Microwave Energy: Powering Sustainable Chemistry for Faster Drug Discovery and Greener Synthesis
This article explores the transformative role of microwave energy in advancing sustainable chemistry, with a focus on applications for researchers and drug development professionals.
Microwave Energy: Powering Sustainable Chemistry for Faster Drug Discovery and Greener Synthesis
Abstract
This article explores the transformative role of microwave energy in advancing sustainable chemistry, with a focus on applications for researchers and drug development professionals. It covers the foundational principles of microwave-assisted synthesis, detailing its mechanisms and advantages over conventional heating. The content provides a methodological guide for applying these techniques to synthesize bioactive heterocycles and nanomaterials, alongside practical troubleshooting and optimization strategies. Finally, it presents a comparative analysis validating the significant enhancements in efficiency, yield, and sustainability, positioning microwave technology as a key enabler for greener pharmaceutical R&D.
The Principles and Promise of Microwave-Assisted Synthesis
The integration of microwave energy into chemical synthesis represents a transformative approach within the broader context of sustainable chemistry. Moving beyond its conventional culinary applications, microwave technology offers a powerful tool for enhancing energy efficiency, reaction speed, and product selectivity in chemical processes. Unlike traditional conductive heating methods that rely on superficial contact and often result in significant energy loss, microwave irradiation enables direct coupling of electromagnetic energy with molecular targets. This fundamental shift in energy transfer mechanism aligns with the core principles of green chemistry by minimizing energy consumption, reducing reaction times, and improving overall process efficiency. The subsequent sections of this whitepaper will provide a comprehensive technical examination of microwave energy's underlying mechanisms, diverse applications, and specific experimental protocols, establishing its critical role in advancing sustainable chemical research and industrial practice.
Fundamental Mechanisms of Microwave Heating
Microwave energy facilitates chemical reactions through fundamentally different mechanisms compared to conventional thermal heating. Understanding these underlying principles is crucial for researchers seeking to exploit microwave chemistry's full potential.
Electromagnetic Energy Transfer
Microwaves represent a form of electromagnetic radiation occupying the frequency spectrum between 300 MHz and 300 GHz, with 2450 MHz being the most commonly used frequency in laboratory and industrial applications due to its optimal penetration depth [1]. This frequency corresponds to a wavelength of approximately 12.2 cm in vacuum. The energy carried by individual microwave photons is remarkably low (approximately 0.037 kcal/mol)—far insufficient to break typical molecular bonds (requiring 80-120 kcal/mol)—confirming that microwave effects are primarily thermal rather than stemming from direct molecular dissociation [1].
Molecular Interaction Mechanisms
Microwave heating occurs through two primary molecular mechanisms that enable efficient energy transfer from the electromagnetic field to the reaction mixture:
- Dipolar Polarization: This mechanism affects molecules possessing a permanent dipole moment. The rapidly oscillating electric field (4.9×10⁹ times per second at 2.45 GHz) causes polar molecules to continuously realign themselves with the alternating field. This molecular rotation generates intense internal friction and collisions with neighboring molecules, resulting in instantaneous, volumetric heating [1] [2].
- Ionic Conduction: This mechanism operates when ionic species are present in the reaction mixture. The oscillating electric field accelerates charged particles through the medium, which then collide with other molecules, converting their kinetic energy into heat. The efficiency of this process increases with temperature as ionic mobility typically increases [1].
Table 1: Comparison of Heating Mechanisms in Chemical Synthesis
| Feature | Conventional Heating | Microwave Heating |
|---|---|---|
| Energy Transfer | Conduction/convection from vessel surfaces | Direct core-to-surface radiation penetration |
| Heating Path | Slow, inward from vessel walls | Rapid, volumetric throughout material |
| Temperature Gradient | Significant (surface hotter than core) | Minimal (potentially inverted gradient) |
| Heating Rate | Limited by thermal conductivity | Nearly instantaneous (10⁻⁹ seconds per cycle) |
| Energy Efficiency | Low (heats vessel and surroundings) | High (selective material coupling) |
| Process Control | Slow response ("instant on-instant off") | Rapid, precise temperature control |
Energy Transfer Dynamics
The exceptional speed of microwave heating stems from fundamental kinetic advantages. Microwave energy transfers every nanosecond (10⁻⁹ seconds), while molecular relaxation occurs on a microsecond timescale (10⁻⁵ seconds) [1]. This creates a non-equilibrium condition where energy delivery outpaces molecular relaxation, leading to instantaneous superheating effects that dramatically enhance reaction kinetics without altering the fundamental activation energy barrier. The "instant on-instant off" characteristic of microwave energy provides researchers with unprecedented control over thermal energy input compared to conventional heating methods.
Advanced Applications in Sustainable Chemistry
The unique advantages of microwave energy have enabled significant advancements across multiple domains of sustainable chemistry, from carbon capture to nanomaterial synthesis.
Energy-Efficient Catalysis and CO₂ Conversion
Recent breakthroughs in microwave-assisted catalysis demonstrate remarkable potential for reducing the energy intensity of industrial chemical processes. Researchers at the University of Tokyo developed a precision microwave technique that achieves energy efficiencies approximately 4.5 times higher than conventional heating methods [3] [4]. This approach utilizes tuned microwave frequencies (approximately 900 MHz) to selectively excite specific atomic sites within zeolite frameworks, where indium ions function as molecular "antennas" that concentrate thermal energy precisely where reactions occur [3]. This localized heating enables demanding transformations like carbon dioxide conversion and methane reforming at significantly lower bulk temperatures, creating pathways for carbon capture and utilization while substantially reducing energy consumption.
Advanced Nanomaterial Synthesis
Microwave irradiation has revolutionized nanomaterial fabrication by enabling unprecedented control over particle size, morphology, and crystallinity. The synthesis of silver nanoparticles for surface-enhanced Raman scattering (SERS) applications exemplifies these advantages. Through Quality by Design (QbD) optimization, researchers identified optimal microwave parameters of 130°C for 3.36 minutes with vigorous stirring (600 rpm) to produce nanoparticles with exceptional uniformity and signal reproducibility (intra- and inter-batch variabilities <15%) [5]. Similar approaches have been successfully applied to create various nanostructured systems, including photocatalytic materials for environmental remediation [6], lipidic and polymeric nanocarriers for drug delivery [2], and metallic nanocomposites for sensing applications.
Enhanced Organic Synthesis Methodologies
Microwave technology has dramatically accelerated and improved countless organic transformations that are fundamental to pharmaceutical research and development. Reaction rate enhancements of up to 1000-fold have been documented compared to conventional heating methods [1]. These dramatic improvements stem from the kinetic advantages of microwave heating, particularly the ability to achieve high instantaneous temperatures at the molecular level while maintaining moderate bulk temperatures. For a reaction with a bulk temperature of 150°C and an activation energy of 50 kcal/mol, calculations indicate that a mere 17°C instantaneous temperature increase enables a 10-fold rate enhancement, while a 56°C increase produces a 1000-fold improvement [1]. This capacity for precise energy delivery makes microwave irradiation particularly valuable for synthesizing complex pharmaceutical intermediates and active ingredients with improved selectivity and yield.
Table 2: Microwave Applications in Sustainable Chemistry
| Application Domain | Key Advancement | Sustainable Benefit |
|---|---|---|
| Carbon Capture & Utilization | CO₂ conversion via zeolite-supported catalysts | 4.5× energy efficiency vs. conventional heating [3] |
| Nanoparticle Synthesis | Optimized silver nanoparticles (130°C, 3.36 min) | <15% batch variability; reduced waste [5] |
| Photocatalyst Design | Controlled crystallinity & morphology | Enhanced pollutant degradation & H₂ production [6] |
| Pharmaceutical Synthesis | Rate enhancements up to 1000× | Reduced reaction times (hours to minutes) [1] |
| Drug Delivery Systems | Lipid/polymer nanoparticle engineering | Improved drug solubility & bioavailability [2] |
Experimental Design and Optimization Protocols
Implementing microwave chemistry requires careful consideration of reaction parameters, vessel selection, and optimization strategies to maximize the technological benefits.
Reaction Vessel and Conditions Selection
The choice between sealed and open vessel configurations represents a fundamental experimental design decision with significant implications for reaction outcomes:
- Pressurized (Sealed Vessel) Reactions: These systems enable superheating of solvents far beyond their atmospheric boiling points (e.g., dichloromethane to 180°C despite a normal bp of 40°C), facilitating dramatic rate enhancements. Certified pressure vessels typically accommodate small reaction volumes (≤10 mL) and provide an inherent inert atmosphere for air- and moisture-sensitive chemistry [7].
- Atmospheric (Open Vessel) Reactions: Conducted in standard round-bottom flasks, these setups allow for larger reaction scales and compatibility with conventional glassware like reflux condensers, addition funnels, and Dean-Stark traps. While rate enhancements are generally more modest than in sealed systems (typically ~10× faster than conventional methods), they enable straightforward translation of traditional synthetic protocols [7].
Solvent Selection Strategy
Solvent polarity critically influences microwave coupling efficiency and therefore reaction performance. The solvent selection process should consider both synthetic requirements and microwave absorption characteristics:
- High Microwave Absorbance: Solvents with high dielectric loss (e.g., water, DMF, NMP, ionic liquids) couple efficiently with microwave radiation, enabling rapid heating [7].
- Medium Microwave Absorbance: Solvents like acetone, dichloroethane, and tertiary alcohols provide moderate heating rates suitable for controlled temperature increases [7].
- Low Microwave Absorbance: Non-polar solvents (e.g., hexane, benzene, toluene) heat poorly under microwave irradiation but can function as thermal buffers for temperature-sensitive reactions by absorbing excess thermal energy while allowing polar reactants to couple directly with microwave energy [7].
Parameter Optimization Framework
Systematic optimization of microwave reaction parameters ensures reproducible and efficient outcomes:
- Temperature: For sealed-vessel reactions, begin approximately 10°C above the conventional reaction temperature. For open-vessel solvent-free reactions on mineral supports, initial temperatures of 200-250°C are appropriate, while reflux conditions should target temperatures at least 50°C above the solvent boiling point [7].
- Time: Initial screening should employ 5-10 minute irradiation times for pressurized and solvent-free reactions. For atmospheric solution-phase reactions, approximate conversions from conventional to microwave conditions are: 4 hours → 10 minutes; 8-18 hours → 30 minutes; >18 hours → 1 hour [7].
- Power: Begin new reactions cautiously at 50W for sealed vessels and 25-50W for solvent-free systems, increasing as needed. Reflux conditions typically require higher power (250-300W) to maintain boiling, while sub-boiling reactions can initiate at 100W [7].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of microwave-assisted chemistry requires specific materials and reagents optimized for microwave energy coupling and reaction performance.
Table 3: Essential Research Reagents for Microwave Chemistry
| Reagent/Material | Function & Application | Technical Considerations |
|---|---|---|
| Zeolite Scaffolds | Porous support with tunable cavities; acts as molecular sponge for reactions | Functionalized with indium ions as microwave antennas; pore size controls selectivity [3] |
| Polar Solvents (DMF, NMP) | High microwave absorption for efficient heating | Dielectric properties determine coupling efficiency; enables rapid temperature ramping [7] |
| Ionic Liquids | Green solvent alternatives with excellent microwave coupling | Combined high polarity and low vapor pressure ideal for high-temperature reactions [7] |
| Solid Mineral Supports | Solvent-free reaction media for "dry" synthesis | Alumina, silica, or clay surfaces concentrate reactants; enable selective heating [7] |
| Metal Nanoparticle Precursors | Silver salts (AgNO₃) for nanostructure synthesis | Microwave enables rapid, uniform nucleation; produces narrow size distribution [5] |
| Heterogeneous Catalysts | Metal-doped supports for selective transformations | Microwave energy targets active sites, reducing bulk temperature requirements [3] |
Microwave energy represents a transformative tool in the pursuit of sustainable chemistry, offering unprecedented control over chemical processes while significantly reducing energy consumption and environmental impact. The precision targeting of molecular active sites, dramatic rate enhancements, and improved product selectivity demonstrated across diverse applications—from carbon utilization to pharmaceutical synthesis—establish microwave technology as a cornerstone of modern green chemistry practices. As research advances in catalyst design, reactor engineering, and process intensification, microwave-assisted approaches will undoubtedly play an increasingly vital role in developing the sustainable chemical technologies required for a circular economy. The continued integration of microwave methodologies with renewable energy sources and flow chemistry systems promises to further enhance the sustainability profile of chemical manufacturing, ultimately contributing to a more efficient and environmentally responsible chemical industry.
Dielectric heating is a thermal process that utilizes high-frequency alternating electric fields, typically in the radio frequency (RF) range of 1–100 MHz or microwave frequencies of 300 MHz to 300 GHz, to generate heat directly within dielectric materials [8] [9]. This phenomenon represents a fundamental shift from conventional heating methods, enabling energy transfer that is both volumetric and instantaneous [10]. Unlike conventional thermal conduction which relies on temperature gradients and slowly penetrates materials from the surface inward, dielectric heating occurs throughout the entire volume of a material simultaneously when exposed to appropriate electromagnetic fields [10].
The significance of dielectric heating extends across numerous fields including sustainable chemistry, drug development, food processing, materials science, and environmental remediation [8] [10] [11]. For researchers and scientists pursuing sustainable chemistry goals, dielectric heating offers a pathway to dramatically reduce energy consumption, decrease reaction times, improve product yields, and minimize waste generation [12]. The pharmaceutical industry has particularly embraced this technology for the synthesis of medicinally privileged heterocycles and active pharmaceutical ingredients, where it enables rapid optimization of reaction conditions and expedites drug discovery timelines [11] [12].
This technical guide examines the fundamental principles, quantitative relationships, and experimental methodologies that underpin dielectric heating, providing researchers with the theoretical and practical foundation necessary to harness this technology for advanced scientific applications.
Fundamental Principles and Mechanisms
The Physics of Dielectric Heating
Dielectric heating operates through the interaction between electromagnetic energy and matter at the molecular level. When dielectric materials are subjected to high-frequency alternating electric fields, two primary mechanisms facilitate the conversion of electromagnetic energy into thermal energy: dipolar polarization and ionic conduction [9] [13].
Dipolar polarization, often referred to as dipole rotation, occurs in materials containing polar molecules that possess a permanent electrical dipole moment [8]. When exposed to an oscillating electric field, these dipoles (such as water molecules) attempt to align themselves with the rapidly changing field direction. The continuous reorientation of molecular dipoles creates molecular friction through collisions with adjacent molecules, dissipating energy as heat throughout the material [8] [9]. This mechanism is particularly effective at higher frequencies (microwave range) and represents the dominant heating mechanism in microwave ovens where water molecules rapidly rotate at frequencies around 2.45 GHz [8].
Ionic conduction occurs when free ions (Na+, K+, Cl−, HCO3−) present in a material are accelerated by the applied electric field [9] [14]. These charged particles gain kinetic energy and move through the medium, colliding with surrounding molecules or atoms inelastically. During these collisions, their kinetic energy converts into thermal energy, increasing the overall temperature of the material [14]. This mechanism becomes increasingly significant at lower frequencies (RF range) and in materials with substantial ionic content, such as biological tissues or electrolyte solutions [9] [14].
The relative contribution of each mechanism depends on the material properties, particularly its dielectric loss factor and ionic conductivity, as well as the frequency of the applied electromagnetic field [8] [9].
Quantitative Analysis of Energy Transfer
The efficiency of dielectric heating is quantified through several key mathematical relationships that enable researchers to predict and optimize heating performance for specific applications.
The power dissipation per unit volume in a material under dielectric heating conditions is given by:
[P = \omega \cdot \varepsilonr'' \cdot \varepsilon0 \cdot E^2]
Where:
- (P) is the power density (W/m³)
- (\omega) is the angular frequency ((\omega = 2\pi f))
- (\varepsilon_r'') is the imaginary part of the complex relative permittivity (dielectric loss factor)
- (\varepsilon_0) is the permittivity of free space ((8.85 × 10^{-12}) F/m)
- (E) is the root-mean-square electric field strength (V/m) [8] [9] [13]
This fundamental equation demonstrates that the heating rate depends linearly on both frequency and the dielectric loss factor, and quadratically on the electric field strength. The total power absorbed throughout a material volume (V) is obtained by integrating the power density over the entire volume, accounting for potential spatial variations in the electric field [13].
The loss tangent ((\tan \delta)) represents another critical parameter, defined as the ratio between the dissipative and storage components of polarization:
[\tan \delta = \varepsilon'' / \varepsilon']
Where (\varepsilon') is the real part of the complex relative permittivity (dielectric constant) [13]. Materials with higher loss tangent values more efficiently convert electromagnetic energy into thermal energy, though extremely high values may limit penetration depth due to preferential surface heating [13].
Table 1: Dielectric Properties of Common Materials at 2.45 GHz and 25°C
| Material | Dielectric Constant (ε') | Loss Factor (ε'') | Loss Tangent (tan δ) |
|---|---|---|---|
| Water | ~78 | ~12 | ~0.15 |
| Ice | ~3.2 | ~0.003 | ~0.001 |
| Ethanol | ~6 | ~1.9 | ~0.32 |
| PVC | ~3 | ~0.03 | ~0.01 |
| Silicon Carbide | ~30 | ~15 | ~0.5 |
| Biological Tissue (muscle) | ~50 | ~15 | ~0.3 |
The penetration depth ((d_p)) defines the distance at which the electromagnetic field intensity decreases to (1/e) (approximately 37%) of its surface value and is calculated as:
[dp = \frac{\lambda0 \sqrt{\varepsilon'}}{2\pi \varepsilon''} = \frac{c}{\sqrt{2} \pi f \sqrt{\varepsilon'}} \sqrt{\sqrt{1 + \tan^2 \delta} - 1}]
Where (\lambda_0) is the wavelength in free space and (c) is the speed of light [13]. This parameter critically determines the effectiveness of dielectric heating for thicker materials, with lower frequencies generally providing greater penetration depths [8] [9].
Dielectric Heating Methodologies and Experimental Protocols
Measurement of Dielectric Properties
Accurate characterization of dielectric properties forms the foundation of any dielectric heating application. The open-ended coaxial probe method has emerged as a standard technique for measuring the dielectric properties of biological tissues, chemical solutions, and various materials [15] [16].
Experimental Protocol: Dielectric Property Measurement Using Open-Ended Coaxial Probe
Instrument Setup: Configure a vector network analyzer (VNA) with an open-ended coaxial probe kit (e.g., Keysight N1501A Dielectric Probe Kit). Calibrate the system using air, short circuit, and distilled water standards following manufacturer specifications [16].
Sample Preparation: Prepare materials with uniform dimensions appropriate for measurement. For solid tissues, cut samples to approximately 2×2×0.5 cm dimensions. For granular materials, ensure consistent packing density. Maintain sample hydration if measuring moisture-dependent properties [15] [16].
Measurement Procedure: Place the probe in firm contact with the sample surface, ensuring no air gaps. Sweep across the frequency range of interest (typically 100 MHz to 20 GHz for comprehensive characterization). Record both the dielectric constant (ε') and loss factor (ε'') at each frequency point. Perform multiple measurements at different sample locations to account for heterogeneity [15] [16].
Temperature Control: For temperature-dependent characterization, implement a temperature chamber or water bath to maintain precise temperature control during measurements. Record dielectric properties at relevant temperature intervals [15].
Data Analysis: Calculate the loss tangent (tan δ) and penetration depth ((d_p)) from the measured ε' and ε'' values. Analyze frequency and temperature dependencies to identify optimal heating conditions [13] [15].
This methodology was successfully employed in a recent study on Cirrhinus microlepis tissues, revealing significant differences in dielectric properties between scales (high loss) and muscle tissue (moderate loss), enabling selective thermal targeting of parasite-infected regions [16].
Experimental Design for Dielectric Heating Applications
Implementing effective dielectric heating requires careful experimental design tailored to specific applications. The following workflow illustrates a generalized approach for developing dielectric heating processes:
Diagram 1: Dielectric Heating Experimental Design
Frequency Selection Guidelines:
- RF Frequencies (1-100 MHz): Preferred for larger, thicker materials with moderate moisture content; provides deeper penetration [8] [9]
- Microwave Frequencies (300 MHz-300 GHz): Ideal for rapid heating of smaller samples with high dielectric loss; regulated to ISM bands (e.g., 915 MHz, 2.45 GHz) [8] [10]
Applicator Design Considerations:
- Parallel Plate Capacitors: Effective for RF heating of sheet materials; creates uniform electric fields between electrodes [8]
- Multimode Cavities: Suitable for microwave heating of irregular shapes; creates standing wave patterns for volumetric heating [8] [10]
- Single Mode Cavities: Provides focused, intense heating for specific small-volume applications [10]
- Horn Antennas: Enables directed microwave energy for selective surface heating; optimal for targeted applications [16]
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Essential Materials and Equipment for Dielectric Heating Research
| Category | Specific Items | Research Function | Application Examples |
|---|---|---|---|
| Dielectric Property Characterization | Vector Network Analyzer, Open-Ended Coaxial Probe, Dielectric Probe Kit, Temperature Chamber | Quantifies ε' and ε'' across frequency and temperature ranges | Material screening, frequency optimization [15] [16] |
| Dielectric Heating Systems | RF Generators (1-100 MHz), Microwave Reactors (2.45 GHz), Waveguide Applicators, Horn Antennas, Multimode Cavities | Delivers controlled electromagnetic energy to samples | Chemical synthesis, material processing, parasite inactivation [10] [12] [16] |
| Process Monitoring | Fiber Optic Temperature Sensors, IR Thermal Cameras, Pressure Sensors, Power Monitors | Provides real-time feedback without electromagnetic interference | Reaction optimization, thermal validation, safety monitoring [15] [16] |
| Specialized Reactors | Sealed Vessels, Flow-Through Systems, Solid-State Supports, Continuous Processing Reactors | Enables specific reaction conditions and scalability | Solvent-free synthesis, continuous manufacturing [10] [12] |
| Reference Materials | Materials with known dielectric properties (water, ethanol, silicon carbide, Teflon) | System calibration and method validation | Measurement verification, comparative studies [15] |
Advanced Applications in Sustainable Chemistry and Drug Development
Sustainable Chemical Synthesis
Dielectric heating has revolutionized modern organic synthesis by enabling dramatic reductions in reaction times, improved yields, and enhanced selectivity. The application of microwave dielectric heating in chemical transformations aligns with multiple principles of green chemistry, including reduced energy consumption, decreased waste production, and improved process safety [12].
In transition metal-catalyzed reactions, which are fundamental to constructing complex molecular architectures, microwave heating has demonstrated remarkable efficacy:
Suzuki Cross-Coupling Reactions: Traditional Suzuki couplings requiring hours or days under conventional heating can be completed in minutes with microwave irradiation. A notable example includes the coupling of aryl chlorides with boronic acids in aqueous media using stable palladium catalysts, achieving excellent yields in just 15 minutes compared to 12-24 hours conventionally [12]. The methodology has been extended to solvent-free conditions and more sustainable nickel-based catalyst systems, significantly reducing both reaction time and environmental impact [12].
Heck Reactions: Microwave-enhanced Heck couplings have achieved remarkable regioselectivity and enantioselectivity (>99% ee) in significantly reduced timeframes. The rapid heating capability enables the use of sensitive catalytic systems that might decompose under prolonged conventional heating [12].
Buchwald-Hartwig Amination: This essential carbon-nitrogen bond forming reaction benefits substantially from microwave irradiation, with copper-based catalytic systems achieving coupling of aryl halides with amines in minutes rather than hours [12].
The sustainable advantages of microwave-assisted synthesis extend beyond mere rate acceleration. The precise control of heating reduces thermal degradation pathways, minimizes side reactions, and enhances overall atom economy. Additionally, the ability to perform reactions under solvent-free conditions or in aqueous media further improves the environmental profile of chemical processes [12].
Pharmaceutical Research and Development
The pharmaceutical industry has embraced dielectric heating technology to accelerate drug discovery and development timelines. Microwave-assisted synthesis has become particularly valuable for constructing medicinally privileged heterocycles, which form the core structural motifs in numerous therapeutic agents [11] [12].
Multi-Component Reactions (MCRs): Dielectric heating has dramatically enhanced the efficiency of MCRs, which are powerful tools for generating molecular diversity in drug discovery. Microwave-assisted Mannich reactions proceed with high stereoselectivity using only 0.5 mol% of proline catalyst, achieving up to 98% enantiomeric excess in significantly reduced timeframes [12]. Similarly, Ugi reactions, which traditionally require 24-48 hours at room temperature, can be completed in minutes under microwave irradiation with improved yields and purity [12].
Solid-Phase Synthesis: Microwave technology has been successfully adapted to solid-phase synthesis methodologies central to combinatorial chemistry and peptide synthesis. The rapid and uniform heating provided by microwave irradiation overcomes the traditional limitations of poor heat transfer in solid-phase systems, enabling more efficient coupling reactions and cleavage steps [12].
Scale-Up Considerations: While early microwave chemistry was limited to small-scale applications, recent advances in continuous flow reactors and scalable multimode systems have enabled the translation of optimized reaction conditions from milligram to kilogram scales without sacrificing efficiency [12]. This capability bridges the critical gap between drug discovery and development, providing a seamless pathway from initial lead optimization to preclinical material preparation.
Environmental and Industrial Applications
Dielectric heating technology has demonstrated significant potential in environmental remediation and sustainable industrial processing:
Catalytic Environmental Remediation: Microwave dielectric heating has been successfully applied to environment-related heterogeneous catalytic systems for decomposing hazardous compounds including hydrogen sulfide and sulfur dioxide [10]. The unique heating characteristics of microwaves create remarkable temperature gradients ("hot spots") within catalyst beds, significantly enhancing reaction rates and shifting apparent equilibrium positions compared to conventional heating methods [10].
Waste Treatment: Microwave thermal regeneration has emerged as an efficient method for treating industrial waste, particularly for regenerating mercury-containing activated carbon catalysts from industrial processes [17]. This approach achieves 99.7% mercury removal while restoring the activated carbon's surface area to 536.2 m²/g, with a 72.75% reduction in activation energy compared to conventional thermal treatment [17].
Food and Agricultural Processing: RF dielectric heating has been implemented for post-harvest pest control in agricultural products, selectively eliminating insects based on their higher moisture content compared to the host material [8] [9]. Similarly, microwave volumetric heating enables rapid drying and processing of food products with improved quality retention compared to conventional thermal processing [8].
Comparative Analysis of Heating Modalities
The distinctive features of dielectric heating become particularly evident when compared with conventional heating methods. The following diagram illustrates the fundamental differences in heat transfer mechanisms and resulting temperature distributions:
Diagram 2: Dielectric vs. Conventional Heating
Table 3: Performance Comparison of Heating Methods for Different Applications
| Application | Conventional Heating Performance | Dielectric Heating Performance | Key Advantages |
|---|---|---|---|
| Organic Synthesis | Reaction times: hours to days; Significant thermal gradients; Potential decomposition | Reaction times: minutes to hours; Uniform heating; Reduced decomposition | 80-90% reduction in processing time; Improved product purity [12] |
| Material Drying | Slow surface-to-center moisture migration; Overheating of surfaces; Long processing times | Rapid volumetric moisture evaporation; Selective heating of water; Self-limiting drying | 50-80% reduction in drying time; 30-50% energy savings [8] [13] |
| Food Processing | Non-uniform pasteurization/sterilization; Overcooking of surfaces; Nutrient degradation | Uniform microbial inactivation; Minimal surface overheating; Improved quality retention | Enhanced safety and quality; Extended shelf life [8] [9] |
| Waste Treatment | Slow thermal desorption; High energy consumption; Incomplete regeneration | Rapid contaminant removal; Selective heating of pollutants; Lower activation energy | Higher removal efficiency (e.g., 99.7% mercury); Reduced energy requirements [17] |
| Polymer Processing | Long curing times; Internal voids from uneven heating; Thermal stress | Rapid uniform curing; Improved physical properties; Reduced internal stresses | Enhanced product quality; Faster production cycles [8] [13] |
Dielectric heating represents a transformative approach to thermal processing that fundamentally differs from conventional heating methods. Through the mechanisms of dipolar polarization and ionic conduction, dielectric heating enables volumetric and instantaneous energy transfer directly within materials, bypassing the limitations of thermal conductivity that govern conventional heating processes [8] [9].
The core principles governing dielectric heating are well-established mathematically, with power dissipation dependent on the square of the electric field strength and linearly related to both frequency and the dielectric loss factor of the material [8] [13]. These relationships provide researchers with precise control over heating characteristics, enabling optimization for specific applications through appropriate frequency selection and field strength adjustment.
For the pharmaceutical and chemical industries, dielectric heating technology offers substantial benefits in sustainable process development, including dramatic reductions in reaction times, improved product yields, enhanced selectivity, and decreased energy consumption [11] [12]. The ability to rapidly explore synthetic pathways and optimize reaction conditions aligns perfectly with the needs of modern drug discovery, while the recent development of scalable systems bridges the critical gap between laboratory research and industrial production.
As dielectric heating technology continues to evolve, emerging applications in environmental remediation, advanced materials processing, and sustainable manufacturing demonstrate the expanding utility of this approach [10] [17]. The integration of dielectric heating with process analytical technologies and real-time monitoring systems promises to further enhance control and reproducibility, solidifying its role as an enabling technology for sustainable chemistry in the 21st century.
For researchers and drug development professionals, mastery of dielectric heating principles and methodologies provides a powerful tool for addressing the dual challenges of efficiency and sustainability in chemical synthesis and material processing. The continued refinement of application-specific protocols and equipment will undoubtedly expand the boundaries of what can be achieved through this unique energy transfer mechanism.
The global shift toward sustainable industrial practices has fundamentally redefined research and development in chemical synthesis. Within this paradigm, microwave-assisted synthesis has emerged as a transformative green technology, offering a pathway to reduce the environmental footprint of chemical manufacturing. Unlike conventional heating methods that are often inefficient and environmentally burdensome, microwave technology leverages direct electromagnetic energy delivery to achieve rapid, efficient, and selective chemical reactions [18]. This technical guide delineates the core advantages of microwave-assisted synthesis—dramatically reduced reaction times, significantly enhanced yields, and improved product purity—within the critical context of sustainable chemistry. Framed for researchers and drug development professionals, this document provides quantitative comparisons, detailed experimental protocols, and an analysis of how this technology aligns with the principles of green chemistry, contributing to more sustainable research and industrial processes.
Core Principles and Sustainable Mechanism
The profound advantages of microwave-assisted synthesis originate from its fundamental mechanism of energy transfer, which is distinct from conventional thermal heating.
Fundamental Mechanism of Microwave Heating
In conventional heating, energy is transferred from an external source (like an oil bath) to the reaction vessel via conduction and convection, creating a temperature gradient from the surface to the core of the mixture. This process is inherently slow and can lead to the decomposition of sensitive compounds on the hot vessel walls [19]. In contrast, microwave irradiation generates heat through two primary mechanisms:
- Dipolar Polarization: Polar molecules in the reaction mixture attempt to align themselves with the oscillating electric field of the microwaves (typically at 2.45 GHz). This rapid reorientation causes molecular friction and collision, generating heat volumetrically throughout the entire sample [20].
- Ionic Conduction: Ions present in the solution migrate under the changing electric field, colliding with other molecules and converting kinetic energy into heat [20].
This volumetric heating enables the reaction mixture to be heated rapidly and uniformly, eliminating the thermal gradient bottleneck of conventional methods and directly coupling energy with the molecules driving the chemical reaction [21].
A Sustainable and Efficient Energy Transfer Pathway
The following workflow illustrates the comparative processes of conventional versus microwave-assisted synthesis, highlighting the points where efficiency and sustainability gains are achieved:
Quantitative Advantages: Data and Comparative Analysis
The theoretical efficiency of microwave-assisted synthesis is consistently borne out by experimental data, demonstrating clear superiority over conventional methods across diverse chemical reactions.
