellent for non-polar compounds, but our target analytes are polar. How can we adjust the process for better selectivity during scale-up?
- Answer: Pure supercritical CO₂ has dissolving power similar to hexane. To extract polar molecules, you must adjust the solvent's polarity.
- Primary Method: Use a co-solvent (modifier). Adding a small amount of a polar solvent like ethanol, methanol, or water significantly enhances the solvating power for polar compounds [17] [1] [53].
- Advanced Control: Precisely tune the temperature and pressure. Since the solvent power of supercritical CO₂ is directly related to its density, which is controlled by temperature and pressure, you can selectively extract specific compounds of interest by fine-tuning these parameters [17] [58].
Experimental Protocols & Optimization Data
Central Composite Face-Centered Design for SFE Optimization
Optimizing SFE parameters, including modifier concentration, is critical for scale-up. The following table summarizes a response surface methodology approach used to optimize the extraction of bioactive compounds from rice bran, balancing yield and bioactive concentration [41].
- Objective: To model the recovery of fatty acids and γ-oryzanol as a function of temperature and pressure.
- Design: Central Composite Face-Centered (CCFC) Design.
- Variables & Levels: Temperature and pressure were coded at three levels (-1, 0, +1).
Table 1: Experimental Design for SFE Optimization
| Experiment No. | Temperature (°C) | Pressure (bar) | Temperature (Coded) | Pressure (Coded) |
|---|---|---|---|---|
| 1 | 40 | 200 | -1 | -1 |
| 2 | 80 | 200 | +1 | -1 |
| 3 | 40 | 500 | -1 | +1 |
| 4 | 80 | 500 | +1 | +1 |
| 5 | 40 | 350 | -1 | 0 |
| 6 | 80 | 350 | +1 | 0 |
| 7 | 60 | 200 | 0 | -1 |
| 8 | 60 | 500 | 0 | +1 |
| 9 | 60 | 350 | 0 | 0 |
citation:4
Results and Workflow: The study identified optimal conditions of 500 bar and 62°C, achieving a mass yield of 17.3% with high concentrations of fatty acids (784.5 mg/g extract) and γ-oryzanol (36.6 mg/g extract). This SFE extract demonstrated higher selectivity for γ-oryzanol and greater efficacy in inhibiting cancer cell growth compared to a conventional n-hexane extract [41]. The logical workflow of this optimization is shown below.
Detailed SFE Protocol for Bioactive Recovery
The following methodology provides a detailed, scalable protocol for SFE, adaptable for modifier use [41].
- Apparatus: SFE system (e.g., Thar SFE-500F-2-C50) with a 500 mL extraction cell and at least one separator with independent temperature and pressure control.
- Raw Material Preparation: 20 g of rice bran (or target biomass) is loaded into the extraction vessel. Glass beads can be added to reduce dead volume and solvent consumption.
- Extraction Process:
- The vessel is pressurized with CO₂ to the target pressure (e.g., 200–500 bar) using a high-pressure pump (e.g., Thar SFC P-50).
- Liquid CO₂ is fed into the vessel at a constant flow rate (e.g., 15 g/min).
- The system is maintained at the target temperature and pressure for the duration of the extraction (e.g., 3 hours) in continuous (dynamic) mode.
- The CO₂ and dissolved extracts are expanded into a fraction collector. The pressure is reduced, causing the CO₂ to revert to gas and separate from the extract, which is collected.
- The CO₂ can be recycled in a closed-loop system [59].
Process Visualization: SFE System Workflow
The diagram below illustrates the key components and workflow of a typical SFE system, highlighting where scale-up challenges often occur.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials and Equipment for SFE Research and Scale-Up
| Item | Function & Relevance to Scale-Up |
|---|---|
| Supercritical CO₂ | Primary extraction solvent. It is GRAS (Generally Recognized as Safe), non-toxic, non-flammable, and leaves no solvent residue. Its tunable density is the key to selectivity [17] [1] [59]. |
| Co-solvents (Modifiers) | Ethanol, Methanol, Water. Used to increase the polarity of SC-CO₂, enabling the extraction of polar compounds. Ethanol is often preferred for food and pharmaceutical applications due to its safety profile [17] [1]. |
| Pneumatic Booster Pump | Critical for medium to large-scale processes. It compresses liquid CO₂ to the required system pressure (e.g., 5,000 psi). Pressure control is achieved via an air regulator [17] [26]. |
| Chiller/Recirculator | Essential for pump efficiency. Cools the pump head to prevent CO₂ from flashing to gas, thereby eliminating cavitation during long-duration, large-volume extractions [17] [26]. |
| Fluid Pre-Heater | Ensures accurate and consistent temperature control of the CO₂/modifier stream before it enters the main vessel, which is crucial for reproducibility in high-flow systems [17] [26]. |
| Back Pressure Regulator / Variable Restrictor | Maintains system pressure above the critical point. It controls the flow rate, and the pump actuates to maintain the set pressure as the restrictor is adjusted [17]. |
| Mass Flow Meter | Measures the real-time flow rate of CO₂. This is vital for dynamically controlling and maintaining the correct ratio of co-solvent modifier to CO₂ in the system [17]. |
The Impact of Modifiers on Post-Extraction Separation and Purification
Technical Support Center: Troubleshooting Guides and FAQs
Frequently Asked Questions
Q1: How does modifier concentration impact the final purity of my extract? Higher modifier concentrations can significantly increase extract purity by enhancing the solubility of target compounds. Research indicates that supercritical fluid extraction (SFE) typically achieves around 95% extract purity, compared to 70-80% with traditional solvent extraction methods [60]. However, excessive modifier can complicate subsequent purification by introducing more co-extracted compounds, requiring careful optimization.
Q2: Why is my post-extraction separation inefficient even with optimal modifier use? Inefficient separation often results from improper modifier selection for your target compounds. For polar compounds like flavonoids, ethanol concentrations of 70-90% are often effective [10]. If separation remains challenging, consider fractional separation with multiple separators in series, adjusting pressure and temperature between stages to selectively precipitate different compound fractions [1].
Q3: What are the practical considerations for using vegetable oil modifiers versus conventional solvents? Vegetable oils like peanut oil can dramatically increase extraction yields—up to three times for compounds like lutein and lycopene [61]. However, they introduce different post-extraction challenges as they remain in the extract, requiring additional separation steps compared to volatile modifiers like ethanol that evaporate more readily [61].
Q4: How can I minimize solvent residue in my final product while maintaining high yield? Supercritical CO₂ with ethanol as a modifier offers an excellent balance. Ethanol is generally recognized as safe (GRAS) and leaves minimal residue when properly separated. Studies show SFE reduces solvent usage by 80-90% compared to traditional methods, significantly reducing residual solvent concerns [60].
Troubleshooting Common Modifier-Related Issues
Problem: Poor extraction yield despite using modifiers
- Potential Cause: Incorrect modifier concentration or type for your target compound polarity
- Solution: Conduct preliminary tests with modifier concentrations from 5-15% for ethanol, or 30-60% for vegetable oils [10] [61]. For polar compounds, ethanol at 80% concentration often provides optimal results [10]
Problem: Co-extraction of unwanted compounds complicating purification
- Potential Cause: Modifier concentration too high, reducing selectivity
- Solution: Implement stepwise extraction with increasing modifier concentrations to selectively extract different compound classes [60]
Problem: Modifier contamination in final product
- Potential Cause: Inefficient separation of modifier from extract
- Solution: Optimize separator pressure and temperature parameters. For ethanol-modified SFE, gradual pressure reduction with intermediate separation stages improves modifier recovery [1]
Table 1: Modifier Optimization for Different Compound Classes
| Compound Class | Optimal Modifier | Concentration Range | Yield Improvement | Purity Achieved | Reference |
|---|---|---|---|---|---|
| Flavonoids (Hops) | Ethanol | 80% | 7.8 mg/g | Not specified | [10] |
| Tanshinones (Danshen) | Peanut Oil | 52% | 1.47-17.13 mg/g | Higher than traditional methods | [61] |
| Bioactive Compounds (Food By-products) | Ethanol | 5-15% | Not specified | ~95% | [60] |
| Polar Bioactives | Ethanol/Methanol | 5-20% | Significant for polar compounds | High with fractionation | [1] |
Table 2: Economic and Environmental Impact of Modifier Use
| Parameter | Traditional Solvent Extraction | SFE with Modifiers | Improvement | Reference |
|---|---|---|---|---|
| Solvent Usage | High | Reduced by 80-90% | Significant reduction | [60] |
| Energy Consumption | High | Reduced by 30-50% | Moderate reduction | [60] |
| Extraction Time | Longer (hours) | 25x faster than Soxhlet | Dramatic reduction | [62] |
| Solvent Residue | Concerning levels | Minimal to none | Significant improvement | [60] |
Experimental Protocols
Protocol 1: Optimizing Ethanol Modifier Concentration for Flavonoid Extraction
Objective: Determine optimal ethanol concentration for maximum flavonoid yield from plant materials.
