Ionic Liquids vs. Supercritical Fluids: A Comparative Guide for Advanced Biomedical Applications
This article provides a comprehensive comparison of ionic liquids and supercritical fluids, two cornerstone technologies in green chemistry, with a focused lens on drug development.
Ionic Liquids vs. Supercritical Fluids: A Comparative Guide for Advanced Biomedical Applications
Abstract
This article provides a comprehensive comparison of ionic liquids and supercritical fluids, two cornerstone technologies in green chemistry, with a focused lens on drug development. It explores their fundamental properties, including the tunable nature of ionic liquids and the density-dependent solvation of supercritical CO2. The content details key methodological applications in pharmaceutical processing, such as drug micronization and energy storage, while addressing practical challenges like solvent purity and prediction of solubility. By synthesizing performance data and current research trends, this analysis offers validation for researchers and scientists selecting the optimal solvent system for specific biomedical and clinical objectives, from nanoparticle design to next-generation drug delivery.
Understanding the Core Properties: From Tunable Salts to Dense Gases
Ionic liquids (ILs) have emerged as a transformative class of materials in chemical research and industrial applications, distinguished by their unique status as organic salts that remain liquid below 100°C. Their evolution from academic curiosities to engineered "designer solvents" represents a paradigm shift in materials science, driven by their modular cation-anion combinations that enable precise tuning of physicochemical properties for specific applications [1]. This design flexibility has propelled their development through four distinct generations, each expanding their functional capabilities while addressing emerging challenges in sustainability and biocompatibility.
The defining characteristic of ILs lies in their structural asymmetry, which disrupts crystal packing and results in low lattice energy, maintaining the liquid state across wide temperature ranges [2]. This fundamental property, combined with their negligible vapor pressure, high thermal stability, and tunable solvation dynamics, positions ILs as superior alternatives to conventional volatile organic solvents across diverse applications from pharmaceuticals to energy storage [1] [3].
When contextualized within broader solvent research, ILs present a compelling contrast to supercritical fluids (SCFs), particularly supercritical carbon dioxide (scCO₂). While SCFs offer tunable density-dependent solvent power through pressure and temperature manipulation [4], ILs provide molecular-level design versatility through cation-anion selection. This distinction establishes complementary roles for these advanced solvent systems, with emerging research even exploring their synergistic combinations, such as using scCO₂ to impregnate ILs into porous supports for enhanced material processing [5].
The Four-Generation Evolution of Ionic Liquids
The development of ionic liquids has progressed through four distinct generations, each marked by significant advancements in functionality and application scope, as systematically detailed in recent comprehensive reviews [1].
Table 1: The Four-Generation Evolution of Ionic Liquids
| Generation | Time Period | Key Characteristics | Primary Applications | Example Systems |
|---|---|---|---|---|
| First Generation | Late 20th Century | Focus on physical properties; green solvent alternatives | Electrochemistry, synthetic chemistry | Chloroaluminate ILs, imidazolium salts |
| Second Generation | 1990s-2000s | Task-specific functionality through ion selection | Catalysis, separation processes, materials synthesis | Functionalized imidazolium, pyrrolidinium ILs |
| Third Generation | 2000s-2010s | Bio-derived components, reduced toxicity, biocompatibility | Pharmaceutical delivery, biomedical applications | Choline-based ILs, API-ILs, glycerol-derived ILs [6] |
| Fourth Generation | 2010s-Present | Sustainability, biodegradability, multifunctionality | Green chemistry, energy storage, precision medicine | Biodegradable ILs, SILs for batteries [3] |
First Generation: Foundation and Physical Properties
The first generation of ILs established their fundamental physical properties and demonstrated their potential as green solvent alternatives to volatile organic compounds. Research during this period focused primarily on their unique physicochemical characteristics, including low vapor pressure, non-flammability, and wide liquid ranges. These early ILs, particularly chloroaluminate systems, provided proof-of-concept for their utility in electrochemistry and as reaction media, though their sensitivity to moisture and limited commercial availability restricted widespread adoption [1].
Second Generation: Task-Specific Functionality
Second-generation ILs represented a significant conceptual advance through the intentional design of task-specific ionic liquids. Researchers began systematically engineering cation-anion pairs to achieve desired properties for particular applications, expanding beyond the limited scope of early systems. This generation saw the development of ILs with tailored acidity/basicity, specific coordination properties, and enhanced catalytic capabilities, enabling their use in specialized separation processes, catalytic transformations, and advanced materials synthesis [1].
Third Generation: Biocompatibility and Pharmaceutical Applications
The third generation marked a pivotal shift toward biocompatibility and pharmaceutical applications, addressing toxicity concerns associated with earlier ILs. This era witnessed the development of ILs derived from biological precursors such as choline, amino acids, and glycerol [6]. A particularly significant innovation was the creation of Active Pharmaceutical Ingredient Ionic Liquids (API-ILs), which convert drug molecules directly into ionic forms to markedly improve solubility, bioavailability, and delivery efficiency while integrating the active agent and delivery vector into a single ionic entity [7]. These advances established ILs as versatile platforms in biomedical sciences, with demonstrated efficacy in drug delivery, biopharmaceutical stabilization, and transdermal transport enhancement [2].
Fourth Generation: Sustainability and Multifunctionality
Current fourth-generation ILs emphasize sustainability, biodegradability, and multifunctionality while maintaining high performance across advanced applications. This generation includes solvate ionic liquids (SILs)—formed through chelation of cationic species by oligoethers paired with charge-diffuse anions—which show exceptional promise for next-generation energy storage systems including lithium-ion, lithium-sulfur, and all-solid-state lithium batteries [3]. Concurrently, research has focused on bio-based ILs derived from renewable feedstocks such as glycerol, designed to combine sustainability with functionality while addressing environmental concerns associated with traditional ILs [6]. These fourth-generation systems represent the cutting edge of IL technology, positioning them as key enablers of sustainable technological advancement.
Properties and Comparative Analysis with Supercritical Fluids
The unique properties of ionic liquids establish them as complementary yet distinct from supercritical fluids in advanced solvent applications. Understanding these differences enables researchers to select appropriate systems for specific technological challenges.
Fundamental Properties of Ionic Liquids
Ionic liquids possess an exceptional combination of physical properties that underpins their utility across diverse applications:
- Tunable Solvation Power: Through rational cation-anion selection, ILs can be engineered to dissolve a wide range of compounds from hydrophobic drugs to hydrophilic biomolecules [7] [2]
- Negligible Vapor Pressure: This property eliminates inhalation exposure risks and enables high-temperature applications without solvent loss [1]
- High Thermal Stability: Many ILs remain stable at temperatures exceeding 300°C, facilitating high-temperature processes [6]
- Wide Electrochemical Windows: Certain IL classes offer potential ranges up to 6V, enabling electrodeposition of reactive elements [3]
- Designer Physicochemical Properties: Density, viscosity, hydrophobicity, and melting point can be systematically tuned through structural modifications [6]
Property Comparison: Ionic Liquids vs. Supercritical Fluids
Table 2: Comparative Properties of Ionic Liquids and Supercritical Fluids
| Property | Ionic Liquids | Supercritical Fluids (scCO₂) | Technological Implications |
|---|---|---|---|
| Tunability Mechanism | Molecular structure via cation-anion selection [1] | Density via pressure/temperature manipulation [4] | ILs: Synthetic design; SCFs: Process control |
| Vapor Pressure | Negligible [1] | Critical pressure: 72.9 bar for CO₂ [4] | ILs: Minimal solvent loss; SCFs: Energy-intensive containment |
| Operating Temperature | Typically <100°C melting point; stable to >300°C [6] | >31.3°C for CO₂ [4] | ILs: Broader liquid range; SCFs: Moderate temperatures required |
| Diffusivity | Moderate (10⁻¹¹-10⁻¹² m²/s) [7] | High (10⁻⁸-10⁻⁹ m²/s) [4] | SCFs superior for mass transfer-limited processes |
| Viscosity | Moderate to high (0.1-189 Pa·s) [6] | Very low (~10⁻⁵ Pa·s) [4] | SCFs offer better flow characteristics in porous media |
| Solvent Power | Wide range through molecular design [7] | Density-dependent [4] | ILs: Broader molecular compatibility; SCFs: Extraction selectivity |
| Environmental Impact | Varies; 4th gen emphasizes biodegradability [6] | scCO₂ considered environmentally benign [4] | Both offer green alternatives to organic solvents |
Synergistic Applications
Emerging research explores synergistic applications combining ILs and SCFs, leveraging their complementary advantages. For instance, supercritical fluid deposition using scCO₂ enables uniform impregnation of ILs into porous supports, with recent modeling advances using the ɛ*-modified Sanchez-Lacombe equation of state achieving improved prediction accuracy for IL solubility in CO₂ + co-solvent systems (average logarithmic AARD of 11.0%) [5]. This hybrid approach marries the precision tunability of ILs with the superior penetration and mass transfer properties of SCFs.