Dramatically Reduced Reaction Times
Microwave irradiation can accelerate chemical reactions by orders of magnitude, reducing processes that require hours or days to mere minutes or seconds. This is due to the rapid and direct superheating of the reaction mixture [20].
Table 1: Comparative Reaction Times in Organic Synthesis
| Reaction Type | Starting Material | Conventional Heating Time | Microwave Heating Time | Time Reduction | Citation |
|---|---|---|---|---|---|
| Amide Coupling | Benzotriazole-5-carbonyl chloride + o-Toluidine | 4 hours | 4.5 minutes | ~98% | [19] |
| Biomass Conversion | Cellulose to Levulinic Acid | 4 hours | 3 minutes | ~99% | [22] |
| Heterocycle Formation | Clauson-Kaas Pyrrole Synthesis | Several hours | 5-10 minutes | ~95% | [20] |
| Nanomaterial Fabrication | Various Metal Nanoparticles | Several hours to days | Seconds to minutes | >90% | [23] |
Enhanced Reaction Yields
The efficient and selective heating provided by microwaves often leads to higher product yields. This can be attributed to reduced thermal decomposition and the ability to reach higher temperatures rapidly, which may favor the desired reaction pathway [21].
Table 2: Comparative Product Yields in Synthesis
| Reaction / Product | Conventional Yield | Microwave Yield | Yield Improvement | Citation |
|---|---|---|---|---|
| N-o-tolyl-1H-benzotriazole-5-carboxamide | 72% | 83% | +11% | [19] |
| Levulinic Acid from Glucose | 6.93% | 9.57% | +38% | [22] |
| Levulinic Acid from Cellobiose | 4.88% | 6.12% | +25% | [22] |
| N-substituted Pyrroles (Clauson-Kaas) | Not Reported | 69-91% | Significant (High-Yield) | [20] |
Improved Product Purity and Selectivity
The "in-core" volumetric heating of microwave irradiation minimizes the temperature gradients that can lead to side reactions and product decomposition. Furthermore, the ability to precisely control temperature and pressure in advanced reactors provides superior command over reaction kinetics [21]. Evidence from 1H and 13C NMR analyses confirms that the microwave-assisted process of converting biomass to levulinic acid improves the purity of the final product by generating fewer by-products compared to the conventional thermochemical reaction [22]. This enhanced purity is a critical advantage in pharmaceutical synthesis, where purification steps are costly and time-consuming.
Experimental Protocols: Illustrative Examples
This protocol for synthesizing a benzotriazole derivative exemplifies the dramatic reduction in reaction time achievable with microwave assistance.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in the Reaction |
|---|---|
| Benzotriazole-5-carbonyl chloride | Electrophilic reactant (acylating agent). |
| o-Toluidine | Nucleophilic reactant (amine). |
| Benzene (as solvent) | Reaction medium. Note: Due to toxicity, alternative green solvents (e.g., ethyl acetate, toluene) should be investigated. |
| Domestic Microwave Oven (Samsung M183DN) | Source of microwave irradiation (180 W). Note: For superior control and safety, dedicated single-mode scientific reactors are recommended. |
| 10% Hydrochloric Acid | Work-up reagent to remove excess o-toluidine. |
| Anhydrous Sodium Sulfate | Drying agent for the organic layer post-work-up. |
Detailed Methodology:
- Reaction Setup: Combine 1 g (5.50 mmol) of benzotriazole-5-carbonyl chloride with 5 mL of benzene in a suitable microwave-transparent vessel. Add an equimolar proportion of o-toluidine dissolved in 10 mL of benzene.
- Conventional Method (For Comparison): Reflux the reaction mixture using a conventional heating mantle for 4 hours.
- Microwave-Assisted Method: Irradiate the reaction mixture in a domestic microwave oven at a power of 180 W for 4 minutes and 30 seconds.
- Work-up (Common to Both):
- After the reaction is complete, add 10% hydrochloric acid to the mixture. This converts any unreacted o-toluidine into a water-soluble hydrochloride salt.
- Separate the organic (benzene) layer and wash it with water (3 × 10 mL).
- Pass the benzene layer through anhydrous sodium sulfate to remove residual water.
- Concentrate the solution by distilling off the benzene to obtain the final product as a light brown crystalline powder.
- Characterization: The product is characterized by its melting point (218°C conventional, 220°C microwave), IR, and 1H NMR spectroscopy.
This protocol highlights the application of microwave synthesis in biomass conversion, a key area of sustainable chemistry.
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in the Reaction |
|---|---|
| Delignified Cellulose (e.g., from rice husk) | Lignocellulosic biomass feedstock. |
| Hierarchical Mn3O4/ZSM-5 Zeolite Catalyst | A heterogeneous catalyst with micro- and mesopores, designed for high activity in biomass conversion. |
| Household Microwave Oven | Source of microwave irradiation (600 W). |
| High-Performance Liquid Chromatography (HPLC) | Analytical instrument for quantifying conversion and yield. |
Detailed Methodology:
- Reaction Setup: Combine delignified cellulose (or model compounds like glucose or cellobiose) with the hierarchical Mn3O4/ZSM-5 catalyst in a microwave vessel.
- Conventional Method (For Comparison): Heat the reaction mixture at 130°C for 4 hours.
- Microwave-Assisted Method: Irradiate the reaction mixture in a household microwave oven at 600 W for 180 seconds (3 minutes).
- Analysis: Analyze the conversion products using HPLC to determine the conversion percentage and levulinic acid yield. As noted in the results, the microwave method not only gives a higher yield of levulinic acid but also results in a product with higher purity, as determined by
1H and 13C NMRanalyses [22].
Sustainability and Economic Impact in Research and Industry
The advantages of microwave-assisted synthesis directly contribute to the core tenets of green and sustainable chemistry, offering both environmental and economic benefits.
Contribution to Green Chemistry Principles
- Prevent Waste: Improved yields and selectivity directly translate to less wasted starting material and a reduced volume of hazardous waste for disposal [23].
- Less Hazardous Chemical Syntheses: The ability to use milder conditions and the facilitation of reactions with less toxic catalysts or solvents align with this principle [20].
- Design for Energy Efficiency: Microwave technology is inherently more energy-efficient. Research from the University of Tokyo demonstrates a system that can be
around 4.5 times more efficientthan conventional heating methods by focusing thermal energy exclusively at atomic active sites within a catalyst [4]. - Reduce Derivatives: Faster reaction times and cleaner profiles can minimize the need for protecting groups, streamlining synthetic routes [21].
The following diagram summarizes the multi-faceted sustainability benefits enabled by microwave-assisted synthesis:
Economic and Industrial Outlook
The economic imperative is clear. The global market for microwave-assisted synthesis is projected to grow significantly, driven by demand in pharmaceuticals, materials science, and sustainability applications [21]. The technology addresses the "productivity crisis" in drug discovery by accelerating the iterative cycle of synthesizing and testing novel compounds, thereby reducing the cost of failure [19]. Continuous-flow microwave reactors are a key innovation, enabling the translation of laboratory-scale successes to industrial production of kilograms of material, bridging the critical gap between R&D and commercial manufacturing [20].
The trajectory of microwave-assisted synthesis points toward even deeper integration with sustainable chemistry. Key future directions include the development of more efficient and scalable microwave systems, such as those using GaN (Gallium Nitride) solid-state technology for precise power control and higher energy efficiency [21]. The synergy of MAE with other green technologies, such as ultrasound or environmentally friendly solvents (e.g., deep eutectic solvents and ionic liquids), is a vibrant area of research [18]. Furthermore, the integration of artificial intelligence and machine learning for process prediction and optimization promises to unlock new levels of efficiency and control [18].
In conclusion, microwave-assisted synthesis is far more than a laboratory curiosity; it is a cornerstone technology for sustainable chemistry. Its demonstrated ability to dramatically reduce reaction times, enhance yields, and improve product purity directly translates to more efficient, economical, and environmentally benign chemical processes. For researchers and drug development professionals, mastering this technology is not merely an optimization of method—it is an essential step toward aligning scientific innovation with the urgent imperative of environmental sustainability.
The principles of green chemistry emphasize the need for energy-efficient processes and the reduction of hazardous waste. Microwave technology has emerged as a transformative tool in this context, offering a fundamentally different approach to heating that enables more sustainable chemical processing. Unlike conventional heating methods that rely on conduction and convection, microwave energy delivers electromagnetic radiation directly to materials, creating heat volumetrically within the substance itself rather than transferring it from external sources [24]. This direct coupling of energy enables unprecedented efficiency in chemical reactions and waste processing while simultaneously reducing the environmental footprint of industrial and research activities.
The sustainability advantages of microwave-assisted chemistry align with multiple principles of green chemistry, particularly energy efficiency and waste reduction. As research in this field has advanced, microwave technology has demonstrated significant potential across diverse applications including organic synthesis, nanomaterials production, drug development, and hazardous waste treatment [2] [25] [24]. This technical guide examines the core mechanisms, quantitative benefits, and practical methodologies that establish microwave chemistry as a cornerstone technology for sustainable scientific innovation.
Fundamental Mechanisms of Microwave Heating
Dielectric Heating Principles
Microwave heating operates through two primary mechanisms: dipolar polarization and ionic conduction. When microwave radiation interacts with materials, the electric field component causes molecular dipoles to align with the oscillating field. At the standard frequency of 2.45 GHz, dipoles reorient themselves approximately 2.5 billion times per second, generating intense internal friction that produces heat volumetrically throughout the material [25]. For ionic species, the electric field induces accelerated movement of ions through the medium, resulting in similar heating effects through collisions. The efficiency of these processes depends critically on a material's dielectric properties, specifically the complex permittivity (ε* = ε' - jε"), where the real part (ε') represents the ability to polarize, and the imaginary part (ε") quantifies the efficiency of converting electromagnetic energy to heat [25].
This direct energy transfer mechanism fundamentally differs from conventional heating, where thermal energy must gradually penetrate from the surface inward, creating thermal gradients and requiring more energy to maintain temperature. Microwave heating generates heat simultaneously throughout the entire volume, eliminating these gradients and providing more uniform temperature distribution [24]. The resulting rapid and specific heating enables chemical processes to proceed with greater efficiency and control while consuming less energy overall.
Material-Microwave Interactions
Materials exhibit three characteristic responses to microwave radiation: transparency, absorption, and reflection. Microwave-transparent materials (e.g., glass, certain ceramics) allow waves to pass through with minimal interaction, while reflective materials (e.g., metals) deflect incident radiation. For sustainable chemistry applications, the most relevant materials are absorbers that efficiently convert microwave energy to heat [25]. The absorption efficiency varies significantly between materials, enabling selective heating where specific components within a mixture can be targeted. This selectivity forms the basis for many energy-efficient microwave applications, including the precise heating of catalytic sites in advanced synthesis and the targeted destruction of hazardous contaminants in waste streams.
Table 1: Material Responses to Microwave Radiation
| Response Type | Interaction Mechanism | Example Materials | Applications in Sustainable Chemistry |
|---|---|---|---|
| Absorption | Converts microwave energy to heat | Water, polar solvents, zeolites | Reaction heating, waste processing |
| Transparency | Minimal interaction, waves pass through | Glass, Teflon, certain ceramics | Reaction vessel materials |
| Reflection | Deflects incident radiation | Metals, graphite | Catalyst supports, reactor components |
Quantitative Assessment of Energy Efficiency
Comparative Energy Consumption
Microwave-assisted processes consistently demonstrate superior energy efficiency compared to conventional methods across diverse applications. Research from the University of Tokyo has documented energy efficiencies approximately 4.5 times higher than conventional techniques for chemical reactions, particularly in CO₂ conversion processes [3] [4]. This dramatic improvement stems from the ability to focus thermal energy specifically at atomic active sites where reactions occur, rather than heating entire reactor systems. In one documented approach, researchers used tuned microwaves (∼900 MHz) to excite indium ions within zeolite frameworks, creating localized heating that reduced the overall energy required for demanding reactions like methane conversion and water decomposition [3].
In industrial waste processing, microwave pyrolysis achieves operating temperatures exceeding 450°C in a fraction of the time required by conventional heating methods, significantly reducing energy consumption [25]. Similarly, in pharmaceutical synthesis and nanomaterial production, microwave methods typically reduce reaction times from hours to minutes while maintaining or improving yields [2] [24]. The cumulative effect of these time savings translates to substantially lower energy demands per unit of product, contributing to more sustainable manufacturing paradigms across multiple sectors.
Table 2: Energy Efficiency Comparison of Microwave vs. Conventional Methods
| Application Area | Conventional Method Energy Use | Microwave Method Energy Use | Efficiency Improvement |
|---|---|---|---|
| Chemical Synthesis (CO₂ Conversion) | Baseline | ~4.5x less energy | ~450% improvement [3] |
| Food Reheating | 0.9 kWh (conventional oven) | 0.12 kWh (microwave) | ~80% reduction [26] |
| Scrap Tyre Pyrolysis | High energy requirement | Reduced time to reach 450°C | Significant time and energy savings [25] |
| Nanomaterial Synthesis | Hours of heating | Minutes of microwave exposure | Dramatic time reduction [24] |
Standby Power Considerations
While microwave processes demonstrate exceptional efficiency during active use, standby power consumption represents an often-overlooked aspect of overall energy efficiency. Modern microwave ovens consume small amounts of electricity (typically 0.6-1.0 watts) when in standby mode, primarily for digital displays and control systems [27]. Recent U.S. Department of Energy regulations have established maximum allowable average standby power standards of 0.6 watts for microwave-only ovens and 1.0 watt for built-in and over-the-range convection microwave ovens [27]. Though this represents a relatively small fraction of total energy consumption, mitigating standby power through strategic power management (e.g., power strips, energy-saving modes) contributes to comprehensive energy conservation strategies in research and industrial settings.
Microwave Applications in Hazardous Waste Reduction
Destruction of Persistent Pollutants
Microwave plasma technology has demonstrated exceptional effectiveness in destroying hazardous industrial emissions, particularly persistent greenhouse gases and volatile organic compounds. Microwave abatement systems achieve destruction and removal efficiencies (DRE) exceeding 99.999% for stable compounds like perfluorocarbons (PFCs), which have global warming potentials thousands of times greater than CO₂ [28]. The high-energy plasma environment generated by microwave radiation effectively breaks strong chemical bonds (e.g., C-F bonds in PFCs) that resist conventional treatment methods. This capability is particularly valuable in semiconductor manufacturing, where microwave systems neutralize potent greenhouse gases like NF₃ and CF₄, preventing their atmospheric release [28].
Additionally, microwave-assisted remediation techniques have successfully treated soil and groundwater contaminated with polychlorinated biphenyls (PCBs) and polycyclic aromatic hydrocarbons (PAHs). The in-situ decomposition of these persistent organic pollutants through microwave energy offers significant advantages over excavation and disposal methods, including reduced secondary waste generation and lower transportation emissions [25]. The targeted nature of microwave heating enables specific contamination hotspots to be treated without disturbing surrounding areas, further minimizing environmental impact.
Waste Valorization and Resource Recovery
Beyond destruction, microwave technology enables waste valorization—converting waste materials into valuable resources. Microwave pyrolysis transforms plastic waste and scrap tires into useful fuels and chemical feedstocks, supporting circular economy principles [25]. The process uses indirect heat in oxygen-limited environments to decompose organic materials into gases, liquids, and chars, with microwave heating providing faster and more controlled heating compared to conventional methods. This approach not only reduces waste volume but also generates recoverable resources, creating economic incentives for waste management.
In carbon capture and utilization, microwave-assisted catalysis converts captured CO₂ into valuable syngas through dry reforming of methane [3] [28]. This dual-approach addresses both waste reduction and resource creation by transforming a climate liability (CO₂) into chemical feedstocks. The precision of microwave heating allows these challenging reactions to proceed at lower overall temperatures than conventional methods, further enhancing the energy efficiency and sustainability of the process.
Experimental Protocols for Sustainable Microwave Chemistry
Microwave-Assisted Synthesis of Nanoarchitectures
The synthesis of nanomaterials benefits significantly from microwave approaches, with demonstrated advantages in yield, purity, and size distribution control. The following protocol outlines a generalized method for microwave-assisted nanomaterial synthesis:
Materials Preparation:
- Precursors: Metal salts, organic ligands, or other precursor compounds appropriate for the target nanomaterial.
- Solvents: Polar solvents (e.g., water, ethanol, DMF) that efficiently couple with microwave energy.
- Stabilizing agents: Surfactants or capping agents to control particle growth and prevent aggregation.
Synthetic Procedure:
- Prepare precursor solution by dissolving reactants in selected solvent system.
- Transfer solution to appropriate microwave reaction vessel (sealed or open, depending on requirements).
- Program microwave reactor with optimized parameters:
- Temperature: Typically 150-200°C for nanoparticle synthesis
- Ramp time: 2-5 minutes to reach target temperature
- Hold time: 5-60 minutes at target temperature
- Stirring: Continuous mixing during reaction
- After reaction completion, cool mixture rapidly using integrated cooling systems.
- Recover product by centrifugation or filtration.
- Purify through washing cycles and characterize using standard analytical techniques.
This method typically produces nanomaterials with more uniform size distributions and higher crystallinity compared to conventional heating, while reducing reaction times from hours to minutes [2] [24]. The rapid, uniform heating prevents the development of thermal gradients that often lead to heterogeneous nucleation and growth in conventional synthesis.
Microwave Abatement of Hazardous Gases
The following protocol describes a laboratory-scale approach for microwave-assisted destruction of hazardous gases, adaptable for research on emission control:
System Configuration:
- Microwave generator: 1.8-3.0 kW system operating at 2.45 GHz
- Plasma reactor: Quartz flow tube with appropriate gas fittings
- Gas delivery system: Mass flow controllers for precise gas mixing
- Byproduct capture: Acid gas scrubber for reaction products
Experimental Procedure:
- Set up microwave plasma system with appropriate safety interlocks and ventilation.
- Establish gas flow rates using mass flow controllers:
- Target gas (e.g., CF₄, NF₃): 100-500 sccm
- Additive gases (e.g., O₂, H₂O): Optimized for specific target compounds
- Activate microwave power and carefully tune for stable plasma formation.
- Monitor system parameters:
- Forward and reflected power
- Gas temperature (optical pyrometer)
- Pressure (in-line gauge)
- Sample effluent gases using appropriate analytical methods (FTIR, GC-MS) to quantify destruction efficiency.
- Treat effluent gases through scrubber system to remove acid gas components (e.g., HF, HCl).
- Calculate destruction and removal efficiency (DRE) based on inlet and outlet concentrations.
This approach achieves DRE values >99% for most hazardous gases, with the potential to exceed 99.999% for optimized systems [28]. The protocol can be scaled for industrial applications while maintaining high destruction efficiencies.
Diagram 1: Hazardous gas abatement workflow.
The Research Toolkit: Essential Reagents and Materials
Successful implementation of sustainable microwave chemistry requires specific reagents and materials optimized for microwave interactions. The following table details key components for constructing effective microwave-assisted processes:
Table 3: Essential Research Reagents for Sustainable Microwave Chemistry
| Reagent/Material | Function | Sustainability Role | Application Examples |
|---|---|---|---|
| Zeolites | Porous support with tunable cavities | Enables precise heating at atomic antenna sites | CO₂ conversion, methane reforming [3] |
| Ionic Liquids | Polar solvents with low vapor pressure | Green solvent alternative, efficient microwave coupling | Synthesis medium for nanomaterials [24] |
| Polar Solvents | Reaction medium (water, ethanol, DMF) | Efficient microwave absorption, replaces toxic solvents | General synthesis, nanoparticle production [29] |
| Mineral Supports | Microwave-transparent supports (alumina, silica) | Enables solvent-free reactions | Supported synthesis, dry media reactions [29] |
| Metallic Catalysts | Active sites for reactions (Indium, Nickel) | Enhances reaction efficiency at lower temperatures | Catalytic reactions, fuel production [3] |
| Carbonaceous Materials | Microwave absorbers (graphite, carbon nanotubes) | Provides efficient heating, enables waste valorization | Pyrolysis, composite materials [25] |
Visualization of Microwave Energy Pathways
The following diagram illustrates the fundamental mechanisms through which microwave energy interacts with materials to enhance sustainability outcomes:
Diagram 2: Microwave energy sustainability pathways.
Microwave technology represents a paradigm shift in sustainable chemistry, offering measurable improvements in energy efficiency and hazardous waste reduction. The documented 4.5-fold increase in energy efficiency for chemical transformations [3], coupled with exceptional destruction efficiencies exceeding 99.999% for hazardous compounds [28], establishes microwave-assisted processes as essential tools for green chemistry initiatives. The precise heating mechanisms, reduced reaction times, and ability to enable solvent-free reactions collectively contribute to a more sustainable approach to chemical synthesis and waste processing.
Future developments in microwave technology will likely focus on scaling laboratory successes to industrial implementation, with particular emphasis on reactor design, process optimization, and integration with renewable energy sources. The ongoing research into specialized catalysts and microwave-absorbing materials will further enhance the efficiency and applicability of these methods across diverse chemical domains. As microwave instrumentation continues to evolve, its role in enabling sustainable chemistry through reduced energy consumption and minimized waste generation will expand, solidifying its position as a cornerstone technology for environmentally responsible scientific progress.
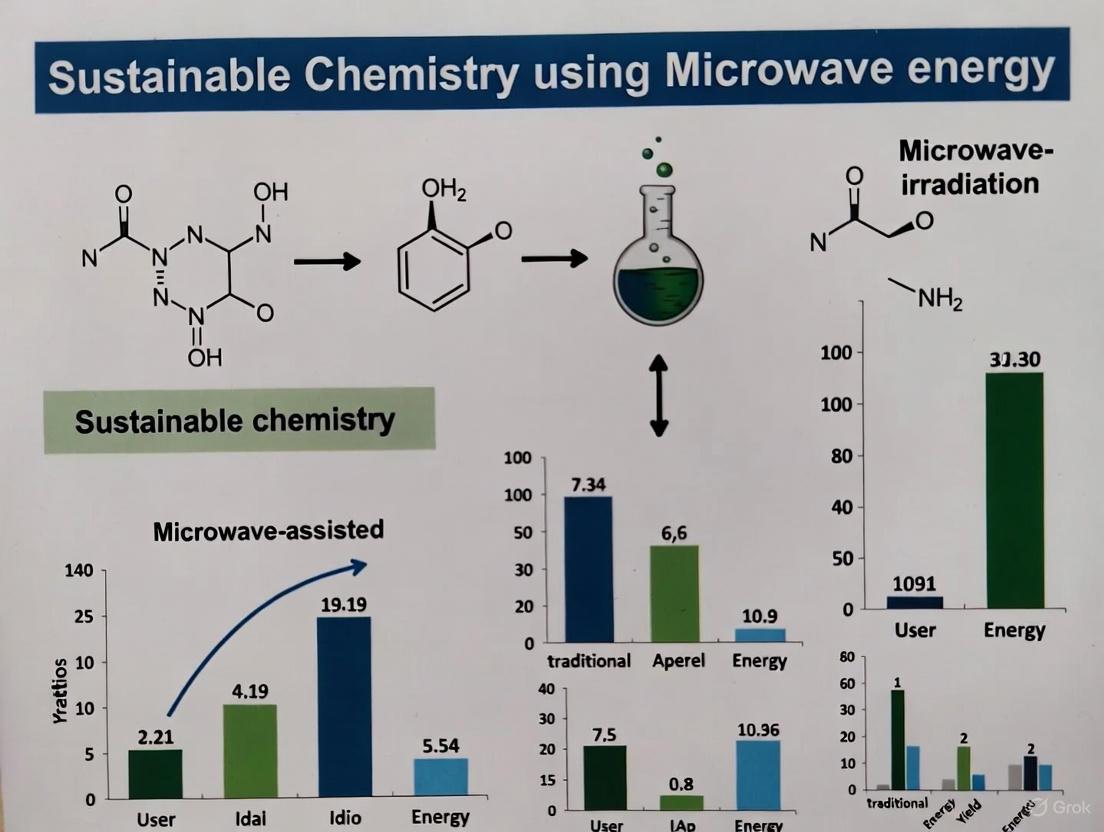
Organic synthesis, defined as the art and science of constructing organic compounds from simpler starting materials, stands as a cornerstone of modern chemistry with profound implications for medicine, materials science, and technology [30] [31]. This discipline has evolved from early explorations of natural substances to the sophisticated, targeted creation of complex molecules. Within contemporary research, a paradigm shift is underway toward sustainable practices, seeking to minimize the environmental impact of chemical processes. In this context, microwave-assisted organic synthesis (MAOS) has emerged as a transformative technology that aligns with the principles of green chemistry by offering dramatic reductions in reaction times, improved energy efficiency, and diminished waste generation [32] [33]. This whitepaper traces the historical foundations of organic synthesis and examines its modern evolution through the lens of microwave energy applications, providing technical guidance for researchers pursuing sustainable synthetic methodologies.
Historical Foundations of Organic Synthesis
The development of organic synthesis represents a remarkable journey from mystical practices to precise molecular construction. This evolution unfolded through several key phases:
From Alchemy to Scientific Chemistry
Before the 19th century, understanding of organic chemicals was primarily embedded in alchemical traditions, where experimentation was often conducted in secrecy with philosophical rather than systematic scientific goals [34]. While alchemists discovered methods to isolate certain organic compounds like acetic acid and citric acid from natural sources, they lacked a coherent theoretical framework for deliberate synthesis [34].
The Demise of Vitalism and Early Synthesis
A pivotal conceptual shift occurred with the abandonment of vitalism—the doctrine that organic compounds could only be produced by living organisms through a "vital force" [30] [34]. This paradigm was definitively challenged in 1828 when Friedrich Wöhler serendipitously synthesized urea, a natural product, from inorganic starting materials (ammonium cyanate) [30] [34]. This seminal demonstration that organic molecules obeyed the same chemical principles as inorganic compounds marked the birth of organic synthesis as a science [30]. The subsequent synthesis of acetic acid by Hermann Kolbe in 1845 further cemented this principle [30].
Industrial Applications and Structural Theory
The late 19th and early 20th centuries witnessed the convergence of synthetic methodology with structural theory. August Kekulé's elucidation of the benzene structure and Archibald Scott Couper's development of structural formulas provided the theoretical foundation for rational synthesis design [30] [34]. Concurrently, practical applications flourished:
- 1856: William Henry Perkin discovered mauveine, the first synthetic dye, creating the foundation for the dye industry [30] [34].
- 1897: Felix Hoffmann synthesized acetylsalicylic acid (aspirin), pioneering the pharmaceutical industry [30] [34].
- Early 20th century: Emil Fischer's work on sugar and purine syntheses earned him the 1902 Nobel Prize, demonstrating the growing sophistication of the field [30].
Table 1: Key Milestones in Early Organic Synthesis
| Year | Scientist | Achievement | Significance |
|---|---|---|---|
| 1828 | Friedrich Wöhler | Synthesis of urea | Challenged vitalism; birth of organic synthesis |
| 1845 | Hermann Kolbe | Synthesis of acetic acid | Extended synthesis to more complex molecules |
| 1856 | William Henry Perkin | Synthesis of mauveine | Founded synthetic dye industry |
| 1897 | Felix Hoffmann | Synthesis of aspirin | Pioneered pharmaceutical synthesis |
| 1902 | Emil Fischer | Sugar and purine syntheses | Advanced complexity achievable in synthesis |
The Emergence of Microwave-Assisted Organic Synthesis
Fundamental Principles
Microwave-assisted organic synthesis utilizes electromagnetic radiation in the frequency range of 0.3-300 GHz, with 2.45 GHz being the standard for laboratory applications due to its optimal penetration depth and regulatory approvals [35] [33]. Unlike conventional conductive heating, which relies on temperature gradients, microwave energy delivers heat volumetrically through two primary mechanisms:
- Dipole rotation: Polar molecules attempt to align with the rapidly oscillating electric field, generating heat through molecular friction [35].
- Ionic conduction: Charged species accelerate under the electric field, colliding with neighboring molecules to produce thermal energy [35].
This direct energy transfer results in instantaneous localized superheating that dramatically enhances reaction kinetics without altering molecular structure, as microwave photon energy (0.037 kcal/mol) is significantly lower than typical bond dissociation energies (80-120 kcal/mol) [35].
Historical Development
The application of microwaves to chemical synthesis began with pioneering work in 1986 by Gedye and Giguere/Majetich, who independently demonstrated that organic reactions in domestic microwave ovens could be dramatically accelerated [33]. Early adoption was limited by safety concerns and equipment limitations, but the mid-1990s saw critical advancements with the introduction of dedicated microwave reactors that provided precise control over temperature, pressure, and power [33]. This technological progress enabled systematic studies and established MAOS as a reliable methodology by the early 2000s, with applications expanding across diverse domains including heterocyclic synthesis, peptide chemistry, and materials science [33].
Microwave Synthesis in Modern Sustainable Chemistry
Green Chemistry Principles
MAOS aligns with multiple principles of green chemistry, making it particularly valuable for sustainable synthesis [33]:
- Energy Efficiency: Direct molecular heating eliminates thermal gradients, reducing energy consumption by up to 85% compared to conventional methods [36].
- Waste Reduction: Enhanced selectivity and fewer by-products decrease purification requirements and solvent usage [32] [33].
- Safer Solvents: MAOS enables reactions in water, ionic liquids, or solvent-free conditions, reducing reliance on hazardous organic solvents [33].
- Atom Economy: Rapid, selective heating facilitates catalytic transformations and multi-component reactions with improved atom utilization [33].
Quantitative Enhancements in Reaction Efficiency
The kinetic advantages of microwave irradiation are demonstrated across diverse reaction classes, with documented rate accelerations of up to 1000-fold compared to conventional heating [35]. These dramatic improvements stem from the ability to achieve high instantaneous temperatures that rapidly overcome activation barriers.
Table 2: Representative Rate Enhancements in Microwave-Assisted Organic Reactions
| Reaction Type | Conventional Time | MW Time | Rate Enhancement | Yield Improvement | Reference |
|---|---|---|---|---|---|
| Aldol Condensation | 3-20 hours | 15-20 minutes | ~12-60x | Comparable or improved | [32] |
| Baker-Venkataraman Rearrangement | >1 hour | Minutes | >15x | >60% maintained | [32] |
| Knoevenagel Condensation | 12-31 hours | 1 hour | ~12-31x | Comparable or improved | [32] |
| Heck Reaction | Hours | <10 minutes | >24x | Significantly improved | [32] |
| Heterocyclic Synthesis | Hours | 5-10 minutes | ~30-60x | >85% | [33] |
The underlying physics of these enhancements can be understood through the Arrhenius equation (k = Ae^(-Ea/RT)), where microwave irradiation primarily affects the temperature parameter. Calculations demonstrate that for a reaction with an activation energy of 50 kcal/mol at a bulk temperature of 150°C, merely a 17°C instantaneous temperature increase produces a 10-fold rate enhancement, while a 56°C increase yields a 1000-fold improvement—readily achievable through microwave superheating [35].
Experimental Protocols and Methodologies
General Workflow for Microwave-Assisted Synthesis
The following diagram illustrates a standardized workflow for developing and optimizing microwave-assisted synthetic protocols:
Representative Experimental Procedures
Objective: Synthesis of chalcones as flavonoid precursors via base-catalyzed aldol condensation.
Reagents:
- 2'-Hydroxyacetophenone (1.0 equiv)
- Benzaldehyde derivatives (1.2 equiv)
- Potassium hydroxide (40% w/v aqueous solution, catalytic amount)
- Ethanol (reaction medium)
Procedure:
- Combine 2'-hydroxyacetophenone (1.0 mmol), benzaldehyde derivative (1.2 mmol), and ethanol (5 mL) in a dedicated microwave reaction vessel.
- Add aqueous KOH solution (0.5 mL) with stirring.
- Seal the vessel and irradiate in a microwave reactor at 120°C for 15 minutes with continuous stirring.
- After cooling, quench the reaction with ice-cold water (10 mL).
- Acidify carefully with dilute HCl to precipitate the product.
- Collect the solid by filtration and recrystallize from ethanol.