Materials:
- Supercritical fluid extraction system with co-solvent pump
- Carbon dioxide (food grade)
- Anhydrous ethanol (analytical grade)
- Plant material (dried, ground, 40-mesh sieve)
- HPLC system for analysis
Methodology:
- Prepare plant material by drying (40°C, 48h), grinding, and sieving through 40-mesh screen
- Load extraction vessel with 10g plant material
- Set SFE base conditions: temperature 50°C, pressure 25 MPa, CO₂ flow rate 2 L/min
- Test ethanol concentrations: 70%, 80%, 90% (v/v) in separate runs
- Maintain static extraction for 10 minutes, followed by dynamic extraction for 90 minutes
- Collect extract in separator at reduced pressure (5 MPa) and temperature (25°C)
- Analyze flavonoid content using HPLC with appropriate standards
- Perform triplicate runs for each condition
Expected Outcomes: Maximum yield expected at 80% ethanol concentration with potential yield decrease at higher concentrations due to altered fluid critical properties [10].
Protocol 2: Using Vegetable Oil Modifiers for Enhanced Lipophilic Compound Recovery
Objective: Evaluate peanut oil as modifier for tanshinone extraction compared to conventional solvents.
Materials:
- SFE system with modifier capability
- Peanut oil (food grade)
- Danshen root powder (Salvia miltiorrhiza)
- Standard compounds (dihydrotanshinone, cryptotanshinone, tanshinone I, tanshinone IIA)
- HPLC system with UV detector
Methodology:
- Prepare danshen roots by drying and grinding to uniform particle size
- Optimize conditions using Response Surface Methodology (RSM) with three factors:
- Flow rate: 2-6 L/min
- Modifier concentration: 40-80%
- Pressure: 35-45 MPa
- Conduct extractions at predicted optimal conditions (approximately 3.23 L/min, 52%, 38.50 MPa)
- Compare results with traditional Soxhlet and ultrasonic extraction
- Quantify individual tanshinones using validated HPLC method
- Calculate extraction yields and compare methodologies
Expected Outcomes: SFE with peanut oil modifier expected to surpass traditional extraction methods in both efficiency and yield, requiring shorter extraction times [61].
Workflow Visualization
Modifier Selection Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Modifier-Enhanced SFE Research
| Reagent/Material | Function | Application Notes | Reference |
|---|---|---|---|
| Supercritical CO₂ | Primary extraction fluid | Non-toxic, non-flammable, critical point at 31°C/74.8 atm | [15] [17] |
| Ethanol (Pharmaceutical Grade) | Polar modifier | GRAS status, enhances polar compound solubility, use at 5-20% concentration | [60] [10] |
| Peanut Oil | Lipophilic modifier | Increases mass transfer of lipophilic compounds, use at 40-80% concentration | [61] |
| Methanol (HPLC Grade) | High-polarity modifier | For challenging polar compounds, requires careful post-extraction removal | [1] |
| Vegetable Oils (Various) | Alternative lipophilic modifiers | Soybean, canola, hazelnut oils for specific applications | [61] |
| Deionized Water | Co-modifier | With ethanol for highly polar compounds, modifies polarity | [10] |
| Back-Pressure Regulator | Pressure control | Maintains system above critical pressure during extraction | [17] |
| Fraction Separators | Product collection | Multiple vessels allow fractional separation by solubility | [1] |
Validation and Comparative Analysis: SFE with Modifiers vs. Conventional Techniques
Quantitative Yield and Efficiency Comparison
The following table summarizes key quantitative data comparing the performance of Supercritical Fluid Extraction (SFE) with modifiers against traditional Soxhlet and Solid-Liquid Extraction methods across various studies.
Table 1: Comparative Performance of SFE vs. Traditional Extraction Methods
| Extraction Method | Extract Purity | Solvent Usage | Energy Consumption | Extraction Time | Key Applications & Observations |
|---|---|---|---|---|---|
| SFE with Modifiers | Up to ~95% purity [60] | 80-90% reduction vs. traditional methods [60] | 30-50% reduction vs. traditional methods [60] | 50-70 min [63] [64] | Higher selectivity for target bioactives; cleaner extracts with less interference [63] [65]. |
| Soxhlet Extraction | ~70-80% purity [60] | High volumes of organic solvents [66] [60] | High (prolonged heating) [60] | Several hours to 18 hours [66] [65] | High gravimetric yield but less selective; darker, more complex extracts [63] [65]. |
| Solid-Liquid Extraction | Information Missing | High volumes of toxic solvents (e.g., methanol, acetone) [63] | Information Missing | Several hours or days [63] | Potential degradation of thermolabile compounds; poorer selectivity [63] [64]. |
Detailed Experimental Protocols
Optimized SFE Protocol for Bioactive Compounds
This protocol is adapted from the extraction of phenolic compounds from Lagerstroemia speciosa leaves [63].
- Equipment: SFE system equipped with a CO₂ pump, co-solvent pump, pre-heater, extraction vessel, pressure control restrictor, and collection vessel.
- Materials: Dried, ground plant material (500 µm particle size), CO₂ (99.9% purity), food-grade ethanol (modifier).
- Procedure:
- Sample Preparation: Load about 10 g of dried biomass into the extraction vessel. Use glass wool to pack both ends of the vessel to prevent clogging.
- System Pressurization & Heating: Pre-heat the system for 15 minutes. Pressurize the vessel with CO₂ and set the temperature.
- Dynamic Extraction:
- Collection: The extract is collected in an amber glass vial. Residual CO₂ evaporates at room temperature, leaving the extract ready for analysis [63].
Conventional Soxhlet Extraction Protocol
This protocol is based on classic Soxhlet extraction used for comparative studies [66] [65].
- Equipment: Soxhlet apparatus, extraction thimble, heat mantle, condenser.
- Materials: Dried sample, organic solvent (e.g., n-hexane, dichloromethane).
- Procedure:
- Sample Loading: Weigh 2-5 g of dried sample and place it in a cellulose extraction thimble.
- Apparatus Assembly: Place the thimble in the Soxhlet extractor. Fill the boiling flask with ~150 mL of solvent and assemble the apparatus.
- Extraction: Heat the solvent for 18 hours. The solvent vapor condenses, drips onto the sample, and the extracted compounds are siphoned back into the boiling flask.
- Concentration: After extraction, the solvent in the boiling flask is evaporated using a rotary evaporator to concentrate the extract [66] [65].