Experimental Protocols and Methodologies
Synthesis of Glycerol-Derived Ionic Liquids
The development of bio-based ILs represents a significant advance in sustainable solvent design. The following protocol outlines the synthesis of glycerol-derived ILs, demonstrating the practical methodology for creating these fourth-generation solvents:
Table 3: Research Reagent Solutions for Glycerol-Derived IL Synthesis
| Reagent/Material | Function | Specifications | Handling Considerations |
|---|---|---|---|
| Glycidyl ethers or Epichlorohydrin | Starting material | Bio-derived platform molecules | Commercial availability varies; epichlorohydrin more accessible [6] |
| Triethylamine | Ammonium cation source | Smallest liquid amine for practical handling | 50% excess recommended for optimal yield [6] |
| Brønsted acid (HCl) | Epoxide activation | Shifts equilibrium toward product formation | Essential for reaction progression; controlled addition critical [6] |
| Anion exchange resins | Anion metathesis | Enables variation of IL properties | For generating ILs with different anions (triflate, bistriflimide, etc.) [6] |
Experimental Workflow:
- Reaction Setup: Charge reactor with glycidyl ether or epichlorohydrin in molar ratio 1:1.5 with triethylamine [6]
- Acid Addition: Slowly add Brønsted acid (HCl) with controlled addition rate to minimize byproduct formation [6]
- Temperature Optimization: React at 80°C for 48 hours—this combination provides optimal yield (82%) while minimizing byproducts [6]
- Byproduct Management: Monitor formation of 1-chloro-3-alkoxypropan-2-ol and triethylammonium chloride byproducts via ¹H NMR [6]
- Anion Exchange: For non-chloride ILs, perform subsequent anion metathesis using appropriate salt solutions or ion-exchange resins [6]
- Purification: Remove volatile components under vacuum with repeated washing and drying to achieve high-purity ILs [6]
Characterization Methods:
- ¹H NMR: Quantify product yield and byproduct formation [6]
- Thermal Analysis (TGA/DSC): Determine thermal stability (up to 672K for glycerol-derived ILs) and phase behavior [6]
- Rheology: Measure viscosity across temperature ranges (0.3-189 Pa·s for glycerol-derived ILs) [6]
- Density Measurement: Characterize density range (1.03-1.40 g·cm⁻³ for glycerol-derived ILs) [6]
Formulation of IL-Based Transdermal Drug Delivery Systems
The development of IL-based transdermal delivery systems for biopharmaceuticals represents a cutting-edge application of third- and fourth-generation ILs:
Nanocarrier Integration Protocols:
IL-Ethosomes (ETs) Formulation:
- Combine dimyristoyl-phosphatidylcholine IL with phospholipids and ethanol [2]
- Hydrate with buffer containing therapeutic agent (e.g., insulin) under controlled temperature [2]
- Process through extrusion to achieve uniform vesicle size (100-200 nm) [2]
- Achieve ~99% encapsulation efficiency with month-long stability at both 4°C and 25°C [2]
IL-in-Oil Micro-/Nanoemulsion Preparation:
Performance Evaluation:
- Skin Permeation Studies: Using Franz diffusion cells with excised skin, demonstrating two-fold increase in skin flux compared with conventional vesicles [2]
- Stability Assessment: Monitor drug encapsulation efficiency and particle size over extended storage periods [2]
- Biological Activity: Verify maintained bioactivity of delivered biopharmaceuticals (e.g., insulin efficacy in diabetic models) [2]
Advanced Applications in Drug Delivery and Beyond
Pharmaceutical and Biomedical Applications
Ionic liquids have revolutionized pharmaceutical approaches through multiple enhancement mechanisms:
- Solubility Enhancement: ILs dramatically improve aqueous solubility of poorly soluble drugs (BCS Class II/IV), addressing a fundamental limitation in pharmaceutical bioavailability [7]
- Transdermal Delivery: IL-based systems enable non-invasive delivery of biopharmaceuticals including proteins, peptides, and nucleic acids by transiently fluidizing stratum corneum lipids without lasting damage [2]
- Stabilization of Biologics: Cholinium ILs elevate melting points of therapeutic proteins like insulin by ≈13°C and monoclonal antibodies by >20°C, markedly delaying unfolding and aggregation [2]
- Active Pharmaceutical Ingredient ILs (API-ILs): Conversion of drug molecules directly into ionic forms integrates active agent and delivery vector into a single ionic entity, improving bioavailability and enabling novel administration routes [7]
Energy Storage and Materials Applications
The unique properties of ILs have enabled advanced energy storage technologies:
- Solvate Ionic Liquids (SILs): Formed through chelation of cations by oligoethers (typically triglyme or tetraglyme) with charge-diffuse anions, SILs exhibit exceptional electrochemical stability for next-generation energy storage [3]
- Battery Applications: SILs demonstrate superior performance in lithium-ion, lithium-sulfur, lithium-air, lithium-redox, and all-solid-state lithium batteries due to high thermal stability, wide electrochemical windows, and tailored ion transport properties [3]
- Advanced Materials Processing: ILs serve as versatile media for polymer synthesis, nanoparticle formation, and material extraction, particularly sustainable processing of biopolymers like cellulose [1]
The evolution of ionic liquids through four generations has transformed them from specialized solvents to versatile platforms for technological innovation. Future development will focus on several key areas:
- AI-Driven Design: Integration of artificial intelligence and computational modeling to accelerate rational design of ILs for specific applications [7]
- Sustainable Feedstocks: Expanded use of bio-derived precursors and emphasis on biodegradable IL structures to enhance environmental compatibility [6]
- Multifunctional Systems: Development of ILs that combine solvent, catalytic, and active pharmaceutical properties in unified platforms [7]
- Hybrid Materials: Creation of IL-based composites and nanostructured materials for enhanced performance in energy, biomedical, and environmental applications [3]
- Clinical Translation: Addressing remaining challenges in biosafety, scalable manufacturing, and regulatory harmonization to enable widespread pharmaceutical adoption [7]
In conclusion, ionic liquids have firmly established themselves as transformative "designer solvents" whose evolution continues to expand their impact across scientific disciplines. Their modular nature provides unparalleled tunability compared to other advanced solvent systems like supercritical fluids, positioning them as enabling technologies for next-generation sustainable processes, precision therapeutics, and advanced energy storage systems. As research advances, the integration of ILs with emerging technologies promises to unlock unprecedented capabilities in molecular design and functional material development.
A supercritical fluid (SCF) represents a unique quasi-state of matter achieved when a substance is heated and compressed above its critical temperature (Tc) and critical pressure (Pc). At this critical point, the distinction between the liquid and gas phases disappears, resulting in a homogeneous fluid that exhibits hybrid properties intermediate between those of liquids and gases [8] [4]. This phenomenon occurs because, at the critical point, the densities of the liquid and vapor phases become equal, and the meniscus separating them vanishes [9] [4]. The phase transition to a supercritical state is not dramatic; rather, it is a gradual change, with no definite boundaries existing below and to the left of the supercritical region on a phase diagram [4].
The supercritical state is characterized by its tunable physicochemical properties. A key feature of SCFs is that their density—and consequently their solvent power—can be continuously adjusted by modifying the system's pressure and temperature without undergoing a phase transition [4]. This tunability, combined with properties such as high diffusivity and low viscosity, makes SCFs exceptionally versatile solvents and reaction media. In the context of ongoing research comparing ionic liquids and supercritical fluids, SCFs offer a distinct approach to green chemistry and sustainable processing, providing an alternative to conventional organic solvents and facilitating the development of more efficient, environmentally friendly industrial processes [10] [4].
Fundamental Properties and Phase Behavior
The Critical Point and Phase Diagrams
The journey to a supercritical state begins with understanding its defining boundary: the critical point. On a standard phase diagram, the critical point is located at the terminus of the liquid-gas equilibrium curve. Beyond this point, the conventional meniscus between liquid and gas phases no longer forms, and the substance enters the supercritical region [9] [4]. Mathematically, the critical point is identified where the first and second partial derivatives of pressure with respect to volume at constant temperature are both zero [4].
The triple point, another crucial feature of phase diagrams, represents the unique set of conditions (temperature and pressure) at which the solid, liquid, and gaseous phases of a substance coexist in equilibrium. It is distinct from the critical point, which relates solely to the liquid-gas boundary [9] [4]. The path to a supercritical fluid involves applying heat to a liquid in a closed container. As temperature increases, the liquid expands, and evaporation occurs, increasing the vapor pressure inside the container. Eventually, a temperature is reached where the densities of the liquid and vapor phases become identical, and the interface between them vanishes—this is the critical point [4].
Characteristic Properties of Supercritical Fluids
Supercritical fluids possess a unique combination of properties that make them technologically valuable, as summarized in Table 1.
Table 1: Characteristic Properties of Supercritical Fluids Compared to Gases and Liquids
| Property | Gas | Supercritical Fluid | Liquid |
|---|---|---|---|
| Density (g/cm³) | ~0.001 | 0.1 - 1.0 [4] | ~1.0 |
| Diffusivity (cm²/s) | ~0.1 | 10⁻³ - 10⁻⁴ [4] | < 10⁻⁵ |
| Viscosity (cP) | ~0.01 | 0.01 - 0.1 [4] | ~1.0 |
This combination of liquid-like density and gas-like transport properties underpins the utility of SCFs. The liquid-like density is responsible for the high solvating power of SCFs, allowing them to dissolve a wide range of compounds. Simultaneously, their gas-like low viscosity and high diffusivity contribute to excellent mass transfer characteristics, superior penetration into porous materials, and favorable hydrodynamic properties [10] [4]. Furthermore, SCFs exhibit low surface tension, enabling them to wet surfaces and penetrate solid matrices very effectively [4].