Key Advantages: Reaction time reduced from 3-20 hours (conventional heating) to 15 minutes with excellent yields and scalability to 500g using multi-vessel systems [32].
Objective: Energy-efficient CO₂ conversion using targeted microwave heating.
Reagents:
- Zeolite support (sponge-like, controlled pore size)
- Indium ions (catalytic antenna sites)
- Reactant gases (CO₂, methane)
Procedure:
- Prepare zeolite-supported indium catalyst by ion exchange.
- Load catalyst into a continuous-flow microwave reactor.
- Tune microwave frequency to ~900 MHz optimized for indium excitation.
- Flow reactant gases through the catalyst bed at optimal space velocity.
- Apply microwave irradiation at controlled power (300W typical).
- Monitor product formation by online GC-MS.
Key Advantages: 4.5× higher energy efficiency versus conventional heating; selective heating of atomic active sites reduces overall energy demand; enables carbon capture and utilization [4].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for Microwave-Assisted Organic Synthesis
| Reagent/Material | Function | Application Examples | Sustainability Considerations |
|---|---|---|---|
| Polar solvents (water, DMF, DMSO) | Efficient microwave absorption | General synthesis, heterocycle formation | Water as green solvent; recycle high-boiling solvents |
| Ionic liquids | Solvent/catalyst with high microwave susceptibility | Transition metal catalysis, oxidation | Reusable reaction media; replace volatile organics |
| Solid mineral supports (alumina, silica) | Solvent-free reaction media | Knoevenagel condensation, rearrangement | Eliminate solvent waste; enable simple product isolation |
| Heterogeneous catalysts (zeolites, supported metals) | Atomic microwave antennas | CO₂ conversion, methane activation | Targeted energy absorption; high stability and reusability |
| Dedicated microwave reactors | Controlled energy input | All microwave-assisted transformations | Precise temperature/pressure control; reproducible results |
Current Research and Future Perspectives
Advanced Applications
Recent research has expanded MAOS applications into emerging domains:
- Nanomaterial Synthesis: Microwave-assisted methods enable precise control over nucleation and growth for metal nanoparticles, carbon quantum dots, and hybrid nanocomposites with applications in catalysis, energy storage, and biomedicine [36].
- Drug Discovery: MAOS facilitates rapid generation of heterocyclic compound libraries for biological screening, with particular utility in synthesizing chromone, quinolone, and flavonoid derivatives with demonstrated bioactivity [32] [33].
- Polymer Chemistry: Controlled microwave irradiation enables precise polymerization kinetics and functional group tolerance for specialized polymers [33].
Sustainability Assessment
Quantitative metrics demonstrate the environmental advantages of MAOS. A comprehensive sustainability assessment comparing microwave-assisted nanomaterial synthesis with conventional methods revealed:
- Energy consumption: Reduced by 60-85% through direct molecular heating [36]
- Reaction time: Shortened by 80-98% across diverse transformations [32] [36]
- Solvent usage: Decreased by 50-90% via solvent-free or concentrated conditions [36] [33]
- E-factor (waste per product unit): Lowered through improved selectivity and reduced purification needs [36]
Future Research Directions
Despite significant progress, challenges remain in scaling MAOS for industrial adoption. Future research priorities include:
- Scalable Reactor Design: Developing continuous-flow microwave systems for industrial-scale production [4] [33].
- Catalyst Optimization: Engineering tailored materials with enhanced microwave susceptibility for specific transformations [4].
- Process Integration: Combining microwave irradiation with other sustainable technologies (photocatalysis, biocatalysis) [33].
- Predictive Modeling: Utilizing artificial intelligence to optimize reaction parameters and predict microwave effects [31].
The following diagram illustrates the strategic roadmap for advancing microwave-assisted synthesis toward broader industrial application:
Organic synthesis has undergone a remarkable evolution from its origins in challenging vitalism to the current era of precision molecular design. Microwave-assisted organic synthesis represents a paradigm shift within this historical continuum, offering tangible solutions to sustainability challenges through dramatic improvements in energy efficiency, reaction rates, and waste reduction. As research addresses current limitations in scalability and catalyst design, MAOS is poised to play an increasingly central role in sustainable chemical manufacturing. For researchers in pharmaceutical development and materials science, mastering microwave-assisted techniques provides a powerful strategy for accelerating discovery while aligning with green chemistry principles. The continued integration of microwave technology with synthetic chemistry promises to yield innovative solutions to global challenges in health, energy, and environmental protection.
Implementing Microwave Techniques: From Drug Scaffolds to Nanomaterials
The pursuit of sustainable chemistry demands technologies that enhance energy efficiency, reduce waste, and improve process safety. Within this framework, microwave-assisted organic synthesis has emerged as a powerful tool for process intensification. A critical decision in designing microwave-assisted reactions is whether to use a pressurized (closed vessel) or an atmospheric (open vessel) system. This choice profoundly impacts reaction kinetics, safety, scalability, and the environmental footprint of the process.
Microwave energy provides a unique advantage by enabling direct, volumetric heating of reaction mixtures, leading to dramatically reduced reaction times and improved yields compared to conventional conductive heating [37]. This guide provides an in-depth technical comparison of pressurized and atmospheric microwave reactions, offering detailed protocols and data to help researchers and development professionals in the pharmaceutical and chemical industries select the optimal setup for their sustainable chemistry goals.
Technical Comparison: Pressurized vs. Atmospheric Systems
The choice between reactor types involves trade-offs between temperature, safety, and application suitability. The table below summarizes the core technical differences.
Table 1: Technical Comparison of Atmospheric vs. Pressurized Microwave Reactors
| Parameter | Atmospheric (Open Vessel) Reactors | Pressurized (Closed Vessel) Reactors |
|---|---|---|
| Maximum Operating Temperature | Limited by solvent boiling point (e.g., ~100°C for water, ~80°C for THF). | Can significantly exceed solvent boiling point (e.g., 200-300°C is common) [38]. |
| Pressure | Ambient (atmospheric) pressure. | Elevated pressure (e.g., up to 19-30 bar reported) [38]. |
| Key Advantages | - Suitable for reactions at reflux.- Easier sampling and addition of reagents.- Lower safety risk. | - Access to superheated conditions, accelerating reaction rates.- Prevention of solvent evaporation or volatile reagent loss.- Enables reactions in non-boiling solvents. |
| Limitations & Risks | - Cannot achieve temperatures above solvent BP.- Not suitable for volatile reagents. | - Requires robust safety protocols for high-pressure containment.- More complex reactor design and operation. |
| Ideal Application Scope | - Reactions requiring gradual solvent evaporation.- Large-scale synthesis with high-boiling-point solvents. | - High-temperature synthesis (e.g., Skraup reaction) [38].- Processes requiring precise temperature/pressure control. |
Impact on Reaction Performance and Sustainability
The reactor environment directly influences reaction outcomes and green chemistry metrics.
Reaction Kinetics and Yield: Pressurized systems enable dramatic rate enhancements. For instance, the quinoline synthesis via a Skraup reaction achieved a 42% yield in a continuous microwave pressurized reactor at 200-220°C and up to 19 bar, a feat not possible at atmospheric pressure [38]. Similarly, studies on heterogeneous catalytic reactions like esterification and hydrolysis showed yield increases of 150% to 200% under microwave heating in pressurized batch reactors compared to conventional methods, attributed to localized superheating at catalytic sites [39].
Energy Efficiency and Selectivity: A key sustainable advantage of microwave heating is its ability to focus energy on specific molecular sites or catalysts. Recent research demonstrates that microwaves tuned to specific frequencies (e.g., ~900 MHz) can excite "atomic antenna" sites within a zeolite catalyst, achieving energy efficiencies approximately 4.5 times higher than conventional heating methods [3] [4] [40]. This selective heating can also alter reaction pathways, favoring thermodynamic products over kinetic ones and improving diastereoselectivity in syntheses [37].
Experimental Protocols and Workflows
Workflow for a Pressurized (Closed Vessel) Reaction
The following diagram illustrates the standard operational workflow for a safe and effective pressurized microwave reaction, typical for small-scale discovery chemistry.
Diagram 1: Pressurized reaction workflow.
Detailed Methodology for a Model Reaction: Quinoline Synthesis [38]
- Reaction: Skraup reaction for quinoline synthesis from glycerol.
- Objective: To demonstrate a continuous, pressurized microwave process for a high-temperature transformation.
- Reagents & Materials:
- Glycerol (30 mmol), Aniline (10 mmol), Sulphuric Acid (30 mmol), Water (10 mL).
- Reactor: A custom PTFE (polytetrafluoroethylene) tubular reactor housed within a single-mode microwave applicator, designed to withstand pressures up to 19 bar and temperatures up to 220°C.
- Procedure:
- Preparation: The liquid feed mixture (glycerol, aniline, acid, water) is loaded into a feed reservoir.
- Pressurization & Flow: The system is pressurized, and the feed is pumped through the PTFE reactor at a controlled flow rate (e.g., 1.33 mL/s) to achieve the desired residence time.
- Microwave Irradiation: Microwave power is applied and regulated based on the measured pressure to maintain the target temperature of 200-220°C.
- Cooling & Collection: The effluent exits the reactor, passes through a cooling system, and is depressurized before collection.
- Key Parameters:
- Temperature: 200-220°C
- Pressure: ≤ 19 bar
- Residence Time: Several minutes (dependent on flow rate and reactor volume)
- Analysis: The collected product is analyzed by GC-MS or NMR to determine the quinoline yield (42% reported in the continuous system [38]).
Workflow for an Atmospheric (Open Vessel) Reaction
The workflow for an atmospheric reaction is simpler, focusing on reflux or concentration.
Diagram 2: Atmospheric reaction workflow.
Detailed Methodology for a Model Reaction: Esterification [39]
- Reaction: Esterification of stearic acid.
- Objective: To compare the efficiency of microwave heating under atmospheric conditions vs. conventional heating.
- Reagents & Materials:
- Stearic Acid, Alcohol (e.g., methanol), Acid Catalyst (e.g., granulated ceramics or montmorillonite).
- Reactor: A microwave system equipped with an open vessel and a reflux condenser.
- Procedure:
- Preparation: The substrates and solid acid catalyst are combined in the open vessel.
- Reflux: The vessel is fitted with a condenser, and microwave irradiation is applied to bring the mixture to a gentle reflux.
- Reaction Monitoring: The reaction is stirred under reflux for a set time (e.g., 120 minutes).
- Work-up: The catalyst is filtered off, and the product is isolated.
- Key Parameters:
- Temperature: ~140°C (solvent-dependent)
- Pressure: Ambient
- Analysis: Yield is determined gravimetrically or via chromatography. A 97% yield was reported for microwave heating versus 83% for classical heating at the same bulk temperature, highlighting the effect of localized superheating of the catalyst [39].
The Scientist's Toolkit: Essential Research Reagents & Materials
The successful implementation of microwave-assisted reactions relies on specialized materials and catalysts.
Table 2: Key Reagent Solutions and Materials for Microwave Chemistry
| Reagent/Material | Function & Rationale | Application Example |
|---|---|---|
| Zeolite-based Catalysts (e.g., In-doped Zeolite) | Acts as a "molecular sponge" and microwave antenna. The indium ions efficiently absorb microwave energy, creating localized hot spots for reaction activation [3] [4]. | CO₂ conversion, methane reforming, and other eco-catalysis reactions requiring high activation energy. |
| Solid Acid Catalysts (e.g., Montmorillonite, Granulated Ceramics) | Provides a highly microwave-absorbent, reusable catalytic surface. Enables "dry media" reactions, reducing solvent waste [39]. | Isomerization of m-xylene, esterification of fatty acids, and other heterogeneous acid-catalyzed reactions. |
| Polar Solvents (e.g., Water, DMF, DMSO) | High dielectric loss factor allows for efficient coupling with microwave energy, leading to rapid heating. | General use as a reaction medium in both atmospheric and pressurized systems. |
| Microwave-Transparent Reactors (PTFE, Quartz Glass) | Allows microwave energy to pass through and interact with the reaction mixture while withstanding high pressure and temperature [38]. | Essential construction material for pressurized reactor vessels in continuous and batch systems. |
Sustainability and Energy Efficiency Analysis
The drive for sustainable chemistry makes the energy efficiency of microwave processes a critical metric.
Focused Energy Delivery: Conventional heating wastes energy by heating reactor walls and the entire volume. In contrast, microwave energy can be focused on atomic active sites within a catalyst. For example, research using zeolite sponges with indium antenna sites demonstrated energy efficiencies 4.5 times higher than conventional methods, directly reducing the carbon footprint of chemical processes [4] [40].
Process Intensification and Waste Reduction: Microwave reactions are significantly faster, reducing overall energy consumption per unit of product. The ability to use solid catalysts and operate under solvent-free or "dry-media" conditions [39] aligns with green chemistry principles by minimizing solvent waste. Furthermore, pressurized systems can facilitate new pathways for carbon capture and utilization, such as converting captured CO₂ into fuel precursors through energy-efficient microwave-activated catalysis [3] [41].
The choice between pressurized and atmospheric microwave reactors is not merely a technical preference but a strategic decision aligned with the principles of sustainable chemistry. Pressurized systems unlock unparalleled access to high-temperature regimes, enabling faster reaction rates and novel synthetic pathways, albeit with greater engineering complexity. Atmospheric systems offer simplicity and safety for reactions at reflux and are often more straightforward to scale.
The future of microwave-assisted synthesis lies in the continued development of intelligent, scalable reactors and tailored catalytic materials that maximize energy efficiency. As research progresses, the integration of microwave technology with renewable energy sources and its application to critical challenges like carbon dioxide recycling and plastic upcycling will further cement its role as a cornerstone of green chemical manufacturing [3] [41]. For today's researcher, a deep understanding of these reactor options is the first step toward designing cleaner, safer, and more efficient chemical processes.
Microwave-Assisted Organic Synthesis (MAOS) has emerged as a cornerstone of green chemistry, revolutionizing synthetic methodologies by offering dramatic reductions in reaction times, improved energy efficiency, and minimized environmental impact [42] [33]. Since its pioneering applications in 1986 by Gedye and Giguere, microwave chemistry has evolved from a laboratory curiosity to an essential tool in research and development, particularly in pharmaceutical and materials science [24] [33]. The fundamental principle of microwave heating involves the direct interaction of electromagnetic radiation with matter, leading to rapid, volumetric heating through two primary mechanisms: dipolar polarization and ionic conduction [24] [42]. This direct energy transfer enables precise thermal control and often results in enhanced reaction rates, higher product yields, and superior purity compared to conventional heating methods [24].
Framed within the broader context of sustainable chemistry, microwave technology directly addresses multiple principles of green chemistry, including energy efficiency, waste reduction, and the use of safer solvents [42] [33]. The ability of microwave irradiation to facilitate reactions under solvent-free conditions or with environmentally benign solvents further solidifies its position as a transformative green technology [43]. This guide provides a comprehensive framework for selecting solvents based on their microwave-absorbing characteristics, enabling researchers to design synthetic protocols that align with both efficiency and sustainability goals.
Theoretical Foundations of Microwave Heating
Microwave Heating Mechanisms
The efficiency of microwave heating is governed by the interaction between the electromagnetic field and the molecular constituents of the reaction mixture. This interaction is quantified by several dielectric parameters:
- Dipolar Polarization: This mechanism affects molecules possessing a permanent dipole moment. When subjected to the oscillating electric field of microwaves (typically at 2.45 GHz), these dipoles attempt to align themselves with the rapidly changing field. The resulting molecular friction generates heat efficiently. The effectiveness of this process depends on the molecular relaxation time—the time taken for a molecule to achieve 63% of its return to randomized disorder after the removal of the microwave field [44] [42].
- Ionic Conduction: This mechanism operates for ionic species or dissolved salts present in the reaction medium. The oscillating electric field causes these charged particles to move back and forth, colliding with neighboring molecules and converting their kinetic energy into thermal energy [24] [42].
The following diagram illustrates the interplay of these mechanisms and the resulting experimental workflow in microwave-assisted synthesis:
Key Dielectric Properties
The capacity of a solvent to convert microwave energy into heat is characterized by three fundamental dielectric properties [44]:
- Dielectric Constant (ε'): Also known as relative permittivity, this parameter measures a solvent's ability to store electrical energy. Solvents with high dielectric constants generally exhibit strong microwave absorption.
- Dielectric Loss (ε''): This quantifies the efficiency with which a solvent dissipates microwave energy as heat. It is the most direct indicator of a solvent's microwave-absorbing capability.
- Loss Tangent (tan δ): Defined as the ratio ε''/ε', the loss tangent represents the dissipation factor of electromagnetic energy. A higher tan δ value indicates more efficient conversion of microwave energy into thermal energy.
These properties are temperature-dependent and typically decrease as temperature rises, influencing heating efficiency during the course of a reaction [44].
Classification of Solvents by Microwave Absorption
Based on their dielectric loss (ε'') values measured at 2450 MHz and room temperature, solvents can be systematically categorized into three distinct groups: high, medium, and low microwave absorbers [44]. This classification provides a practical framework for solvent selection in microwave-assisted synthesis.
Table 1: Dielectric Properties and Classification of Common Laboratory Solvents [44]
| Solvent | Dielectric Constant (ε') | Dielectric Loss (ε'') | Loss Tangent (tan δ) | Absorption Category |
|---|---|---|---|---|
| Ethylene Glycol | 37.0 | 28.46 | 0.720 | High |
| Ethanol | 24.3 | 22.87 | 0.941 | High |
| DMSO | 46.7 | 21.45 | 0.459 | High |
| Methanol | 32.7 | 20.86 | 0.638 | High |
| Nitrobenzene | 34.8 | 15.00 | 0.431 | High |
| 2-Propanol | 19.9 | 11.00 | 0.553 | Medium |
| Water | 80.4 | 9.89 | 0.123 | Medium |
| DMF | 37.7 | 6.07 | 0.161 | Medium |
| Acetonitrile | 37.5 | 2.33 | 0.062 | Medium |
| Acetone | 20.7 | 1.94 | 0.094 | Medium |
| Dichloromethane | 9.1 | 0.53 | 0.058 | Low |
| Chloroform | 4.8 | 0.11 | 0.023 | Low |
| Ethyl Acetate | 6.0 | 0.06 | 0.010 | Low |
| Tetrahydrofuran (THF) | 7.6 | 0.05 | 0.007 | Low |
| Toluene | 2.4 | 0.01 | 0.004 | Low |
| Hexane | 1.9 | 0.00 | 0.000 | Low |
High Microwave-Absorbing Solvents
Solvents classified as high absorbers exhibit dielectric loss (ε'') values greater than 14.00 [44]. This category includes strongly polar solvents such as:
- Short-chain alcohols (methanol, ethanol, ethylene glycol)
- Dipolar aprotic solvents (DMSO, DMF)
- Specialized solvents (nitrobenzene)
These solvents couple exceptionally well with microwave energy, achieving rapid temperature increases within seconds of irradiation [44]. While this property enables ultra-fast heating, it necessitates careful temperature monitoring to prevent violent reflux or decomposition, especially in sealed vessels. From a green chemistry perspective, ethanol and ethylene glycol represent more sustainable options within this category due to their favorable environmental and safety profiles compared to solvents like DMF or DMSO [45].
Medium Microwave-Absorbing Solvents
Medium absorbers possess dielectric loss values typically between 1.00 and 13.99 [44]. This category includes:
- Water
- Longer-chain alcohols (2-propanol, butanols)
- Polar aprotic solvents (acetonitrile)
- Ketones (acetone)
Water deserves special attention as it demonstrates unique behavior under microwave irradiation. Despite its high dielectric constant (80.4), its dielectric loss (9.89) places it firmly in the medium absorption category [44]. Under pressurized conditions at elevated temperatures, water undergoes significant physicochemical changes—its dielectric constant decreases, and it begins to behave more like an organic solvent, enhancing the solubility of non-polar compounds [44]. This property makes superheated water an exceptionally green reaction medium for microwave-assisted synthesis.
Low Microwave-Absorbing Solvents
Solvents with dielectric loss values below 1.00 are classified as low absorbers [44]. This group includes:
- Chlorinated solvents (dichloromethane, chloroform)
- Ethers (THF, diethyl ether)
- Hydrocarbons (toluene, hexane)
- Ethyl acetate
These solvents heat slowly under microwave irradiation due to their limited ability to couple with the microwave field [44] [33]. While they can be heated to temperatures well above their conventional boiling points in sealed vessels, their use in microwave chemistry often requires the addition of polar additives or the use of passive heating elements to achieve efficient heating. From a sustainability standpoint, many low-absorbing solvents (particularly chlorinated compounds) present significant environmental and health concerns and should be replaced with greener alternatives where possible [45].
The Scientist's Toolkit: Materials and Reagents
Table 2: Essential Research Reagents and Equipment for Microwave-Assisted Synthesis
| Item | Function & Application in Microwave Chemistry |
|---|---|
| Dedicated Microwave Reactor | Provides precise control over temperature, pressure, and power; essential for reproducible and safe experimentation [33]. |
| Sealed Vessels | Enable reactions to be performed at temperatures significantly above the normal boiling point of solvents, expanding synthetic possibilities [44]. |
| Ionic Liquids | Serve as green, non-volatile, and highly microwave-absorbing solvents or catalysts for various transformations [44]. |
| Silicon Carbide (SiC) Reactors | Function as passive heating elements for low-absorbing solvents, enabling reactions that would otherwise not couple with microwave energy [24]. |
| Ethanol & Water | Green solvents of choice for sustainable microwave synthesis; offer excellent safety profiles and microwave absorption characteristics [45] [33]. |
| Solid Supports (e.g., silica, alumina) | Enable solvent-free microwave reactions through reagent immobilization, eliminating solvent use entirely [43]. |
Sustainable Solvent Selection and Experimental Design
Green Chemistry Principles in Solvent Selection
Aligning solvent choice with the 12 Principles of Green Chemistry requires careful consideration of multiple factors beyond microwave absorption efficiency. The guiding principles for sustainable solvent selection include [45] [33]:
- Prefer renewable and bio-based solvents (e.g., ethanol, 2-propanol, glycerol) over petroleum-derived alternatives
- Prioritize human and environmental health by choosing solvents with favorable toxicity profiles
- Minimize solvent use through solvent-free microwave approaches or reduced volumes
- Select biodegradable solvents to reduce environmental persistence
The following diagram outlines a decision-making workflow for selecting sustainable solvents in microwave-assisted synthesis:
Safer Solvent Substitutions
Advancements in green chemistry have identified safer alternatives to many traditional hazardous solvents. The following substitutions are recommended for microwave applications [45]:
- Replace dichloromethane with ethyl acetate/heptane mixtures or 2-methyltetrahydrofuran (2-MeTHF)
- Replace hexane with heptane (safer toxicological profile)
- Replace DMF/DMAc/NMP with acetonitrile or biorenewable solvents like Cyrene (dihydrolevoglucosenone) or γ-valerolactone (GVL)
- Replace diethyl ether and THF with 2-MeTHF or tert-butyl methyl ether (safer peroxide formation profile)
Experimental Protocol: Microwave-Assisted Reaction Optimization
This general protocol can be adapted for various microwave-assisted transformations:
Reaction Vessel Preparation:
- Select appropriate sealed vessel or open container based on reaction requirements
- For a 10 mL total volume, typically use 1-3 mmol of limiting reagent in 5-8 mL of solvent
- Add magnetic stir bar for adequate mixing
Solvent Selection and Optimization:
- Primary choice: Select water or ethanol for green synthesis [33]
- Secondary choice: If reaction necessitates, use acetonitrile or 2-propanol
- Tertiary choice: Consider solvent mixtures for tuning polarity and absorption
Microwave Irradiation Parameters:
- Set initial temperature 20-30°C above conventional reaction temperature
- Program ramp time of 1-3 minutes to reach target temperature
- Adjust hold time at reaction temperature (typically 5-30 minutes for most reactions)
- Use moderate stirring (300-600 rpm) to ensure homogeneity
Reaction Monitoring and Control:
- Monitor temperature and pressure in real-time using built-in sensors
- Use external cooling to maintain precise temperature control
- Employ power modulation (typically 50-150W for small-scale reactions) to prevent overshooting
Work-up and Isolation:
- Allow vessel to cool to room temperature before opening
- For sealed vessels, release pressure slowly in a fume hood
- Transfer reaction mixture and proceed with standard isolation techniques
Advanced Applications and Emerging Trends
Solvent-Free Microwave Chemistry
Solvent-free microwave reactions represent the ultimate green approach in synthetic chemistry, completely eliminating solvent-related environmental impacts [43]. These methodologies include:
- Reactions between neat reagents: Particularly effective for solid-solid and solid-liquid systems where reagents can directly interact under microwave irradiation
- Supported reactions: Using microwave-transparent inorganic supports (silica, alumina, or clay) that adsorb organic reagents, providing high surface area for reactions without solvent mediation
- Phase-transfer catalysis: Enabling reactions between reagents in different phases without solvent dissolution
Solvent-free conditions often result in enhanced reaction rates, cleaner product profiles, and simplified workup procedures, as purification often involves simple extraction or filtration [43].
Microwave-Assisted Extraction (MAE)
Beyond synthetic applications, microwave technology has revolutionized extraction processes for natural products and functional compounds from biological matrices [46]. Key advantages include:
- Reduced extraction times (minutes instead of hours)
- Lower solvent consumption (50-90% reduction compared to conventional methods)
- Higher extraction yields and preservation of thermolabile compounds
- Improved selectivity for target compounds
The efficiency of MAE depends on the dielectric properties of both the plant material and the extraction solvent, with ethanol-water mixtures often providing optimal green extraction media [46].
The strategic selection of solvents based on their microwave-absorbing characteristics represents a critical aspect of sustainable chemistry design. This guide provides a comprehensive framework for categorizing solvents according to their dielectric properties while aligning with green chemistry principles. The integration of microwave technology with solvent-free approaches, aqueous systems, and bio-based solvents offers a powerful pathway toward more sustainable synthetic practices. As microwave chemistry continues to evolve, its synergy with green chemistry principles will undoubtedly yield increasingly innovative solutions for the challenges of 21st-century chemical synthesis.
Nitrogen- and oxygen-containing heterocycles such as quinoline, coumarin, and isatin represent privileged scaffolds in medicinal chemistry, forming the core structures of numerous pharmaceutical agents and natural products. These compounds exhibit a broad spectrum of biological activities, including anticancer, antimicrobial, antiviral, and anti-inflammatory properties [47] [48] [49]. Traditional synthetic approaches to these heterocycles, including the Skraup, Paal-Knorr, Fischer, Pechmann, and Knoevenagel reactions, often necessitate harsh conditions, extended reaction times, and hazardous solvents, resulting in limited functional group tolerance and environmental concerns [47] [49].
The field of organic synthesis has undergone a paradigm shift with the emergence of non-conventional energy sources, particularly microwave irradiation, which aligns with the principles of green chemistry by enhancing synthetic efficiency while reducing environmental impact [50] [33]. Microwave-assisted organic synthesis (MAOS) provides rapid, volumetric heating that frequently leads to dramatic reductions in reaction times, improved yields, enhanced selectivity, and diminished energy consumption [47] [48] [33]. This technical guide examines contemporary, efficient synthetic methodologies for constructing bioactive quinoline, coumarin, and isatin derivatives, emphasizing microwave-assisted and other sustainable approaches that support the broader objective of sustainable chemistry through microwave energy research.
Sustainable Synthetic Methodologies: Principles and Practices
The limitations of traditional heating methods have catalyzed the development of alternative energy sources that offer greater control, efficiency, and sustainability. Microwave irradiation, ultrasound, and mechanochemical approaches constitute the forefront of these green synthetic technologies.
Microwave-Assisted Organic Synthesis (MAOS) operates on the principle of dielectric heating, where polar molecules or solvents align with the rapidly oscillating electric field (typically at 2.45 GHz), generating heat through molecular friction [47] [33]. This mechanism enables direct energy transfer to the reactants, achieving reaction temperatures rapidly and uniformly throughout the sample. The efficiency of microwave absorption depends on the solvent's dissipation factor (tanδ = ε′′/ε′), with polar solvents like water, methanol, and DMF being excellent microwave absorbers [47]. Modern dedicated microwave reactors (e.g., CEM Discover SP, Biotage Initiator+, Anton Paar Monowave 400) provide precise control over temperature, pressure, and power, enabling reproducible and scalable synthesis from milligram to multigram scales [47].
Sonochemical Synthesis utilizes ultrasonic irradiation (20 kHz-1 MHz) to drive chemical reactions through acoustic cavitation—the formation, growth, and implosive collapse of microbubbles in liquid media. This phenomenon generates localized extreme conditions (temperatures up to 5000 K and pressures exceeding 1000 bar) that enhance reaction rates, improve mass transfer, and frequently increase product yields [50] [47]. Ultrasound-assisted synthesis is particularly advantageous for heterogeneous reactions and multicomponent transformations.
Mechanochemical Synthesis employs mechanical forces (e.g., compression, shear, impact) to induce chemical transformations, typically using ball mills (e.g., Retsch PM series, Fritsch Pulverisette) [47]. This solvent-minimized or solvent-free approach eliminates the environmental burden associated with volatile organic solvents and can access novel reaction pathways and polymorphs not available through conventional solution chemistry.
Quantitative Comparison of Conventional vs. Non-Conventional Methods
The advantages of non-conventional synthesis are evident when comparing reaction parameters and outcomes with traditional methods.
Table 1: Performance Comparison of Conventional Heating vs. Microwave-Assisted Synthesis
| Reaction Type | Traditional Conditions | Microwave Conditions | Yield (%) Traditional | Yield (%) Microwave |
|---|---|---|---|---|
| Quinoline Synthesis (Friedländer) | Reflux, 2-4 hours | 100°C, 4 minutes | 60-70% | 80-95% [48] |
| Coumarin-Heterocycle Formation | Reflux, 6-12 hours | 120-150°C, 10-30 minutes | 40-75% | 85-98% [50] |
| Isatin-Hydrazone Formation | Reflux, 3-5 hours | 80-100°C, 5-15 minutes | 50-70% | 85-95% [51] |
| N-Heterocyclic Compound Synthesis | 80-120°C, 6-24 hours | 100-150°C, 5-20 minutes | 45-80% | 75-95% [47] |
Table 2: Green Chemistry Metrics for Non-Conventional Synthesis Methods
| Method | Energy Consumption | Reaction Time Reduction | Solvent Usage | Atom Economy | Waste Reduction |
|---|---|---|---|---|---|
| Microwave | Low (direct energy transfer) | 10-100x faster | Often solvent-free or green solvents | High | Significant [33] |
| Ultrasound | Moderate | 5-50x faster | Aqueous media possible | High | Moderate to high [47] |
| Mechanochemical | Low | 3-20x faster | Solvent-free | High | Significant [47] |
Quinoline Synthesis: Modern Green Approaches
Quinoline, a benzopyridine scaffold, constitutes a fundamental structural motif in numerous bioactive molecules, including the antimalarials chloroquine and hydroxychloroquine, and the anticancer agent camptothecin [52] [48]. Recent advances have focused on sustainable, atom-economical routes to functionalized quinolines.
Microwave-Assisted One-Pot Quinoline Synthesis
Ahmed and colleagues developed an efficient, solvent-free three-component domino reaction for synthesizing quinolin-4-ylmethoxychromen derivatives using ytterbium(III) chloride catalysis under microwave irradiation [48]. This method exemplifies the power of MAOS in multicomponent reactions, achieving excellent yields (80-95%) in merely 4 minutes at 100°C, compared to 60 minutes with conventional heating.
Experimental Protocol:
- Reaction Setup: Combine equimolar quantities of propargylated-flavone or coumarin (1a-1b, 1 mmol), aldehyde (3a-g, 1 mmol), and aniline (2a-e, 1 mmol) in a microwave vessel.
- Catalyst Addition: Add YbCl₃ (5 mol%) to the reaction mixture.
- Microwave Conditions: Irradiate the open vessel at 100°C for 4 minutes without solvent.
- Work-up: Upon completion (monitored by TLC), triturate the crude product with ethanol.