Troubleshooting Common SFE Issues
Table 2: Frequently Asked Questions and Troubleshooting for SFE
| Question / Issue | Probable Cause | Solution |
|---|---|---|
| Low extraction yield of target compound. | Incorrect pressure/temperature for compound solubility; lack of modifier for polar compounds. | Optimize pressure and temperature using experimental design (e.g., RSM). For polar compounds, add a polar modifier like ethanol (5-15%) [17] [64]. |
| The pump cavitates or operates inefficiently. | Liquid CO₂ flashing to gas in the pump head. | Ensure the pump head is cooled using a chiller to maintain liquid state of CO₂ [17]. |
| The system cannot maintain stable pressure during dynamic flow. | Inefficient heating or excessive flow rate. | Use a fluid pre-heater to regulate CO₂ temperature before it enters the extraction vessel [17]. |
| How can I selectively extract different compound classes from one sample? | Non-optimized, single-step extraction. | Use a sequential extraction strategy: first, extract non-polar compounds with pure SC-CO₂ at lower pressure, then extract polar compounds by adding a modifier and increasing pressure [60] [65]. |
| Extract collection problems or clogging in the restrictor. | Precipitated extract in the restrictor. | Heat the restrictor valve electrically (e.g., 20°C above extraction temperature) to prevent clogging [63]. |
Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for SFE Experiments
| Reagent / Material | Function in SFE | Application Notes |
|---|---|---|
| Supercritical CO₂ | Primary solvent; possesses gas-like diffusivity and liquid-like density. | GRAS (Generally Recognized As Safe), non-flammable, and easily separated from the extract. Its solvating power is tunable with pressure and temperature [17] [60]. |
| Ethanol (as modifier) | Polar co-solvent that increases the solubility of mid- to high-polarity compounds in SC-CO₂. | Commonly used at 5-15% concentration. It is non-toxic and GRAS, making it suitable for food and pharmaceutical applications [63] [17] [64]. |
| Diatomaceous Earth (for SLE) | Solid support for Supported Liquid Extraction, an alternative to LLE. | Provides a high-surface-area interface for partitioning analytes from an aqueous phase to an organic solvent, effectively preventing emulsion formation [31]. |
| Sodium Chloride (Brine) | "Salting out" agent to break emulsions in Liquid-Liquid Extraction. | Increases the ionic strength of the aqueous phase, forcing surfactant-like compounds to separate into one phase and breaking the emulsion [31]. |
Experimental Workflow and Optimization Strategy
The following diagrams illustrate the typical workflow for a comparative extraction study and the systematic approach to optimizing SFE parameters.
Diagram 1: Comparative Extraction Study Workflow
Diagram 2: SFE Parameter Optimization Strategy
This technical support center provides targeted guidance for researchers working to optimize modifier concentrations in Supercritical Fluid Extraction (SFE), with a specific focus on preserving thermolabile bioactive compounds.
Frequently Asked Questions (FAQs)
FAQ 1: Why is modifier concentration critical for extracting thermolabile compounds? The primary supercritical fluid, CO2, is non-polar and has limited ability to dissolve many polar bioactive compounds. Modifiers like ethanol alter the polarity of the supercritical solvent, thereby enhancing its solvating power for a wider range of molecules. More efficient extraction reduces the need for prolonged high-temperature exposure, which is a key factor in degrading heat-sensitive (thermolabile) compounds [27] [62]. Precise control over modifier concentration allows for selective and efficient extraction, minimizing the thermal stress on the target analytes.
FAQ 2: What is a typical starting point for ethanol modifier concentration? For initial method development, a concentration of 5-10% ethanol is a common and effective starting point. Research on hemp seed oil showed that 10% ethanol significantly boosted the yield of phenolic compounds without negatively affecting oil quality [27]. Furthermore, studies on marigold extraction have successfully used ethanol concentrations in the range of 15-30% (sometimes with added water) to sequentially extract compounds of different polarities [67].
FAQ 3: How does modifier concentration interact with other SFE parameters? Modifier concentration does not work in isolation; it has significant interactions with pressure and temperature. For instance, increasing the pressure generally enhances solubility by increasing fluid density, while temperature has a dual effect on vapor pressure and fluid density [62]. The optimal modifier level must be determined in conjunction with these other parameters, often through statistical experimental design, to find the true global optimum for your specific sample and target compounds [27] [67].
FAQ 4: Can I use a multi-step SFE process with different modifier concentrations? Yes, a sequential-selective SFE (S3FE) process is a powerful strategy. This involves first extracting non-polar compounds with pure or low-modifier CO2, followed by a second step with a higher modifier concentration to extract polar compounds. This approach was successfully used to recover both triterpendiol esters and the flavonoid narcissin from marigold flowers in a single, integrated process [67].
Troubleshooting Guides
Issue 1: Low Extraction Yield of Target Bioactive Compound
| Possible Cause | Diagnostic Steps | Corrective Action |
|---|---|---|
| Insufficient Modifier Polarity | Review logP of target compound; if polar, current modifier may be inadequate. | Increase ethanol concentration (e.g., from 5% to 10-15%) or consider ethanol/water mixtures for highly polar compounds [67]. |
| Suboptimal Pressure/Temperature | Check if fluid density is too low for solvation power. | Use experimental design (e.g., Box-Behnken) to simultaneously optimize pressure, temperature, and modifier percentage [27]. |
| Inadequate Extraction Time | Analyze kinetic data; yield may not have reached plateau. | Extend the static extraction time or the total process time in dynamic mode to ensure complete extraction. |
Issue 2: Degradation of Thermolabile Compounds in the Extract
| Possible Cause | Diagnostic Steps | Corrective Action |
|---|---|---|
| Excessive Temperature | Verify that set temperature does not exceed the known degradation point of the compound. | Lower the extraction temperature, even if it requires a compensatory increase in pressure or modifier concentration to maintain yield [62]. |
| Prolonged Extraction Time | Evaluate if the extraction time is longer than necessary for satisfactory yield. | Optimize the extraction time to the minimum required, as SFE is typically faster than traditional methods [62]. |
| Reactive Co-solvent | Confirm that the chosen modifier does not react with the target compound. | Switch to a greener, non-reactive solvent like ethanol, which is safe for food and pharmaceutical applications [27]. |
Issue 3: Poor Selectivity and Co-extraction of Unwanted Matrix Components
| Possible Cause | Diagnostic Steps | Corrective Action |
|---|---|---|
| Excessively High Modifier Concentration | The modifier is too strong, dissolving a wide range of compounds. | Reduce the modifier percentage or employ a sequential extraction strategy, starting with a low-modifier step to remove non-polar interferents first [67]. |
| Incorrect Particle Size | Fines may cause channeling or excessive surface area contact. | Optimize the plant material's particle size (e.g., ~500 μm) to ensure efficient and selective contact with the fluid [27]. |
Experimental Protocol: Optimizing Modifier Concentration for Hemp Seed Oil
This detailed protocol is adapted from a study that successfully enhanced the recovery of phenolic compounds in hemp seed oil using ethanol-modified supercritical CO2 [27].
1. Aim: To maximize the yield and bioactive compound content (e.g., total phenols, tocopherols) of hemp seed oil by optimizing SFE parameters, with a focus on ethanol modifier concentration.
2. Experimental Design and Setup
- Design: Employ a Response Surface Methodology (RSM) with a Box-Behnken Design (BBD). This statistical approach efficiently models the interaction between multiple variables [27].
- Variables and Ranges:
- Temperature: 30 - 60 °C
- Pressure: 10 - 20 MPa
- Time: 120 - 300 min
- Ethanol Modifier: 2.5% - 20% (v/v)
- Material Preparation: Hemp seeds should be crushed and sieved to a uniform particle size of 500 μm [27].
- SFE Setup: The CO2 fluid flow rate should be maintained at a constant 0.25 kg/h throughout the experiments [27].
3. Optimization and Analysis
- Execution: Run the extractions as per the BBD experimental matrix.
- Analysis of Responses: For each experimental run, measure the following responses:
- Oil yield (g/100 g fresh seeds)
- Total Phenolic Content (TPC, mg GAE/kg oil)
- Total tocopherols (mg/kg oil)
- Oxidative Stability Index (OSI, hours)
- Identification: Use HPLC-DAD/ESI-MS2 to identify and quantify individual phenolic compounds in the optimized extract [27].
4. Expected Outcome: Under the identified optimal conditions (e.g., 50°C, 20 MPa, 244 min, 10% ethanol), the study achieved an oil yield of 30.13%, with a TPC of 294.15 mg GAE/kg and total tocopherols of 484.38 mg/kg [27].
Research Reagent Solutions
This table lists key materials and reagents essential for SFE experiments focused on modifier optimization.
| Item | Function / Relevance in SFE |
|---|---|
| Supercritical CO2 | The primary extraction fluid. It is non-toxic, non-flammable, and leaves no solvent residue, making it ideal for high-purity extracts [62]. |
| Ethanol (Food Grade) | The most common green modifier. It is safe, tunable, and effectively increases the solubility of polar bioactive compounds in supercritical CO2 [27] [67]. |
| Plant Material (e.g., Hemp Seed) | The source of target bioactive compounds. Must be prepared with a controlled and uniform particle size (e.g., 500μm) for reproducible extractions [27]. |
| Response Surface Methodology Software | Statistical software (e.g., Design-Expert, Minitab) is crucial for designing experiments and modeling complex parameter interactions to find optimal conditions [27]. |
| HPLC-DAD/ESI-MS2 System | Used for the precise identification and quantification of individual bioactive compounds in the final extract, confirming the success of the extraction protocol [27]. |
SFE Modifier Optimization Workflow
The diagram below outlines the logical workflow for developing an SFE method optimized for thermolabile compounds.