Common Supercritical Fluids and Their Parameters
Various substances can be brought to a supercritical state. Their critical properties determine the required operating conditions and potential applications. Table 2 lists several common supercritical fluids.
Table 2: Critical Properties of Common Supercritical Fluids [4]
| Fluid | Critical Temperature, Tc (°C) | Critical Pressure, Pc (bar) | Critical Density, δc (g/cm³) |
|---|---|---|---|
| Xenon (Xe) | 16.6 | 58.4 | 1.10 |
| Trifluoromethane (CHF₃) | 25.9 | 46.9 | 0.52 |
| Carbon Dioxide (CO₂) | 31.3 | 72.9 | 0.47 |
| Nitrous Oxide (N₂O) | 36.5 | 72.5 | 0.45 |
| Sulfur Hexafluoride (SF₆) | 45.5 | 37.1 | 0.74 |
| Ammonia (NH₃) | 132.5 | 112.5 | 0.24 |
| Pentane | 196.6 | 33.3 | 0.23 |
Among these, supercritical carbon dioxide (scCO₂) is the most widely used. Its relatively low critical temperature (31.3°C) and manageable critical pressure (72.9 bar) make it suitable for processing heat-sensitive compounds, including many pharmaceuticals and biologics [10] [4]. As a non-toxic, non-flammable, and readily available solvent, scCO₂ aligns perfectly with the principles of green chemistry, offering a sustainable alternative to conventional organic solvents [11] [10] [4].
Supercritical Fluids in Pharmaceutical Research and Drug Development
The unique properties of supercritical fluids have positioned them as powerful tools in pharmaceutical research, particularly for addressing challenges related to drug solubility, bioavailability, and particle engineering.
Enhancing Drug Solubility and Bioavailability
A significant hurdle in drug development is the poor aqueous solubility of many active pharmaceutical ingredients (APIs), which limits their absorption and bioavailability in the body [10]. Supercritical fluid technology offers a green and efficient strategy to overcome this. By controlling the particle size and morphology of drugs, SCF processes can significantly enhance their dissolution rates and permeability [10]. For instance, reducing drug particles to the micron or nanometer scale increases their surface area-to-volume ratio, leading to faster dissolution and improved therapeutic efficacy [10].
Technologies like Super-stable Homogeneous Intermix Formulating Technology (SHIFT) and Super-Table Pure-Nanomedicine Formulation Technology (SPFT) have been developed using supercritical fluids to disperse and micronize insoluble drugs. These methods overcome the limitations of traditional techniques such as milling and crystallization, which often cause thermal degradation or result in uneven particle size and organic solvent residues [10]. In one application, SHIFT was used to create a homogeneous dispersion of Indocyanine Green (ICG), an FDA-approved tracer, in Lipiodol (an oil-based embolic agent). This formulation demonstrated superior stability and photothermal conversion efficiency compared to a crude mixture, highlighting its potential for guiding precise surgical resection of tumors [10].
Advanced Particle Engineering Techniques
Several sophisticated SCF-based processes are employed for drug particle formation and encapsulation, each with distinct mechanisms and applications.
Rapid Expansion of Supercritical Solution (RESS): This method leverages the pressure-dependent solvation power of an SCF. The API is dissolved in the supercritical fluid, and the solution is then rapidly expanded through a nozzle into a low-pressure chamber. This sudden drop in pressure causes extreme supersaturation, leading to the precipitation of the solute as uniform, fine particles [10]. RESS is particularly suitable for compounds with reasonable solubility in scCO₂.
Supercritical Anti-Solvent (SAS): For substances insoluble in scCO₂, the SAS process is highly effective. The API is first dissolved in a conventional organic solvent. This solution is then sprayed into a vessel filled with scCO₂, which acts as an anti-solvent. The scCO₂ is miscible with the organic solvent but not with the solute, causing the API to precipitate as fine particles. The scCO₂ also strips away the organic solvent, resulting in products with minimal solvent residue [10]. This technique is widely used to produce drug-loaded nanoparticles and microparticles.
Precipitation from Gas Saturated Solution (PGSS): In PGSS, an SCF (commonly scCO₂) is dissolved into a liquid or melted substrate (e.g., a polymer or drug) to form a gas-saturated solution. This mixture is then depressurized through a nozzle. The rapid release of the dissolved gas causes a sharp temperature drop ( Joule-Thomson effect) and rapid precipitation of the substrate, forming composite particles or powders [10]. PGSS is advantageous for processing viscous materials and creating solid dispersions.
These particle formation technologies are pivotal in developing advanced drug delivery systems (DDSs), including those for targeted therapy, controlled release, and improved stability of thermolabile compounds [10] [12].
Experimental Methodologies for Supercritical Fluid Research
Research into supercritical fluids and their applications requires specialized equipment and protocols to handle high-pressure conditions and accurately measure material properties.
Core Experimental Setups
A fundamental tool in SCF research is the high-pressure variable-volume view-cell. These custom-designed systems, capable of withstanding pressures up to 60 MPa and temperatures up to 180°C, are equipped with movable pistons to vary internal volume (and thus pressure) and sapphire windows for visual observation [8]. Researchers use them to determine phase boundaries—the specific pressure-temperature conditions where miscibility and demixing occur for polymer solutions or fluid mixtures. This is critical for applications like polymer synthesis, purification, and particle formation [8]. Phase state assessment can be done visually or, more precisely, by measuring the variation in transmitted light intensity as a function of temperature and pressure [8].
Advanced versions incorporate a magnetically coupled mixer and a pair of sapphire windows for laser-light scattering experiments. This setup allows investigation of the dynamics of phase separation, determining whether the process follows a nucleation and growth (NG) or spinodal decomposition (SD) mechanism by analyzing the angular distribution and time evolution of scattered light intensities [8]. This has direct practical implications: NG at low polymer concentrations leads to particle formation, while SD at the critical concentration can yield interconnected porous networks or foams [8].
Assessment of Thermal, Mechanical, and Transport Properties
Understanding how SCFs interact with materials requires probing changes in fundamental properties.
Thermal Transitions: High-Pressure Torsional Braid Analysis (HP-TBA) is a unique technique for assessing changes in a polymer's thermal transitions, such as its glass transition temperature (Tg) or melting temperature (Tm), when exposed to an SCF like CO₂ [8]. A glass braid impregnated with the polymer is suspended in a high-pressure chamber and set into oscillation. The decay of these oscillations is analyzed to generate information on relative rigidity and mechanical damping. The dissolution of CO₂ in the polymer facilitates chain motions, depressing the Tg, which is a critical parameter for processes like foaming [8].
Viscosity: A high-pressure rotational viscometer is used to determine the viscosity of polymer solutions and other fluids at pressures up to 60 MPa. This system can also reveal the plasticizing effect of SCFs; for example, the sorption of scCO₂ in polymers like polydimethylsiloxane (PDMS) leads to significant reductions in viscosity [8].
Foaming: Specialized foaming cells are used to produce micro- or nano-porous polymer networks. These can be designed for free-foaming explorations or for confined foaming within molds. A gradient foaming cell allows experiments under a temperature gradient at a fixed pressure, enabling the study of how temperature influences foam morphology. Data from HP-TBA on Tg depression is often used to rationally select optimal foaming pressures and temperatures [8].
Visualization of Supercritical Fluid Processes and Properties
Phase Diagram of a Pure Substance
Supercritical Fluid Experimental Workflow
The Scientist's Toolkit: Essential Reagents and Materials
Successful experimentation with supercritical fluids relies on a suite of specialized reagents, materials, and equipment. Table 3 details key components of a research toolkit for this field.
Table 3: Research Reagent Solutions for Supercritical Fluid Research
| Reagent/Material | Function/Application | Critical Properties / Notes |
|---|---|---|
| Supercritical Carbon Dioxide (scCO₂) | Primary solvent for extraction, particle formation (RESS, PGSS), and as an anti-solvent (SAS). Prized for being non-toxic, non-flammable, and recyclable. [10] [4] | Tc = 31.3°C, Pc = 72.9 bar [4] |
| Co-solvents (e.g., Ethanol, Methanol) | Modifies the polarity and solvation power of scCO₂, enhancing the solubility of polar compounds. [12] | Must be miscible with scCO₂. |
| Pharmaceutical Compounds (e.g., Rifampin, Sirolimus) | Model drugs for solubility measurement and particle engineering studies to improve bioavailability. [11] [12] | Often BCS Class II/IV with poor aqueous solubility. |
| Biodegradable Polymers (e.g., PLGA, PCL) | Used in SAS and PGSS processes to fabricate drug-loaded microparticles and nanoparticles for controlled release. [8] [10] | Must be soluble in scCO₂ or an organic solvent. |
| Ionic Liquids (e.g., imidazolium-based) | Co-solvents or novel media in SCF processes; studied for CO₂ capture and sequestration; subject to ML property prediction. [13] [14] | Negligible vapor pressure, high thermal stability. |
| High-Pressure View Cells | Core apparatus for visualizing phase behavior and determining miscibility boundaries of fluid mixtures. [8] | Equipped with sapphire windows and movable pistons. |
| Variable-Volume Dual-Piston Systems | Advanced view cells for studying phase separation dynamics via light scattering in viscous polymer solutions. [8] | Includes magnetically coupled mixer. |
Advanced Modeling and Machine Learning in SCF Research
The experimental determination of properties like drug solubility in scCO₂ or surface tension of ionic liquids can be costly and time-consuming. This has driven the adoption of advanced computational models as efficient predictive tools.