- Purification: Filter the solid product and recrystallize from ethanol to afford pure quinolin-4-ylmethoxychromen derivatives (4a-n and 5a-e) [48].
This protocol demonstrates several green chemistry advantages: solvent-free conditions, minimal catalyst loading, excellent atom economy (95%), and recyclability of the catalytic system.
C-H Bond Functionalization Strategies
Modern quinoline synthesis increasingly employs selective C-H functionalization to streamline the construction of complex quinoline architectures. Recent methodologies enable direct functionalization of the quinoline core through transition-metal catalysis, providing efficient access to structurally diverse analogs with enhanced pharmacological profiles [53]. These approaches typically employ palladium, rhodium, or ruthenium catalysts with directing groups to achieve regioselective C-H activation, followed by coupling with various partners including alkenes, alkynes, and aryl halides.
Diagram: Strategic Approaches to Quinoline Synthesis
Coumarin Heterocycles: Innovative Synthetic Strategies
Coumarins (benzopyran-2-ones) represent an important class of oxygenated heterocycles with widespread natural occurrence and diverse pharmacological activities, including anticoagulant, anticancer, antimicrobial, and antioxidant effects [50] [49]. Contemporary synthetic methodologies have evolved beyond classical approaches like the Pechmann and Perkin reactions to include transition-metal-catalyzed transformations, photoredox processes, and multicomponent reactions.
Microwave- and Ultrasound-Assisted Coumarin Synthesis
Green synthesis techniques have been extensively applied to coumarin-heterocycle formation. Microwave-assisted conditions typically achieve reaction times of 10-30 minutes at 120-150°C with yields exceeding 85%, a significant improvement over conventional heating which often requires 6-12 hours with moderate yields [50]. Similarly, ultrasound-assisted coumarin synthesis proceeds under milder conditions (room temperature to 80°C) with reduced reaction times (20-60 minutes) and improved yields compared to traditional methods [50].
Experimental Protocol: Ultrasound-Assisted Synthesis of Coumarin-Pyrazole Hybrids
- Reaction Setup: Charge a round-bottom flask with 4-hydroxycoumarin (1 mmol), chalcone derivative (1 mmol), and phenylhydrazine (1 mmol).
- Solvent Conditions: Add ethanol (5-10 mL) as a green solvent.
- Ultrasound Parameters: Subject the reaction mixture to ultrasonic irradiation (35-40 kHz) at 60°C for 45 minutes.
- Reaction Monitoring: Track progress by TLC (ethyl acetate/hexane, 3:7).
- Work-up and Isolation: Upon completion, concentrate the reaction mixture under reduced pressure and collect the solid product by filtration.
- Purification: Recrystallize the crude product from ethanol to afford pure coumarin-pyrazole hybrids in excellent yields (85-90%) [50].
Transition-Metal-Catalyzed Coumarin Functionalization
Recent advances in transition-metal catalysis have enabled direct C-H functionalization of coumarin scaffolds, providing efficient access to structurally diverse analogs. Representative methodologies include:
- Palladium-catalyzed C-H activation: Direct coupling of phenols with acrylates to form coumarin cores [49].
- Rhodium-catalyzed oxidative annulation: Coupling of aryl thiocarbamates with internal alkynes to construct coumarin derivatives [49].
- Silver-mediated radical cyclization: Synthesis of 3-substituted coumarins from alkynoates and α-keto acids via radical processes [49].
These catalytic methods typically proceed under milder conditions with superior regioselectivity and functional group tolerance compared to classical approaches.
Isatin-Based Heterocycles: Sustainable Routes to Privileged Scaffolds
Isatin (1H-indole-2,3-dione) is a privileged nitrogen-containing heterocycle with demonstrated importance in anticancer drug discovery. Marketed isatin-based drugs include sunitinib, nintedanib, and ponatinib, which target receptor tyrosine kinases involved in oncogenic signaling [51] [48]. Sustainable synthesis of isatin hybrids has been achieved through microwave-assisted and solvent-free methodologies.
Microwave-Assisted Isatin-Hybrid Synthesis
Experimental Protocol: Isatin-Thiazole-Coumarin Hybrid Synthesis
- Step 1 - Thiosemicarbazone Formation: Reflux a mixture of 5-chloroisatin (1 mmol, 2) and thiosemicarbazide (1 mmol) in ethanol (10 mL) for 2 hours. Cool and filter to obtain the thiosemicarbazone intermediate (3) in 91% yield [54].
- Step 2 - Hybrid Formation: Combine the thiosemicarbazone intermediate (3, 1 mmol) with 3-(2-bromoacetyl)-6-bromocoumarin (4, 1 mmol) in acetic acid (5 mL). Reflux the reaction mixture for 4-5 hours. Cool and pour into ice-cold water with stirring. Collect the precipitated solid by filtration and recrystallize from ethanol to afford the target isatin-thiazole-coumarin hybrid (5) in 90% yield [54].
Microwave Optimization: This two-step sequence can be significantly accelerated under microwave irradiation. For step 1, microwave at 80°C for 5-10 minutes provides comparable yield to conventional heating. For step 2, microwave at 120°C for 8-12 minutes affords the hybrid in similar yield but with substantially reduced reaction time [51] [54].
Molecular Hybridization Strategies for Enhanced Bioactivity
Molecular hybridization of isatin with complementary pharmacophores represents a powerful strategy for developing multitargeted therapeutic agents with improved efficacy and potential to overcome drug resistance [51] [54]. Representative hybrids include:
- Isatin-triazole-coumarin hybrids: Demonstrate tubulin polymerization inhibition (IC₅₀ ≈ 1-5 μM) and overcome multidrug resistance in prostate and breast cancers [51].
- Isatin-hydrazone derivatives: Suppress Bcl-2 expression, activate caspases, and induce ROS-mediated apoptosis in breast cancer cells [51].
- Isatin-thiazole-coumarin hybrids: Exhibit potent cytotoxic activity against MCF-7 (IC₅₀ = 10.85 μg/mL) and MDA-MB-231 (IC₅₀ = 14.45 μg/mL) breast cancer cell lines, along with anticholinesterase inhibition (IC₅₀ = 0.0998 μg/mL) and anti-MRSA activity (MIC = 1.3 μg/mL) [54].
Diagram: Synthetic Routes to Isatin-Based Hybrid Molecules
Experimental Protocols: Essential Methodologies
General Procedure for Microwave-Assisted Heterocycle Synthesis
- Reaction Vessel Preparation: Charge a dedicated microwave vessel with substrates (0.1-1.0 mmol scale) and appropriate solvent (2-5 mL per mmol substrate) if required. For solvent-free reactions, mix substrates thoroughly.
- Catalyst/Additive Introduction: Add catalyst (1-10 mol%) and any necessary additives to the reaction mixture.
- Microwave Parameters: Program the microwave reactor with optimized conditions: temperature (80-150°C), hold time (5-30 minutes), fixed hold mode, and appropriate stirring rate (medium to high).
- Reaction Execution: Irradiate the reaction mixture according to programmed parameters while monitoring pressure development.
- Work-up Procedure: After irradiation and cooling, dilute the reaction mixture with appropriate solvent (ethyl acetate or water) and transfer to separation equipment if needed.
- Purification Techniques: Purify crude products using standard techniques (recrystallization, column chromatography, or trituration) to afford pure heterocyclic products [47] [48] [33].
Representative Multicomponent Reaction: Solvent-Free Quinoline Synthesis
Materials:
- Propargylated coumarin (1 mmol)
- Aromatic aldehyde (1 mmol)
- Aniline derivative (1 mmol)
- YbCl₃ (5 mol%)
Procedure:
- Mixing: Combine all solid materials in a mortar and grind thoroughly for 2-3 minutes.
- Microwave Processing: Transfer the mixture to a microwave vessel and irradiate at 100°C for 4 minutes.
- Isolation: After cooling, triturate the crude material with cold ethanol (2-3 mL).
- Purification: Filter the solid product and recrystallize from ethanol to afford pure quinoline derivative [48].
The Scientist's Toolkit: Essential Research Reagents and Equipment
Table 3: Key Research Reagent Solutions for Heterocycle Synthesis
| Reagent/Catalyst | Function | Application Examples | Green Alternatives |
|---|---|---|---|
| Yb(OTf)₃ / YbCl₃ | Lewis acid catalyst | Quinoline synthesis, coumarin formation | Recyclable, low toxicity [48] |
| Palladium Catalysts | C-H activation, cross-coupling | Coumarin functionalization, quinoline derivatization | Low loading, supported versions [49] |
| 3-(2-Bromoacetyl)coumarin | Electrophilic coupling partner | Isatin-thiazole-coumarin hybrid synthesis | - [54] |
| Thiosemicarbazide | Thiazole precursor | Isatin-thiazole hybrid formation | - [54] |
| Thiohydantoins | Heterocyclic coupling partner | Isatin-hydantoin hybrid synthesis | - [54] |
Table 4: Essential Equipment for Non-Conventional Synthesis
| Equipment | Key Features | Representative Models | Optimal Application |
|---|---|---|---|
| Microwave Reactor | Temperature/pressure control, magnetic stirring | CEM Discover SP, Biotage Initiator+, Anton Paar Monowave 400 | Small-scale optimization, library synthesis [47] |
| Ultrasonic Bath | Temperature control, adjustable frequency | Branson, Elma, Crest ultrasonic cleaners | Heterogeneous reactions, catalyst preparation [50] |
| Planetary Ball Mill | Variable speed, multiple jar materials | Retsch PM100/PM400, Fritsch Pulverisette 7 | Solvent-free synthesis, mechanochemistry [47] |
| Microwave Reactor Scale-Up Systems | Continuous flow, larger vessel capacity | Milestone Ethos EASY, CEM MARS 6 | Gram-scale synthesis, process development [33] |
The integration of microwave-assisted and other non-conventional synthesis methods has revolutionized the preparation of bioactive heterocycles, enabling rapid, efficient, and sustainable access to quinoline, coumarin, and isatin scaffolds. These methodologies align with green chemistry principles by reducing reaction times, improving energy efficiency, minimizing solvent consumption, and enhancing product yields and purity. The experimental protocols and strategies outlined in this technical guide provide researchers with practical tools for implementing these sustainable approaches in drug discovery and development.
Future directions in this field will likely focus on several key areas: (1) integration of microwave chemistry with continuous flow systems for scalable manufacturing; (2) development of hybrid approaches combining microwave irradiation with biocatalysis or photocatalysis; (3) advancement of computational modeling and AI-assisted reaction optimization; and (4) expansion of solvent-free and mechanochemical methods for complete elimination of volatile organic solvents. As these sustainable methodologies continue to evolve, they will further accelerate the discovery and development of novel therapeutic agents based on privileged heterocyclic scaffolds while reducing the environmental footprint of chemical synthesis.
The fabrication of nanomaterials through green synthesis principles represents a paradigm shift in sustainable chemistry, aligning with global efforts to minimize environmental impact in scientific research and industrial production. This approach leverages microwave energy as a core tool to drive rapid, efficient, and eco-friendly nanomaterial synthesis. Microwave-assisted synthesis (MAS) has emerged as a superior alternative to conventional methods, offering precise control over reaction parameters, significantly reduced energy consumption, and shorter reaction times, thereby embodying the principles of green chemistry [55]. The integration of MAS with bio-based precursors—such as plant extracts and food waste—further enhances the sustainability profile by utilizing renewable resources and minimizing hazardous waste generation [56] [57]. This whitepaper provides an in-depth technical guide for researchers and scientists on the microwave-assisted green synthesis of two critical classes of nanomaterials: metal nanoparticles and carbon quantum dots (CQDs), framing the discussion within the broader thesis of advancing sustainable chemistry through innovative energy application.
Microwave-Assisted Green Synthesis of Metal Nanoparticles
Synthesis Mechanism and Workflow
The microwave-assisted green synthesis of metal nanoparticles utilizes plant-derived extracts as both reducing and stabilizing agents. The polar molecules in the plant extract, such as water and phytochemicals, couple efficiently with microwave radiation, leading to rapid and uniform heating. This instantaneous heating promotes the nucleation and growth of nanoparticles, while the phytoconstituents (e.g., alkaloids, terpenoids, flavonoids) reduce metal ions and cap the newly formed nanoparticles, preventing agglomeration [56]. The following diagram illustrates the core experimental workflow for synthesizing metal nanoparticles, such as silver nanoparticles (AgNPs), using a plant extract.
Detailed Experimental Protocol: Silver Nanoparticles (AgNPs) fromTrigonella hamosaL.
This protocol details the synthesis of AgNPs using Trigonella hamosa L. leaf extract via microwave assistance, achieving nanoparticles with an average size of 14 nm [56].
- Precursor Preparation:
- Plant Extract: Wash and dry the aerial parts of Trigonella hamosa L. Prepare an aqueous extract, typically by boiling dried leaves in deionized water, followed by filtration to remove particulate matter.
- Metal Salt Solution: Prepare an aqueous solution of silver nitrate (AgNO₃). Concentrations between 1-25 mM can be used for optimization [56] [57].
- Reaction Procedure:
- Combine the plant extract with the AgNO₃ solution in a defined ratio (e.g., 2-8 mL extract per fixed volume of AgNO₃ solution) [56] [57].
- Place the mixture in a suitable microwave reactor vessel.
- Irradiate the mixture using a programmed microwave protocol. For instance, use a power setting of 50-100 W and a temperature maintained above the boiling point of the solvent for a short duration (e.g., 5-10 minutes) [29] [55]. The rapid heating leads to the formation of a colloidal suspension indicated by a color change.
- Purification and Recovery:
- Allow the reaction mixture to cool to room temperature.
- Purify the synthesized AgNPs by centrifugation (e.g., at 15,000 rpm for 15-20 minutes) to form a pellet. Discard the supernatant and re-disperse the pellet in deionized water or an organic solvent. Repeat this process 2-3 times to remove any unreacted ions or plant residues.
- The final purified nanoparticles can be stored as a suspension or as a dry powder after freeze-drying.
Key Research Reagent Solutions
The following table summarizes the essential materials and their functions in the green synthesis of metal nanoparticles.
Table 1: Essential Reagents for Green Synthesis of Metal Nanoparticles
| Reagent / Material | Function in the Synthesis Process | Example & Notes |
|---|---|---|
| Metal Salt Precursor | Source of metal ions for reduction into nanoparticles. | Silver nitrate (AgNO₃) for AgNPs [56]. Use high-purity grades. |
| Plant Extract | Acts as both a reducing agent and a stabilizing capping ligand. | Trigonella hamosa L. leaf extract [56] or pineapple leaves waste extract [57]. |
| Reaction Solvent | Medium for the reaction; water is preferred in green synthesis. | Deionized water. Its polarity enables efficient microwave coupling [29]. |
| Purification Solvents | For washing and purifying the synthesized nanoparticles. | Deionized water, ethanol, or acetone [56]. |
Microwave-Assisted Green Synthesis of Carbon Quantum Dots (CQDs)
Synthesis Mechanism and Workflow
The synthesis of CQDs from carbon-rich natural precursors like starch involves microwave-assisted hydrothermal carbonization. Microwave irradiation causes the rapid decomposition and carbonization of the precursor, leading to the formation of small, fluorescent carbon nuclei. Further microwave treatment facilitates their growth and surface functionalization, which determines their fluorescence properties [58]. The process can yield CQDs with multi-color fluorescence depending on the synthesis parameters and post-synthesis processing.
Detailed Experimental Protocol: Fluorescent CDs from Starch
This protocol, adapted from published research, produces CDs with a high fluorescence quantum yield of up to 30% [58].
- Precursor Preparation:
- Carbon Source: Use common food-grade starch as the carbon precursor.
- Reaction Medium: Prepare an aqueous phosphoric acid (H₃PO₄) solution. The acid acts as a catalyst for the hydrolysis and dehydration of starch.
- Reaction Procedure:
- Disperse the starch in the aqueous phosphoric acid solution under stirring to form a homogeneous mixture.
- Transfer the mixture to a microwave reactor vessel suitable for hydrothermal reactions.
- Subject the mixture to microwave irradiation. The synthesis requires heating for a specific duration to efficiently form blue-fluorescent CDs. For example, irradiation at 300 W for a defined period (e.g., several minutes) can be applied. The formation of aggregates, which can be extracted to emit orange/green fluorescence, may occur with extended heating [58].
- Purification and Recovery:
- After cooling, the crude product can be neutralized and purified by dialysis (using a dialysis membrane with a molecular weight cutoff of 500-1000 Da) against deionized water for 24-48 hours to remove excess acid and small molecular by-products.
- Alternatively, liquid-liquid extraction can be used: the reaction mixture can be extracted with an organic solvent like toluene to isolate aggregated CDs that exhibit different fluorescence (orange in toluene, green in water) [58].
- The final aqueous solution of CDs can be stored at 4°C.
Key Research Reagent Solutions
Table 2: Essential Reagents for Green Synthesis of Carbon Quantum Dots
| Reagent / Material | Function in the Synthesis Process | Example & Notes |
|---|---|---|
| Carbon Source | Provides the carbon for the dot structure; often a biomass. | Starch [58]. Other sources include food waste or plant materials. |
| Reaction Medium / Catalyst | Facilitates decomposition and carbonization; can passivate the CD surface. | Aqueous phosphoric acid (H₃PO₄) [58]. |
| Purification Materials | For separating and purifying the synthesized CDs from the reaction mixture. | Dialysis membranes, filter membranes, or organic solvents for extraction [58]. |
Characterization and Performance Evaluation
Essential Characterization Techniques
Confirming the successful synthesis, size, morphology, and surface chemistry of the nanomaterials is crucial. The following techniques are standard in the field.
- UV-Vis Spectroscopy: Used to confirm nanoparticle formation by detecting characteristic absorption peaks. For AgNPs, this is the Surface Plasmon Resonance (SPR) band around 400-430 nm [56]. For CDs, it shows absorption bands related to carbon core and surface functional groups.
- FTIR (Fourier-Transform Infrared) Spectroscopy: Identifies functional groups present on the nanoparticle surface, confirming the role of biomolecules as capping and stabilizing agents [56] [59] [57]. It is a rapid and non-destructive method for surface chemistry analysis.
- XRD (X-Ray Diffraction): Determines the crystallinity and phase of the synthesized nanomaterials. The XRD pattern for AgNPs shows characteristic peaks for face-centered cubic (fcc) silver [56].
- HR-TEM (High-Resolution Transmission Electron Microscopy): Provides direct information on the size, shape, and morphology of the nanoparticles. It is the primary technique for confirming nanoscale dimensions (e.g., 14 nm AgNPs) and near-spherical shapes [56].
- Fluorescence Spectroscopy: Essential for characterizing CDs, used to measure fluorescence quantum yield and emission/excitation profiles [58].
Quantitative Performance Data
The efficacy of green-synthesized nanomaterials is demonstrated through application-specific performance metrics. The following tables summarize quantitative data from recent studies.
Table 3: Photocatalytic Degradation Performance of AgNPs [56]
| Pollutant | Light Source | Degradation Percentage (%) | Key Synthesis Factor |
|---|---|---|---|
| Methylene Blue (MB) Dye | Sunlight | 96.2% | Microwave-synthesized AgNPs (14 nm) |
| Methylene Blue (MB) Dye | Visible Lamp | 94.9% | Microwave-synthesized AgNPs (14 nm) |
| Paracetamol (PCA) | Sunlight | 94.5% | Microwave-synthesized AgNPs (14 nm) |
| Paracetamol (PCA) | Visible Lamp | 92.0% | Microwave-synthesized AgNPs (14 nm) |
Table 4: Antimicrobial Activity of AgNPs from Pineapple Leaves [57]
| Bacterial Strain | Relative Efficacy | Key Synthesis Factor |
|---|---|---|
| Escherichia coli | Greater activity with microwave-assisted synthesis | Spherical nanoparticles (40-150 nm) |
| Bacillus subtilis | Greater activity with microwave-assisted synthesis | Spherical nanoparticles (40-150 nm) |
| Staphylococcus aureus | Greater activity with microwave-assisted synthesis | Spherical nanoparticles (40-150 nm) |
| Minimum Inhibitory Concentration (MIC) | 60 μg/mL | - |
Sustainability and Safety Considerations
Environmental and Economic Benefits
Microwave-assisted green synthesis aligns with several United Nations Sustainable Development Goals by offering substantial environmental and economic advantages over conventional routes.
- Reduced Energy Consumption: MAS protocols often require reaction times of minutes instead of hours or days, leading to a dramatic decrease in overall energy use [55].
- Waste Minimization: The use of water as a solvent, plant extracts as reagents, and the high efficiency of MAS collectively minimize the generation of hazardous waste [55].
- Utilization of Waste Streams: The capability to use agricultural waste (e.g., pineapple leaves) as precursor materials adds a circular economy dimension, reducing raw material costs and waste [57].
Nanomaterial Safety and Handling
Despite their green synthesis route, nanomaterials require careful handling due to potential health and environmental risks that are not yet fully understood [60] [61].
- Exposure Controls:
- Engineering Controls: Conduct all powder handling and liquid aerosol-generating steps (e.g., sonication, centrifugation) in ventilated enclosures such as chemical fume hoods or Class II Biological Safety Cabinets with HEPA-filtered exhaust [60].
- Administrative Controls: Implement standard operating procedures that minimize the generation of dusts and aerosols. Use good housekeeping practices to prevent surface contamination.
- Personal Protective Equipment (PPE):
- Wear appropriate gloves (e.g., nitrile), lab coats, and safety glasses. If engineering controls cannot reliably contain exposure, use respiratory protection equipped with HEPA filters (e.g., N95 mask or higher) [60].
- Spill Management and Waste Disposal:
- Develop specific procedures for managing nanomaterial spills. Collect spill debris as hazardous waste.
- Dispose of all nanomaterial waste, including contaminated gloves and consumables, through a designated hazardous waste program, as the environmental impact regulations are still evolving [60].
The global industrial sector is undergoing a fundamental transition toward sustainability and decarbonization, driving the need for innovative processes that reduce energy consumption and greenhouse gas emissions. Within this context, precision microwave heating has emerged as a transformative technology for enabling sustainable chemistry. Unlike conventional thermal heating, which relies on conductive and convective heat transfer from surfaces, microwave energy delivers electromagnetic waves that interact directly with materials at the molecular level. This interaction enables volumetric heating, where energy penetrates the entire material simultaneously, leading to dramatically faster heating rates, significantly reduced energy consumption, and enhanced process efficiency across various industrial applications [62].
The application of microwave technology aligns with the core principles of green chemistry by minimizing solvent use, reducing reaction times, improving product yields, and lowering overall energy requirements. This whitepaper examines two critical applications of precision microwave heating that contribute to a circular economy: clean hydrogen production and CO2 conversion. These applications demonstrate the potential of microwave-driven processes to transform industrial manufacturing into a more environmentally responsible enterprise while addressing key challenges in sustainable energy and carbon management [36].
Microwave Technology Fundamentals
Core Mechanisms of Microwave-Material Interactions
Microwave heating operates through distinct physical mechanisms that enable its superior efficiency compared to conventional heating methods. The primary mechanisms include:
- Dipolar Polarization: When microwave radiation interacts with polar molecules possessing a permanent dipole moment, these molecules attempt to align themselves with the rapidly oscillating electric field. This continuous reorientation generates molecular friction and collision, resulting in efficient and rapid heating throughout the material volume [63].
- Ionic Conduction: In systems containing ionic species, the applied electric field induces accelerated movement of these charged particles. The resulting collisions between ions and neighboring molecules convert kinetic energy into heat, further enhancing the heating efficiency, particularly in solutions with dissolved salts or ionic liquids [63].
The efficiency of microwave heating for any given material is governed by its dielectric properties, quantified by the complex permittivity (ε = ε′ - jε″). The real component (dielectric constant, ε′) represents the material's ability to store electrical energy, while the imaginary component (dielectric loss factor, ε″) quantifies the efficiency of converting electromagnetic energy into heat. The loss tangent (tanδ = ε″/ε′) serves as a key indicator of a material's microwave absorption capacity [64].
Comparative Advantages Over Conventional Heating
- Volumetric Heating: Energy delivery occurs throughout the material simultaneously rather than progressing slowly from the surface inward, drastically reducing thermal cycling time [62].
- Selective Heating: Materials with different dielectric properties heat at different rates, enabling targeted heating of specific components within a mixture [65].
- Rapid Reaction Kinetics: Microwave heating often enhances reaction rates by factors of thousands compared to traditional methods, completing processes in minutes rather than hours [63].
- Energy Efficiency: Direct energy transfer to the reaction mixture minimizes losses to the environment and reaction vessels, reducing overall power consumption [62].
- Precision Control: Advanced microwave systems provide precise regulation of temperature, pressure, and power parameters, enabling reproducible and optimized reaction conditions [33].
Microwave-Driven Clean Hydrogen Production
Microwave Plasma Reforming of Greenhouse Gases
Experimental Protocol: A novel atmospheric-pressure microwave plasma system operating at 2.45 GHz has been developed for dry reforming of methane (DRM). The system utilizes a waveguide-based plasma source without a dielectric discharge tube, incorporating a swirling gas flow and operating without catalysts. The experimental setup involves introducing controlled mixtures of CO₂ and CH₄ into the plasma region, where microwave energy (typically 2-6 kW) converts these greenhouse gases into hydrogen-enriched syngas. Gas composition is analyzed using gas chromatography, with performance metrics including H₂ concentration, conversion rates, and energy yield calculated based on flow rates and input power [66].
Table 1: Performance Metrics of Microwave Plasma Hydrogen Production from CO₂/CH₄ Mixture [66]
| Parameter | Value | Impact Factor |
|---|---|---|
| H₂ Concentration | 33% | Microwave power beneficial; flow rate detrimental |
| CH₄ Conversion Rate | 46% | Dependent on power and flow conditions |
| H₂ Production Rate | 118 g/h | Optimized at specific CO₂/CH₄ ratio |
| Energy Yield of H₂ | 30 g/kWh | Superior to conventional thermochemical methods |
The underlying chemical process is dry methane reforming: CH₄ + CO₂ → 2H₂ + 2CO, ΔH₂₉₈K = 247 KJ/mol [66]
This approach consumes two major greenhouse gases simultaneously while producing valuable syngas, representing a dual environmental benefit.
Microwave-Assisted Thermochemical Hydrogen Production
Experimental Protocol: Researchers have developed a microwave-assisted thermochemical process for hydrogen production using Gd-doped ceria (CeO₂) as a redox-active material. The experimental methodology involves subjecting the metal oxide to microwave irradiation at specific frequencies, which creates oxygen vacancies—defects in the crystal structure where oxygen atoms are missing. These vacancies are essential for splitting water molecules in subsequent steps. The material is then exposed to steam, where water molecules dissociate at the vacancy sites, producing hydrogen and regenerating the oxide. The cycle repeats with alternating reduction and oxidation steps [67].
Table 2: Comparative Analysis of Conventional vs. Microwave Hydrogen Production [67]
| Production Method | Temperature Requirement | Time Requirement | Energy Input |
|---|---|---|---|
| Conventional Thermal | Up to 1,500°C | Several hours | 100% thermal |
| Microwave-Assisted | Below 600°C | Minutes | 25% thermal, 75% microwave |
This microwave-driven process achieves a 60% reduction in temperature requirement and cuts processing time from hours to minutes while replacing approximately 75% of the thermal energy needed with more efficient microwave energy [67].
Microwave-Assisted CO2 Capture and Conversion
Synthesis of Advanced CO2 Adsorbents
Experimental Protocol: Microwave-assisted synthesis of metal-organic frameworks (MOFs) and porous polymers has demonstrated remarkable efficiency for creating high-performance CO₂ adsorbents. For example, bimetallic NiCo-MOF-74 can be synthesized by combining metal precursors and organic linkers in appropriate solvents and subjecting the mixture to microwave irradiation under controlled temperature and pressure conditions. The rapid, uniform heating achieved through microwave energy enables precise crystal growth and formation of highly porous structures with exceptional CO₂ adsorption capacity [64].
Table 3: Performance of Microwave-Synthesized CO₂ Adsorbents [64]
| Adsorbent Material | Synthesis Time | Surface Area | CO₂ Uptake Capacity |
|---|---|---|---|
| NiCo-MOF-74 | 24x faster than conventional | 1,147 m²/g | 6.68 mmol/g at 25°C, 1 bar |
| NENP-1 (N-doped polymer) | 30 minutes | Not specified | 2.97 mmol/g |
The microwave synthesis of NiCo-MOF-74 achieves a 24-fold reduction in production time compared to conventional methods while maintaining excellent structural properties and adsorption capacity [64].
Microwave-Driven Regeneration and Catalytic Conversion
Experimental Protocol: For adsorbent regeneration, saturated CO₂ capture materials are placed in a microwave reactor and subjected to controlled irradiation. The microwave energy selectively targets the adsorbed CO₂ molecules and the binding sites, facilitating rapid desorption at significantly lower temperatures than conventional thermal regeneration. For catalytic conversion, microwave reactors are loaded with CO₂ and appropriate catalysts (e.g., for the Boudouard reaction), where selective heating activates catalytic sites and promotes specific reaction pathways [64].
- Regeneration Efficiency: Microwave regeneration of MCM-48 mesoporous silica completes in just three minutes—a 75% reduction in time compared to conventional methods. For amine solutions, microwave-assisted regeneration of a 30 wt% MEA solution lowers energy consumption by approximately 33% [64].
- Catalytic Performance: The microwave-assisted Boudouard reaction (CO₂ + C → 2CO) achieves 81.9% CO₂ conversion at 900°C. With the addition of 5% BaCO₃ as a catalyst, conversion increases to 99% with a reduction in activation energy to 46.3 kJ·mol⁻¹, approximately 31% of that required by conventional heating [64].
Industrial Reactor Design and Scale-Up Considerations
Microwave-Heated Cyclic Simulated Moving Bed Reactor
Experimental Protocol: A novel microwave heating-assisted cyclic simulated moving bed reactor (MWH-CSMBR) has been proposed for industrial-scale syngas production. The system incorporates multiple reaction zones that cyclically undergo reduction and oxidation steps while exposed to microwave radiation. Computational modeling integrates bed hydrodynamics, mass transfer, reaction kinetics, and temperature distribution with Maxwell's equations to simulate electromagnetic wave distribution. The reactor design was validated experimentally and optimized for a plant processing 50 tonnes/day of methane, achieving nearly complete (≥0.98) methane conversion with only four reactors and ensuring uniform electromagnetic distribution with >97% microwave absorption by the loaded oxygen carrier beds [65].
Key technical challenges in scaling up microwave-assisted processes include:
- Limited capacity of individual MW generators (max ~65-100 kW per magnetron)
- Ensuring uniform electromagnetic wave distribution throughout large-volume reactors
- Accurate temperature measurement in electromagnetic fields
- Maximizing microwave absorption by bed inventories
- Preventing sparking, arc formation, and microwave leakage [65]
The MWH-CSMBR design addresses the magnetron limitation by implementing a cyclic system that requires fewer microwave generators while maintaining high efficiency and conversion rates [65].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents and Materials for Microwave Applications
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| Gd-doped Ceria (CeO₂) | Redox material for thermochemical H₂ production | Creates oxygen vacancies under microwave irradiation [67] |
| Fe₃O₄ (Magnetite) | Oxygen carrier in chemical looping reforming | Magnetic, strong MW absorption, enables selective heating [65] |
| NiCo-MOF-74 | High-performance CO₂ adsorbent | Bimetallic MOF with high surface area and CO₂ uptake [64] |
| Bimetallic Catalysts | Dry reforming catalysis | Enhance reaction rates and selectivity in microwave fields [66] |
| Ionic Liquids | Green solvents for microwave reactions | High polarity, excellent MW absorption, low vapor pressure [63] |
| SiC (Silicon Carbide) | Microwave absorber for non-reactive environments | Strong MW susceptor, enables heating of low-loss materials [65] |
Process Visualization and Workflows
Microwave-Assisted Dry Reforming Process
Thermochemical Hydrogen Production Cycle
Microwave-Material Interaction Mechanisms
Precision microwave heating represents a paradigm shift in sustainable chemical processing, offering substantial advantages for clean hydrogen production and CO₂ conversion. The technology demonstrates compelling benefits through reduced energy consumption, faster reaction kinetics, lower operating temperatures, and enhanced product selectivity compared to conventional thermal processes. The experimental protocols and performance data summarized in this whitepaper provide a foundation for researchers and industrial practitioners to advance these applications toward commercial implementation.