SFE Parameter Relationships
This diagram illustrates how key SFE parameters interact to influence the final outcome of the extraction process, which is central to troubleshooting and optimization.
Troubleshooting Guides
Troubleshooting Liquid-Liquid Extraction in Sample Preparation
Problem: Emulsion Formation during Liquid-Liquid Extraction (LLE) Emulsion formation is a common issue that can halt extraction progress, trap analytes, and lead to quantitative errors [31].
- Prevention: Gently swirl the separatory funnel instead of shaking vigorously to reduce agitation while maintaining extraction surface area [31].
- Resolution Techniques:
- Salting Out: Add brine or salt water to increase the ionic strength of the aqueous layer, forcing surfactant-like molecules to separate into one phase [31].
- Filtration: Pass the emulsion through a glass wool plug or a phase separation filter paper (highly silanized) to isolate the desired layer [31].
- Centrifugation: Use centrifugation to isolate the emulsion material in the residue [31].
- Solvent Adjustment: Add a small amount of a different organic solvent to adjust solvent properties and break the emulsion [31].
- Alternative Technique: For samples prone to emulsions, use Supported Liquid Extraction (SLE). The aqueous sample is applied to a solid support (e.g., diatomaceous earth), creating an interface for extraction with an organic solvent percolating through, which prevents emulsion formation [31].
Problem: Analyte Loss or Low Recovery
- Strong Adsorption to Particulates: Filter samples through a fine filter or centrifuge to remove particulates before extraction.
- Protein Binding: For samples like plasma, consider protein precipitation prior to LLE to release protein-bound drugs [31].
Troubleshooting Supercritical Fluid Extraction (SFE) Modifier Optimization
Problem: Low Yield of Target Bioactive Compounds This often indicates the supercritical CO₂'s solvating power is insufficient for the target polar compounds.
- Solution: Optimize Co-solvent (Modifier) Concentration
- Purpose: Modifiers like ethanol or methanol enhance the solubility of polar compounds (e.g., polyphenols) in supercritical CO₂ [17] [68].
- Method: A co-solvent pump is used to introduce the modifier. It can be added to the sample vessel before extraction or pumped simultaneously with CO₂ at a set ratio [17].
- Optimization Strategy: To maintain a fixed modifier percentage (e.g., 5% ethanol) during dynamic flow, calculate 5% of the vessel's volume. Use a mass flow meter to gauge CO₂ outflow and add back ethanol at 5% of this volume to maintain the concentration [17].
Problem: Poor Extraction Efficiency for Polar Compounds
- Check Modifier Type and Polarity: Neat supercritical CO₂ has dissolving properties similar to hexane. For polar molecules, use polar modifiers like ethanol, methanol, or water [17] [68].
- Optimize Pressure and Temperature: Solubility in SFE increases with pressure (due to higher fluid density) and can be manipulated with temperature. Use a pre-heater for accurate temperature control, especially at high flow rates [53] [17].
Problem: Inconsistent Results Between Extractions
- Ensure Proper System Equilibration: Always use a pre-heater to regulate the temperature of CO₂ and co-solvent before they enter the main vessel, ensuring consistent and reproducible extraction conditions [17].
- Monitor Pump Performance: Ensure the pump head is cooled with a chiller to prevent CO₂ from flashing to gas, which causes cavitation and inefficient pumping [17].
Frequently Asked Questions (FAQs)
Q1: What are the core advantages of using SFE over traditional solvent extraction for bioactivity studies?
- Selectivity: The solvating power of supercritical CO₂ can be finely tuned by adjusting pressure and temperature, allowing selective extraction of target compounds [53] [69] [68].
- Environmental Friendliness: SFE uses non-toxic, non-flammable CO₂, eliminating the need for hazardous organic solvents. CO₂ is easily recycled and leaves no toxic residues [53] [69] [68].
- Mild Conditions: SFE operates at relatively low temperatures (e.g., 31-60°C for CO₂), preserving heat-sensitive bioactive compounds from degradation [53] [69].
- Residue-Free Extracts: Supercritical CO₂ reverts to gas upon depressurization, yielding pure, solvent-free extracts critical for pharmaceutical and food applications [69] [68].
Q2: Why is carbon dioxide the most common solvent in SFE? CO₂ is preferred because it is inexpensive, non-toxic, non-flammable, and readily available in high purity. Its critical temperature (31.1°C) and pressure (73.8 bar) are mild, making it suitable for heat-sensitive compounds. It is also approved by regulatory bodies (FDA, GRAS) for use in food and pharmaceutical products [53] [17] [68].
Q3: When and why are modifiers (co-solvents) used in SFE? Modifiers are used to increase the solubility of polar compounds in supercritical CO₂. Neat CO₂ is excellent for non-polar compounds, but its polarity can be enhanced by adding small amounts (typically 1-15%) of polar solvents like ethanol or methanol. This expands SFE's application to a wider range of bioactive compounds, such as polyphenols and glycosides [17] [68].
Q4: What are the main disadvantages of SFE technology?
- High Initial Investment: SFE equipment is complex and more expensive than traditional solvent extraction systems [69] [68].
- High-Pressure Operation: Requires specialized equipment and training for safe operation [68].
- Limited Mass Transfer: The low viscosity of supercritical fluids can sometimes limit mass transfer rates, potentially leading to longer extraction times for some matrices [69].
Experimental Protocols for Key Bioassays
Antioxidant Activity Assays
DPPH (2,2-Diphenyl-1-picrylhydrazyl) Radical Scavenging Assay
- Principle: Measures the ability of antioxidants to donate hydrogen, reducing the stable purple DPPH radical to a yellow-colored diphenylpicrylhydrazine. The degree of discoloration correlates with antioxidant potency.
- Protocol Summary:
- Prepare serial dilutions of the SFE extract.
- Add a known volume of each dilution to a methanolic solution of DPPH.
- Incubate the mixture in the dark at room temperature for 30 minutes.
- Measure the absorbance of the solution at 517 nm.
- Calculate the percentage of DPPH scavenging activity and determine the IC₅₀ value (concentration required to scavenge 50% of DPPH radicals) [70] [71].
FRAP (Ferric Reducing Antioxidant Power) Assay
- Principle: Measures the reduction of ferric iron (Fe³⁺) to ferrous iron (Fe²⁺) by antioxidants in an acidic medium, forming a blue-colored complex.
- Protocol Summary:
- Prepare the FRAP reagent (acetate buffer, TPTZ solution, and FeCl₃ solution).
- Mix the SFE extract with the FRAP reagent and incubate.
- Measure the absorbance at 593 nm.
- Quantify antioxidant potential against a standard curve of ferrous sulfate (FeSO₄) or Trolox [71].
ABTS (2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) Assay
- Principle: Measures the ability of antioxidants to scavenge the pre-formed ABTS⁺ radical cation, which has a characteristic blue-green color.
- Protocol Summary:
- Generate the ABTS⁺ radical by reacting ABTS solution with potassium persulfate and incubating in the dark.
- Dilute the ABTS⁺ solution to a specific absorbance at 734 nm.
- Mix the SFE extract with the diluted ABTS⁺ solution and incubate.
- Measure the decrease in absorbance at 734 nm and calculate the Trolox Equivalent Antioxidant Capacity (TEAC) [70].
Table 1: Summary of Key Antioxidant Assays
| Assay | Mechanism | Key Reagents | Detection Wavelength | Example IC₅₀ from Literature |
|---|---|---|---|---|
| DPPH | Radical Scavenging | DPPH radical in methanol | 517 nm | 49.4 μg/mL (Prionosciadium dissectum callus methanolic extract) [70] |
| FRAP | Reductive Potential | TPTZ, FeCl₃, Acetate buffer | 593 nm | Reported as FRAP value (e.g., µM Fe²⁺/g) [71] |
| ABTS | Radical Scavenging | ABTS, Potassium Persulfate | 734 nm | 10.0 μg/mL (Prionosciadium dissectum callus methanolic extract) [70] |
Anti-inflammatory Activity Assays
Nitric Oxide (NO) Inhibition Assay in RAW 264.7 Macrophages
- Principle: Lipopolysaccharide (LPS)-stimulated macrophages produce excess nitric oxide (NO), a key inflammatory mediator. This assay measures the extract's ability to inhibit NO production.