Predicting Drug Solubility in scCO₂
Machine learning (ML) models have demonstrated remarkable accuracy in predicting drug solubility in supercritical CO₂, a critical parameter for designing pharmaceutical processes. Ensemble frameworks combining multiple ML regressors have shown particular promise. For example, one study used an ensemble of Extreme Gradient Boosting (XGBoost), Light Gradient Boosting (LGBM), and CatBoost Regression, optimized with bio-inspired algorithms, achieving a predictive accuracy of R² = 0.9920 [11]. Another study found that the XGBoost model alone could achieve an R² value of 0.9984 and a root mean square error (RMSE) of 0.0605 for predicting the solubility of 68 different drugs [12].
These models typically use input features such as temperature (T), pressure (P), CO₂ density (ρ), and drug-specific properties like molecular weight (MW), melting point (Tm), critical temperature (Tc), critical pressure (Pc), and acentric factor (ω) [11] [12]. The ability of ML models to learn complex, non-linear relationships from data directly, without relying on predefined physical equations, gives them superior predictive accuracy and generalization across diverse drug-solvent systems compared to traditional thermodynamic models [12].
Estimating Properties of Ionic Liquids
In parallel research on ionic liquids (ILs), which are often studied alongside or in conjunction with SCFs, machine learning is also making significant inroads. For instance, ensemble models like Extra Trees (ET), Random Forest (RF), and Decision Trees (DT), optimized with the Harmony Search (HS) algorithm, have been successfully employed to predict the surface tension of ILs at different temperatures [14]. In one study, the ET model achieved the highest performance with an R² of 0.979 and a low mean absolute percentage error (MAPE) of 2.05E-02 [14]. These models serve as valuable tools for estimating IL properties, saving significant time and cost associated with experimental measurements, and facilitating their application in green processing [14].
Supercritical fluids, particularly supercritical CO₂, represent a unique and versatile state of matter whose properties can be finely tuned for specific applications. Their hybrid liquid-gas characteristics underpin their utility across a wide range of fields, with pharmaceutical research and green chemistry standing out as major beneficiaries. The ability of SCF technology to enable precise particle engineering, enhance drug solubility, and eliminate organic solvent residues aligns perfectly with the modern demand for sustainable and efficient industrial processes. As research progresses, the integration of advanced machine learning models with traditional experimental methodologies is creating a powerful paradigm for accelerating the discovery and optimization of SCF-based applications, solidifying their critical role in the future of material science, drug development, and green engineering.
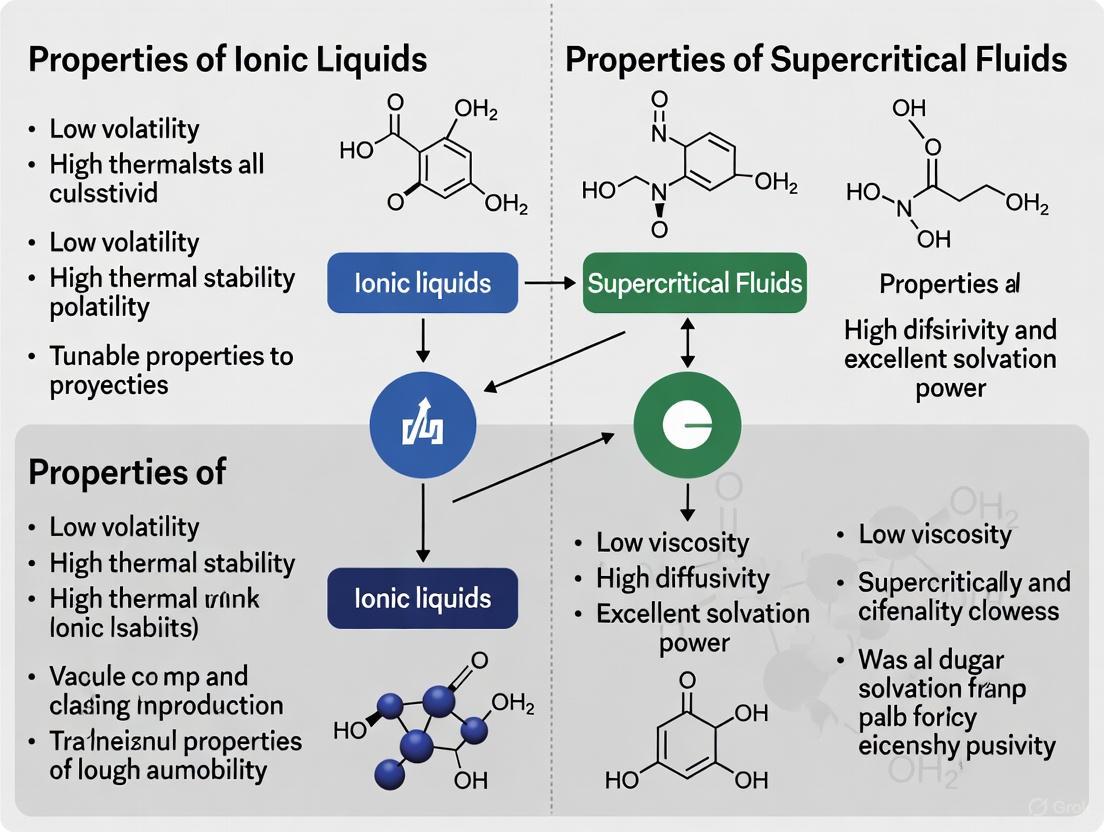
In the pursuit of advanced materials for scientific and industrial applications, ionic liquids (ILs) and supercritical fluids (SCFs) have emerged as two distinct classes of substances with unique properties. Ionic liquids are organic or organic-inorganic salts that are liquid below 100°C, characterized by an combination of organic cations and organic/inorganic anions [15]. In contrast, supercritical fluids are substances maintained at temperatures and pressures above their critical point, where they exhibit hybrid properties between liquids and gases [16] [17]. Understanding their fundamental physicochemical properties—volatility, viscosity, diffusivity, and thermal stability—is essential for selecting the appropriate medium for specific applications ranging from drug development to green energy technologies. This whitepaper provides a structured comparison of these key properties to guide researchers and scientists in their experimental design and application development.
Core Property Comparison: Ionic Liquids vs. Supercritical Fluids
Table 1: Comparative overview of key physicochemical properties of Ionic Liquids and Supercritical Fluids.
| Property | Ionic Liquids (ILs) | Supercritical Fluids (SCFs) | Technical Implications |
|---|---|---|---|
| Volatility | Extremely low vapor pressure, virtually non-volatile at standard conditions [15] | Density/pressure-dependent; exists as a single fluid phase above critical point [16] | ILs: Suitable for high-temperature/vacuum processes; reduced solvent loss and inhalation risk [15]. SCFs: Tuning pressure allows control over solvation power; enables supercritical extraction [16] [17]. |
| Viscosity | Relatively high (e.g., 500–1000 mPa·s), decreases with increasing temperature [15] | Very low, gas-like (e.g., 0.05–0.1 mPa·s for SC-CO₂) [16] [17] | ILs: High viscosity can limit mass transfer and flow, requiring system design adaptations [15]. SCFs: Low viscosity reduces pumping energy and improves penetration into porous materials [17]. |
| Diffusivity | Low, liquid-like (e.g., ~0.001 mm²/s) [15] | High, intermediate between liquids and gases (e.g., 0.01–0.1 mm²/s) [16] [17] | ILs: Slower diffusion rates can kinetically limit reactions or dissolution processes. SCFs: Enhanced mass transfer leads to faster extraction and reaction kinetics compared to liquids [17]. |
| Thermal Stability | High thermal stability; decomposition temperatures often exceed 400°C [1] [18] | Stability is a function of critical temperature; SC-CO₂ is stable, but SC-H₂O can be corrosive at high T [16] | ILs: Excellent for high-temperature catalysis, lubrication, and as heat transfer fluids [1] [15]. SCFs: Enables processes like supercritical water oxidation; stability is system-specific [16]. |
Table 2: Quantitative comparison of physical properties with common states of matter. Data is representative; specific values vary by compound and conditions [16] [17].
| State of Matter | Density (kg/m³) | Viscosity (μPa·s) | Diffusivity (mm²/s) |
|---|---|---|---|
| Gases | 1 | 10 | 1–10 |
| Supercritical Fluids | 100–1000 | 50–100 | 0.01–0.1 |
| Liquids | 1000 | 500–1000 | 0.001 |
| Ionic Liquids | ~1000–1400 [15] | ~500,000–1,000,000 [15] | ~0.001 [15] |
Experimental Characterization Protocols
Accurate measurement of these properties is fundamental for research and application development. Below are detailed methodologies for key characterization experiments cited in recent literature.