Future research should prioritize several key areas: developing advanced materials optimized specifically for microwave interactions, designing scalable reactor systems that maintain efficiency at industrial capacities, and establishing comprehensive life-cycle assessments to quantify the full environmental benefits of microwave-assisted processes. As microwave technology continues to evolve, its integration with other sustainable energy sources and process intensification strategies will further solidify its role as a cornerstone of green chemistry and sustainable industrial manufacturing.
Optimizing Reaction Parameters and Overcoming Common Challenges
Microwave-assisted synthesis (MAS) has emerged as a cornerstone of sustainable chemistry, offering significant advantages over conventional heating methods through reduced energy consumption, shorter reaction times, and minimized waste generation [33]. This green chemistry approach aligns with multiple United Nations Sustainable Development Goals, particularly those related to affordable and clean energy, industry innovation, and responsible consumption and production [23]. The fundamental principle underlying MAS is dielectric heating, where microwave irradiation in the 0.3-300 GHz frequency range (typically 2.45 GHz for laboratory applications) causes rapid rotation of polar molecules, leading to efficient internal heat generation [23] [33]. This direct energy transfer mechanism enables precise control over reaction parameters—temperature, time, and power—which collectively determine the success and sustainability of chemical synthesis. For researchers in drug development and materials science, mastering these parameters is essential for developing efficient, scalable, and environmentally responsible synthetic protocols that can replace traditional energy-intensive methods.
The transition to microwave-assisted methods represents a paradigm shift in synthetic chemistry, moving toward processes with dramatically reduced environmental footprints. By providing rapid, uniform heating that often leads to higher yields, improved selectivity, and cleaner reaction profiles, MAS addresses several key principles of green chemistry simultaneously [33]. This technical guide provides a comprehensive framework for optimizing the core parameters of microwave-assisted reactions, enabling researchers to harness the full potential of this technology for advancing sustainable chemistry goals across pharmaceutical development, nanomaterial fabrication, and other specialized chemical syntheses.
Fundamental Parameters in Microwave-Assisted Synthesis
The Interplay of Temperature, Time, and Power
In microwave-assisted synthesis, temperature, time, and power represent interconnected variables that collectively determine reaction efficiency, selectivity, and sustainability. Temperature serves as the primary control parameter in most modern microwave reactors, with the system automatically adjusting power output to maintain the user-defined setpoint [29]. This approach ensures reproducible results by directly controlling the critical thermodynamic variable affecting reaction kinetics. Power determines the rate at which thermal energy is delivered to the reaction mixture, with higher power levels enabling more rapid heating but potentially causing decomposition of sensitive compounds if not properly controlled [29]. The time parameter in MAS is typically dramatically reduced compared to conventional methods, with reactions that traditionally require hours or days often completing within minutes or even seconds under optimized microwave conditions [33].
The relationship between these parameters is complex and reaction-specific. Higher power settings enable faster heating rates and shorter times to reach target temperatures, but may require careful optimization to prevent overshooting or thermal degradation. For temperature-sensitive reactions, moderate power with extended time may yield superior results. The dielectric properties of the reaction mixture significantly influence how efficiently microwave energy is converted to thermal energy, making solvent selection and reagent properties important considerations in parameter optimization [29]. Modern microwave reactors with real-time temperature and pressure monitoring capabilities allow researchers to precisely track the relationship between these parameters throughout the reaction process, enabling data-driven optimization.
Reaction Vessel Configuration and Solvent Selection
The configuration of the reaction vessel represents another critical parameter in microwave-assisted synthesis. Pressurized (closed vessel) reactions enable heating of solvents far beyond their atmospheric boiling points (e.g., dichloromethane to 180°C despite its normal boiling point of 40°C), which dramatically accelerates reaction rates—up to 1000-fold in some cases [29]. These systems are ideal for small-scale reactions (typically 7-10 mL maximum volume) and provide an inherent inert atmosphere for air- and moisture-sensitive reagents. In contrast, atmospheric (open vessel) reactions allow for larger reaction scales and compatibility with standard laboratory glassware like round-bottom flasks with reflux condensers [29]. While offering more modest rate enhancements (approximately 10-fold), open vessel systems enable conventional techniques such as continuous addition, distillation, or use of Dean-Stark traps.
Solvent selection critically influences parameter optimization through dielectric properties that determine microwave absorption efficiency. High microwave-absorbing solvents like water, DMF, and NMP enable rapid heating, while low-absorbing solvents like hexane and toluene heat inefficiently unless polar reagents are present [29]. Recent advances in green chemistry have expanded the use of solvent-free microwave reactions, where reagents are adsorbed onto mineral supports or used in neat conditions, eliminating environmental concerns associated with volatile organic solvents [29] [33]. The following table summarizes the microwave-absorption characteristics of common solvents:
Table: Microwave-Absorption Characteristics of Common Laboratory Solvents
| Absorption Category | Solvent Examples | Heating Efficiency | Typical Applications |
|---|---|---|---|
| High | Water, DMF, NMP, Ethylene Glycol | Excellent | Rapid heating; most synthetic applications |
| Medium | Acetonitrile, DMSO, Alcohols | Good | Controlled heating; temperature-sensitive reactions |
| Low | Hexane, Toluene, Diethyl Ether, Chloroform | Poor | Heat sink applications; reactions with polar reagents |
Systematic Parameter Optimization Framework
Step-by-Step Optimization Methodology
Developing optimized microwave reaction parameters requires a systematic approach that balances reaction efficiency with sustainability considerations. The following workflow provides a logical progression for parameter optimization:
Diagram 1: A systematic workflow for optimizing microwave reaction parameters through iterative refinement.
Initial Parameter Selection: Begin optimization by setting initial parameters based on conventional reaction conditions. For pressurized reactions, start with a temperature approximately 10°C above the conventional reaction temperature, a time of 5-10 minutes, and a conservative power level of 50 W [29]. For atmospheric reactions, heating times can be estimated using conversion guidelines: conventional 4-hour reactions typically require 10 minutes under microwave conditions, 8-18 hour reactions need approximately 30 minutes, and reactions exceeding 18 hours may require up to 1 hour [29]. Power settings for open-vessel reactions vary significantly with application: 25-50 W for solvent-free reactions, 100 W for sub-boiling temperatures, and 250-300 W for reflux conditions [29].
Iterative Refinement Process: After initial testing, systematically adjust each parameter based on reaction outcome. If no product formation occurs, increase temperature in 10-20°C increments or extend reaction time by 50-100% [29]. For reactions showing decomposition or byproduct formation, reduce temperature or power settings. The power parameter is particularly important to optimize—while high power (250-300 W) enables rapid heating, some reactions benefit from moderate power (50-150 W) applied for longer durations to prevent thermal degradation [29]. Modern microwave reactors with simultaneous cooling capabilities can maintain high power levels while preventing overheating, nearly doubling yields in some lower-yielding reactions [29].
Advanced Optimization Strategies
Beyond basic parameter adjustment, several advanced strategies can enhance optimization outcomes. Temperature ramping protocols gradually increase temperature throughout the reaction, which can improve selectivity in multi-step transformations. Power cycling techniques alternate between high and low power settings to maintain temperature control in highly exothermic reactions. Gradient methods simultaneously test multiple parameter combinations in a single run, dramatically accelerating optimization workflows [29].
For sustainable chemistry applications, optimization should incorporate green metrics alongside reaction efficiency. The GreenPrep MW Score represents one such approach, evaluating chemical parameters, technology variables, and workflow automation to quantify environmental impact [68]. Lifecycle assessment considerations should influence parameter selection, favoring conditions that minimize total energy consumption (time-temperature integral), reduce solvent volume, and enable catalyst recycling [23].
Parameter Tables for Specific Applications
Nanomaterial Synthesis Parameters
Microwave-assisted synthesis has proven particularly valuable for nanomaterial fabrication, where precise parameter control enables tailored morphologies and properties. The following table summarizes optimized parameters for key nanomaterials with applications in sustainable technologies:
Table: Optimized Microwave Parameters for Sustainable Nanomaterial Synthesis
| Nanomaterial | Temperature (°C) | Time | Power (W) | Key Findings | Sustainability Benefits |
|---|---|---|---|---|---|
| Reduced Graphene Oxide (rGO) [69] | 120-140 | 5 min | 300 | 94.56% reduction efficiency; Specific surface area: 845.6 m²/g | Rapid, energy-efficient alternative to conventional methods |
| Metal Nanoparticles [23] | Varies by metal | 1-10 min | 150-600 | Improved size/shape control vs. conventional heating | Reduced hazardous waste generation |
| Carbon Quantum Dots (CQDs) [23] | 150-200 | 10-30 min | 200-500 | Tunable fluorescence properties | Compatibility with green precursors |
| Hybrid Nanocomposites [23] | 100-180 | 5-20 min | 200-400 | Enhanced functional properties | Reduced energy consumption & reaction times |
Organic Synthesis and Pharmaceutical Applications
Microwave-assisted organic synthesis (MAOS) has revolutionized pharmaceutical development by accelerating reaction optimization and enabling sustainable manufacturing approaches. The following table outlines parameter guidelines for key reaction types:
Table: Microwave Parameters for Sustainable Organic Synthesis
| Reaction Type | Typical Temperature Range (°C) | Typical Time Range | Solvent System | Green Chemistry Advantages |
|---|---|---|---|---|
| Cyclization Reactions(e.g., Friedländer synthesis) [33] | 120-180 | 5-10 min | Ethanol, Water, or Solvent-free | Yields >85%; Reduced solvent waste |
| Heterocyclic Synthesis(e.g., coumarins, pyrazolopyrimidines) [33] | 100-160 | 5-15 min | Water, PEG, or Solvent-free | Atom economy; Benign solvents |
| Peptide Coupling [33] | 50-90 | 10-20 min | DMF, DCM, or Solvent-free | Reduced racemization; Higher yields |
| Cross-Coupling Reactions(e.g., Suzuki, Heck) [33] | 120-200 | 5-15 min | Water, DMF, or Toluene/Water | Reduced catalyst loading |
Experimental Protocols for Key Sustainable Chemistry Applications
Protocol: Microwave-Assisted Hydrothermal Synthesis of Reduced Graphene Oxide for Energy Storage
This optimized protocol demonstrates the sustainable synthesis of high-quality reduced graphene oxide (rGO) for electrochemical energy storage applications, achieving superior results in just 5 minutes compared to conventional methods requiring several hours [69].
Materials and Reagents:
- Graphene oxide aqueous dispersion (2 mg/mL)
- Deionized water
- Microwave reactor with temperature and pressure control
- Teflon-lined microwave reaction vessels
- Vacuum filtration apparatus
Experimental Procedure:
- Reaction Setup: Transfer 100 mL of graphene oxide dispersion (2 mg/mL) to a 200 mL Teflon-lined microwave reaction vessel. Seal the vessel according to manufacturer specifications.
- Parameter Configuration: Program the microwave reactor with the following optimized parameters [69]:
- Temperature: 140°C
- Microwave Power: 300 W
- Reaction Time: 5 minutes
- Stirring: Continuous (medium speed)
- Reaction Execution: Initiate the microwave program. Monitor temperature and pressure throughout the process. The system will typically reach the target temperature within 1-2 minutes.
- Product Isolation: After completion and cooling, open the reaction vessel and collect the resulting black precipitate. Filter through a 0.22 μm membrane filter and wash with deionized water (3 × 50 mL).
- Drying: Dry the product at 60°C under vacuum for 12 hours to obtain pristine rGO.
Characterization and Validation: Validate successful synthesis through the following characterization techniques [69]:
- UV-Vis Spectroscopy: Should show a red shift in absorption to approximately 268 nm
- FTIR Analysis: Demonstrates effective removal of oxygen functionalities
- BET Surface Area Analysis: Should reveal a high specific surface area (~845.6 m²/g) with hierarchical mesoporosity
- Electrical Conductivity: Optimal results show conductivity of ~13,486 S/m
- Thermal Stability: TGA should show 80% residual mass up to 480°C
Protocol: Scalable Preparation of Microwave-Assisted Reduced Graphene Oxide via Spatial Configuration Engineering
This advanced protocol addresses the challenge of non-uniform heating in scaled-up microwave synthesis, enabling batch production of high-quality rGO with minimal quality variability [70].
Materials and Specialized Equipment:
- Graphene oxide precursor
- Custom microwave reactor with spatial configuration engineering capabilities
- Corona-discharge-free reaction chamber
- Specially designed sample holders for fully exposed irradiation areas
Experimental Procedure:
- Spatial Configuration: Arrange the graphene oxide precursor in the reaction chamber using spatial configuration engineering to ensure uniform microwave exposure. This critical step minimizes hot spots and ensures consistent product quality.
- Parameter Optimization: Employ the following parameters, optimized through numerical simulation [70]:
- Microwave Power: Adjust based on batch size (typically 500-1000 W for 70 g batches)
- Temperature: Ramp to 150°C over 2 minutes
- Reaction Time: 8-10 minutes total
- Atmosphere: Inert gas (N₂ or Ar)
- Process Monitoring: Monitor for corona discharge phenomena, which indicate non-uniform field distribution and require parameter adjustment.
- Product Collection: After reaction completion, allow the system to cool under inert atmosphere before collecting the product.
Quality Assessment: High-quality m-rGO should exhibit [70]:
- ID/IG ratio ≤ 0.12 (Raman spectroscopy)
- Electrical conductivity ≥ 13,486 S/m
- Batch-to-batch consistency with yields of approximately 70 g per batch
- Excellent performance in composite applications (e.g., EMI shielding effectiveness of 40 dB at 90 μm thickness)
The Scientist's Toolkit: Essential Research Reagent Solutions
Table: Essential Reagents and Equipment for Microwave-Assisted Sustainable Synthesis
| Category | Item | Specifications | Sustainable Chemistry Function |
|---|---|---|---|
| Green Solvents [29] [33] | Water | Deionized, degassed | High microwave absorption; Non-toxic alternative to organic solvents |
| Ionic Liquids | e.g., [BMIM][BF4], [EMIM][OAc] | Low volatility; Tunable polarity; Recyclable catalysts/solvents | |
| Ethanol | Biobased, anhydrous | Renewable solvent for extraction and synthesis | |
| Eco-Friendly Precursors [23] | Plant Extracts | Standardized compositions | Natural reducing/capping agents for nanoparticle synthesis |
| Biomolecules | Amino acids, carbohydrates | Sustainable precursors for carbon quantum dots | |
| Catalysts [23] [33] | Heterogeneous Catalysts | Supported metals, zeolites | Recyclable alternatives to homogeneous catalysts |
| Biocatalysts | Immobilized enzymes | Sustainable catalysis under mild conditions | |
| Specialized Equipment [29] | Microwave Reactors | Temperature/pressure control | Enables precise parameter optimization |
| Reaction Vessels | Teflon-lined, sealed | Allows superheating of solvents for accelerated kinetics |
Scaling Up and Industrial Applications
Overcoming Scale-Up Challenges
Translating optimized laboratory parameters to industrial production presents significant challenges in microwave-assisted synthesis. The primary obstacle is non-uniform heating in larger reaction volumes, which can lead to inconsistent product quality and variable yields [70]. Recent advances in spatial configuration engineering address this limitation by optimizing the arrangement of reactants within the microwave field to ensure uniform energy distribution [70]. Industrial microwave reactors now incorporate numerical simulation tools to model electromagnetic field distribution and predict heating patterns before physical scale-up attempts.
Continuous flow microwave reactors represent another solution to scale-up challenges, enabling sustained production while maintaining the benefits of microwave activation [33]. These systems circumvent the volume limitations of batch microwave reactors by passing reaction mixtures through a focused microwave zone, combining the advantages of microwave heating with continuous processing benefits including improved heat transfer, better temperature control, and easier product isolation [33]. Market analysis indicates growing adoption of these technologies, with the microwave chemical reactor market projected to reach USD 880 million by 2032, driven largely by pharmaceutical and advanced materials applications [71].
Sustainability Assessment and Green Metrics
Quantifying the environmental benefits of microwave-assisted synthesis requires comprehensive sustainability assessment tools. The GreenPrep MW Score represents a specialized metric for evaluating microwave-assisted sample preparation procedures, considering chemical parameters, technology variables, and workflow automation [68]. This and similar green metrics enable researchers to objectively compare the environmental performance of microwave-assisted protocols against conventional methods, considering factors such as energy consumption, waste generation, and reagent toxicity.
Lifecycle assessment studies consistently demonstrate that microwave-assisted synthesis offers significant environmental advantages over conventional methods, including reduced energy consumption (shorter reaction times), minimized waste generation (higher selectivity), and decreased solvent usage (enabled by solvent-free or aqueous conditions) [23]. These benefits align microwave chemistry with circular economy principles by promoting resource efficiency and reducing environmental impact across the chemical lifecycle. As microwave technology continues to evolve toward industrial-scale applications, its integration with other green chemistry approaches (biocatalysis, renewable feedstocks, process intensification) will further enhance sustainability profiles across pharmaceutical and materials manufacturing sectors [23] [33].
The strategic optimization of temperature, time, and power parameters in microwave-assisted synthesis represents a critical pathway toward more sustainable chemical manufacturing. By systematically applying the optimization frameworks, reference parameters, and experimental protocols outlined in this guide, researchers can harness the full potential of microwave technology to accelerate reaction kinetics, improve product selectivity, and reduce environmental impact. The ongoing development of specialized microwave reactors with enhanced control capabilities, coupled with advanced sustainability assessment tools, continues to expand the applications of microwave-assisted synthesis across pharmaceutical development, nanomaterial fabrication, and industrial chemical production. As microwave technology evolves to address scale-up challenges through spatial engineering and continuous flow systems, its role in enabling sustainable chemistry aligned with circular economy principles will continue to grow, ultimately transforming chemical manufacturing into a more efficient and environmentally responsible enterprise.
The transition from small laboratory-scale experiments to large-scale industrial manufacturing, known as scale-up, is a critical and complex multi-step journey in chemical process development. The primary objective is to facilitate the efficient and safe production of desired products at a volume high enough to meet market demand while maintaining process sustainability, safety, and economic viability [72]. Within the evolving paradigm of sustainable chemistry, innovative energy solutions like microwave technology are emerging as transformative tools. This guide details the core strategies, experimental protocols, and safety considerations essential for a successful scale-up, with specific emphasis on integrating microwave techniques to enhance efficiency and support green transformation goals in industrial chemistry [3].
Foundational Principles of Scale-Up
A successful scale-up process is characterized by a seamless transition that is not only about meeting production targets but also about ensuring the process is sustainable, safe, and economically viable [72]. The World Health Organization's ExpandNet framework, while designed for health interventions, offers a valuable structure for conceptualizing scale-up more broadly, emphasizing key elements including the intervention attributes, the user organization, the environment, and the resource team [73]. A holistic view of success includes:
- Scaling Inputs: Securing sustained partner buy-in and resources.
- Scaling Outputs: Achieving sustained, quality implementation.
- Scaling Outcomes: Attaining increased and equitable reach, alongside improved organization and system capacity.
- Scaling Context: Navigating partner mental models and ensuring context-specific adaptation [73].
Core Scale-Up Strategies and Methodologies
Key Strategic Pillars from Bench to Plant
Implementing a robust scale-up strategy requires meticulous planning and execution across several core technical areas.
Table 1: Essential Pillars for Successful Scale-Up
| Strategic Pillar | Core Objective | Key Activities |
|---|---|---|
| Thermodynamics & Kinetics Characterization [72] | Understand optimal and safe reaction conditions; predict behavior at larger scales. | Determine heat flow (ΔH), reaction rates, and optimize parameters (T, p, pH). Study mixing effects. |
| Process Reproducibility [72] | Ensure process consistency and build robust statistical data foundation. | Conduct repeated experiments using automated parallel reactors to minimize human error. |
| Raw Material & Product Handling [72] | Adapt process for cost-effective, lower-purity industrial feedstocks. | Test different raw material grades; study decomposition and storage stability. |
| Systematic Safety Assurance [72] | Identify and mitigate risks of thermal runaway and hazardous events. | Perform calorimetry in small-scale reactors; understand heat and mass transfer limitations. |
| Pilot Testing [72] | Fine-tune process parameters and inform final reactor design. | Operate at an intermediate scale to bridge the gap between lab and full production. |
An Experimental Protocol for Process Characterization
A standardized protocol is critical for acquiring the deep knowledge needed for scale-up.
1. Objective: To characterize the thermodynamics, kinetics, and safety parameters of a target chemical reaction suitable for scale-up, with emphasis on microwave-specific conditions.
2. Materials and Equipment:
- Automated benchtop reactor system (e.g., H.E.L Group parallel reactors) with precise control over temperature, pressure, and agitation [72].
- Microwave reactor system tunable to frequencies ~900 MHz - 2.45 GHz, capable of precise power delivery [3].
- In-situ analytical probes (e.g., FTIR, Raman) for real-time reaction monitoring.
- Calorimetry equipment for measuring heat of reaction.
3. Methodology:
- Parameter Scoping: Perform initial reactions in the benchtop reactor across a defined range of temperatures, pressures, and stirring rates to establish a performance baseline (yield, selectivity) [72].
- Microwave Optimization: Transfer the reaction to the microwave system. Methodically vary microwave power (W) and frequency (MHz) while holding other parameters constant to identify conditions that maximize energy efficiency and yield [3].
- Mixing Analysis: Deliberately simulate suboptimal mixing conditions in the benchtop reactor (e.g., reduced agitation speed) to assess impact on yield and by-product formation [72].
- Calorimetry & Safety Screening: In the benchtop reactor, use calorimetry to determine the reaction enthalpy (ΔH) and adiabatic temperature rise. Identify temperature thresholds for decomposition and secondary reactions [72].
- Raw Material Variability: Repeat the optimized reaction protocol using lower-purity, industrial-grade reactants to quantify impact on yield and purity [72].
4. Data Analysis:
- Calculate key metrics: Conversion (%), Yield (%), Selectivity (%), Space-Time-Yield (kg m⁻³ h⁻¹).
- Compare energy efficiency (kJ kg⁻¹) between conventional and microwave heating.
- Model the reaction kinetics to determine rate constants and orders.
The following workflow diagram visualizes this iterative experimental protocol:
The Sustainable Chemistry Context: Microwave Energy
Precision Microwave Technology
Conventional heating methods are often wasteful, dispersing thermal energy throughout the entire reactor vessel. Microwave technology offers a paradigm shift by focusing energy directly on the reaction sites. A recent breakthrough technique uses microwaves tuned to specific frequencies (e.g., ~900 MHz) to excite single atomic "antenna" sites within a catalyst material, such as zeolite [3]. This achieves energy efficiencies approximately 4.5 times higher than conventional heating [3].
Applications and Workflow for Microwave Scaling
This precision heating enables demanding reactions like CO₂ conversion into fuel and methane conversion at lower overall temperatures, key pathways for carbon recycling in a green chemical industry [3]. The method involves functionalizing a zeolite catalyst with metal ions (e.g., Indium) that act as microwave antennas.
Table 2: Key Reagent Solutions for Microwave Eco-Catalysis
| Reagent/Material | Function in the Process | Sustainable Benefit |
|---|---|---|
| Zeolite Catalyst (e.g., with Indium ions) | Porous support structure; metal ions act as localized microwave antennas. | Enables lower energy input for reactions; can be designed for reusability. |
| Tuned Microwave Reactor (~900 MHz) | Provides focused energy to excite catalyst antennas, not the entire reactor. | Dramatically increases energy efficiency (≈4.5x) compared to conventional heating [3]. |
| CO₂ Feedstock | Reactant for conversion into useful fuel products. | Facilitates carbon capture and recycling, contributing to a circular economy [3]. |
The following diagram illustrates the core mechanism of this advanced microwave technique:
Quantitative Data for Scale-Up Projections
Transitioning a process to industrial scale requires careful projection of key performance indicators. The tables below summarize critical quantitative data for planning.
Table 3: Projected Market and Performance Metrics for Industrial Scaling (2025-2033)
| Metric Category | Projected Value / Range | Context and Notes |
|---|---|---|
| Global Industrial Equipment Market Growth [74] | CAGR of 4.6% - 7% | Compound Annual Growth Rate (CAGR) from 2025-2033, indicating a stable expansion environment. |
| Energy Efficiency via Microwaves [3] | ≈ 4.5x improvement | Achieved energy efficiency compared to conventional heating methods in lab-scale research. |
| Cost Reduction from Scaling & Automation [75] | Up to 25% reduction | Reported in automotive manufacturing case studies through robotics and data analytics integration. |
Table 4: Economic and Scaling Formulae for Decision-Making
| Formula Name | Equation | Application in Scale-Up |
|---|---|---|
| Average Cost per Unit [75] | Average Cost = (Fixed Costs + Variable Costs) / Number of Units Produced |
Illustrates the principle of economies of scale, where increasing production volume reduces unit cost. |
| Competitive Advantage [75] | Competitive Advantage = Cost Savings / Investment in Scaling |
A simple model to assess the return on investment from scaling and cost-reduction initiatives. |
Scaling chemical processes from the lab bench to industrial plant is a complex but manageable journey that demands a rigorous, data-driven approach. By leveraging foundational strategies—deep process characterization, rigorous safety testing, and systematic piloting—and embracing innovative technologies like precision microwave energy, researchers and drug development professionals can navigate this path successfully. The integration of microwave techniques not only enhances energy efficiency but also aligns the scale-up process with the overarching principles of sustainable chemistry. Future success will depend on continued optimization of catalyst design, reactor engineering, and the integration of these advanced processes with renewable energy sources, paving the way for a more efficient and environmentally responsible chemical industry [3].
Microwave-assisted organic synthesis (MAOS) represents a cornerstone technology for sustainable chemistry, offering dramatic reductions in reaction times, improved yields, and diminished environmental impact. However, suboptimal yields frequently stem from inadequate energy coupling between microwave radiation and the reaction mixture, a phenomenon governed by the polarity of solvents, reagents, and transition states. This whitepaper provides an in-depth technical guide for researchers troubleshooting yield issues in MAOS. By synthesizing recent advances in mechanistic understanding and computational predictions, we establish a structured framework for diagnosing and rectifying poor energy coupling. The guidance is framed within the broader thesis that mastering these parameters is essential for developing truly sustainable synthetic protocols in pharmaceutical and fine chemical industries.
The integration of microwave energy into organic synthesis aligns with the core principles of Green Chemistry by promoting energy efficiency, reducing reaction times, and minimizing solvent waste [76] [18]. Conventional conductive heating is often slow and inefficient, as heat must penetrate vessel walls before reaching the reaction mixture. In contrast, microwave irradiation delivers energy volumetrically through direct interaction with molecules, enabling instantaneous superheating [37]. This can lead to remarkable rate enhancements—up to 1000-fold in some cases—and can alter product selectivity by providing access to high-energy transition states typically inaccessible via conventional heating [37].
Despite these advantages, the practical implementation of MAOS often encounters the challenge of poor energy coupling, where the microwave radiation fails to transfer energy efficiently to the reaction components. This results in insufficient heating, slow reaction rates, low yields, and irreproducible results. The efficiency of this energy transfer is predominantly dictated by the dielectric properties of the reaction medium, specifically the polarity of solvents, substrates, and intermediates [77] [37]. This guide details a systematic approach to diagnosing and resolving these issues, positioning microwave chemistry as a reliable pillar for sustainable research and development.
Theoretical Foundations of Microwave Heating
Understanding the mechanism of microwave heating is prerequisite for effective troubleshooting. Microwave energy, operating at 2450 MHz, transfers heat via two primary mechanisms:
- Dipole Rotation: Polar molecules attempt to align with the rapidly oscillating electric field. This molecular rotation causes in-phase friction and collision, resulting in instantaneous and efficient heating [37]. The coupling efficiency depends on the molecule's polarity and its ability to reorient within the field.
- Ionic Conduction: Ions or charged species in solution migrate under the influence of the electric field, colliding with neighboring molecules and converting kinetic energy into heat. The efficiency of this process increases with temperature [37].
A critical concept is the penetration depth, which is the depth at which the power of the microwave radiation is reduced to half its original value. This dictates the scale-up potential of a microwave reaction. It is crucial to recognize that the energy in microwave photons (0.037 kcal/mol) is far too low to break typical covalent bonds (80-120 kcal/mol), confirming that microwave effects are primarily thermal/kinetic in nature, not molecular [37].
The Arrhenius equation ((k = Ae^{-Ea/RT})) provides a kinetic framework for understanding rate enhancements. While microwaves do not alter the fundamental activation energy ((Ea)) or the pre-exponential factor ((A)), they dramatically increase the instantaneous temperature ((T)) at the molecular level. This localized superheating is responsible for the observed accelerations [37]. For a reaction with an (E_a) of 50 kcal/mol, a 100-fold rate increase requires an instantaneous temperature increase of only about 35°C above the measured bulk temperature [37].
Diagnosing Poor Energy Coupling: A Polarity-Centric Framework
Poor yields in MAOS often originate from a mismatch between the energy provided by the microwave field and the system's ability to absorb it. The following table outlines common symptoms, their underlying causes, and initial diagnostic steps.
Table 1: Symptoms and Diagnosis of Poor Energy Coupling
| Symptom | Potential Cause | Diagnostic Check |
|---|---|---|
| Low reaction temperature despite high microwave power | Low polarity solvent or reagents incapable of effective dipole rotation | Determine solvent polarity (dielectric constant, loss tangent); consider adding a microwave susceptor |
| Slow reaction rate despite apparent bulk heating | Inefficient energy transfer to key reactants or failure to couple with polar transition state | Calculate the polarity of reactants and proposed transition states computationally [77] |
| Reaction performs better in conventional heating | "Wall effects" or thermal degradation; microwave power may be too high, leading to decomposition | Use simultaneous microwave cooling to allow higher power delivery without overheating [37] |
| Inconsistent results between batches | Non-uniform heating or formation of "hot spots" | Ensure efficient stirring; consider adding a material like silicon carbide to improve heating homogeneity [77] |
Computational studies have quantified the ideal parameters for successful MAOS. The activation energy ((E_a)) of the reaction and the polarity (μ, dipole moment) of the species involved are decisive factors [77].
Table 2: Quantitative Guidelines for Microwave Improvement Based on Computational Studies [77]
| Reaction Type | Activation Energy (Ea) | Expected Microwave Effect | Required Conditions |
|---|---|---|---|
| Type A | < 20 kcal mol⁻¹ | Not significantly improved | Reaction is already facile under conventional heating. |
| Type B | 20 - 30 kcal mol⁻¹ | Significantly improved | Can be performed without harsh conditions. |
| Type C | > 30 kcal mol⁻¹ | Improved only with susceptors | Requires polar additives (ionic liquids) or solvents for "flash heating." |
Furthermore, for significant microwave improvement, the polarity of the participating species should ideally fall between 7 and 20 Debye [77]. Reactions involving a polar mechanism, where the polarity increases from the reactants to the transition state (a "late transition state" according to Hammond's postulate), are particularly well-suited for acceleration [76] [77].
Optimization Strategies and Experimental Protocols
Solvent Selection and Engineering
The solvent is a primary coupler of microwave energy. Its selection is the most critical parameter for success.