- Detailed Protocol:
- Cell Culture: Maintain RAW 264.7 murine macrophage cells in DMEM medium supplemented with 10% Fetal Bovine Serum (FBS) [71].
- Cell Viability Assessment (MTT Assay): Seed cells in a 96-well plate. Treat with a range of SFE extract concentrations (e.g., 5–100 µg/mL) for 24 hours. Add MTT reagent and incubate. Dissolve the formed formazan crystals with DMSO and measure absorbance at 570 nm to ensure tested concentrations are non-cytotoxic [71].
- NO Production Inhibition: Seed cells and pre-treat with non-cytotoxic concentrations of the SFE extract for a set time (e.g., 1-2 hours). Stimulate inflammation by adding LPS (e.g., 1 µg/mL) and incubate for 18-24 hours [70] [71].
- NO Measurement (Griess Reaction): Mix cell culture supernatant with an equal volume of Griess reagent (sulfanilamide and NEDD). Incubate and measure absorbance at 540 nm [71].
- Data Analysis: Calculate NO concentration from a sodium nitrite standard curve. Determine the IC₅₀ value for NO inhibition [71].
Gene and Protein Expression Analysis of Inflammatory Markers
- Principle: Confirms the anti-inflammatory mechanism by quantifying the effect of the extract on the expression of key inflammatory genes and proteins.
- Protocol Summary:
- Treatment: Treat LPS-stimulated RAW 264.7 cells with the SFE extract.
- mRNA Analysis (qRT-PCR): Extract total RNA, reverse transcribe to cDNA, and perform quantitative PCR to measure mRNA levels of iNOS, COX-2, IL-6, and TNF-α [71].
- Protein Analysis (Western Blot): Lyse treated cells, separate proteins via SDS-PAGE, transfer to a membrane, and probe with antibodies against iNOS and COX-2 proteins to assess protein expression levels [71].
Table 2: Summary of Key Anti-inflammatory and Antiproliferative Assays
| Assay Type | Model System | Key Measured Endpoints | Example IC₅₀ / Result from Literature |
|---|---|---|---|
| Anti-inflammatory | RAW 264.7 Macrophages | Inhibition of NO production | 24.4 μg/mL (Belog Plus polyherbal extract) [71] |
| Anti-inflammatory | RAW 264.7 Macrophages | Inhibition of COX-1 / LOX activity | 97 kIU/g d.w. (COX-1), 720 kIU/g d.w. (LOX) (Turkey tail microcapsules) [72] |
| Gene/Protein Expression | RAW 264.7 Macrophages | Down-regulation of iNOS, COX-2, IL-6, TNF-α mRNA and protein | Significant reduction at 6.25–25 µg/mL (Belog Plus extract) [71] |
| Antiproliferative | HT-29 Human Colorectal Cancer Cells | Cell viability inhibition (MTT assay) | High sensitivity reported (Turkey tail microcapsules) [72] |
Antiproliferative Activity Assay
MTT Cell Viability Assay in Cancer Cell Lines
- Principle: Measures the reduction of yellow MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) to purple formazan by metabolically active cells. The amount of formazan produced is proportional to the number of viable cells.
- Protocol Summary:
- Cell Seeding: Seed human cancer cell lines (e.g., HT-29 colorectal adenocarcinoma) in a 96-well plate and allow to adhere [72] [70].
- Treatment: Treat cells with a range of SFE extract concentrations (e.g., up to 100 µg/mL) for a defined period (e.g., 48 hours) [70].
- MTT Incubation: Add MTT solution to each well and incubate to allow formazan crystal formation.
- Solubilization: Remove the medium and dissolve the formazan crystals in DMSO.
- Absorbance Measurement: Measure the absorbance at 570 nm. Calculate the percentage of cell viability and the IC₅₀ value (concentration that inhibits cell viability by 50%) [70].
Signaling Pathways in Bioactivity
Experimental Workflow for SFE Bioactivity Validation
Research Reagent Solutions
Table 3: Essential Reagents and Materials for Bioactivity Validation
| Category | Reagent / Material | Function / Application | Key Details / Examples |
|---|---|---|---|
| Extraction | Supercritical CO₂ | Primary solvent for SFE; non-toxic, tunable solvation power. | Critical point: 31.1°C, 73.8 bar [53]. |
| Extraction | Modifiers (Co-solvents) | Enhance solubility of polar compounds in SFE. | Ethanol, Methanol (common, food-grade); typically 1-15%[v citation:5] [68]. |
| Cell Culture | RAW 264.7 Cells | Murine macrophage cell line for anti-inflammatory assays. | Used for NO production and expression analysis of iNOS, COX-2, IL-6, TNF-α [70] [71]. |
| Cell Culture | HT-29 Cells | Human colorectal cancer cell line for antiproliferative assays. | A common model for screening anticancer properties [72]. |
| Cell Culture | DMEM / RPMI 1640 Media | Cell culture growth medium. | Supplemented with Fetal Bovine Serum (FBS) [71]. |
| Assay Kits & Reagents | DPPH Radical | Free radical for antioxidant scavenging assays. | Dissolved in methanol; absorbance measured at 517nm [70] [71]. |
| Assay Kits & Reagents | Griess Reagent | Detects Nitric Oxide (NO) as nitrite concentration. | Used in anti-inflammatory assays with RAW 264.7 cells; absorbance at 540nm [71]. |
| Assay Kits & Reagents | MTT Reagent | Measures cell viability and proliferation. | Converted to formazan by living cells; absorbance at 570nm [70] [71]. |
| Assay Kits & Reagents | LPS (Lipopolysaccharide) | Potent inflammatory stimulator for macrophages. | Used to induce NO and inflammatory cytokine production in anti-inflammatory assays [71]. |
Frequently Asked Questions (FAQs)
Q1: What is the primary economic advantage of replacing traditional organic solvents with supercritical CO₂ in extraction processes? The primary economic advantage stems from significant operational savings and environmental cost avoidance. Supercritical CO₂ (scCO₂) is non-toxic, readily available, and can be recovered and reused in a closed-loop system, drastically reducing solvent consumption and waste disposal costs associated with hazardous organic solvents [59] [30]. Furthermore, it eliminates the expense of removing toxic solvent residues from final products, a critical requirement in the pharmaceutical and food industries [59].
Q2: How does the use of a modifier like ethanol in scCO₂ extraction align with the goal of reducing hazardous solvents? While the core goal is to reduce hazardous solvents, the selective use of green modifiers like ethanol makes the process more universally applicable. Ethanol is considered environmentally benign, safe for human health, and is approved for use in food and pharmaceutical products [10] [1]. By using scCO₂ modified with ethanol, researchers can extract a wider range of polar compounds without resorting to more toxic solvents like methanol or hexane, thus maintaining a favorable environmental and safety profile [10] [20].
Q3: A common issue in SFE is low extraction yield for target polar compounds. Is this a failure of the scCO₂ system, and how can it be corrected? Low yield for polar compounds is not a system failure but a reflection of the non-polar nature of pure scCO₂ [20]. This issue can be corrected by systematically optimizing the concentration of a polar co-solvent, or modifier. Ethanol is the preferred choice for this purpose. The modifier increases the polarity of the supercritical fluid, enhancing the solubility of polar target compounds like flavonoids or polyphenols [10] [1]. Optimization experiments should test different ethanol concentrations (e.g., 70%, 80%, 90%) to identify the level that maximizes yield without compromising the extract's purity or the process's green credentials [10].
Q4: During method development, my extracts show undesirable co-extraction of impurities. How can I improve selectivity? Selectivity can be finely tuned by manipulating SFE parameters. You can leverage a fractional separation strategy by connecting multiple separators in series. By setting different pressures and temperatures in each separator, you can precipitate different compound fractions based on their solubility [1]. Additionally, carefully adjusting the density of the scCO₂ by changing the pressure and temperature allows for selective extraction. Starting at lower pressures can help extract volatile oils first, followed by higher pressures for lipids, reducing co-extraction from the outset [20].
Troubleshooting Guides
Issue: Inconsistent Extraction Yields Despite Fixed Parameters
Possible Causes and Solutions:
Cause 1: Uncontrolled Particle Size of Biomass.