Protocol for Thermal Property Analysis of Ionic Liquids
1. Objective: To determine the melting point ((Tm)), glass transition temperature ((Tg)), and thermal decomposition temperature of an ionic liquid. 2. Methodology:
- Differential Scanning Calorimetry (DSC):
- Procedure: Weigh 5–10 mg of the IL sample in a sealed, high-pressure crucible. Perform a temperature scan from -150°C to 150°C at a controlled rate of 10°C/min under a nitrogen atmosphere. Cycle the temperature (cool-heat-cool) to identify reversible transitions.
- Data Interpretation: The melting temperature ((Tm)) is identified as the onset of the endothermic peak during the first heating scan. The glass transition temperature ((Tg)) appears as a step change in the heat flow curve during the second heating scan, indicating a transition from a brittle glassy state to a viscous liquid [18].
- Thermogravimetric Analysis (TGA):
- Procedure: Load 5–20 mg of the IL sample into a platinum pan. Heat the sample from room temperature to 600–800°C at a rate of 10°C/min under an inert nitrogen atmosphere.
- Data Interpretation: The thermal decomposition temperature is typically reported as the temperature at which 5% or 10% mass loss occurs ((T{d5}) or (T{d10})), indicating the onset of degradation [18].
Protocol for Phase Equilibrium and Solubility Modeling in Supercritical Systems
1. Objective: To model and predict the solubility of a solid compound (e.g., an Ionic Liquid) in a supercritical fluid (e.g., CO₂) with a co-solvent. 2. Methodology:
- System Setup: Utilize a high-pressure view cell or a saturation apparatus to experimentally gather phase equilibrium data for the ternary system (IL + CO₂ + co-solvent) across a range of pressures and temperatures.
- Modeling with ε-Modified Sanchez-Lacombe Equation of State (ε-mod SL-EoS):
- Procedure: Determine pure component parameters for the IL, CO₂, and the co-solvent by correlating high-pressure density and vapor pressure data. Fit binary interaction parameters for each pair (IL-CO₂, CO₂-co-solvent, IL-co-solvent) using available binary phase equilibrium data.
- Prediction: Use the determined parameters in the ε*-mod SL-EoS to predict the phase equilibrium and solubility of the IL in the supercritical CO₂ + co-solvent mixture. This lattice-fluid-based model has demonstrated superior predictive accuracy for systems involving heavy molecules like ILs compared to cubic equations of state like Peng-Robinson [5].
Property Interrelationships and Phase Behavior
The defining characteristics of ionic liquids and supercritical fluids are intrinsically linked to their molecular interactions and phase behavior. The following diagram illustrates the distinct pathways and governing factors that lead to the formation and unique properties of each.
The Scientist's Toolkit: Essential Research Reagents and Materials
The experimental study and application of ionic liquids and supercritical fluids require specific reagents and equipment. The following table details key items essential for research in this field.
Table 3: Essential research reagents, materials, and their primary functions.
| Item Name | Function/Application | Relevant System |
|---|---|---|
| Imidazolium-based ILs (e.g., [BmIm][BF₄]) | Versatile, widely-studied ILs used as solvents, electrolytes, and catalysts [15]. | Ionic Liquids |
| Sulfonate-based Anions (e.g., Triflate, Tosylate) | Anions contributing to high electrochemical stability and low viscosity in ILs [18]. | Ionic Liquids |
| High-Purity Carbon Dioxide (CO₂) | The most common supercritical fluid due to its mild critical point and non-toxic nature [17]. | Supercritical Fluids |
| Co-solvents (e.g., Methanol, Ethanol) | Added in small quantities to modify the solvation power of supercritical CO₂ and enhance solubility [5]. | Supercritical Fluids |
| Differential Scanning Calorimeter (DSC) | Instrument for measuring phase transitions (melting point, glass transition) of ionic liquids [18]. | Characterization |
| Thermogravimetric Analyzer (TGA) | Instrument for determining the thermal decomposition temperature and stability of materials [18]. | Characterization |
| High-Pressure View Cell/Reactor | Essential apparatus for visualizing and studying phase behavior and solubility in supercritical systems [5]. | Supercritical Fluids |
Ionic liquids and supercritical fluids represent two powerful yet fundamentally different classes of materials, each defined by a unique profile of volatility, viscosity, diffusivity, and thermal stability. The selection between them is not a matter of superiority but of application-specific suitability. Ionic liquids, with their negligible volatility, high thermal stability, and tunable chemistry, are ideal for applications as electrolytes, stable catalytic media, and advanced materials [1] [15]. Supercritical fluids, particularly CO₂, offer unparalleled advantages in processes requiring high diffusivity, low viscosity, and tunable solvation power, such as green extraction and particle formation [16] [17]. A deep understanding of these properties, coupled with robust experimental protocols for their characterization, enables researchers to push the boundaries of drug development, energy storage, and sustainable chemical processes. The future of this field lies in the continued refinement of these materials and the development of intelligent, application-specific designs.
In modern pharmaceutical research, the limitations of conventional solvents pose significant challenges, particularly for new drug candidates which often exhibit poor aqueous solubility, leading to inadequate dissolution profiles and subtherapeutic bioavailability [7]. The evolution of green chemistry has catalyzed the search for sustainable, efficient, and precisely controllable alternatives. Among the most promising are Ionic Liquids (ILs) and Supercritical Fluids (SCFs), which offer unparalleled tunability of fundamental physicochemical properties. This principle of tunability—the intentional and rational modification of a solvent's polarity, viscosity, and solvation power—is transforming drug delivery systems, extraction processes, and material synthesis. By moving beyond the fixed properties of traditional organic solvents, researchers can now design customized solvent environments optimized for specific therapeutic agents and industrial applications, thereby overcoming persistent pharmacological and processing barriers [7] [10] [19]. This technical guide examines the core mechanisms behind this tunability and its practical implementation in advanced research and development.
Ionic Liquids: Designer Solvents for Pharmaceutical Engineering
Ionic Liquids (ILs) are organic salts that remain liquid below 100 °C, characterized by their modular composition of bulky, asymmetric organic cations and inorganic or organic anions [2] [20]. This structural foundation grants them a remarkable spectrum of tunable properties.
Fundamental Tunability Mechanisms
The properties of ILs are primarily engineered through strategic cation-anion pairing and functionalization of ion side chains.
- Polarity and Hydrophilicity/Lipophilicity: The polarity of an IL can be finely adjusted to range from highly polar to non-polar by selecting appropriate ion pairs. For instance, ILs incorporating acetate or chloride anions tend to be hydrophilic, while those with bis(trifluoromethylsulfonyl)imide ([Tf₂N]⁻) or hexafluorophosphate ([PF₆]⁻) anions are typically hydrophobic [2] [21]. This allows for the custom design of ILs to solubilize specific drug molecules, whether hydrophilic biopharmaceuticals or hydrophobic small molecules.
- Viscosity: The viscosity of ILs is a critical parameter affecting diffusion rates and process efficiency. High viscosity often results from strong van der Waals forces and hydrogen bonding networks, particularly in ILs with long alkyl chains on the cation. Viscosity can be reduced by selecting ions with weaker cohesive energies, such as those containing fluorinated anions, or by increasing thermal energy [20].
- Solvation Power: The exceptional solvation power of ILs stems from their complex and diverse intermolecular interactions, including ionic bonds, hydrogen bonds, van der Waals forces, and π-π stacking [7]. The strength and type of these interactions can be modulated via ion selection to dissolve a wide array of solutes, from small-molecule active pharmaceutical ingredients (APIs) to large proteins and nucleic acids.
Advanced IL Formulations: API-ILs and Bio-ILs
Tunability extends beyond mere solvent properties to the creation of new chemical entities.
- API-Ionic Liquids (API-ILs): This innovative approach involves converting a drug molecule itself into an ionic liquid form by pairing an acidic or basic API with a biocompatible counterion. This strategy can dramatically improve the drug's solubility, thermal stability, and bioavailability while circumventing issues of polymorphism [7] [21]. For example, transforming ranitidine or paracetamol into an IL with a docusate counterion has yielded new forms with enhanced properties [21].
- Biocompatible ILs (Bio-ILs): Third-generation ILs are designed with biological and environmental compatibility in mind. These often utilize cations derived from natural, low-toxicity precursors such as choline, amino acids, or organic acids [2] [21]. Choline-based ILs, for instance, have demonstrated exceptional biocompatibility and are particularly effective for stabilizing biologics and enhancing mucosal permeability [7].
Table 1: Key Cations and Anions for Tunable Ionic Liquid Design
| Role | Component | Representative Examples | Impact on Properties |
|---|---|---|---|
| Cations | Imidazolium | 1-butyl-3-methylimidazolium ([BMIM]⁺) | Good structural adaptability; can be tuned for hydrophobicity. |
| Cholinium | Choline ([Ch]⁺) | High biocompatibility; effective for biologics and permeation. | |
| Phosphonium | Tetraalkylphosphonium ([P₆₆₆₁₄]⁺) | High thermal stability; often used in polymer applications. | |
| Pyrrolidinium | 1-butyl-1-methylpyrrolidinium ([BMP]⁺) | High electrochemical stability; useful in energy storage. | |
| Anions | Halides | Chloride (Cl⁻), Bromide (Br⁻) | Tend to be hydrophilic; can increase viscosity and melting point. |
| Fluorinated | [Tf₂N]⁻, [PF₆]⁻, [BF₄]⁻ | Hydrophobic; generally lower viscosity and increased stability. | |
| Carboxylates | Acetate [OAc]⁻, Geranate [Ger]⁻ | Hydrophilic; good hydrogen bond acceptors; can be bioactive. | |
| Amino Acids | Alaninate, Prolinate | Biocompatible; can be derived from natural sources. |
Supercritical Fluids: Tunability Through Thermodynamics
Supercritical Fluids (SCFs) are substances heated and compressed above their critical temperature (T~c~) and pressure (P~c~), entering a state that exhibits properties of both liquids and gases. Their most significant advantage is that their physicochemical properties can be dramatically altered through simple changes in temperature and pressure.