Table 3: Solvent Selection and Modification Strategies
| Strategy | Protocol | Mechanism & Rationale |
|---|---|---|
| High Polarity Solvents | Use solvents like DMF, NMP, water, or DMSO. | High dielectric loss enables efficient dipole rotation. |
| Solvent-Free Conditions | Neat reactants are adsorbed on solid mineral supports (e.g., alumina, silica). | Eliminates solvent waste (Green Chemistry) and maximizes energy coupling directly with reactants [76]. |
| Mixed Solvent Systems | Add a small volume of a high-polarity solvent (e.g., ionic liquid) to a low-polarity medium. | The polar additive acts as a "molecular radiator," transferring energy to the bulk solution [77]. |
| Use of Microwave Susceptors | Add materials like silicon carbide (SiC), graphite, or ionic liquids to the reaction mixture. | Susceptors absorb microwaves intensely and transfer heat convectively, enabling reactions of non-polar systems [77]. |
Protocol: Optimizing a Reaction with Poor Solvent Coupling
- Initial Setup: Run the reaction in a low-polarity solvent (e.g., toluene, dioxane) under microwave irradiation at a set temperature (e.g., 150°C). Observe the time required to reach the target temperature and the final yield.
- Strategy A - Solvent Swap: Replace the solvent with a high-polarity alternative (e.g., switch from toluene to DMF). Repeat the reaction, noting the improved heating rate and any change in yield.
- Strategy B - Additive Use: To the original low-polarity solvent, add 1-5% vol. of a microwave susceptor like an ionic liquid ([BMIM]Cl) or a few silicon carbide chips. Repeat the reaction. The susceptor will facilitate heating, leading to a faster ramp time and potentially higher yield.
Harnessing Polar Mechanisms and Transition States
Beyond the solvent, the intrinsic polarity of the reaction itself can be leveraged. A protocol for computational pre-screening is highly recommended.
Protocol: Computational Pre-Screening for Microwave Suitability
- Modeling: Use computational chemistry software (e.g., Gaussian, Spartan) to calculate the dipole moments (μ) of the reactants, proposed transition states, and products at an appropriate level of theory (e.g., B3LYP/6-31G*).
- Analysis: Plot the relative polarity along the reaction coordinate. A reaction is likely to be enhanced by microwaves if the dipole moment increases significantly in the transition state compared to the ground state [77].
- Activation Energy Calculation: Compute the activation energy ((Ea)). Reactions with (Ea) between 20-30 kcal mol⁻¹ are prime candidates for MAOS without requiring extreme conditions [77].
- Experimental Design: If the calculations are favorable, proceed with microwave experimentation using the solvent and additive strategies outlined above.
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key materials used to optimize and troubleshoot microwave-assisted reactions.
Table 4: Research Reagent Solutions for Microwave Chemistry Optimization
| Reagent/Material | Function & Mechanism | Application Example |
|---|---|---|
| Ionic Liquids (e.g., [BMIM][BF₄], [BMIM]Cl) | High polarity enables intense microwave absorption, acting as a "molecular radiator" or catalyst [77]. | Added in small quantities (1-5% vol.) to low-polarity solvents to dramatically improve heating. |
| Silicon Carbide (SiC) | Inorganic microwave susceptor; absorbs radiation and transfers heat via conduction, eliminating "wall effects" [77]. | Used as a reaction vessel insert or chips to ensure uniform, efficient heating of non-polar mixtures. |
| Deep Eutectic Solvents (DES) | Biodegradable, renewable solvents with tunable polarity; align with Green Chemistry principles [18]. | Used as a green, reactive solvent medium for microwave-assisted extraction and synthesis. |
| Water | High dielectric loss makes it an excellent, safe, and sustainable microwave absorber. | Used as a green solvent for microwave-promoted hydrolyses or polar cycloadditions. |
| Solid Mineral Supports (e.g., alumina, silica) | Provide a high-surface-area, solvent-free environment for reactions; can also act as a catalyst. | Used for "dry media" reactions where neat reactants are adsorbed onto the support [76]. |
Mastering solvent polarity and energy coupling is not merely a technical troubleshooting exercise but a fundamental requirement for advancing sustainable chemistry through microwave research. By adopting the diagnostic framework and optimization strategies outlined here—including computational pre-screening, strategic solvent selection, and the use of targeted susceptors—researchers can transform underperforming reactions into efficient, high-yielding processes.
The future of MAOS is inextricably linked to the principles of Safe and Sustainable by Design (SSbD) [78]. This involves integrating computational hazard screening and Life Cycle Assessment (LCA) early in reaction development. The convergence of microwave synthesis with predictive modeling, artificial intelligence for process optimization [18], and greener solvent systems paves the way for a new era of chemical production that is not only efficient and selective but also environmentally responsible and inherently safe. For the pharmaceutical industry and beyond, these tools are indispensable for minimizing environmental impact while maximizing synthetic efficiency.
In the pursuit of sustainable chemistry, microwave energy has emerged as a transformative technology for accelerating chemical synthesis while reducing environmental impact. Unlike conventional conductive heating, microwave irradiation delivers energy directly to molecules through dielectric heating mechanisms, enabling dramatic rate enhancements and improved product yields [29]. However, a significant challenge persists: the risk of thermal decomposition due to uncontrolled power delivery and inefficient heat dissipation. This technical guide examines the critical role of precisely controlled power levels and simultaneous cooling technology in preventing molecular decomposition, thereby advancing microwave-assisted sustainable chemistry.
The transition from fossil fuels to cleaner energy sources has positioned microwave chemistry as a key enabling technology for greener synthetic pathways [67] [79]. Microwave processes effectively convert electrical energy into chemical reactions and can be driven by renewable energy sources, contributing to carbon-neutral chemical industries [79]. Within this framework, preventing decomposition through advanced thermal management not only improves reaction efficiency but also aligns with global sustainability goals by minimizing waste and energy consumption.
Theoretical Foundations of Microwave Heating
Dielectric Heating Mechanisms
Microwave-assisted heating operates through two primary mechanisms that transform electromagnetic energy into thermal energy. The first involves the coupling of the oscillating electric field with permanent dipoles in molecules, causing them to rotate rapidly and generate heat through molecular friction. The second mechanism occurs through the interaction of the electric field with ions present in the reaction mixture, promoting their conduction and generating heat through ionic friction [80]. These dual mechanisms enable volumetric heating, where energy penetrates and heats the entire sample simultaneously rather than transferring heat from the surface inward as with conventional methods.
The efficiency of these dielectric heating mechanisms depends on the dielectric properties of the materials, characterized by the complex relative permittivity (ε̄) [81]:
where the real part (ε′) represents the dielectric constant (energy storage capacity), the imaginary part (ε″) signifies the dielectric loss (energy conversion to heat), ω is the angular frequency, and ϑ is the temperature [81]. Materials with higher loss factors (ε″) more efficiently convert microwave energy into heat, making them ideal candidates for microwave-assisted synthesis.
Thermal Runaway and Decomposition Risks
A critical challenge in microwave chemistry is thermal runaway, a self-accelerating process where localized overheating leads to increasingly rapid temperature rises that can ultimately cause decomposition of desired products [80]. This phenomenon occurs when the heat generated within the material exceeds the rate of heat dissipation to the surroundings. The non-uniform electric field distribution in multimode microwave cavities, common in laboratory systems, can create hot and cold spots that exacerbate this problem [80].
The degree of thermal runaway (ΔT) can be quantitatively described through modeling of temperature evolution within the reaction medium [80]. In microwave pyrolysis of biomass, for example, thermal runaway values ranging from 1K to 102K have been observed, highlighting the significant decomposition risks without proper control mechanisms [80]. The comprehensive evaluation coefficient (CEC), calculated as the ratio of power absorption efficiency (PAE) to the coefficient of variation (COV) of temperature distribution, provides a valuable metric for assessing a sample's usability while minimizing decomposition risks [80].
Power Control Methodologies
Strategic Power Parameter Selection
Precise control of microwave power represents the first critical strategy for preventing decomposition. The initial power setting must be carefully selected based on reaction type, vessel configuration, and sensitivity of reagents [29]. For unknown reactions or thermally sensitive compounds, starting with lower power levels (25-50W) allows assessment of the reaction's coupling efficiency with microwave energy before committing to higher power delivery [29].
Table 1: Recommended Initial Power Settings for Different Reaction Conditions
| Reaction Type | Vessel Configuration | Starting Power | Temperature Control Setpoint | Rationale |
|---|---|---|---|---|
| New/Uncertain Reactions | Closed vessel | 50 W | 10°C above conventional method | Prevents rapid pressure rise; allows assessment of coupling efficiency [29] |
| Solvent-free on mineral oxides | Open vessel | 25-50 W | 200°C (not exceeding 250°C) | Prevents scorching of solid mixtures [29] |
| Reflux conditions | Open vessel | 250-300 W | 50°C above solvent boiling point | Ensures constant power for maintained reflux [29] |
| Conventional mimic | Open vessel | 100 W | Same as conventional method | Gentle heating for temperature-sensitive reactions [29] |
The relationship between power input and temperature stability follows a critical threshold pattern. Research has identified a critical power level "where the heat generated in the penetration depth can no longer be transferred to the rest of the sample" [80]. Exceeding this threshold guarantees thermal runaway and potential decomposition. For biomass materials like wood, sugarcane bagasse, orange peel, and palm oil, this critical power varies significantly based on dielectric properties and sample geometry [80].
Dynamic Power Modulation
Advanced microwave systems employ dynamic power modulation, where the system continuously adjusts power output to maintain a set temperature throughout the reaction. As the temperature approaches the target setpoint, the power is automatically reduced to prevent overshoot and decomposition [29]. This closed-loop control system represents a significant advancement over early microwave reactors that operated at fixed power levels.
The transition from fixed to modulated power delivery enables precise maintenance of optimal reaction conditions while preventing thermal degradation. In practice, "as the temperature reaches the set value, the power is reduced so that the reaction mixture does not bypass that temperature point" [29]. This dynamic adjustment occurs throughout the reaction, with power maintained at lower levels to sustain the target temperature once achieved.
Simultaneous Cooling Technology
Principles and Mechanisms
Simultaneous cooling technology represents a paradigm shift in microwave reaction control, addressing the fundamental challenge of excessive heat accumulation. This innovative approach involves cooling the reaction vessel externally while microwave energy continues to be applied internally. The technology leverages the fact that microwave energy transfer occurs on a nanosecond timescale, far faster than conventional heating methods [29].
The simultaneous cooling mechanism enables a unique decoupling of the microwave energy input from the bulk temperature of the reaction mixture. While microwave energy continues to activate molecules through direct dielectric heating, the cooling system removes excess thermal energy that would otherwise lead to decomposition. This maintains the reaction mixture at the optimal temperature range while allowing continuous microwave energy delivery.
Benefits for Reaction Efficiency
The implementation of simultaneous cooling technology produces dramatic improvements in reaction outcomes, particularly for thermally sensitive compounds. Research has demonstrated that "simultaneous cooling of the reaction vessel during a reaction will ensure a constant, high power level for direct molecular heating" [29]. This sustained high-power delivery without thermal degradation nearly doubles percent yields in some lower-yielding reactions [29].
The table below summarizes key experimental parameters and outcomes achievable with simultaneous cooling technology:
Table 2: Reaction Performance Metrics with Simultaneous Cooling Technology
| Parameter | Conventional Microwave | With Simultaneous Cooling | Improvement |
|---|---|---|---|
| Sustained Power Delivery | Limited by temperature rise | Maintained at high levels | Up to 100% increase [29] |
| Reaction Yield | Variable, decomposition-limited | Significantly enhanced | Nearly doubled in some reactions [29] |
| Temperature Control | Oscillations around setpoint | Precise maintenance | Not quantified |
| Decomposition Products | Significant in sensitive reactions | Substantially reduced | Not quantified |
The combination of continuous high-power microwave delivery with effective heat dissipation creates an optimal environment for challenging synthetic transformations that would otherwise succumb to thermal degradation under conventional microwave or thermal conditions.
Experimental Protocols and Methodologies
Power Optimization Procedure
Establishing decomposition-free microwave reactions requires systematic optimization of power parameters. The following step-by-step protocol ensures safe and effective power calibration:
Initial Setup: Select appropriate vessel type (closed or open) based on reaction scale and solvent requirements [29]. For closed vessels, ensure proper sealing and pressure tolerance.
Baseline Power Selection: Refer to Table 1 for recommended starting power based on reaction configuration. Begin conservatively, especially for new or uncertain reactions.
Temperature Monitoring: Implement real-time temperature monitoring using infrared sensors or fiber-optic probes. Record the time taken to reach the target temperature.
Power Adjustment: If the reaction struggles to reach target temperature within 2-3 minutes, incrementally increase power by 25W intervals until appropriate heating kinetics are achieved [29].
Stability Assessment: Maintain the reaction at target temperature for the desired duration, observing power modulation patterns and temperature stability.
Validation Run: Conduct a control experiment at the optimized parameters to verify reproducibility and assess product quality and decomposition levels.
This methodology ensures that power levels remain below the critical threshold where thermal runaway initiates while providing sufficient energy for efficient reaction kinetics.
Simultaneous Cooling Integration
The integration of simultaneous cooling requires specialized microwave apparatus equipped with cooling mechanisms, typically compressed air or liquid cooling systems. The implementation protocol includes:
System Configuration: Activate the cooling mechanism in the microwave system settings, typically through the instrument software interface.
Cooling Parameter Setup: Set the cooling intensity to maintain the desired reaction temperature. Initial recommendations suggest moderate cooling power with adjustment based on observed temperature stability.
Power-Cooling Balance: Utilize the maximum microwave power setting while relying on the cooling system to maintain isothermal conditions. This represents a fundamental shift from conventional power-limited approaches.
Real-time Monitoring: Observe both temperature and power delivery profiles throughout the reaction. Optimal conditions show high, sustained microwave power with stable temperature maintenance.
Post-reaction Analysis: Compare yield and purity metrics with conventional microwave and thermal methods to quantify improvements.
This protocol leverages the finding that "simultaneous cooling of the reaction vessel during a reaction will ensure a constant, high power level for direct molecular heating" [29].
Research Reagent Solutions and Essential Materials
Table 3: Essential Research Reagents and Materials for Decomposition-Free Microwave Chemistry
| Item | Function/Significance | Application Notes |
|---|---|---|
| Gd-doped ceria (CeO₂) | Benchmark material for hydrogen production; demonstrates 60% reduction temperature with microwaves [67] | Microwave energy replaces 75% of thermal energy needed; enables rapid oxygen vacancy formation [67] |
| Polar solvents (DMF, DMSO, Water) | High microwave coupling efficiency due to substantial loss factors (ε″) [29] | Enables rapid heating; requires careful power control to prevent decomposition |
| Low-boiling point solvents (DCM, Ether) | Can be superheated in sealed vessels (DCM to 180°C) [29] | Enables novel high-temperature chemistry; requires precise pressure and temperature control |
| Mineral oxides (alumina, silica) | Supports for solvent-free reactions; enable efficient microwave coupling [29] | Particularly effective for adsorbed reagents; requires mechanical stirring for homogeneity |
| Ionic liquids | Exceptional microwave absorption; enable low-temperature reactions [29] | Can create extremely rapid heating; necessitates advanced power modulation |
| Biomass materials (wood, sugarcane, orange peel) | Feedstock for sustainable chemical processes; variable dielectric properties [80] | Requires pre-screening of dielectric properties for process optimization |
Visualization of Control Systems
Microwave Power Control Logic
Simultaneous Cooling Mechanism
The integration of controlled power levels and simultaneous cooling technology represents a sophisticated approach to preventing decomposition in microwave-assisted chemistry. These advanced control strategies enable researchers to harness the full potential of microwave energy while minimizing the risks of thermal degradation that have historically limited its application for sensitive synthetic transformations. Through the methodologies outlined in this technical guide—including systematic power optimization, dynamic power modulation, and simultaneous cooling integration—scientists can achieve unprecedented control over reaction conditions.
Within the broader context of sustainable chemistry, these decomposition prevention technologies assume even greater significance. By enabling higher yields, reducing energy consumption through more efficient processes, and minimizing waste from side reactions, controlled microwave chemistry aligns perfectly with the principles of green chemistry and sustainable manufacturing. As microwave technology continues to evolve, particularly with integration of AI for predictive control and real-time optimization, these foundational strategies for preventing decomposition will remain essential for researchers pursuing sustainable synthetic methodologies across pharmaceutical development, materials science, and energy applications.
The pursuit of sustainable chemistry necessitates the development of efficient processes that minimize waste, energy consumption, and time. Rapid reaction optimization has emerged as a critical discipline in modern chemical research and development, directly supporting these goals by dramatically accelerating the scoping and refinement of synthetic methods. This paradigm is particularly transformative within pharmaceutical and fine chemical industries, where the speed of process development directly impacts research costs, material sustainability, and ultimately, the delivery of new therapeutics [82] [83].
The integration of microwave energy into chemical synthesis represents a cornerstone of this accelerated, sustainable approach. Microwave-assisted synthesis offers a pathway to dramatically reduced reaction times—from hours to minutes—while simultaneously improving energy efficiency and product yields [69] [67]. This guide details the advanced methodologies, experimental protocols, and computational tools that enable researchers to leverage short timeframes for efficient method scoping, thereby embedding the principles of green chemistry directly into the research and development lifecycle.
Core Methodologies for Accelerated Optimization
Several high-throughput and automated strategies form the backbone of modern rapid reaction optimization.
High-Throughput Experimentation (HTE)
HTE utilizes reaction miniaturization and automation to rapidly explore vast chemical spaces. Platforms like the phactor software enable researchers to design and analyze arrays of chemical reactions in 24, 96, 384, or 1,536-well plates. This software interfaces with liquid handling robots and chemical inventories to create a closed-loop workflow for HTE-driven research, facilitating the discovery of new reactivities and the optimization of known reactions with unprecedented speed [84]. In practice, phactor has been used to discover novel reactions, such as a deaminative aryl esterification, and to optimize key steps in pharmaceutical syntheses, generating heatmaps and multiplexed pie charts for facile evaluation of results [84].
Automated Computational and Kinetic Modeling
Beyond physical experimentation, computational frameworks can autonomously identify reaction models and kinetic parameters. One novel methodology uses transient flow data from automated flow chemistry platforms. A library of candidate reaction models is generated, and Mixed Integer Linear Programming (MILP) is used to simultaneously identify the correct reaction model and optimize its kinetic parameters. This approach achieves comprehensive process understanding with minimal user input and is applicable to data from any set of experiments [82]. For more accessible kinetic analysis, Variable Time Normalization Analysis (VTNA) is a valuable technique implemented in spreadsheets to determine complex reaction orders without extensive mathematical derivation, providing critical insight into reaction mechanisms and efficiency [85].
Machine Learning-Driven Optimization
Machine learning algorithms, particularly Bayesian Optimization (BO), have revolutionized self-optimizing reaction platforms. BO uses a probabilistic model, an acquisition function, and an optimization algorithm to iteratively suggest high-performing experiments. Multi-Task Bayesian Optimization (MTBO) enhances this by incorporating data from previous, related optimization campaigns (auxiliary tasks). A multitask Gaussian process model learns correlations between different chemical transformations, allowing for the acceleration of new reaction optimization by leveraging historical data, which leads to significant savings in precious materials and time [83].
Table 1: Core Methodologies for Rapid Reaction Optimization
| Methodology | Key Principle | Primary Application | Key Advantage |
|---|---|---|---|
| High-Throughput Experimentation (HTE) [84] | Miniaturization & parallelization of reactions | Reaction discovery & condition scoping | Explores vast parameter space (solvent, catalyst, etc.) rapidly |
| Automated Kinetic Modeling [82] | Autonomous model identification & parameter fitting | Fundamental process understanding | Minimizes user input; provides mechanistic insight |
| Multi-Task Bayesian Optimization [83] | Leverages historical data for new optimizations | Accelerated optimization of related reactions | Reduces experimental budget and material use |
Microwave-Assisted Synthesis for Sustainable Chemistry
Microwave energy provides a direct and efficient means of heating reactions, leading to profound enhancements in sustainability profile.
Principles and Sustainable Advantages
Microwave irradiation directly energizes molecules, enabling rapid and uniform heating throughout the reaction mixture. This often results in dramatically reduced reaction times and lower energy consumption compared to conventional conductive heating. The efficiency of microwave heating can lower the thermal energy required for a reaction by up to 75%, as demonstrated in the microwave-assisted thermochemical reduction of metal oxides for clean hydrogen production [67]. Furthermore, the ability to perform reactions at lower temperatures while maintaining high efficiency helps preserve structural integrity of sensitive materials and catalysts [69].
Protocol: Microwave-Assisted Hydrothermal Synthesis of Reduced Graphene Oxide (rGO)
Reduced graphene oxide (rGO) is a promising material for electrochemical energy storage. Traditional synthesis methods are often environmentally harmful and time-consuming. The following microwave-assisted protocol outlines a rapid, green alternative [69].
- Objective: To synthesize high-quality rGO in a rapid, energy-efficient, and scalable manner.
- Materials: Graphite oxide precursor, deionized water.
- Equipment: Microwave reactor with temperature and power control (e.g., CEM Mars or Biotage Initiator+), Teflon-lined autoclave vessels, vacuum filtration setup.
- Procedure:
- Preparation: Disperse graphite oxide in deionized water to form a homogeneous aqueous suspension via sonication.
- Reaction: Transfer the suspension to a Teflon-lined autoclave suitable for the microwave reactor. Seal the vessel.
- Microwave Irradiation: Place the vessel in the microwave reactor and initiate the following program:
- Power: 300 W
- Temperature: 140 °C
- Time: 5 minutes
- Use magnetic stirring during the irradiation process.
- Work-up: After the reaction is complete and the vessel has cooled, collect the resulting solid product via vacuum filtration.
- Purification: Wash the solid thoroughly with deionized water and ethanol to remove any impurities.
- Drying: Dry the purified rGO in an oven at 60-80 °C overnight.
- Analysis & Notes: The resulting rGO exhibits a high reduction efficiency (94.56 wt%), a large specific surface area (845.6 m²/g), and improved electrical conductivity, making it ideal for supercapacitors and batteries. This method is a testament to how microwave energy can achieve in minutes what traditional methods take hours to accomplish, with superior material properties [69].
Table 2: Optimized Parameters for Microwave rGO Synthesis [69]
| Parameter | Optimal Condition | Impact on Reaction |
|---|---|---|
| Microwave Power | 300 W | Balances efficient deoxygenation with morphology retention |
| Reaction Temperature | 120–140 °C | Drives efficient reduction of oxygen functional groups |
| Reaction Time | 5 minutes | Enables rapid synthesis while preserving structural integrity |
Experimental Protocols for Key Techniques
Protocol: Automated Photoredox Reaction Optimization
Photoredox catalysis is a powerful tool for forging challenging bonds but can be difficult to optimize due to parameters like light intensity and wavelength. The Photoredox Optimization (PRO) reactor automates this process [86].
- Objective: To rapidly optimize challenging photoredox-mediated cross-coupling reactions.
- Materials: Substrates, photocatalyst, reagents, solvent.
- Equipment: PRO reactor (provides precise light irradiance control to temperature-controlled, optically thin reaction volumes), high-intensity laser source, liquid handler, IR-MALDESI-MS for high-throughput analysis.
- Procedure:
- Platform Setup: The PRO reactor is configured to control light irradiance and temperature for reactions volumes of less than 10 μL.
- Reaction Array Design: Design a 384-reaction array varying key parameters such as photocatalyst, light intensity, stoichiometry, and solvent.
- Automated Execution & Analysis: The liquid handler prepares the reaction array in a microplate. After irradiation in the PRO reactor, crude products are automatically transferred to a new microplate for analysis.
- High-Throughput Analysis: IR-MALDESI-MS quantifies reaction outcomes across the 384-well plate in under 6 minutes.
- Validation: Promising conditions are scaled up to validate isolated yields.
- Analysis & Notes: This platform has successfully identified improved conditions for decarboxylative cross-couplings (up to 58% isolated yield) and enabled the discovery of novel conditions for previously unsuccessful transformations [86].
Protocol: Green Chemistry and Kinetic Analysis Spreadsheet
A combined analytical approach using a customized spreadsheet allows for the simultaneous determination of kinetics, solvent effects, and green metrics to guide optimization towards more sustainable conditions [85].
- Objective: To thoroughly understand and optimize a reaction for green chemistry principles using kinetic and solvation data.
- Materials: Reaction components, a range of solvents for testing.
- Equipment: NMR spectrometer or other analytical tool for concentration monitoring, spreadsheet software (e.g., Microsoft Excel) with the custom reaction optimization template.
- Procedure:
- Data Collection: Perform the reaction of interest in different solvents and at different initial reactant concentrations. Monitor the concentration of a key reactant or product over time using ( ^1H ) NMR.
- Kinetic Analysis (VTNA): Input concentration-time data into the "Kinetics" worksheet. Test different potential reaction orders; the correct orders will cause the data from different initial conditions to overlap onto a single curve. The spreadsheet will calculate the rate constant (( k )) for each experiment.
- Solvent Effect Analysis (LSER): In the "Solvent effects" worksheet, input the calculated ( \ln(k) ) values and the Kamlet-Abboud-Taft solvatochromic parameters (( \alpha ), ( \beta ), ( \pi^* )) for each solvent. Perform a multiple linear regression to derive an equation correlating reaction rate with solvent properties.
- Solvent Greenness Evaluation: The "Solvent selection" and "Solvent list" worksheets compare the predicted performance of solvents (from the LSER) against their greenness scores from guides like the CHEM21 Solvent Selection Guide.
- Metrics Prediction: Use the "Metrics" worksheet to predict conversion and calculate green metrics like Reaction Mass Efficiency (RME) and Optimum Efficiency for proposed conditions.
- Analysis & Notes: This integrated method was validated for reactions like aza-Michael addition, revealing that polar, hydrogen bond accepting solvents accelerate the reaction and identifying dimethyl sulfoxide (DMSO) as a high-performing, though not ideal, solvent [85].
Visualization of Workflows
The following diagrams illustrate the core logical relationships and experimental workflows for the key methodologies discussed.
Diagram 1: Automated Kinetic Modeling Workflow
Diagram 2: Multi-Task Bayesian Optimization Cycle
The Scientist's Toolkit
A successful rapid optimization campaign relies on a suite of specialized reagents, equipment, and software.
Table 3: Essential Research Reagent Solutions and Tools
| Tool / Reagent | Function / Purpose | Example Use Case |
|---|---|---|
| Microwave Reactor [69] | Provides rapid, controlled, and uniform heating for chemical reactions. | Accelerated hydrothermal synthesis of reduced graphene oxide (rGO). |
| Autonomous Flow Chemistry Platform [82] [83] | Automated system for continuous reagent mixing and reaction under controlled conditions (temp, flow rate). | Self-optimization of C–H activation reactions for medicinal chemistry. |
| Photoredox Optimization (PRO) Reactor [86] | Automated platform for high-throughput photoredox screening with precise light and temperature control. | Optimization of decarboxylative cross-couplings. |
| Liquid Handling Robot [84] | Automates precise dispensing of liquids for preparation of reaction arrays in microplates. | Enabling high-throughput experimentation (HTE) in 384- or 1536-well formats. |
| phactor Software [84] | Software for designing, executing, and analyzing high-throughput experiment arrays. | Managing metadata and results for reaction discovery campaigns. |
| Summit Package [83] | Open-source Python package for reaction optimization, includes Multi-Task BO algorithms. | Implementing Bayesian Optimization strategies for chemical reactions. |
| Gd-doped Ceria (CeO₂) [67] | A benchmark metal oxide for thermochemical water splitting. | Microwave-assisted clean hydrogen production. |
| Variable Time Normalization Analysis (VTNA) Spreadsheet [85] | A tool for determining reaction orders and rate constants from concentration-time data. | Understanding reaction mechanism and kinetics for green chemistry optimization. |
Evidence and Efficacy: Validating Microwave Chemistry's Superior Performance
The integration of microwave (MW) energy into chemical synthesis represents a transformative approach for advancing sustainable chemistry. This paradigm shift moves beyond traditional thermal heating methods, offering dramatic improvements in efficiency, energy consumption, and reaction speed. As the pharmaceutical and energy sectors face increasing pressure to develop greener manufacturing processes, microwave-assisted synthesis has emerged as a powerful solution that aligns with the principles of green chemistry. By enabling reactions that are faster, cleaner, and more energy-efficient, microwave technology directly addresses key environmental challenges while maintaining high synthetic efficiency [33].
The fundamental advantage of microwave chemistry lies in its heating mechanism. Unlike conventional conductive heating that relies on surface-to-core heat transfer, microwave irradiation delivers energy volumetrically through direct interaction with molecular dipoles and ions. This dielectric heating mechanism eliminates thermal gradients, reduces wall effects, and provides instantaneous, precise temperature control [24] [87]. The result is a dramatic acceleration of reaction rates—in best cases up to 1000-fold—alongside significant temperature reductions exceeding 60% for certain transformations [88] [89] [90]. These quantitative benefits position microwave chemistry as a cornerstone technology for sustainable industrial processes.
Quantitative Case Studies: Documented Acceleration and Temperature Reduction
Pharmaceutical Synthesis: 1000-Fold Rate Acceleration
Within pharmaceutical research and development, microwave-assisted organic synthesis (MAOS) has demonstrated extraordinary rate enhancements that fundamentally change synthetic planning and execution.
Table 1: Documented Cases of Extreme Rate Acceleration in Pharmaceutical Synthesis
| Reaction Type/System | Conventional Duration | MW-Irradiated Duration | Acceleration Factor | Key Quantitative Benefit | Citation |
|---|---|---|---|---|---|
| General organic transformations | Hours to days | Minutes | Up to 1000-fold | Reaction rates accelerated 1,000-fold for best cases | [88] |
| Diverse purine library synthesis | 12-24 hours | Minutes | ~100-fold | Rapid optimization identified optimum conditions in minutes instead of hours | [88] |
| 1,2,4-oxadiazoles optimization | Not specified | 10 minutes (2-10 min range) | Significant | Statistical design revealed optimum reaction time of 10 min | [88] |
| Aspirin synthesis | Not specified | Reduced time | Not specified | Yield increased from 85% to 97% with higher purity | [24] [87] |
The documented 1000-fold acceleration enables medicinal chemists to rapidly explore chemical space and optimize reaction conditions with unprecedented speed. This acceleration directly translates to reduced development timelines for new pharmaceutical entities, demonstrating how microwave technology addresses the industry's need for increased efficiency in the drug discovery and development process [88].
Clean Energy Applications: 60% Temperature Reduction
In the energy sector, microwave-assisted processes have achieved dramatic temperature reductions that overcome fundamental limitations in clean energy production, particularly for hydrogen generation.
Table 2: Documented Temperature Reductions in Materials and Energy Applications
| Process/Reaction | Conventional Temperature | MW-Irradiated Temperature | Temperature Reduction | Key Quantitative Benefit | Citation |
|---|---|---|---|---|---|
| Hydrogen production (Gd-doped ceria reduction) | >1,500°C | <600°C | >60% | Microwave energy replaced 75% of thermal energy requirement | [89] [90] |
| NO decomposition (CuO-Cu-ZSM-5 catalyst) | 550°C | 300-350°C | ~45% | NO conversion increased from 65% to 98% at lower temperature | [91] |
| NO decomposition (BaMnO3 catalyst) | 650°C | 250°C | ~62% | NO conversion increased from 45.4% to 93.7% at lower temperature | [91] |
| H2S decomposition (CoS/γ-Al2O3/BaMn0.2Cu0.8O3) | Not specified | 200°C lower | Significant | Temperature decreased by hundreds of degrees centigrade | [91] |
The 60% temperature reduction demonstrated in hydrogen production technology represents a breakthrough for sustainable energy. By lowering the reduction temperature of Gd-doped ceria from >1,500°C to below 600°C, microwave irradiation addresses the key limitation of conventional thermochemical methods—their extreme energy intensity and associated costs [89] [90]. This temperature reduction directly enables more economical and scalable clean hydrogen production.
Experimental Protocols & Methodologies
Protocol for Microwave-Assisted Hydrogen Production via Oxygen Vacancy Creation
The POSTECH research team developed a specialized protocol for microwave-assisted hydrogen production that achieves the documented 60% temperature reduction through rapid creation of oxygen vacancies [89] [90].