- Solution: Ensure the raw biomass is ground and sieved to a consistent particle size (e.g., ≤ 0.42 mm, 40-mesh) [10]. A uniform particle size ensures reproducible mass transfer and diffusion rates.
Cause 2: Fluctuations in Modifier Delivery.
Cause 3: Incomplete Extraction of the Modifier-analyte Complex.
- Solution: If using a static modifier introduction method, ensure an adequate equilibration time to allow the modifier to swell the matrix and interact with the analyte. Alternatively, use a dynamic mode where the modified CO₂ continuously flows through the sample [1].
Issue: Blockages in the Restrictor or Back Pressure Regulator
Possible Causes and Solutions:
Cause 1: Precipitation of Extracted Material or Ice.
- Solution: This is often caused by the significant cooling from adiabatic expansion of CO₂ [20]. Ensure the restrictor or back pressure regulator is properly heated. Use an electrically heated jacket or a hairdryer for analytical-scale systems to prevent the freezing of water or the precipitation of heavy compounds.
Cause 2: Presence of Waxes or Lipids in the Extract.
- Solution: Implement a pre-extraction step using pure scCO₂ at a specific pressure to remove non-polar lipids and waxes before adding the modifier for the target polar compounds [20]. This "defatting" step prevents these compounds from causing blockages later.
The table below summarizes key experimental data from orthogonal tests on optimizing ethanol-modified scCO₂ for flavonoid extraction from waste hops. This provides a reference for designing your experiments [10].
Table 1: Optimal Conditions for SFE of Flavonoids from Waste Hops
| Factor | Level 1 | Level 2 | Level 3 | Optimal Value |
|---|---|---|---|---|
| Extraction Temperature (°C) | 40 | 50 | 60 | 50 |
| Extraction Pressure (MPa) | 20 | 25 | 30 | 25 |
| Amount of Modifier (wt%) | 30 | 40 | 50 | 50 |
| Ethanol Concentration (%) | 70 | 80 | 90 | 80 |
Reported maximum yield under optimal conditions: 7.8 mg/g [10].
Detailed Experimental Protocol: Optimizing Ethanol Modifier Concentration
This protocol is adapted from a study optimizing the extraction of flavonoids from waste hops using scCO₂ with ethanol as a modifier [10].
1. Objective: To determine the optimal concentration of ethanol modifier in scCO₂ for maximizing the yield of target bioactive compounds (e.g., flavonoids).
2. Materials and Reagents:
- Biomass: SC-CO2 extracted waste hops, pulverized and sieved (40-mesh, Dp ≤ 0.42 mm).
- Solvent: Food-grade carbon dioxide (CO₂).
- Modifier: Anhydrous ethanol (food grade or HPLC grade).
- Apparatus: Supercritical fluid extraction system equipped with a co-solvent pump, refrigeration unit, 1-5 L extraction vessel, and separators.
3. Methodology:
- Sample Preparation: Load a known mass of pre-processed biomass (e.g., 100g) into the extraction vessel.
- Fixed Parameters: Set the following baseline conditions for all experimental runs:
- Extraction Time: 90 minutes.
- Extraction Temperature: 50 °C.
- Extraction Pressure: 25 MPa.
- CO₂ Flow Rate: Maintain a constant mass flow rate.
- Variable Parameter (Ethanol Concentration): Perform a series of extractions where the concentration of ethanol in the modifier stream is varied. Test at least three levels, for example: 70%, 80%, and 90% (v/v) aqueous ethanol.
- Modifier Delivery: Use the duplex pump to introduce the ethanol modifier at a fixed ratio to the biomass (e.g., 50% w/w of the sample mass).
- Extraction and Collection: Initiate the dynamic extraction process. The mixture of extract and solvent is passed into a separator where pressure is reduced, causing the extract to precipitate. Collect the extract from the separator.
- Analysis: Weigh the extracted mass. Analyze the extract for your target compounds using an appropriate analytical method (e.g., HPLC-MS for flavonoids) [10].
4. Data Analysis: Plot the yield of the target compound(s) against the ethanol concentration. The concentration that yields the highest recovery is considered optimal for that specific matrix and target.
Workflow for Modifier Optimization
The diagram below outlines the logical workflow for systematically optimizing modifier concentration in SFE research.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents for SFE with Modifier Optimization
| Item | Function/Explanation | Example/Specification |
|---|---|---|
| Supercritical CO₂ | Primary extraction solvent; non-toxic, tunable, and leaves no residue [59] [30]. | Food-grade or 99.99% purity. |
| Ethanol (Modifier) | Polar co-solvent that enhances the solubility of medium to high polarity compounds in scCO₂ [10] [20]. | Anhydrous, ACS grade or higher. |
| Biomass | The raw material containing the target compounds. | Dried, ground, and sieved to a consistent particle size (e.g., 40-mesh) [10]. |
| Analytical Standards | Used for calibrating instruments to identify and quantify target compounds in the extract [10]. | e.g., Xanthohumol for hops analysis. |
| HPLC-MS System | For analyzing extract composition, verifying compound identity, and assessing purity [10]. | Equipped with C18 column and APCI or ESI source. |
Frequently Asked Questions (FAQs)
FAQ 1: What is the primary advantage of using supercritical CO₂ over traditional organic solvents like n-hexane? Supercritical CO₂ is considered a green solvent because it is non-toxic, non-flammable, and leaves no harmful solvent residues in the final extract. It replaces solvents like n-hexane, resulting in cleaner extracts and a reduced environmental impact. The solvating power of supercritical CO₂ can be precisely tuned by adjusting pressure and temperature, allowing for highly selective extractions that are difficult to achieve with conventional solvents. [53] [17]
FAQ 2: When and why should I use a co-solvent like ethanol in SFE? Neat supercritical CO₂ has dissolving properties similar to hexane, making it ideal for non-polar compounds. A co-solvent (or modifier) like ethanol is used to increase the solubility of more polar molecules. The addition of even a small amount of co-solvent can significantly enhance the extraction yield of polar compounds, such as certain phenolic acids and flavonoids, which would not be efficiently extracted with CO₂ alone. [17] [73]
FAQ 3: How do I maintain a fixed co-solvent concentration during a dynamic extraction? To maintain a fixed percentage of co-solvent (e.g., ethanol) in the extraction vessel during dynamic flow, you must replace the co-solvent that is flushed out by the flowing CO₂. A simple method is to use a mass flow meter to gauge the volume of CO₂ leaving the vessel. Your co-solvent pump should then be set to add back a volume of ethanol equal to the desired percentage of the CO₂ flow volume. For example, to maintain a 5% co-solvent level, you would add 5 mL of ethanol for every 100 mL of CO₂ that flows out. [17]
FAQ 4: What are common causes of low extraction yield, and how can I troubleshoot them? Low yields can often be attributed to:
- Insufficient Solvating Power: The density of supercritical CO₂, which governs its solvating power, is highly dependent on pressure and temperature. An increase in pressure at constant temperature increases fluid density and solute solubility. Ensure your operating pressure is high enough for your target compounds. [53]
- Mass Transfer Limitations: The unique properties of SCFs (low viscosity, high diffusivity) generally enhance mass transfer, but the matrix of the raw material can still pose a barrier. Grinding the material to increase surface area can improve recovery. [74]
- Incorrect Polarity: For polar analytes, not using a co-solvent, or using an insufficient amount, can lead to poor recovery. Consider adding or increasing the percentage of a polar co-solvent like ethanol. [17] [73]
FAQ 5: Why is a pre-heater recommended for the CO₂ fluid before it enters the extraction vessel? A pre-heater is crucial for maintaining accurate and stable temperatures during extraction, especially at high flow rates. Without a pre-heater, the incoming liquid CO₂ can cool the system, making it difficult for the vessel heaters alone to maintain the set temperature. This temperature fluctuation can affect the density of the supercritical fluid and lead to irreproducible results. A pre-heater ensures the CO₂ and any co-solvent reach the desired temperature before contacting the sample. [17]
Troubleshooting Guides
Problem 1: Emulsion Formation During Extract Collection
- Description: Emulsions can form during the collection of the extract, particularly when the sample matrix is high in surfactant-like compounds (e.g., phospholipids, free fatty acids, or proteins). This creates a stable mid-zone that prevents clean separation of phases. [31]
- Solutions:
- Prevention during SFE: Gently swirl the collection vessel instead of using vigorous shaking to reduce agitation.