The Supercritical State and Its Tunable Parameters
- Solvation Power and Density: The solvation power of an SCF is highly dependent on its density. Near the critical point, small adjustments in temperature or pressure result in large changes in density. Since density is directly correlated with solvating strength, this allows for precise control over a supercritical fluid's dissolving power. This is the foundational principle for supercritical extraction and particle formation techniques [10] [12].
- Transport Properties: SCFs possess gas-like low viscosity and high diffusivity. This combination allows them to penetrate porous matrices much more effectively than liquids, leading to faster mass transfer and more efficient processes [10].
- Surface Tension: Supercritical fluids exhibit zero surface tension, which enables them to infuse into delicate structures, such as biological tissues or porous drug carriers, without causing structural damage [12].
Supercritical Carbon Dioxide as the Model Tunable Fluid
Supercritical CO₂ (scCO₂) is the most widely used supercritical fluid due to its accessible critical point (T~c~ = 31.1°C, P~c~ = 7.38 MPa), non-toxicity, non-flammability, and low cost [10] [12]. Its inherent low polarity makes it an ideal solvent for non-polar compounds. However, its polarity and solvation power can be significantly enhanced through two primary methods:
- Modulation of Pressure and Temperature: Increasing the pressure of scCO₂ at a constant temperature increases its density and, consequently, its solvating power for non-polar molecules.
- Use of Polar Co-solvents (Modifiers): Small amounts of a polar co-solvent, such as ethanol or methanol, can be added to scCO₂ to dramatically extend its solubility range to include more polar molecules, such as many pharmaceuticals [10] [22].
Table 2: Tunable Properties of Supercritical CO₂ and Common Modifiers
| Property | Typical Range in scCO₂ | Tuning Mechanism | Impact on Pharmaceutical Processing |
|---|---|---|---|
| Density | 0.1 - 0.9 g/mL | Adjust temperature and pressure. | Directly controls solvation power for extraction and recrystallization. |
| Viscosity | 0.01 - 0.1 mPa·s | Varies with density (T & P). | Governs mass transfer and diffusion rates in porous materials. |
| Diffusivity | 10⁻⁷ - 10⁻⁸ m²/s | Higher than liquids, varies with T & P. | Enhances extraction speed and efficiency compared to liquid solvents. |
| Polarity | Low (non-polar) | Addition of polar co-solvents (e.g., 1-10% ethanol). | Enables dissolution of polar and medium-polarity drug molecules. |
| Surface Tension | Zero | N/A | Allows impregnation of fragile biological structures and aerogels. |
Comparative Analysis: ILs vs. SCFs in Application
While both ILs and SCFs are tunable solvents, their application landscapes differ significantly based on their inherent characteristics.
Table 3: Comparative Analysis of Ionic Liquids and Supercritical Fluids
| Characteristic | Ionic Liquids (ILs) | Supercritical Fluids (SCFs) |
|---|---|---|
| Tunability Mechanism | Chemical structure (cation/anion combination). | Thermodynamic state (Temperature & Pressure). |
| Primary Tunable Properties | Polarity, viscosity, hydrophilicity/lipophilicity, chemical functionality. | Density, solvation power, transport properties. |
| Vapor Pressure | Negligible (non-volatile) [20]. | High (part of the supercritical state). |
| Typical Operating Conditions | Ambient temperature and pressure. | High pressure (tens of MPa), moderate temperature. |
| Key Pharmaceutical Applications | Drug solubilization, API-IL synthesis, transdermal delivery enhancers, stabilizers for biologics [7] [2]. | Drug micronization (RESS, SAS, PGSS), extraction of natural products, impregnation of matrices [10]. |
| Green Chemistry Considerations | High; reusable, but requires assessment of toxicity and biodegradability (esp. for 3rd Gen. ILs) [19] [21]. | High; SCF CO₂ is non-toxic and reusable, but energy-intensive for pressurization. |
Experimental Protocols for Solvent Tunability
Protocol: Synthesis of an Active Pharmaceutical Ingredient-Ionic Liquid (API-IL)
This protocol outlines the metathesis reaction for creating a novel drug-based ionic liquid, which can enhance the solubility and bioavailability of a poorly soluble API [21].
Objective: To synthesize a hydrophobic drug-based IL, such as Lidocaine Docusate. Principle: An ion-exchange reaction between a salt of the drug cation (e.g., Lidocaine HCl) and a silver or ammonium salt of the desired anion (e.g., Sodium Docusate).
Materials and Reagents:
- Lidocaine hydrochloride (API salt)
- Sodium docusate (source of hydrophobic anion)
- Dichloromethane (DCM, anhydrous)
- Deionized water
- Magnetic stirrer and stir bar
- Separatory funnel
- Rotary evaporator
- High-vacuum desiccator
Procedure:
- Dissolution: Dissolve Lidocaine HCl (1 equivalent) and Sodium Docusate (1 equivalent) in separate beakers using a mixture of deionized water and DCM (1:1 v/v).
- Mixing and Reaction: Combine the two solutions in a separatory funnel and shake vigorously for 2-4 hours at room temperature to allow the ion-exchange reaction to proceed to completion.
- Phase Separation: Allow the mixture to separate into two distinct phases. The newly formed, hydrophobic Lidocaine Docusate IL will partition into the lower organic (DCM) layer, while the inorganic salt byproduct (e.g., NaCl) will remain in the upper aqueous layer.
- Isolation: Drain and collect the organic layer.
- Purification: Wash the organic phase several times with small volumes of deionized water to remove any residual inorganic salts or starting materials.
- Solvent Removal: Evaporate the DCM under reduced pressure using a rotary evaporator to obtain the pure Lidocaine Docusate IL as a viscous liquid.
- Drying: Further dry the product under high vacuum for at least 24 hours to remove trace water and solvent. The resulting IL can be characterized by NMR, FT-IR, and DSC to confirm structure and the absence of crystallinity.
Protocol: Drug Micronization via Supercritical Antisolvent (SAS) Precipitation
This protocol describes a common SCF process for producing uniform, micro- or nano-sized drug particles to improve dissolution rates and bioavailability [10].
Objective: To produce fine particles of a drug (e.g., Rifampin) using scCO₂ as an antisolvent. Principle: The drug is dissolved in an organic solvent (e.g., DMSO). This solution is sprayed into a vessel filled with scCO₂. The scCO₂ is completely miscible with the organic solvent but not with the drug, causing extremely high supersaturation and the precipitation of fine, uniform drug particles.
Materials and Reagents:
- Model drug (e.g., Rifampin)
- Organic solvent (e.g., Dimethyl sulfoxide - DMSO)
- High-purity CO₂ gas
- SAS apparatus consisting of: high-pressure precipitation vessel, CO₂ pump, solution pump, co-solvent pump, back-pressure regulator, and temperature controllers.
Procedure:
- Solution Preparation: Dissolve the drug in the organic solvent to form a saturated or near-saturated solution.
- System Pressurization and Heating: Fill the precipitation vessel with scCO₂ and bring it to the desired operating temperature and pressure (e.g., 40°C, 10 MPa) using the CO₂ pump and heating jacket.
- Precipitation: Pump the drug solution through a fine nozzle into the precipitation vessel at a constant flow rate. The scCO₂ acts as an antisolvent, causing instantaneous precipitation of the drug as fine particles.
- Washing: Continue to pump pure scCO₂ through the vessel for a set time to wash away any residual organic solvent from the precipitated particles.
- Depressurization: Slowly depressurize the vessel to atmospheric pressure.
- Collection: Collect the dry, solvent-free micronized powder from the vessel's frit or bottom. The particles can be characterized by SEM for morphology, laser diffraction for particle size distribution, and XRD to determine any changes in crystallinity.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Reagents and Materials for IL and SCF Research
| Category | Item | Function/Application |
|---|---|---|
| Ionic Liquid Synthesis | Choline Chloride / Geranic Acid | Synthesis of biocompatible ILs (e.g., CAGE) for transdermal delivery [7] [2]. |
| 1-Alkyl-3-Methylimidazolium Salts | Versatile precursors for creating a wide range of ILs with tunable properties. | |
| Docusate (DOSS) Salt | Common counterion for creating hydrophobic API-ILs [21]. | |
| Silver Bis(trifluoromethylsulfonyl)imide | Reagent for halide-free metathesis reactions in high-purity IL synthesis. | |
| SCF Processing | Supercritical Fluid Chromatography (SFC) Grade CO₂ | High-purity carbon dioxide with dedicated dip tubes for SCF applications. |
| HPLC/GC-MS Grade Modifiers (e.g., Ethanol, Methanol) | Polar co-solvents for tuning scCO₂ polarity and dissolving a wider range of drugs [22]. | |
| High-Pressure Precipitation Vessel | Core component of RESS, SAS, and PGSS systems for particle engineering. | |
| Nozzles (Laser-drilled Orifices, Coaxial) | Critical for creating rapid expansion or mixing in SCF particle formation [10]. | |
| Analytical & Formulation | In-situ FTIR / UV-Vis Probes | For real-time monitoring of solubility and reactions in high-pressure SCF systems. |
| High-Pressure Phase Equilibrium Cell | For measuring solubility data of drugs in scCO₂, essential for process design. | |
| Lipids (e.g., DMPC) & Surfactants | For formulating IL-loaded nanocarriers like ethosomes and lipid nanoparticles [2]. |
Advanced Modeling and Visualization
Machine Learning for Solubility Prediction
Accurately predicting drug solubility in tunable solvents like scCO₂ is crucial for process design. Advanced machine learning (ML) models have demonstrated superior performance over traditional thermodynamic models. Ensemble frameworks combining XGBoost (XGBR), Light Gradient Boosting (LGBR), and CatBoost Regression (CATr), optimized with bio-inspired algorithms, have achieved predictive accuracies of R² > 0.99 [11] [12]. These models use inputs such as temperature, pressure, drug molecular weight, and melting point to capture the complex, non-linear relationships governing solubility, providing a rapid and reliable computational tool for pharmaceutical engineers.