Materials and Equipment:
- Gd-doped ceria (CeO2) as benchmark hydrogen production material
- Dedicated microwave reactor with temperature and pressure control
- Microwave frequency: 2.45 GHz (standard ISM band)
- Inert atmosphere chamber for reaction control
Procedure:
- Prepare Gd-doped ceria samples with controlled particle size and morphology
- Load sample into microwave reactor under controlled atmosphere
- Apply microwave irradiation at precisely controlled power levels
- Ramp temperature to below 600°C (compared to conventional 1,500°C requirement)
- Maintain temperature for minutes (compared to hours in conventional methods)
- Monitor oxygen vacancy formation in real-time using appropriate analytical techniques
- Validate oxygen vacancy concentration post-reaction through thermodynamic modeling
Key Innovation: The protocol leverages the direct coupling of microwave energy with the ceramic material to create oxygen vacancies—defects in the material structure essential for splitting water into hydrogen—in minutes rather than hours. This rapid process at lower temperature was further validated with a thermodynamic model that provided insight into the microwave-driven reaction mechanism [89].
Protocol for Pharmaceutical Library Synthesis Using Microwave Acceleration
The documented 1000-fold acceleration in pharmaceutical synthesis employs specialized protocols for rapid reaction optimization and library generation [88].
Materials and Equipment:
- Single-mode or multi-mode microwave reactor with temperature control
- Sealed reaction vessels appropriate for small-scale (0.2mL to 5mL) synthesis
- Polar solvents with high dielectric constants for efficient microwave coupling
- Catalysts (e.g., YbCl3 for domino reactions)
- Building blocks (e.g., propargylated-flavones, aldehydes, anilines)
Procedure:
- Charge reaction vessels with substrates, catalysts, and solvents
- Seal vessels and place in microwave reactor
- Program temperature and pressure parameters based on initial scoping studies
- Irradiate with microwave energy for short durations (typically minutes)
- Employ rapid cooling after irradiation through built-in cooling systems
- Parallel process multiple reactions simultaneously using rotor systems
- Analyze results and iterate conditions for optimization
Key Innovation: This protocol enables rapid reaction scoping where reaction times are typically in the order of minutes, allowing facile optimization of reaction parameters including time, temperature, reagents, and solvents. The incorporation of analytical tools for real-time data acquisition further aids in reaction optimization [88].
Theoretical Foundation: Mechanisms Behind the Dramatic Enhancements
The extraordinary quantitative benefits observed in microwave-assisted processes stem from fundamental theoretical principles that distinguish microwave irradiation from conventional heating methods.
Dielectric Heating and Volumetric Energy Transfer
Microwave heating operates through dielectric mechanisms, primarily dipolar polarization and ionic conduction. When microwave radiation at 2.45 GHz interacts with polar molecules or ionic species, the electric field component causes rapid molecular reorientation (dipolar polarization) or ion migration (ionic conduction). This molecular motion generates heat instantaneously throughout the reaction volume rather than transferring heat gradually from the surface inward [24] [87]. This volumetric heating eliminates thermal gradients and wall effects that plague conventional heating methods, explaining the dramatically reduced processing times.
Reduction in Apparent Activation Energy
Groundbreaking research has demonstrated that microwave irradiation directly reduces the apparent activation energy (Ea') required for chemical reactions. Studies on NO decomposition and H2S splitting showed Ea' reductions from 71-123 kJ/mol to 20-25 kJ/mol under microwave irradiation [91]. This phenomenon challenges the classical view of microwaves as merely a heating method and reveals that microwave energy partially transforms to directly reduce the energy barrier for reactions. The proposed model of interactions between microwave electromagnetic waves and molecules provides a quantitative framework for estimating this Ea' reduction, positioning microwave irradiation as a distinct form of power energy for chemical activation [91].
Oxygen Vacancy Formation in Energy Materials
In hydrogen production, the dramatic temperature reduction achieved through microwave irradiation stems from enhanced formation of oxygen vacancies—defects in the material structure where oxygen atoms are missing from their lattice sites. Conventional methods require extreme temperatures (>1,500°C) and extended time to create these vacancies, but microwave energy facilitates their formation in minutes at temperatures below 600°C [89] [90]. The microwave electromagnetic field directly interacts with the crystal lattice, lowering the energy required for oxygen vacancy creation and thereby enabling more efficient thermochemical hydrogen production.
The Scientist's Toolkit: Essential Research Reagents & Materials
Successful implementation of microwave-accelerated reactions requires specific materials and equipment designed to optimize microwave energy transfer and reaction control.
Table 3: Essential Research Reagents and Materials for Microwave-Assisted Synthesis
| Item/Category | Specific Examples | Function & Importance | Application Context |
|---|---|---|---|
| Polar Solvents | Water, DMSO, ethanol, ionic liquids | Efficient microwave absorption through high dielectric loss factors; enable rapid heating | General MAOS, green chemistry applications |
| MW-Absorbing Catalysts | YbCl₃, CuO-Cu-ZSM-5, BaMnO₃, Gd-doped ceria | Enhance microwave absorption and catalytic activity; enable lower temperature transformations | Heterocyclic synthesis, hydrogen production, gas phase reactions |
| Dedicated MW Reactors | Single-mode systems, multi-mode parallel systems | Provide controlled temperature/pressure conditions, homogeneous microwave field, magnetic stirring | Pharmaceutical R&D, materials synthesis, reaction optimization |
| Specialized Reaction Vessels | Sealed tubes, microtiter plates, continuous-flow cells | Enable safe operation at elevated temperatures and pressures; facilitate parallel processing | High-throughput chemistry, process development, scale-up studies |
| Susceptors & MW-Absorbing Additives | Silicon carbide, activated carbon, specialized ceramics | Absorb and transfer microwave energy to low-absorbing materials; enable heating of non-polar systems | Gas-phase reactions, non-polar solvent systems, specialized syntheses |
The selection of appropriate solvents is particularly critical, as efficient microwave absorption requires materials with high dielectric loss factors (ε''). Solvents like dimethyl sulfoxide (DMSO, ε'' ≈ 37) are classified as "lossy" and efficiently convert microwave energy to heat, while non-polar solvents like hexane (ε'' ≈ 0.05) are essentially microwave-transparent and heat poorly [87]. Similarly, the strategic use of microwave-absorbing catalysts and additives can dramatically enhance reaction efficiency by creating localized "hotspots" or facilitating specific molecular activation pathways [91].
Workflow & Process Integration
Implementing microwave-assisted synthesis requires careful planning and optimization to maximize the dramatic benefits documented in case studies. The following workflow outlines the key stages for developing and optimizing microwave-accelerated processes.
This systematic approach ensures that researchers fully leverage the unique advantages of microwave irradiation while maintaining control over reaction parameters. The workflow emphasizes the importance of initial assessment and scoping studies, followed by systematic optimization and careful scale-up. At each stage, the fundamental principles of microwave-matter interactions guide decision-making, particularly regarding solvent selection, catalyst design, and parameter optimization.
The quantitative case studies presented in this technical guide—documenting 1000-fold rate accelerations and 60% temperature reductions—provide compelling evidence for microwave energy as a transformative technology in sustainable chemistry. These dramatic enhancements stem from fundamental microwave-specific effects including volumetric heating, reduced activation energies, and selective molecular activation. The experimental protocols and theoretical frameworks outlined herein offer researchers practical pathways to implement these advanced synthetic methodologies.
Looking forward, microwave-assisted synthesis is poised to play an increasingly critical role in green chemistry initiatives across pharmaceutical manufacturing, materials science, and clean energy production. Emerging applications in continuous-flow microwave reactors, microwave-biocatalysis hybrid systems, and microwave-assisted nanoparticle synthesis demonstrate the expanding scope of this technology [24] [33]. As quantification of green metrics becomes increasingly important in industrial settings, the demonstrated reductions in energy consumption, reaction times, and waste generation position microwave chemistry as an essential component of sustainable chemical manufacturing.
The ongoing development of more sophisticated microwave reactor systems with enhanced process control and monitoring capabilities will further accelerate adoption across diverse chemical industries. By integrating microwave technology with other green chemistry approaches such as solvent-free reactions, renewable feedstocks, and catalytic processes, researchers can continue to advance the overarching goal of sustainable chemical synthesis—maximizing efficiency while minimizing environmental impact.
This whitepaper provides a technical comparison between microwave irradiation and conventional conductive heating within the context of pharmaceutical synthesis. Framed by the principles of sustainable chemistry, this analysis examines the fundamental mechanisms, energy efficiency, and practical implications of each heating method. Data synthesized from recent studies demonstrate that microwave-assisted synthesis can reduce reaction times by at least 60% and lower energy consumption compared to conventional methods, while conductive heating offers superior temperature uniformity [92] [19]. The selection of heating technology presents a significant opportunity for drug development professionals to minimize the environmental footprint of their processes, aligning with broader goals of sustainable chemistry through optimized energy research.
The efficacy and sustainability of pharmaceutical synthesis are profoundly influenced by the chosen heating method. Conventional conductive heating and microwave irradiation represent two fundamentally different approaches to energy transfer, each with distinct implications for reaction kinetics, product purity, and overall energy efficiency.
Conventional conductive heating relies on the external application of thermal energy. Heat is generated by an external source (e.g., a heating mantle, oil bath, or hot plate) and must then be transferred to the reaction vessel via conduction, and subsequently to the reaction mixture via conduction and convection [92]. This process is inherently slow and inefficient, as it depends on the temperature gradients and the thermal conductivity of the vessel and reaction materials. A significant amount of energy is wasted in heating the reactor walls and the surrounding environment rather than the reactants themselves [4].
Microwave irradiation, in contrast, employs electromagnetic radiation to deliver energy directly and volumetrically to the reactants. Microwave ovens operate at a frequency of 2450 MHz, causing polar molecules and ions in the reaction mixture to oscillate rapidly. This oscillation generates heat internally through molecular friction and dielectric loss, a process known as dielectric heating [92] [93]. This mechanism allows for exceptionally rapid and direct heating of the entire reaction volume simultaneously, bypassing the limitations of thermal conductivity.
The following diagram illustrates the fundamental differences in how these two methods deliver energy to a reaction mixture.
Quantitative Comparative Analysis
A direct, side-by-side comparison of key performance metrics is essential for evaluating the two heating methodologies. The data below, compiled from recent research, highlights the trade-offs between speed, efficiency, and control.
Table 1: Direct Performance Comparison of Heating Methods
| Performance Metric | Microwave Heating | Conventional Conductive Heating | Source |
|---|---|---|---|
| Typical Reaction Time | 4 minutes | 4 hours | [19] |
| Heating Rate | Very fast, instant heating | Slow, dependent on thermal transfer | [92] |
| Energy Efficiency | High; energy transferred directly to reactants | Lower; significant heat loss to surroundings | [94] [4] |
| Temperature Uniformity | Can suffer from hotspots and spikes | Uniform and consistent delivery | [95] |
| Crude Purity (18mer peptide) | 51.7% | 52.0% | [95] |
| Crude Purity (39mer peptide) | 30.5% | 37.0% | [95] |
| Moisture Removal Efficiency | 93.3% (at 85°C for naproxen-sodium) | Information not explicitly provided in search results | [96] |
The data in Table 1 reveals a complex picture. While microwave heating offers dramatic reductions in reaction time, its impact on crude purity can be variable. For instance, a study on Solid Phase Peptide Synthesis (SPPS) found that for a 39mer peptide, conduction heating resulted in a 37.0% crude purity compared to 30.5% for microwave heating, suggesting that for more complex syntheses, the consistent temperature of conduction heating may help minimize side reactions like racemization [95].
Table 2: Suitability Analysis for Reaction Types
| Reaction Characteristic | Suitable for Microwave? | Notes | |
|---|---|---|---|
| Polar Solvents/Reagents | Yes (Highly Suitable) | Efficiently absorb microwave energy (e.g., water, DMF) | [93] |
| Non-Polar Solvents | No (Less Suitable) | Poorly absorb microwave energy (e.g., hexane, toluene) | [93] |
| Reactions Requiring High Uniformity | Potentially Problematic | Risk of hotspots and non-uniform electromagnetic fields | [95] [92] |
| Heat-Sensitive Compounds | Conditional | Rapid heating may be detrimental without precise control | [97] |
Detailed Experimental Protocols
To illustrate the practical application of these heating methods, below are detailed protocols for a model reaction and a common pharmaceutical unit operation.
Protocol A: Synthesis of N-o-tolyl-1H-benzo[d][1,2,3]triazole-5-carboxamide
This protocol for synthesizing a benzotriazole derivative provides a direct comparison of conditions and outcomes [19].
Objective: To compare the efficiency of microwave-assisted synthesis versus conventional conductive heating for the formation of a benzotriazole carboxamide.
Reaction Scheme: Benzotriazole-5-carbonyl chloride + o-toluidine → N-o-tolyl-1H-benzo[d][1,2,3]triazole-5-carboxamide
Materials & Reagents:
- Benzotriazole-5-carbonyl chloride: Starting material, acylating agent.
- o-Toluidine: Nucleophile, reacts with acyl chloride to form amide bond.
- Benzene: Solvent, reaction medium. (Note: Due to toxicity, alternative solvents may be explored in modern applications).
- 10% Hydrochloric Acid: Quenching agent, removes excess o-toluidine.
- Anhydrous Sodium Sulfate: Drying agent, removes residual water from organic layer.
Procedure:
- Reaction Setup: Mix benzotriazole-5-carbonyl chloride (1 g, 5.50 mmol) with 5 ml benzene in a suitable reaction vessel. Add an equimolar proportion of o-toluidine dissolved in 10 ml of benzene.
- Heating Method (Conventional):
- Attach a reflux condenser to the vessel.
- Heat the reaction mixture using a heating mantle or oil bath (conductive heating) under reflux for 4 hours [19].
- Heating Method (Microwave):
- Transfer the reaction mixture to a sealed microwave vessel.
- Irradiate the mixture at 180 W for 4 minutes and 30 seconds [19].
- Work-up (Common to Both Methods):
- After the reaction is complete, add 10% hydrochloric acid to the mixture to convert any excess o-toluidine into its water-soluble hydrochloride salt.
- Separate the benzene layer and wash it with water (3 × 10 ml).
- Dry the benzene layer over anhydrous sodium sulfate.
- Concentrate the solution by distillation or evaporation to obtain the product as a light brown crystalline powder.
Results:
- Yield (Conventional): 72%
- Yield (Microwave): 83%
- Reaction Time Reduction: >98% with microwave irradiation.
Protocol B: Microwave Drying of Pharmaceutical Powder (Naproxen-Sodium)
Drying is a critical, energy-intensive step in pharmaceutical manufacturing. This protocol details a microwave approach for drying a moisturized drug formulation [96].
Objective: To efficiently remove moisture from a formulated naproxen-sodium granule using microwave irradiation and characterize the impact on the final powder.
Materials:
- Naproxen-Sodium API: Active Pharmaceutical Ingredient.
- Microcrystalline Cellulose: Excipient, provides bulk and stability.
- Povidone (Polyvinylpyrrolidone): Binder, promotes granule formation.
- Deionized Water: Solvent for wet granulation.
Procedure:
- Formulation & Moisturizing: Prepare a formulation containing naproxen-sodium, microcrystalline cellulose, and povidone. Wet the powder mixture using deionized water via a wet granulation process.
- Microwave Drying: Place the moisturized granules in a microwave dryer. Dry the granules at a controlled temperature of 85°C.
- Characterization: Analyze the dried granules using:
- Moisture Analyzer: To determine final moisture content and removal efficiency.
- Powder X-ray Diffraction (PXRD): To assess changes in crystallinity.
- Scanning Electron Microscopy (SEM): To examine particle morphology and size.
- Fourier-Transform Infrared Spectroscopy (FTIR): To confirm chemical stability.
Results:
- Moisture Removal Efficiency: 93.3% at 85°C.
- Final Moisture Content: Reduced to 0.84%.
- Impact: While effective at moisture removal, high microwave drying temperatures can lead to changes in particle morphology and crystallinity, underscoring the need for precise temperature control [96].
The Scientist's Toolkit: Essential Research Reagents & Equipment
Successful implementation of these heating methods requires specific materials and instrumentation. The following table lists key items and their functions in the context of the discussed experiments.
Table 3: Key Reagents and Equipment for Heating Experiments
| Item | Function/Description | Relevance to Heating Methods |
|---|---|---|
| Polar Solvents (e.g., DMF, Water) | Solvents with high dielectric loss; efficiently absorb microwave energy. | Essential for efficient microwave heating. [92] [93] |
| Dedicated Microwave Reactor | Laboratory instrument providing controlled microwave irradiation with temperature and pressure monitoring. | Ensures reproducibility and safety in MAOS; avoids hotspots of domestic ovens. [93] |
| Zeolite-supported Catalysts | Spongelike materials with controlled cavity sizes; contain antenna sites (e.g., Indium ions). | Used in advanced microwave catalysis to focus thermal energy at specific active sites. [4] |
| Benzotriazole-based Intermediates | Versatile scaffolds acting as leaving groups, directors, or precursors in synthesis. | Model compound for comparing reaction efficiency between heating methods. [19] |
| Vacuum Tray Dryer (Conduction) | Dryer where trays are directly heated via conduction from warming shelves under vacuum. | Provides uniform, gentle heating for sensitive pharmaceuticals; an alternative to microwave drying. [97] |
The integration of microwave energy into pharmaceutical synthesis represents a significant stride toward sustainable chemistry. The primary advantages are profound: a reduction in reaction times by at least 60%, lower overall energy consumption, and decreased solvent usage through improved efficiency [92] [93]. One study highlighted that microwave heating could achieve a temperature rise in water using significantly less energy than a conventional hotplate, demonstrating its superior energy transfer efficiency [94]. Furthermore, the ability of microwaves to enable reactions under milder conditions and facilitate the use of greener solvents aligns with multiple principles of green chemistry [93].
However, this guide demonstrates that a one-size-fits-all approach is not optimal. The choice between microwave and conductive heating is application-dependent. Microwave irradiation excels in accelerating discovery chemistry, optimizing reactions, and reducing processing times for many transformations. In contrast, conventional conductive heating, particularly as implemented in direct-heating tray dryers, remains a robust and often superior choice for processes requiring extreme temperature uniformity and gentle handling of heat-sensitive active pharmaceutical ingredients (APIs) [95] [97].
For the drug development professional, the strategic integration of both technologies offers the most promising path forward. Microwave reactors can be used for rapid route scouting and optimization, while conduction-based systems can be employed for scaled-up, cGMP-sensitive processes where consistency is paramount. Future innovations, such as the combination of microwave heating with other technologies like acoustic cavitation or flow chemistry, will further enhance the sustainability profile of pharmaceutical manufacturing [92]. By making informed, context-driven decisions on heating methodologies, researchers can significantly advance the dual goals of pharmaceutical innovation and environmental stewardship.
The rapid integration of microwave technology into chemical synthesis and analysis represents a paradigm shift toward more sustainable laboratory practices. As the scientific community urgently moves to develop processes that are not aggressive to the environment nor to all forms of life, the principles of Green Chemistry provide an essential framework for evaluation [68]. Microwave-assisted synthesis offers significant environmental advantages through rapid, uniform heating that substantially reduces energy consumption, reaction times, and hazardous waste generation compared to conventional methods [36]. However, these claimed benefits require rigorous validation through standardized assessment tools. Green metrics transform subjective claims of sustainability into quantifiable, comparable data, enabling researchers to make informed decisions that align with the principles of sustainable chemistry [98].
The integration of green metrics is particularly crucial for microwave-derived protocols, which are often promoted as "green" alternatives without comprehensive life-cycle assessment. As microwave technology finds expanding applications in nanomaterial fabrication [24], sample preparation for elemental analysis [68], and pharmaceutical development, systematic sustainability assessment ensures that these protocols genuinely reduce environmental impact rather than simply shifting the environmental burden to other aspects of the process. This technical guide provides researchers with a comprehensive framework for applying established green metrics to microwave-assisted protocols, enabling meaningful sustainability assessments that support the broader thesis of sustainable chemistry through microwave energy research.
Green Metric Tools for Sustainability Assessment
Several validated metric systems have been developed to evaluate the environmental impact of chemical processes. Each tool employs distinct parameters and scoring mechanisms, allowing researchers to select the most appropriate assessment method for their specific application.
Table 1: Comparison of Major Green Metric Assessment Tools
| Metric Tool | Assessment Scope | Scoring System | Key Assessed Parameters | Best Applications |
|---|---|---|---|---|
| GreenPrep MW Score [68] | Microwave-assisted sample preparation | Comprehensive scoring of chemical, technology, and workflow parameters | Acid consumption, energy use, waste generation, automation | Elemental analysis sample preparation |
| AGREE Index [98] [99] | Entire analytical procedure | 0-1 scale (closer to 1 = greener) | 12 principles of green analytical chemistry | Comparative method assessment |
| GAPI [98] [99] | Five-stage analytical process | Pentagram visualization with color coding | Sample collection to instrumentation | Life-cycle impact assessment |
| Eco-Scale [99] | Penalty-based assessment | 100-point scale (higher = greener) | Reagents, energy, waste, toxicity | Quick procedural assessment |
| NEMI [98] | Environmental impact | Binary classification (pass/fail) | Persistence, bioaccumulation, toxicity, waste | Preliminary screening |
Specialized Metric for Microwave Applications: GreenPrep MW Score
The GreenPrep MW Score represents a specialized metric designed specifically for evaluating microwave-assisted sample preparation procedures for elemental analysis [68]. This tool evaluates the whole analytical procedure, considering how chemical parameters, technology variables, and workflow automation can be optimized, and how these factors impact the green metrics score. Unlike generic assessment tools, GreenPrep MW Score accounts for microwave-specific parameters including:
- Chemical Parameters: Acid consumption, reagent toxicity, and waste generation
- Technology Variables: Energy efficiency, heating uniformity, and vessel design
- Workflow Automation: Throughput, manual intervention, and safety features
The GreenPrep MW Score serves as a complementary strategy to existing metrics, helping identify specific points for improvement in microwave-assisted procedures rather than replacing sound metrics already proposed for evaluating the sample preparation step [68]. Application of this metric has been demonstrated for diverse sample matrices including foods, multivitamin tablets, and spodumene, providing comparative data for sustainability optimization.
Applying Green Metrics to Microwave-Assisted Synthesis
Sustainability Advantages of Microwave Chemistry
Microwave-assisted synthesis provides several quantifiable advantages that directly contribute to improved green metric scores across assessment platforms:
- Rate Enhancement: Microwave irradiation typically reduces reaction times from hours to minutes or even seconds due to reduced activation energy barriers, directly decreasing energy consumption [24].
- High Product Yield: Shorter reaction times and uniform heating minimize undesirable side products, increasing atom economy [24].
- High Product Purity: Reduced formation of side products eliminates purification steps, minimizing solvent waste and energy use [24].
- High Reproducibility: Uniform electromagnetic field distribution ensures consistent results, reducing failed experiments and material waste [24].
- Expanded Reaction Conditions: Precise temperature control enables optimization of reaction parameters for maximum efficiency [24].
The fundamental mechanism of microwave heating—dielectric heating through dipolar polarization or ionic conduction—enables these advantages by providing instantaneous, uniform heating throughout the reaction mixture rather than relying on convective heat transfer from vessel walls [24]. This direct energy transfer eliminates thermal gradients, providing a reliable nucleation and growth environment that leads to uniform size distribution in nanomaterial synthesis [24].
Quantitative Sustainability Assessment in Nanomaterial Synthesis
The application of green metrics to microwave-assisted nanomaterial synthesis reveals substantial environmental advantages over conventional methods. In the synthesis of metal nanoparticles, carbon quantum dots, and hybrid nanocomposites, microwave protocols demonstrate superior performance across multiple sustainability parameters [36]. The integration of microwave assistance with eco-friendly precursors, including plant extracts, biomolecules, and ionic liquids, further enhances the green credentials of these protocols.
Table 2: Quantitative Comparison of Microwave vs. Conventional Heating for Nanomaterial Synthesis
| Parameter | Conventional Method | Microwave Method | Green Metric Improvement |
|---|---|---|---|
| Reaction Time | 2-12 hours | 5-30 minutes | 70-95% reduction |
| Energy Consumption | 300-800 W·h | 50-150 W·h | 75-85% reduction |
| Solvent Volume | 50-200 mL | 10-30 mL | 70-85% reduction |
| Temperature | 80-250°C | 60-150°C | 20-40% reduction |
| Product Yield | 65-85% | 85-97% | 15-40% improvement |
| Side Products | Significant | Minimal | Reduced purification needs |
The sustainability assessment of microwave-assisted synthesis extends beyond laboratory-scale evaluations to consider industrial viability. Green chemistry metrics applied to MAS protocols demonstrate not only reduced environmental impact but also improved economic feasibility through energy savings, reduced waste disposal costs, and decreased processing times [36]. When applied to the synthesis of aspirin, microwave assistance increased yield from 85% to 97% while eliminating purification steps, demonstrating the interconnected benefits of reduced resource consumption and improved process efficiency [24].
Experimental Protocols and Methodologies
Microwave-Assisted Sample Preparation for Elemental Analysis
The application of the GreenPrep MW Score to microwave-assisted sample preparation involves a systematic evaluation of all process parameters. The following protocol outlines the key steps for implementing and assessing this green metric:
Materials and Equipment:
- Microwave digestion system (cavity microwave oven or single reaction chamber system)
- Analytical balance (±0.1 mg precision)
- Appropriate digestion vessels
- High-purity acids (HNO₃, HCl, HF based on sample matrix)
- Certified reference materials for validation
Procedure:
- Sample Preparation: Weigh 0.1-0.5 g of homogeneous sample into digestion vessel. For organic matrices, preliminary drying and grinding may be required.
- Acid Addition: Add minimal acid volume required for complete digestion. For plant materials, typically 5-8 mL HNO₃; for geological samples, 3 mL HNO₃ + 1 mL HF.
- Microwave Program: Implement a stepped temperature program:
- Ramp to 120°C over 10 minutes, hold for 5 minutes
- Ramp to 180°C over 8 minutes, hold for 10 minutes
- Ramp to 200°C over 5 minutes, hold for 15 minutes
- Cooling and Dilution: Cool vessels to room temperature, carefully release pressure, transfer digestates to volumetric flasks, and dilute to volume with deionized water.
- Analysis: Proceed with elemental analysis via ICP-OES or ICP-MS.
Green Metric Assessment:
- Calculate GreenPrep MW Score based on:
- Acid consumption (mL/sample)
- Energy consumption (kW·h/sample)
- Total process time (including cooling)
- Waste generation (mL/sample)
- Safety considerations (pressure/temperature limits)
- Compare scores against benchmark values for similar matrices
- Identify parameters with lowest scores for optimization
Microwave-Assisted Synthesis of Carbon Quantum Dots
The following protocol demonstrates the application of green metrics to nanomaterial synthesis, highlighting sustainability advantages:
Materials:
- Citric acid (1.0 g)
- Ethylenediamine (0.34 mL)
- Deionized water (50 mL)
- Ethanol for purification
Procedure:
- Reaction Mixture Preparation: Dissolve citric acid (1.0 g) in deionized water (50 mL) in a microwave-safe vessel. Add ethylenediamine (0.34 mL) under stirring.
- Microwave Synthesis: Place vessel in microwave reactor. Program conditions: 180°C for 30 minutes at 300 W power.
- Product Recovery: Cool reaction mixture to room temperature. Filter through 0.22 μm membrane to remove large particles.
- Purification: Dialyze against deionized water using 1000 Da molecular weight cut-off membrane for 24 hours.
- Characterization: Analyze using UV-Vis spectroscopy, fluorescence spectroscopy, and TEM.
Green Metric Assessment:
- AGREE Calculator Inputs:
- Sample preparation: 2 (minimal preparation)
- Sample amount: 10 (1.0 g starting material)
- Energy consumption: 8 (0.15 kW·h total)
- Reagent toxicity: 7 (low toxicity precursors)
- Waste generation: 8 (aqueous waste, minimal)
- Expected AGREE Score: 0.75-0.85 (significantly greener than conventional methods)
Visualization of Assessment Workflows
Green Metric Integration in Microwave Protocol Development
The following diagram illustrates the systematic integration of green metrics throughout the development and optimization of microwave-assisted protocols:
Comparative Green Assessment Workflow
This diagram outlines the decision process for selecting appropriate green metrics based on research objectives and protocol type:
The Researcher's Toolkit: Essential Materials and Reagents
Table 3: Research Reagent Solutions for Sustainable Microwave Protocols
| Reagent/Material | Function | Green Characteristics | Application Examples |
|---|---|---|---|
| Ionic Liquids [24] | Green solvents and catalysts | Non-volatile, recyclable, low toxicity | Nanomaterial synthesis, catalysis |
| Deep Eutectic Solvents [99] | Sustainable extraction media | Biodegradable, low cost, low toxicity | Sample preparation, natural product extraction |
| Water as Solvent [36] | Reaction medium | Non-toxic, non-flammable, inexpensive | Hydrothermal synthesis, green extraction |
| Plant Extracts [36] | Natural reducing/capping agents | Renewable, biodegradable, non-toxic | Metallic nanoparticle synthesis |
| Solid-Supported Reagents [24] | Heterogeneous catalysis | Recyclable, reduced waste, easy separation | Organic synthesis, functionalization |
Future Perspectives and Implementation Challenges
Advancing Green Metric Methodologies
The evolution of green metrics continues with the emergence of whiteness assessments that balance environmental impact with analytical functionality. The Whiteness Assessment Criteria (WAC) align more closely with sustainable development than other methods due to their holistic perspective, seeking to balance environmental impact with functionality rather than unconditionally increasing greenness at the expense of performance [98]. This balanced approach is particularly relevant for microwave-derived protocols in regulated industries like pharmaceutical development, where method performance cannot be compromised.
Future developments in green metric applications will likely include:
- Integration of Artificial Intelligence: Machine learning algorithms for predicting green metric scores during method development
- Automated Assessment Tools: Direct integration of sustainability metrics into laboratory information management systems
- Standardized Reporting Frameworks: Industry-wide acceptance of specific metrics for comparative studies
- Circular Economy Metrics: Assessment of resource recovery and recyclability within microwave protocols
Implementation in Developing Countries
The adoption of green metrics in developing countries presents both challenges and opportunities. While resource constraints may limit access to advanced microwave instrumentation, the fundamental principles of green chemistry can be adapted to local contexts [98]. Microwave-assisted methods often provide particular benefits in these settings due to their reduced energy requirements and minimal infrastructure needs compared to conventional laboratory equipment. Knowledge transfer and capacity building through international collaborations play crucial roles in advancing sustainable chemistry practices globally [98].
The application of green metrics to microwave-derived protocols provides an essential framework for validating sustainability claims and guiding method optimization. Tools such as the GreenPrep MW Score, AGREE, GAPI, and Eco-Scale offer complementary approaches for quantifying environmental impact across different applications and development stages. As microwave technology continues to evolve as a cornerstone of sustainable chemistry practice, rigorous assessment using these metrics ensures genuine environmental benefits rather than perceived advantages. By integrating sustainability assessments throughout method development, researchers can contribute meaningfully to the transformation of chemical processes toward alignment with circular economy principles and reduced environmental impact.
Microwave-assisted organic synthesis (MAOS) has evolved from a laboratory curiosity to a cornerstone of modern sustainable chemistry. While its advantages in accelerating reaction rates are well-documented, this whitepaper delves into its transformative role in unlocking chemical transformations that are inherently inefficient, impractical, or entirely inaccessible via conventional thermal methods. By providing intense, instantaneous, and volumetric heating, microwave irradiation fundamentally alters reaction pathways and energy landscapes. This in-depth technical guide explores the mechanistic foundations of this phenomenon, supported by quantitative data and detailed protocols, framing MAOS as an indispensable tool for researchers and drug development professionals dedicated to advancing green chemistry principles.