- Salting Out: Add brine or salt water to the collection mixture. This increases the ionic strength of the aqueous layer and can break the emulsion by forcing surfactant molecules into one phase. [31]
- Filtration or Centrifugation: Pass the mixture through a phase separation filter paper or a glass wool plug. Alternatively, centrifugation can isolate the emulsion material in the residue. [31]
- Solvent Adjustment: Add a small amount of a different organic solvent (e.g., ethanol or ethyl acetate) to adjust the solvent properties and break the emulsion. [31]
Problem 2: Poor Selectivity for Target Compounds
- Description: The extract contains a wide range of compounds instead of being enriched in the desired bioactive molecules.
- Solutions:
- Optimize Pressure and Temperature: Systematically adjust pressure and temperature to manipulate the density and solvating power of the supercritical CO₂. Higher pressures generally increase density and improve the solubility of heavier molecules. [4] [53]
- Implement a Two-Step Extraction: Use a comprehensive two-step SFE process. The first step uses neat CO₂ or CO₂ with a low percentage of co-solvent (e.g., 2%) to extract non-polar compounds (e.g., volatile terpenes, lipids). The conditions are then automatically switched to a higher co-solvent percentage (e.g., 44%) in the second step to extract more polar compounds (e.g., flavonoids, phenolic acids). This yields two distinct, selective fractions within a single run. [73]
- Review Co-solvent Choice: Ensure the co-solvent's polarity matches that of your target compounds. Ethanol is a common, food-safe choice for medium-polarity molecules.
Problem 3: System Pressure Cannot Be Maintained
- Description: The pump struggles to maintain the set pressure during dynamic flow, or the system experiences pressure drops.
- Solutions:
- Check for Blockages: Inspect the variable restrictor (or back-pressure regulator) and tubing for particulates or waxy extracts that may be causing a blockage.
- Verify Pump Cooling: Ensure the pump head chiller is functioning correctly. Inefficient cooling can cause liquid CO₂ to flash to gas in the pump head (cavitation), leading to poor pumping efficiency and pressure instability. [17]
- Confirm CO₂ Supply: Check that your CO₂ tank has adequate pressure and volume. A helium headspace tank or a properly functioning chiller is necessary to prevent cavitation by ensuring the CO₂ remains in a liquid state as it enters the pump. [17]
Experimental Protocols & Data
Protocol: Optimizing SFE for γ-Oryzanol from Rice Bran
This methodology is adapted from a published study on recovering γ-oryzanol-rich extracts with improved bioactivity. [4]
1. Objective: To identify optimal SFE conditions (pressure and temperature) for the selective extraction of γ-oryzanol and fatty acids from rice bran, and to compare the bioactivity of the SFE extract with a conventional n-hexane extract.
2. Materials and Equipment:
- Raw Material: Rice bran (e.g., Japônica variety). Store at -20°C protected from light until use. [4]
- Equipment: Supercritical fluid extraction system (e.g., 500 mL extraction cell), CO₂ source (99.95% purity), co-solvent pump, pre-heater, chilled collection vessel. [4]
- Chemicals: n-hexane (for conventional extraction), ethanol (as a potential co-solvent), analytical standards (e.g., γ-oryzanol, fatty acids). [4]
3. Experimental Design:
- A Central Composite Face-Centered Design (CCF) was employed to optimize the process. [4]
- Independent Variables:
- Pressure: 200 - 500 bar
- Temperature: 40 - 80 °C
- Dependent Variables (Responses): Global mass yield, concentration of γ-oryzanol (mg/g extract), concentration of fatty acids (mg/g extract).
4. Methodology:
- Sample Preparation: Load 20 g of rice bran into the extraction vessel. Add glass beads to reduce dead volume. [4]
- Extraction Procedure:
- Set the CO₂ flow rate to 15 g/min. [4]
- Pressurize the vessel and set the temperature according to the experimental design.
- Conduct a 3-hour continuous (dynamic) extraction.
- Collect the extract in a chilled vessel.
- Weigh the extract to determine the global yield.
- Analysis: Analyze the extracts for γ-oryzanol and fatty acid content using appropriate analytical techniques (e.g., HPLC, GC).
- Bioactivity Assay: Evaluate the antioxidant activity (e.g., by measuring the reduction of reactive oxygen species in Caco-2 cells) and antiproliferative activity (e.g., against colorectal cancer cells) of the optimal SFE extract and a traditional n-hexane extract. [4]
5. Key Results and Optimal Conditions: The study identified the optimal extraction conditions at 500 bar and 62 °C. The results from the optimal SFE extraction compared to conventional n-hexane extraction are summarized below: [4]
Table 1: Comparison of SFE and n-Hexane Extracts from Rice Bran
| Parameter | n-Hexane Extraction | SFE (Optimal Conditions) |
|---|---|---|
| Global Yield (%) | 18.0% | 17.3% |
| γ-Oryzanol (mg/g extract) | 18.0 | 36.6 |
| γ-Oryzanol (mg/g rice bran) | 3.3 | 6.3 |
| Fatty Acids (mg/g extract) | 784.5 | 784.5 (approximate, based on 135.70 mg/g RB and 17.3% yield) |
| Fatty Acids (mg/g rice bran) | 130.14 | 135.70 |
| Antiproliferative Activity (EC₅₀) | 1.15 mg/mL | 0.9 mg/mL |
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for SFE Experimentation
| Item | Function/Explanation |
|---|---|
| Supercritical CO₂ | The primary extraction fluid. GRAS (Generally Recognized As Safe), non-flammable, and leaves no toxic residue. Its solvating power is tunable. [53] [17] |
| Co-solvents (e.g., Ethanol, Methanol) | Polar modifiers added to CO₂ to enhance the extraction efficiency of mid- to high-polarity compounds. Ethanol is preferred for food and pharmaceutical applications due to its safety profile. [17] [73] |
| Analytical Standards | Pure compounds (e.g., γ-oryzanol, CBD, THC, phenolic acids) used for quantification and method validation via calibration curves. [4] [54] |
| Diatomaceous Earth (for SLE) | A solid support used in Supported Liquid Extraction, an alternative technique to troubleshoot emulsion-prone samples from liquid matrices. [31] |
| Phase Separation Filter Paper | Highly silanized paper used to isolate a specific liquid phase (aqueous or organic) and break emulsions during extract work-up. [31] |
Workflow and Conceptual Diagrams
SFE Optimization and Troubleshooting Workflow
The following diagram outlines a logical pathway for developing and troubleshooting a supercritical fluid extraction method.
Comprehensive Two-Step SFE Procedure
This diagram illustrates the automated two-step SFE procedure for the sequential extraction of non-polar and polar compounds from a single sample. [73]
Conclusion
The strategic optimization of modifier concentration is a powerful lever to enhance the efficiency and selectivity of Supercritical Fluid Extraction, transforming it into a tailored platform for recovering high-value bioactive compounds. By integrating foundational knowledge with robust methodological design and troubleshooting insights, researchers can consistently produce extracts with superior bioactivity profiles, as validated against conventional methods. The future of SFE in drug development lies in advancing continuous processing, exploring novel GRAS co-solvent mixtures, and integrating with in-line analytics for real-time process control. This green technology is poised to play a pivotal role in developing sustainable and effective therapeutic agents, meeting the stringent demands of both modern pharmaceuticals and clinical research.