Workflow Visualization
The following diagram illustrates the integrated research and development workflow for applying tunable solvents in drug development, from initial design to final formulation.
Diagram 1: Integrated R&D Workflow for Tunable Solvents. This chart outlines the parallel pathways for developing drug delivery systems using Ionic Liquids and Supercritical Fluids, highlighting their distinct tuning mechanisms that converge on formulation and evaluation.
The following diagram details the sequential mechanism by which Ionic Liquids enhance transdermal drug delivery, a key application area.
Diagram 2: IL Transdermal Delivery Mechanism. This flowchart visualizes the step-by-step process by which Ionic Liquids facilitate the skin penetration of drugs, particularly large biopharmaceuticals, by reversibly modifying the skin's barrier properties.
The tunability of Ionic Liquids and Supercritical Fluids represents a paradigm shift in pharmaceutical sciences, moving from a reliance on solvents with fixed properties to the engineering of customized solvation environments. The principle of tuning polarity, viscosity, and solvation power—whether through the rational selection of ion pairs in ILs or the precise control of temperature and pressure in SCFs—provides powerful levers to overcome longstanding challenges in drug delivery and processing. As computational prediction models become more sophisticated and our understanding of biological interactions deepens, the precision and application of these versatile solvents will continue to expand. Their integration is paving the way for next-generation, patient-centric therapeutics with enhanced efficacy, stability, and compliance profiles.
A supercritical fluid (SCF) is a substance that exists at a temperature and pressure above its critical point, the unique state where the distinct liquid and gas phases cease to exist [16]. This state was first discovered by Baron Charles Cagniard de la Tour in 1822 through his experiments with a sealed cannon barrel [16]. In the supercritical region, the substance possesses hybrid properties that are intermediate between those of a liquid and a gas, making it an exceptionally versatile medium for numerous industrial and scientific applications, from decaffeination of coffee to advanced drug development [16].
This state of matter is not just a laboratory curiosity; it occurs naturally throughout the universe. Supercritical water is found issuing from hydrothermal vents on the ocean floor, known as "black smokers" [16]. The atmospheres of planets like Venus and the gas giants Jupiter and Saturn are also believed to contain supercritical fluids [16]. The intrinsic interest in SCFs, combined with their practical utility in green chemistry, positions them as a critical area of modern research, particularly when their properties are contrasted with or enhanced by other advanced materials like ionic liquids [23] [24].
Critical Parameters and Physicochemical Properties
Defining the Critical Point
The critical point of a pure substance is defined by its critical temperature (Tc) and critical pressure (Pc). Above the critical temperature, a gas cannot be liquefied no matter how much pressure is applied [16]. The critical pressure is the vapor pressure of the substance at its critical temperature [9]. At this precise point, the densities of the liquid and gas phases become identical, and the boundary between the two phases disappears, resulting in a single, homogeneous supercritical phase [16] [9].
Properties of Supercritical Fluids
Supercritical fluids exhibit a unique combination of liquid-like and gas-like properties, as summarized in the table below. This blend of characteristics enables superior mass transfer and dissolution capabilities.
Table 1: Comparison of Typical Physical Properties of Gases, Supercritical Fluids, and Liquids [16]
| State of Matter | Density (kg/m³) | Viscosity (μPa·s) | Diffusivity (mm²/s) |
|---|---|---|---|
| Gases | 1 | 10 | 1 – 10 |
| Supercritical Fluids | 100 – 1000 | 50 – 100 | 0.01 – 0.1 |
| Liquids | 1000 | 500 – 1000 | 0.001 |
Key properties of SCFs include:
- Density: SCFs have densities comparable to liquids, which grants them good solvating power [16].
- Diffusivity: Their diffusivity is about an order of magnitude higher than that of liquids, leading to better mass transfer and penetration into porous structures [16] [25].
- Viscosity: SCFs have viscosities much closer to those of gases than liquids, which reduces resistance to flow [16] [25].
- Tunable Solvent Strength: A defining feature of SCFs is that their properties can be "tuned" with small changes in pressure or temperature, especially near the critical point [16]. For instance, a slight increase in pressure can cause a large increase in density, which directly enhances its ability to dissolve materials [16].
- No Surface Tension: Since there is no liquid/gas phase boundary, supercritical fluids lack surface tension [16].
Critical Parameters of Common Substances
The conditions required to achieve the supercritical state vary significantly from one substance to another. The following table provides the critical parameters for several common solvents used as supercritical fluids.
Table 2: Critical Parameters of Common Supercritical Fluids [16]
| Solvent | Molecular Mass (g/mol) | Critical Temperature (K) | Critical Pressure (MPa (atm)) | Critical Density (g/cm³) |
|---|---|---|---|---|
| Carbon dioxide (CO₂) | 44.01 | 304.1 | 7.38 (72.8) | 0.469 |
| Water (H₂O) | 18.015 | 647.096 | 22.064 (217.755) | 0.322 |
| Methane (CH₄) | 16.04 | 190.4 | 4.60 (45.4) | 0.162 |
| Ethane (C₂H₆) | 30.07 | 305.3 | 4.87 (48.1) | 0.203 |
| Propane (C₃H₈) | 44.09 | 369.8 | 4.25 (41.9) | 0.217 |
| Ethanol (C₂H₅OH) | 46.07 | 513.9 | 6.14 (60.6) | 0.276 |
Among these, supercritical carbon dioxide (scCO₂) is exceptionally prominent in research and industry. Its low critical temperature (31.1 °C) and modest critical pressure (7.4 MPa) make it relatively easy and safe to handle in a laboratory or industrial setting [25]. Furthermore, CO₂ is non-flammable, of low toxicity, inexpensive, and environmentally benign, as it often utilizes CO₂ captured as a by-product from other processes [25].
Phase Diagrams: A Graphical Representation
Understanding the Phase Diagram
A phase diagram is a graphical representation that shows the stable states of a substance (solid, liquid, gas) at different temperatures and pressures. Understanding this diagram is fundamental to visualizing the supercritical state.
Figure 1 illustrates a generic pressure-temperature (P-T) phase diagram. Key elements include [9]:
- The Boiling Curve (Liquid-Gas boundary): This curve separates the liquid and gas regions and represents the conditions under which the two phases coexist in equilibrium. It defines the boiling point of the liquid at any given pressure.
- The Critical Point: The boiling curve terminates at the critical point. Above this temperature and pressure, the liquid and gas phases are no longer distinct.
- The Supercritical Fluid Region: This is the area of the diagram at temperatures and pressures above the critical point. A substance in this region is a supercritical fluid.
- The Triple Point: The single point at which the solid, liquid, and gas phases coexist in equilibrium [9].
The Phase Diagram of Carbon Dioxide
The phase diagram for carbon dioxide is of particular practical importance. Unlike water, the melting curve for CO₂ has a positive slope, meaning the solid (dry ice) sublimes directly to a gas under standard atmospheric pressure [16] [9].
Figure 2 highlights the phase behavior of CO₂. A key feature is the dramatic change in density that occurs near the critical point. At a temperature just above the critical temperature (e.g., 310 K), a small increase in pressure results in a very large, almost vertical, increase in density, transforming the fluid from a gas-like to a liquid-like consistency [16]. This drastic change in a fundamental property like density is what allows for the fine-tuning of solvent strength.
Experimental Protocols and Methodologies
Generating a Supercritical Fluid
The general protocol for creating and utilizing a supercritical fluid, particularly scCO₂, for an extraction or reaction process involves the following steps:
- Pressurization: The substance (e.g., CO₂) is drawn from a supply cylinder as a liquefied gas. It is then pressurized above its critical pressure using a high-pressure pump.
- Heating: The pressurized fluid is passed through a heating unit to raise its temperature above the critical temperature. At this stage, it becomes a supercritical fluid.
- Contact and Processing: The supercritical fluid is introduced into an extraction vessel or reaction chamber containing the target material (e.g., plant matter for extraction, a substrate for impregnation). The tunable solvent properties are exploited by carefully controlling the system's pressure and temperature.