The drive for sustainable chemistry has catalyzed a paradigm shift in synthetic methodology, moving beyond incremental improvements to seek transformative technological advances. Microwave-assisted synthesis aligns perfectly with this goal, directly addressing multiple principles of green chemistry, including energy efficiency, waste reduction, and the use of safer solvents [93]. First reported in 1986, MAOS has matured into a sophisticated discipline, offering more than mere acceleration of known reactions [93]. The core distinction lies in the heating mechanism: conventional heating (CH) relies on conductive thermal transfer from surface to core, often resulting in thermal gradients and inefficient energy utilization. In contrast, microwave irradiation delivers energy directly to molecules throughout the reaction volume via dipole rotation and ionic conduction, enabling instantaneous "in-core" heating [100]. This fundamental difference is not merely a matter of kinetics but can be the decisive factor in achieving novel chemical transformations, particularly those involving thermally sensitive intermediates or requiring precise energy input at the molecular level. For synthetic chemists, this represents a powerful strategy to push the boundaries of accessible chemistry, facilitating the discovery of new reaction pathways and the synthesis of complex architectures under markedly greener conditions.
Fundamental Mechanisms: Beyond Simple Heating
Dielectric Heating and Molecular Interaction
Microwave energy interacts with matter through two primary mechanisms: dipole rotation and ionic conduction [100]. Dipole rotation involves the rapid reorientation of polar molecules in an oscillating electromagnetic field, generating heat through molecular friction. Ionic conduction involves the accelerated movement of dissolved ions, which collide with neighboring molecules. The efficiency of these processes is governed by the material's dielectric properties. The loss tangent (tan δ) is a critical parameter, defining a substance's ability to convert microwave energy into heat [100]. A high tan δ indicates efficient absorption.
The energy transfer in microwave irradiation occurs on a nanosecond timescale, far outpacing the heat transfer in conventional heating [29]. This direct coupling can lead to the superheating of solvents well above their standard boiling points, especially in sealed vessels, dramatically altering reaction kinetics and thermodynamics [100]. While the existence of "non-thermal" microwave effects remains debated, the consensus is that the observed dramatic rate enhancements are primarily due to these rapid thermal/kinetic effects [100].
The Atomic Focus: Precision Heating at Active Sites
Recent pioneering research has demonstrated that microwave energy can be concentrated at the atomic scale. A groundbreaking study from the University of Tokyo showed that microwaves can be tuned to selectively excite single atomic active sites within a catalyst, such as indium ions embedded in a zeolite framework [4]. These ions act as "atomic microwave antennas," localizing thermal energy precisely where the reaction occurs. This technique achieved an energy efficiency approximately 4.5 times greater than conventional heating methods [4]. This selective activation is a quintessential example of a transformation unachievable with traditional heating, where energy is dissipated throughout the entire reactor. This approach is particularly valuable for demanding reactions like CO₂ conversion and methane activation, opening new avenues for eco-catalysis with a significantly reduced energy footprint [4].
Table 1: Dielectric Properties of Common Solvents in Microwave Synthesis
| Solvent | Dielectric Loss (ε") | Absorptivity Category | Boiling Point (°C) |
|---|---|---|---|
| Ethanol | ~20 | High | 78 |
| Water | ~10 | Medium | 100 |
| DMF | ~10 | Medium | 153 |
| Dichloromethane | ~0.4 | Low | 40 |
| Toluene | ~0.1 | Low | 111 |
| Hexane | ~0.02 | Low | 69 |
Quantitative Validation: Comparative Data in Novel Synthesis
The theoretical advantages of MAOS are borne out by direct, quantitative comparisons with conventional methods across diverse chemical domains. The following data highlights its pivotal role in enabling efficient syntheses.
Accessing Privileged Heterocycles in Drug Discovery
The synthesis of nitrogen-containing heterocycles like triazoles is a critical task in pharmaceutical chemistry. Conventional methods often suffer from long reaction times and harsh conditions. Microwave irradiation has proven uniquely effective in this domain.
Table 2: Comparative Synthesis of 1,2,4-Triazole and Piperidine Derivatives [101]
| Method | Reaction Time | Yield (%) | Conditions |
|---|---|---|---|
| Conventional Heating | 33-90 minutes | 82 | Thermal heating |
| Microwave Irradiation | 33-90 seconds | 82 | Microwave, 300 W |
In another example, the synthesis of 4-(benzylideneamino)-3-(1-(2-fluoro-[1,1′-biphenyl]-4-yl)ethyl)-1H-1,2,4-triazole-5(4H)-thione derivatives was completed in 10–25 minutes with a 97% yield under microwave radiation, compared to 290 minutes for a 78% yield using conventional heating [101]. This dramatic compression of reaction time while improving yield is a hallmark of MAOS, enabling rapid exploration of structure-activity relationships in drug development.
Advanced Materials and Polymer Chemistry
The benefits of MAOS extend beyond small molecules to the realm of materials science. In the synthesis of superabsorbent agar-based magnetic composite hydrogels (AMHs) for drug delivery, microwave-assisted free radical polymerization yielded products with remarkable swelling capacities of up to 19,840% [102]. Furthermore, in nanomaterials, a microwave solvothermal synthesis (MSS) route was successfully used to produce aluminum-doped ZnO nanocrystals in a non-polar hydrocarbon solvent—a challenging feat due to the solvent's low microwave absorptivity [103]. This demonstrates the versatility of MAOS in facilitating reactions even in typically microwave-transparent media, which is crucial for designing advanced materials like IR emissivity modulation devices.
Experimental Protocols: A Guide to Method Development
General Workflow for Microwave Reaction Setup
The following diagram outlines the core decision-making process for developing a microwave-assisted synthesis method.
Objective: To rapidly synthesize a complex 1,2,4-triazole and piperidine-containing derivative via microwave irradiation.
Reagents and Materials:
- Precursor: 5-{1-[(4-methoxyphenyl)sulfonyl]-4-piperidinyl}-4-phenyl-4H-1,2,4-triazole-3-thiol
- Various acid chlorides (for N-substitution)
- Base: Triethylamine (TEA) or similar
- Solvent: Anhydrous DMF or acetonitrile (medium microwave absorbers)
- Microwave reactor capable of controlled temperature and power output (e.g., Biotage Initiator+)
Procedure:
- Reaction Setup: In a dedicated microwave vial (e.g., 2-5 mL), combine the triazole-thiol precursor (1.0 mmol) and the selected acid chloride (1.2 mmol). Add anhydrous solvent (3-5 mL) and a base like TEA (2.0 mmol) to scavenge HCl produced.
- Sealing and Atmosphere: Seal the vial with the appropriate pressure-resistant cap. Purge the headspace with an inert gas (e.g., nitrogen or argon) for 1-2 minutes to create an oxygen-free environment, crucial for radical stability and preventing oxidation [102] [29].
- Microwave Irradiation: Place the vial in the microwave reactor. Program the method based on the general workflow:
- Temperature: Set 150-170°C (superheating above solvent bp).
- Time: Set a 60-second irradiation time.
- Power: Use a fixed power of 300 W or use dynamic power control to regulate temperature.
- Stirring: Enable high-speed magnetic stirring throughout the run.
- Reaction Work-up: After irradiation and controlled cooling, carefully depressurize the vial. Transfer the reaction mixture to a round-bottom flask. Dilute with water and ethyl acetate, then extract the product. Purify the crude material using standard techniques like column chromatography or recrystallization.
Key Considerations: This protocol reduces the reaction time from over an hour to mere seconds while maintaining high yield, showcasing a key advantage of MAOS [101].
The Scientist's Toolkit: Essential Research Reagent Solutions
The successful implementation of microwave-assisted synthesis relies on a set of key reagents and materials designed to efficiently couple with microwave energy and facilitate desired transformations.
Table 3: Essential Reagents and Materials for Microwave-Assisted Synthesis
| Item | Function & Rationale | Example Applications |
|---|---|---|
| Polar Solvents (High ε") | Efficiently absorb microwave energy, enabling rapid heating. Act as "molecular radiators." | DMSO, Methanol, Ethanol for rapid heating to high temperatures [29] [100]. |
| Ionic Liquids | Excellent microwave absorbers via ionic conduction; often used as solvents or catalysts. | Enabling solvent-free conditions or catalyzing reactions like cycloadditions [100]. |
| Solid Mineral Supports | In solvent-free synthesis, they absorb MW energy and provide a large surface area for reagents. | Alumina, silica, clay used for synthesis of heterocycles and other compounds [29]. |
| Dedicated Microwave Vials | Certified pressure vessels allow safe superheating of low-boiling solvents. | Enabling reactions in solvents like DCM at 180°C [104] [29]. |
| Molecular Radiators | "Microwave-absorbing" reagents/catalysts that heat efficiently even in low-absorbing solvents. | Polar or ionic catalysts that transfer heat locally to reactants [100]. |
Microwave-assisted synthesis has unequivocally transcended its role as a mere laboratory convenience. It is a powerful, enabling technology that validates novel chemical transformations by providing a unique energy input mechanism fundamentally different from traditional heating. From selectively exciting atomic antennae in catalysts to achieving unprecedented efficiency in the synthesis of complex pharmaceutical heterocycles and advanced materials, MAOS provides a direct and versatile pathway to innovation. For the research and drug development community, embracing this technology is not just an option for improving efficiency; it is a strategic imperative for accessing new chemical space and pioneering sustainable synthetic methodologies that align with the critical principles of green chemistry. The continued exploration of microwave energy promises to unlock further frontiers, solidifying its place as a cornerstone of modern chemical synthesis.
The principles of green chemistry provide a framework for developing chemical processes that minimize environmental impact and promote sustainability. Within this framework, microwave-assisted organic synthesis (MAOS) has emerged as a transformative technology that directly addresses two critical areas: the reduction of energy consumption and solvent waste generation [33]. For researchers, scientists, and drug development professionals, adopting microwave energy represents a convergence of technological innovation and environmental responsibility, enabling more efficient synthesis pathways while significantly lowering the ecological footprint of chemical research and production [105].
This technical guide examines the substantial economic and environmental advantages of microwave-assisted synthesis through quantitative data comparisons, detailed experimental protocols, and analytical frameworks tailored for implementation in research and industrial settings. The convergence of microwave technology with green chemistry principles is paving the way for more sustainable biomedical applications and pharmaceutical development [105] [106].
Quantitative Analysis of Energy and Waste Reduction
Microwave-assisted synthesis offers substantial improvements in process efficiency compared to conventional heating methods. The table below summarizes key quantitative benefits documented across various synthetic applications:
Table 1: Quantitative Comparison of Conventional vs. Microwave-Assisted Synthesis
| Parameter | Conventional Method | Microwave-Assisted Method | Improvement | Application Context |
|---|---|---|---|---|
| Reaction Time | Hours (1-24h) [33] | Minutes (5-10 min) [33] | Up to 98% reduction [33] | Heterocyclic synthesis (e.g., Quinolines) [33] |
| Energy Consumption | High (Prolonged heating) [23] | Low (Direct molecular heating) [23] | Significantly lower [23] | Nanomaterial fabrication [23] |
| Chemical Yield | Often moderate (e.g., ~75%) [106] | Frequently high (82-97%) [106] | Up to 29% increase [106] | 2-Aminobenzoxazoles synthesis [106] |
| Solvent Volume | Often substantial [33] | Reduced or solvent-free [33] | Major reduction [33] | Various organic transformations [33] |
The economic implications of these improvements are profound for drug development and industrial applications. Reduced reaction times translate directly to lower energy costs and increased laboratory or production throughput [33]. The dramatic acceleration of reaction kinetics—from hours to minutes—enables researchers to rapidly iterate and optimize synthetic pathways, potentially shortening development timelines for new pharmaceutical compounds [106].
From an environmental perspective, MAOS contributes to waste minimization at multiple levels. The technology's compatibility with solvent-free conditions or greener solvents like water directly reduces the use and generation of hazardous substances [33] [23]. Additionally, improved reaction selectivity and reduced byproduct formation decrease the environmental burden associated with waste disposal and purification processes [33]. The combination of these factors positions microwave-assisted synthesis as a cornerstone technology for achieving broader sustainability goals in chemical research and manufacturing [105].
Experimental Protocols for Microwave-Assisted Synthesis
Microwave-Assisted Synthesis of Metallic Nanoparticles
The synthesis of nanomaterials exemplifies the advantages of microwave-assisted approaches through precise size control and reduced energy intensity compared to conventional methods [23].
Detailed Methodology
- Step 1: Precursor Preparation - Dissolve metal salt precursors (e.g., HAuCl₄ for gold nanoparticles) in a suitable green solvent such as deionized water or plant extracts [23].
- Step 2: Reaction Mixture Formulation - Add reducing agents (e.g., plant-derived polyphenols) and stabilizing agents (e.g., biodegradable polymers) to the precursor solution under constant agitation [23].
- Step 3: Microwave Irradiation - Transfer the reaction mixture to dedicated microwave reactors. Apply microwave irradiation at a frequency of 2.45 GHz with precise temperature control (typically 80-150°C) for short durations (30 seconds to 10 minutes) [23].
- Step 4: Product Recovery - Recover nanoparticles through centrifugation or filtration after cooling the reaction mixture to room temperature [23].
Critical Parameters for Success
- Dielectric Properties: Select solvents and reagents with appropriate polarity to ensure efficient microwave energy absorption [33] [23].
- Temperature Control: Utilize reactors with integrated temperature monitoring to prevent localized overheating and ensure reproducible results [23].
- Scalability Considerations: Implement continuous-flow microwave systems to overcome batch size limitations for industrial-scale production [23].
Metal-Free Synthesis of 2-Aminobenzoxazoles
This protocol demonstrates the application of microwave energy in conjunction with metal-free catalysis and green solvents to achieve efficient C–N bond formation [106].
Detailed Methodology
- Step 1: Reaction Setup - Combine benzoxazole derivatives and amine partners in the ionic liquid 1-butylpyridinium iodide ([BPy]I) as both catalyst and reaction medium [106].
- Step 2: Oxidant Addition - Introduce tert-butyl hydroperoxide (TBHP) as an oxidant, with acetic acid as an additive to facilitate the reaction [106].
- Step 3: Microwave Irradiation - Subject the reaction mixture to microwave irradiation at room temperature for significantly reduced time (minutes versus hours under conventional heating) [106].
- Step 4: Product Isolation - Extract the desired 2-aminobenzoxazole products through simple phase separation, with the ionic liquid medium available for recovery and reuse [106].
Advantages Over Conventional Approaches
This method eliminates the need for toxic transition metal catalysts like copper or silver, which pose environmental and toxicity concerns [106]. The combination of microwave acceleration and ionic liquid media enables reaction efficiency at room temperature, further reducing energy requirements compared to conventional thermal approaches that often require elevated temperatures [106].
Workflow Visualization
The following diagram illustrates the typical workflow and decision-making process for implementing a microwave-assisted synthetic protocol, highlighting the convergence of sustainability and efficiency goals.
Diagram 1: Microwave Synthesis Workflow
This workflow emphasizes the critical decision points in implementing microwave-assisted synthesis. The initial assessment of microwave suitability depends primarily on the presence of polar reactants or solvents that can efficiently couple with microwave energy [33]. The selection of green solvents aligns with the principles of sustainable chemistry, while parameter optimization ensures maximum efficiency and minimal environmental impact [23] [106].
Research Reagent Solutions
The successful implementation of microwave-assisted green synthesis requires specific reagents and materials that align with sustainability principles while enabling efficient energy transfer.
Table 2: Essential Reagents for Microwave-Assisted Green Synthesis
| Reagent/Material | Function | Green Attributes | Example Applications |
|---|---|---|---|
| Ionic Liquids(e.g., 1-Butylpyridinium iodide) | Catalyst and reaction medium [106] | Negligible vapor pressure, recyclable, non-flammable [106] | Metal-free C–N bond formation [106] |
| PEG (Polyethylene Glycol) | Biocompatible solvent and phase-transfer catalyst [106] | Biodegradable, low toxicity, reusable [106] | Synthesis of tetrahydrocarbazoles and pyrazolines [106] |
| Dimethyl Carbonate (DMC) | Green methylating agent [106] | Non-toxic, biodegradable alternative to methyl halides [106] | O-Methylation of phenolic compounds [106] |
| Water | Green solvent [33] | Non-toxic, non-flammable, inexpensive [33] | Microwave-assisted heterocyclic synthesis [33] |
| Plant Extracts | Source of natural reducing/capping agents [23] | Renewable, biodegradable, non-hazardous [23] | Synthesis of metal nanoparticles [23] |
The reagents listed represent a shift toward bio-based and environmentally benign alternatives that maintain or even enhance synthetic efficiency while reducing environmental impact [23] [106]. Ionic liquids, for instance, serve dual roles as catalysts and solvents, enabling metal-free transformations under microwave irradiation [106]. Similarly, polyethylene glycol (PEG) provides a biodegradable medium that facilitates various heterocyclic syntheses with improved yields and reduced waste generation [106].
Microwave-assisted synthesis represents a technologically advanced and environmentally responsible approach that directly addresses the economic and ecological challenges facing modern chemical research and pharmaceutical development. The significant reductions in energy consumption through rapid, volumetric heating mechanisms, coupled with minimized solvent waste through green media alternatives, establish this methodology as a cornerstone of sustainable chemistry [33] [23].
The integration of microwave technology with green chemistry principles—including the use of bio-based solvents, metal-free catalysts, and renewable precursors—creates a powerful framework for advancing pharmaceutical synthesis and nanomaterial fabrication [23] [106]. As research continues to address scalability challenges and refine sustainability metrics, microwave-assisted approaches are poised to transform chemical manufacturing into a more efficient, economical, and environmentally benign enterprise [105] [23]. For researchers and drug development professionals, adopting these methodologies offers a pathway to simultaneously achieve scientific excellence and environmental stewardship.
Conclusion
Microwave-assisted synthesis stands as a cornerstone of modern sustainable chemistry, offering a validated pathway to drastically improve efficiency and reduce the environmental footprint of chemical research and production. By enabling faster reaction times, higher yields, and access to novel chemical space, this technology directly accelerates drug discovery, particularly in the synthesis of critical heterocyclic scaffolds. The successful application in nanomaterial fabrication and clean energy reactions further underscores its versatility. For biomedical research, the future lies in scaling these proven lab-scale benefits to industrial production, integrating microwave processes with renewable energy, and further exploring its potential to revolutionize the synthesis of complex active pharmaceutical ingredients (APIs) and biodegradable materials, ultimately fostering a more sustainable and efficient pipeline from bench to clinic.
References
- 1. Theory of Microwave Heating for Organic Synthesis [https://cem.com/cn/microwave-chemistry/theory]
- 2. Recent advances in microwave-assisted nanocarrier ... [https://www.sciencedirect.com/science/article/abs/pii/S1773224723006949]
- 3. New Microwave Technique Could Turn CO2 Into Fuel Far ... [https://scitechdaily.com/new-microwave-technique-could-turn-co2-into-fuel-far-more-efficiently/]
- 4. Microwaves for energy-efficient chemical reactions [https://www.u-tokyo.ac.jp/focus/en/press/z0508_00427.html]
- 5. Optimisation of a Microwave Synthesis of Silver ... [https://pubmed.ncbi.nlm.nih.gov/39065020/]
- 6. Advanced microwave synthesis strategies for innovative ... [https://www.sciencedirect.com/science/article/pii/S2468025724003194]
- 7. Getting Started with Microwave Synthesis [https://cem.com/cn/microwave-chemistry/getting-started]
- 8. Dielectric heating [https://en.wikipedia.org/wiki/Dielectric_heating]
- 9. Dielectric Heating - an overview [https://www.sciencedirect.com/topics/food-science/dielectric-heating]
- 10. Review Applications of microwave dielectric heating in ... [https://www.sciencedirect.com/science/article/abs/pii/S0020169306001034]
- 11. Synthesis of Medicinally Privileged Heterocycles through ... [https://pubmed.ncbi.nlm.nih.gov/28240166/]
- 12. Microwave dielectric heating in modern organic synthesis ... [https://www.intechopen.com/chapters/17021]
- 13. Dielectric heating [https://grokipedia.com/page/Dielectric_heating]
- 14. Biological Effects of RF Fields [https://mriquestions.com/rf-biological-effects.html]
- 15. Effect of microwave vacuum puffing conditions on the ... [https://www.nature.com/articles/s41598-025-06253-w]
- 16. Development of a Dielectric Heating System for Selective ... [https://www.mdpi.com/2076-3417/15/10/5466]
- 17. Dielectric properties, processes, kinetics, and mechanism ... [https://www.sciencedirect.com/science/article/abs/pii/S0009250925000636]
- 18. Microwave-assisted extraction: Recent advances in ... [https://www.sciencedirect.com/science/article/pii/S2772826925000677]
- 19. Comparison of Conventional and Microwave-assisted ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4007255/]
- 20. Benefits and applications of microwave-assisted synthesis of ... [https://pubs.rsc.org/en/content/articlehtml/2020/ra/d0ra01378a]
- 21. RF Energy Industrial I Microwave-Driven Chemical Reactions [https://rfhic.com/industries-rf-energy/industrial/revolutionizing-chemical-synthesis-with-microwave-technology/]
- 22. Comparative study of microwave-assisted versus ... [https://www.sciencedirect.com/science/article/pii/S2588913323000157]
- 23. Microwave-assisted synthesis of nanomaterials: a green ... [https://pubs.rsc.org/en/content/articlehtml/2025/su/d5su00584a]
- 24. Microwave chemistry, recent advancements, and eco ... [https://www.sciencedirect.com/science/article/abs/pii/S2588842020300055]
- 25. Microwave technology for energy-efficient processing of ... [https://www.sciencedirect.com/science/article/abs/pii/S0306261904001011]
- 26. Microwave Energy Usage and Power Saving Tips [https://utilitiesnow.com/blog/microwave-energy-usage-and-power-saving-tips/]
- 27. Energy Conservation Standards for Microwave Ovens [https://www.federalregister.gov/documents/2023/06/20/2023-12958/energy-conservation-program-energy-conservation-standards-for-microwave-ovens]
- 28. Microwave Abatement: A Transformative Technology for ... [https://www.mks.com/microwave-based-abatement-for-net-zero-solutions]
- 29. Getting Started with Microwave Synthesis [https://cem.com/microwave-chemistry/getting-started]
- 30. Organic synthesis: the art and science of replicating the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3899854/]
- 31. Organic Synthesis: From Concept to Creation [https://www.openaccessjournals.com/articles/organic-synthesis-from-concept-to-creation-17883.html]
- 32. Microwave Irradiation: Alternative Heating Process for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8541360/]
- 33. Microwave-Assisted Organic Synthesis: An Eco-Friendly ... [https://www.mdpi.com/1424-8247/18/11/1692]
- 34. The History of Organic Chemicals [https://www.boronmolecular.com/the-history-of-organic-chemicals/]
- 35. Theory of Microwave Heating for Organic Synthesis [https://cem.com/fr/microwave-chemistry/theory]
- 36. a green chemistry perspective and sustainability assessment [https://pubs.rsc.org/en/content/articlelanding/2025/su/d5su00584a]
- 37. Theory of Microwave Heating for Organic Synthesis [https://cem.com/microwave-chemistry/theory]
- 38. Microwaves under pressure for the continuous production ... [https://www.sciencedirect.com/science/article/abs/pii/S0920586114007895]
- 39. Atmospheric Pressure Microwave Assisted Heterogeneous ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6149456/]
- 40. Microwaves for energy-efficient chemical reactions [https://www.eurekalert.org/news-releases/1101470]
- 41. Exploring the versatile roles of microwave energy in ... [https://www.sciencedirect.com/science/article/pii/S0016236125014097?dgcid=rss_sd_all]
- 42. Microwaves in Organic Synthetic Chemistry- A Greener ... [https://www.ajgreenchem.com/article_178730.html]
- 43. Solvent-free microwave reactions towards significant ... [https://www.sciencedirect.com/science/article/pii/S2773223124000190]
- 44. Solvent Choice for Microwave Synthesis [https://cem.com/cn/microwave-chemistry/solvent-choice]
- 45. Solvents for a Safer, Sustainable Lab [https://ehs.usc.edu/research/lab/sustainability-in-research-labs-at-usc/solvents-for-a-safer-sustainable-lab/]
- 46. Microwave-assisted extraction of functional compounds from ... [https://bioresources.cnr.ncsu.edu/resources/microwave-assisted-extraction-of-functional-compounds-from-plants-a-review/]
- 47. Recent advances in non-conventional synthesis of N ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra06028a?page=search]
- 48. Recent developments on microwave-assisted organic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10628940/]
- 49. Coumarins as Versatile Scaffolds: Innovative Synthetic ... [https://www.sciencedirect.com/science/article/abs/pii/S0022286025030704]
- 50. and ultrasound-assisted green synthesis of coumarin ... [https://www.sciencedirect.com/science/article/pii/S1878535221006699]
- 51. A comprehensive review and recent advances on isatin-based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12415693/]
- 52. A review on quinolines: New green synthetic methods and ... [https://www.sciencedirect.com/science/article/pii/S092809872500096X]
- 53. Synthesis of quinoline mimics via C–H bond ... [https://pubs.rsc.org/en/content/articlelanding/2025/ob/d4ob02013h]
- 54. Design, eco-friendly synthesis, and molecular docking studies ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra04722f]
- 55. -assisted Microwave of synthesis : a nanomaterials chemistry... green [https://pubs.rsc.org/En/content/articlelanding/2025/su/d5su00584a]
- 56. Microwave assisted green synthesis of silver nanoparticles ... [https://www.nature.com/articles/s41598-025-21112-4]
- 57. Microwave-assisted green synthesis of silver nanoparticles ... [https://www.sciencedirect.com/science/article/pii/S2666790823000654]
- 58. Microwave-assisted synthesis and formation mechanism of ... [https://www.sciencedirect.com/science/article/pii/S2666893922000366]
- 59. Recent advances on the characterization of nanoparticles ... [https://www.sciencedirect.com/science/article/pii/S0165993616300103]
- 60. Nanomaterials | Environment, Health and Safety - Cornell EHS [https://ehs.cornell.edu/research-safety/chemical-safety/specific-chemical-hazards/nanomaterials]
- 61. Toxicity and Environmental Risks of Nanomaterials [https://pmc.ncbi.nlm.nih.gov/articles/PMC2844666/]
- 62. Microwave Technology: Powering the Green Industrial ... [https://www.mks.com/microwave-green-processing-solutions]
- 63. Microwave-Assisted Organic Synthesis: A Green Chemistry ... [https://www.ijrpas.com/HTMLPaper.aspx?Journal=International%20Journal%20of%20Research%20in%20Pharmacy%20and%20Allied%20Science;PID=2025-4-6-5]
- 64. Microwave-assisted carbon capture and conversion [https://www.sciencedirect.com/science/article/abs/pii/S2213343725045816]
- 65. A novel microwave heating-assisted reactor for industrial ... [https://www.sciencedirect.com/science/article/abs/pii/S0960148125011152]
- 66. Hydrogen fuel production from CO2/CH4 mixture using a ... [https://www.nature.com/articles/s41598-025-21309-7]
- 67. Clean hydrogen in minutes: Microwaves deliver ... [https://www.sciencedaily.com/releases/2025/01/250121125907.htm]
- 68. Green Metric for Microwave-Assisted Sample Preparation ... [https://www.sciencedirect.com/science/article/pii/S2772582025000683]
- 69. Systematic Optimization of High-Throughput Microwave ... [https://www.sciencedirect.com/science/article/pii/S2667056925001300]
- 70. Scalable Preparation of High-Quality Microwave-Assisted ... [https://pubmed.ncbi.nlm.nih.gov/40641258/]
- 71. Microwave Chemical Reactor Market Outlook 2025-2032 [https://www.intelmarketresearch.com/microwave-chemical-reactor-market-3507]
- 72. 5 Top Bench- Scale | H.E.L Group Strategies [https://helgroup.com/blog/5-top-bench-scale-strategies/]
- 73. Scale-up influences and definitions of scale-up 'success' [https://pmc.ncbi.nlm.nih.gov/articles/PMC11812037/]
- 74. Industrial Scales 2025-2033 Analysis: Trends, Competitor ... [https://www.archivemarketresearch.com/reports/industrial-scales-177549]
- 75. in Scale : Best Tips to Cut Cost Industry [https://www.numberanalytics.com/blog/industry-scale-tips-cut-cost]
- 76. Solvent-free microwave organic synthesis as an efficient ... [https://www.sciencedirect.com/science/article/pii/S1631074804000281]
- 77. Influence of Polarity and Activation Energy in Microwave– ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4522181/]
- 78. Progress and future outlook towards a safe ... [https://www.nature.com/articles/s42004-025-01785-8]
- 79. Emerging Microwave Chemistry – Celebrating Work from ... [https://www.frontiersin.org/research-topics/60368/emerging-microwave-chemistry---celebrating-work-from-the-5th-global-congress-on-microwave-energy-applicationsundefined]
- 80. Heating of biomass in microwave household oven [https://www.sciencedirect.com/science/article/abs/pii/S0360544220325792]
- 81. Experimental Validation of Realistic Measurement Setup ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11435978/]
- 82. Rapid, automated determination of reaction models and ... [https://www.sciencedirect.com/science/article/pii/S1385894720331454]
- 83. Accelerated Chemical Reaction Optimization Using Multi-Task ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10214532/]
- 84. Rapid planning and analysis of high-throughput ... [https://www.nature.com/articles/s41467-023-39531-0]
- 85. Reaction Optimization for Greener Chemistry with a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9738638/]
- 86. High-throughput experimentation for photoredox cross ... [https://pubs.rsc.org/en/content/articlelanding/2025/re/d5re00241a]
- 87. Microwave chemistry [https://grokipedia.com/page/Microwave_chemistry]
- 88. Microwave-assisted synthesis in the pharmaceutical ... [https://www.ddw-online.com/microwave-assisted-synthesis-in-the-pharmaceutical-industry-a-current-perspective-and-future-prospects-1563-200608/]
- 89. Hydrogen in Minutes: The Microwave Innovation Changing ... [https://scitechdaily.com/hydrogen-in-minutes-the-microwave-innovation-changing-clean-energy/]
- 90. Hydrogen produced in minutes: Microwave technology ... [https://interestingengineering.com/energy/microwave-assisted-hydrogen-production-method]
- 91. A new type of power energy for accelerating chemical ... [https://www.nature.com/articles/srep25149]
- 92. Prospects and Challenges of Microwave-Combined ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7913569/]
- 93. Microwave-Assisted Organic Synthesis: An Eco-Friendly ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12655639/]
- 94. Comparison of Heating Efficiency by Microwave and [https://web.njit.edu/~mitra/green_chemistry/EXP_1.htm]
- 95. Comparison of Microwave and Conduction Heating ... [https://www.csbio.com/blog/conductionandmicrowave]
- 96. Microwave heating temperatures and pharmaceutical ... [https://www.sciencedirect.com/science/article/abs/pii/S2214785319334364]
- 97. Why Direct Heating Trays are important in Pharmaceutical ... [https://powdersystems.com/2024/04/why-direct-heating-trays-important-in-pharmaceutical-manufacturing/]
- 98. Green metrics and green analytical applications [https://www.sciencedirect.com/science/article/pii/S2772577424000685]
- 99. Green metrics for analytical methodologies [https://www.sciencedirect.com/science/article/abs/pii/S0165993624000803]
- 100. Microwave-Assisted Synthesis: Can Transition Metal ... [https://www.mdpi.com/1420-3049/27/13/4249]
- 101. Recent advances in microwave-assisted synthesis of triazoles ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d4ra06886f]
- 102. Microwave-assisted synthesis of superabsorbent agar ... [https://www.sciencedirect.com/science/article/pii/S2590123025006309]
- 103. A new microwave-assisted synthesis route to produce ... [https://www.sciencedirect.com/science/article/pii/S1385894725017875]
- 104. Initiator+ Microwave Synthesis [https://www.biotage.com/initiator-microwave-synthesis]
- 105. Sustainable chemistry approaches for biomedical ... [https://www.sciencedirect.com/science/article/pii/S2950477525000205]
- 106. metal-free, bio-based, and microwave-assisted methods [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2025.1656935/full]