References
- 1. Recent Advances in Supercritical Fluid Extraction of Natural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7504334/]
- 2. Carbon Dioxide Supercritical Extraction - an overview [https://www.sciencedirect.com/topics/engineering/carbon-dioxide-supercritical-extraction]
- 3. Green Extraction of Plant Materials Using Supercritical CO2 [https://www.mdpi.com/2223-7747/13/16/2295]
- 4. Optimization of Supercritical Fluid Extraction for the ... [https://www.mdpi.com/2076-3921/14/2/206]
- 5. Supercritical CO2 Extraction Process [https://www.co2extractionmachine.com/supercritical-co2-extraction-process]
- 6. Dry ice forming when extracting from supercritical CO2 [https://future4200.com/t/dry-ice-forming-when-extracting-from-supercritical-co2/45074]
- 7. CO2 Extractor Maintenance Guide [https://www.co2supercriticalextractionmachine.com/co2-supercritical-extraction-equipment/1703082926.html]
- 8. Optimization of supercritical fluid extraction from waste oil ... [https://ui.adsabs.harvard.edu/abs/2025JEST..tmp..450M/abstract]
- 9. Supercritical Fluid Extraction - an overview [https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/supercritical-fluid-extraction]
- 10. Optimization of conditions for supercritical fluid extraction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1390442/]
- 11. Supercritical Fluid Extraction—A Sustainable and Selective ... [https://www.mdpi.com/2076-3417/15/11/5914]
- 12. Optimization of supercritical fluid extraction of bioactive ... [https://www.sciencedirect.com/science/article/pii/S0896844624000469]
- 13. Process optimization for the supercritical carbondioxide ... [https://www.nature.com/articles/s41598-021-89772-6]
- 14. Methodological Optimization of Supercritical Fluid ... [https://www.mdpi.com/2076-3921/11/7/1248]
- 15. Supercritical Fluid Extraction - an overview [https://www.sciencedirect.com/topics/chemical-engineering/supercritical-fluid-extraction]
- 16. It's All About Selectivity [https://www.chromatographyonline.com/view/its-all-about-selectivity]
- 17. FAQ - Supercritical Fluid Technologies [https://www.supercriticalfluids.com/technical-resources/faq/]
- 18. Supercritical Extraction Techniques for Obtaining ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11171758/]
- 19. Supercritical Fluid Extraction - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/supercritical-fluid-extraction]
- 20. Supercritical fluid extraction [https://en.wikipedia.org/wiki/Supercritical_fluid_extraction]
- 21. Supercritical CO2 Extraction in Pharmaceuticals [https://www.buffaloextracts.com/knowledge/supercritical-co2-extraction-in-pharmaceuticals-driving-innovation-and-purity-standards/]
- 22. Supercritical fluid extraction of cannabinoids and their ... [https://www.sciencedirect.com/science/article/pii/S2212982024002427]
- 23. Effect of the polar modifiers on supercritical extraction ... [https://www.sciencedirect.com/science/article/abs/pii/S1383586613004139]
- 24. Supercritical Fluid Extraction - an overview [https://www.sciencedirect.com/topics/chemistry/supercritical-fluid-extraction]
- 25. Optimization of supercritical fluid extraction of polar ... [https://www.sciencedirect.com/science/article/pii/S2212982023000513]
- 26. FAQs Supercritical Fluids - ::::::Ocean Tech:::::: [http://www.oceantech.co.in/faq.html]
- 27. Optimizing ethanol-modified supercritical CO₂ extraction ... [https://www.nature.com/articles/s41598-025-91441-x]
- 28. Optimization of supercritical fluid extraction of essential oils ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4486533/]
- 29. Optimization and Comparison of Five Methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6253841/]
- 30. Supercritical CO2 technology for biomass extraction: Review [https://www.sciencedirect.com/science/article/pii/S0926669025008945]
- 31. Tips for Troubleshooting Liquid–Liquid Extractions [https://www.chromatographyonline.com/view/tips-troubleshooting-liquid-liquid-extractions]
- 32. Review Experimental design of supercritical fluid extraction [https://www.sciencedirect.com/science/article/abs/pii/S0260877413005128]
- 33. An enhanced response surface methodology (RSM ... [https://www.sciencedirect.com/science/article/abs/pii/S0305054898000148]
- 34. 5.3.3.6.1. Central Composite Designs (CCD) [https://www.itl.nist.gov/div898/handbook/pri/section3/pri3361.htm]
- 35. Box–Behnken design [https://en.wikipedia.org/wiki/Box%E2%80%93Behnken_design]
- 36. Factors Affecting CO2 Supercritical Fluid Extraction [https://www.toptiontech.com/info/factors-affecting-co2-supercritical-fluid-extr-103062716.html]
- 37. New Trends in Supercritical Fluid Technology and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10254675/]
- 38. Coumarin derivatives as acetyl- and butyrylcholinestrase ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6488543/]
- 39. Identification of Potent Acetylcholinesterase Inhibitors as ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10974877/]
- 40. Extraction of γ-Oryzanol from defatted rice bran using ... [https://www.sciencedirect.com/science/article/abs/pii/S0957582020318048]
- 41. Optimization of Supercritical Fluid Extraction for the Recovery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11852124/]
- 42. A Bibliometric Analysis of the Supercritical CO2 Extraction ... [https://www.mdpi.com/2311-7524/10/11/1185]
- 43. Optimization of extraction in supercritical fluids in obtaining ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11666378/]
- 44. Application of an artificial neural network model for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0926669018305661]
- 45. Evaluation of the response surface and hybrid artificial ... [https://www.sciencedirect.com/science/article/abs/pii/S1876107015004915]
- 46. Artificial intelligence-assisted optimization of extraction ... [https://www.nature.com/articles/s41598-025-25130-0]
- 47. A hybrid artificial neural network and multi-objective ... [https://www.nature.com/articles/s41598-024-83029-8]
- 48. Optimization of extraction in supercritical fluids in obtaining ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2024.1491479/full]
- 49. Optimization of accelerated solvent extraction of zeaxanthin ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11319554/]
- 50. Optimization of fucoxanthin extraction obtained from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9558218/]
- 51. How to Optimize Supercritical CO2 Extraction Parameters [https://www.co2extractionmachine.com/blog/how-to-optimize-supercritical-co2-extraction-parameters-a-practical-guide.html]
- 52. Parameter Effects and Optimisation in Supercritical Fluid ... [https://www.mdpi.com/2297-8739/9/12/385]
- 53. Supercritical Fluid Extraction: A Green and Sustainable ... [https://www.ajgreenchem.com/article_221328.html]
- 54. Optimization of the supercritical extraction and ... [https://www.sciencedirect.com/science/article/pii/S2212982024003421]
- 55. Control strategies for a supercritical fluid extraction process [https://www.sciencedirect.com/science/article/abs/pii/0009250995002952]
- 56. Supercritical CO2 extraction, chemical composition, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9360582/]
- 57. Supercritical CO2 extraction of solid-state cultivation ... [https://www.sciencedirect.com/science/article/pii/S2772577424000119]
- 58. Supercritical Fluid Extraction as a Potential ... [https://www.mdpi.com/2673-4591/37/1/115]
- 59. Your Guide To Supercritical Fluid CO₂ Extraction [https://neomagroup.com/scfe]
- 60. Valorization of bioactive compounds from food by-products ... [https://www.sciencedirect.com/science/article/abs/pii/S0308814625015286]
- 61. Optimization of supercritical fluid extraction ... [https://www.sciencedirect.com/science/article/abs/pii/S0263876222000752]
- 62. Supercritical Fluids Extraction: Benefits and Applications [https://extraktlab.com/supercritical-fluids-extraction/]
- 63. of Optimization of valuable compounds... supercritical fluid extraction [https://pubs.rsc.org/en/content/articlehtml/2025/fb/d5fb00395d]
- 64. Supercritical fluid extraction (SFE) optimization of trans ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7054580/]
- 65. Comparisons of soxhlet extraction, pressurized liquid ... [https://pubmed.ncbi.nlm.nih.gov/11045502/]
- 66. Comparison of supercritical fluid extraction and Soxhlet ... [https://www.sciencedirect.com/science/article/abs/pii/S0045653505000330]
- 67. Exploring the Sequential-Selective Supercritical Fluid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10609265/]
- 68. Supercritical Extraction Techniques for Obtaining ... [https://www.mdpi.com/2304-8158/13/11/1713]
- 69. Supercritical Fluid Extraction: Advantages and Disadvantages [https://www.co2extractionmachine.com/blog/supercritical-co2-fluid-extraction-top-5-pros-and-cons.html]
- 70. Antioxidant, Anti-Inflammatory, and Antiproliferative Activity of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12073327/]
- 71. Evaluation of antioxidant and anti-inflammatory properties, ... [https://www.sciencedirect.com/science/article/pii/S2667031324001362]
- 72. Antioxidant, anti-inflammatory, and antiproliferative ... [https://www.sciencedirect.com/science/article/pii/S0308814625036003]
- 73. Comprehensive two-step supercritical fluid extraction for ... [https://pubs.rsc.org/en/content/articlelanding/2024/gc/d4gc00371c]
- 74. On the mass transfer of supercritical fluids, specifically ... [https://www.sciencedirect.com/science/article/pii/S1385894724040087]