- Separation and Deposition: The SCF, now laden with dissolved solutes, is transferred to a separate chamber. Here, a phase transition is induced by decreasing the pressure and/or temperature, causing the solvent power to drop and the solute to precipitate for collection. The now-gasified CO₂ can often be recycled or vented.
Predicting Solubility: The Peng-Robinson Equation of State
A critical aspect of experimental design is predicting the solubility of materials in supercritical fluids. For complex systems, such as those involving ionic liquids in scCO₂, thermodynamic models are essential. The Peng-Robinson Equation of State (PR-EoS) is a widely used model for this purpose [22].
The methodology involves:
- Parameter Determination: The binary interaction parameters for the PR-EoS are determined using correlations from binary phase-equilibrium data (e.g., for IL + CO₂, CO₂ + co-solvent, and IL + co-solvent pairs) [22].
- Critical Temperature Adjustment: A key to accuracy is the use of a re-determined critical temperature for the ionic liquid as a pure component parameter within the model [22].
- Prediction: Once the binary parameters are established, the model can predict the effects of temperature, pressure, and co-solvent concentration on IL solubilities in the supercritical fluid phase without needing additional fitting parameters for the ternary system [22]. This approach has been shown to achieve acceptable accuracy, with average absolute relative deviations below 23% for solubility predictions, providing a valuable tool for process development [22].
The Research Context: Ionic Liquids vs. Supercritical Fluids
Ionic Liquids as Green Solvents
Ionic liquids (ILs) are salts that are liquid at relatively low temperatures (often below 100 °C). They are composed entirely of cations and anions and possess a unique set of properties, including negligible vapor pressure (non-volatility), non-flammability, high thermal stability, and tunable physicochemical characteristics [24]. This "tunability" allows for the design of "task-specific ionic liquids" by selecting different combinations of cations and anions [23] [24].
Synergistic IL-SCF Systems
While both ILs and SCFs are considered green solvents, their combination creates powerful synergistic systems for advanced applications. A fundamental and highly useful characteristic of IL-scCO₂ systems is their unique phase behavior: CO₂ is highly soluble in most ionic liquids, whereas ionic liquids have negligible solubility in scCO₂ [24].
This property is exploited in several key processes:
- Extraction and Recovery: A solute can be extracted from an IL using scCO₂. The CO₂ dissolves into the IL, modifying its properties and facilitating the release of the solute, which is then carried away by the scCO₂ stream. Because the IL itself does not dissolve, it remains as a pure, non-volatile residue after the CO₂ is depressurized [24].
- Separation and Purification: Organic solvents or reaction products can be separated from ILs using scCO₂. The scCO₂ strips the volatile components out, leaving the ionic liquid behind for reuse [24].
- Impregnation and Materials Processing: The low solubility of ILs in scCO₂ is a key factor in supercritical fluid deposition (SCFD) processes used to impregnate porous substrates with ionic liquids. However, for this application, accurately predicting the often-low solubility of ILs in scCO₂ (especially with co-solvents) becomes critical, which is where thermodynamic models like the Peng-Robinson EoS are vital [22].
The following diagram illustrates the logical workflow for developing a process that utilizes this synergistic relationship.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for Supercritical Fluid and Ionic Liquid Research
| Reagent/Material | Function & Application in Research |
|---|---|
| Supercritical Carbon Dioxide (scCO₂) | The workhorse supercritical fluid. Used as a tunable solvent for extraction (e.g., decaffeination), as a reaction medium, and as a carrier fluid for impregnation and particle formation due to its mild critical point and benign properties [16] [25]. |
| Co-solvents (Modifiers)(e.g., Methanol, Ethanol, Acetone) | Small quantities of organic solvents added to scCO₂ to significantly enhance its solubility for polar compounds, which are often poorly soluble in pure scCO₂. This is crucial for expanding the range of applicable solutes [22]. |
| Ionic Liquids (ILs)(e.g., Imidazolium, Pyridinium-based) | Non-volatile, tunable solvents used as advanced media for reactions, catalysis, and separations. In SCF research, they function as non-volatile receiving phases for SCF extraction or as target materials for impregnation into substrates [23] [24]. |
| High-Pressure Pumps(e.g., Syringe Pumps) | Essential equipment for pressurizing solvents like CO₂ to levels above their critical pressure, a prerequisite for creating and maintaining the supercritical state. |
| Titania (TiO₂) / Silica (SiO₂) Supports | Common porous solid supports used in studies on supercritical fluid deposition (SCFD) for creating supported ionic liquid phases (SILPs) or catalysts, where the SCF is used to transport and deposit the IL within the pores [22]. |
| Peng-Robinson Equation of State (PR-EoS) | A key thermodynamic model used to predict high-pressure phase equilibria and solute solubilities in supercritical fluids, crucial for designing and optimizing SCF processes without excessive trial-and-error experimentation [22]. |
Driving Innovation in the Lab: Key Pharmaceutical and Energy Applications
Supercritical Fluid (SCF) technology has emerged as an effective and environmentally benign alternative to traditional pharmaceutical manufacturing processes over the past few decades [26]. This technology operates active pharmaceutical ingredients (APIs) alone or in combination with biodegradable polymeric carriers under high-pressure conditions to enhance physical properties, particularly bioavailability [26]. The foundation of SCF technology lies in utilizing substances at temperatures and pressures above their critical point, where they exhibit unique properties intermediate between gases and liquids [27]. Carbon dioxide (CO₂) is the most widely used supercritical fluid due to its modest critical parameters (Tc = 31.1 °C, Pc = 7.38 MPa), non-toxicity, non-flammability, and economic viability [26] [27].
The interest in SCF technology for particle engineering stems from significant limitations of conventional micronization techniques, including broad particle size distribution, thermal and chemical degradation of drugs, excessive use of organic solvents, and residual solvent contamination in final products [26] [27]. In contrast, SCF processes can produce micro- and nano-sized particles with controlled size distribution, smooth surfaces, and enhanced bioavailability while avoiding these drawbacks [26]. The technology is particularly valuable for addressing challenges associated with poorly soluble APIs, as approximately 32% of all active ingredients are classified as Biopharmaceutics Classification System (BCS) Class 2 drugs with high permeability but poor water solubility [28].
This technical guide explores three principal SCF techniques—RESS, SAS, and PGSS—within the broader context of advanced solvent research that includes ionic liquids. While both SCFs and ionic liquids represent green alternatives to conventional organic solvents, they offer distinct advantages and applications in pharmaceutical engineering [29]. The following sections provide detailed methodologies, applications, and comparative analysis of these transformative particle engineering technologies.
Fundamental Principles of Supercritical Fluids
Definition and Unique Properties
A supercritical fluid exists as a single phase above its critical temperature (Tc) and critical pressure (Pc), where the distinction between liquid and gas phases disappears [27]. In this state, SCFs exhibit physical properties intermediate between those of liquids and gases, including liquid-like densities and gas-like viscosities and diffusivities [26] [28]. These hybrid properties enable SCFs to possess exceptional solvating power while maintaining high mass transfer capabilities, making them ideal for particle formation processes [27].
The tunability of SCF properties represents their most significant advantage for pharmaceutical applications. By making slight adjustments to temperature and/or pressure near the critical point, the density, solvent strength, transport properties, and dielectric constant of SCFs can be finely controlled [26] [27]. This allows precise manipulation of the precipitation environment during particle formation processes, enabling control over critical particle characteristics such as size, morphology, and crystal form [30].
Supercritical Carbon Dioxide as the Preferred Solvent
Among various compounds that can reach supercritical states, CO₂ has become the predominant choice for pharmaceutical applications for several compelling reasons [27]. Its critical point is easily achievable (31.1°C, 7.38 MPa), making it suitable for processing thermally labile pharmaceuticals [26]. CO₂ is non-flammable, inert, and recognized as safe by regulatory authorities including the US Food and Drug Administration [26]. Additionally, its high vapor pressure means residual CO₂ is not harmful to human health, with a threshold limit value (TLV) of 5000 ppm—significantly higher than many organic solvents like acetone (750 ppm) or pentane (600 ppm) [27].
While supercritical CO₂ (scCO₂) excels as a solvent for non-polar compounds, its effectiveness with polar molecules is limited [27]. This solubility challenge can be addressed through the use of co-solvents (modifiers) such as ethanol, acetone, or methanol, which are miscible with scCO₂ at moderate pressures and can enhance solubility of polar pharmaceutical compounds [27]. The ability to fine-tune solvent properties through pressure, temperature, and co-solvent adjustments provides pharmaceutical scientists with a powerful toolkit for optimizing particle engineering processes.
SCF-based particle formation processes are classified according to the role of the supercritical fluid in the system, primarily as a solvent, anti-solvent, or solute [26] [31]. The following table summarizes the fundamental characteristics of the three primary techniques discussed in this guide:
Table 1: Classification and Basic Principles of Major SCF Particle Engineering Techniques
| Technique | Role of SCF | Mechanism | Key Advantage | Primary Limitation |
|---|---|---|---|---|
| RESS (Rapid Expansion of Supercritical Solutions) | Solvent [26] | Dissolution of API in SCF followed by rapid expansion through nozzle causing supersaturation and precipitation [28] | Produces solvent-free products without need for organic solvents [28] | Limited to compounds with sufficient solubility in scCO₂ [26] |
| SAS (Supercritical Anti-Solvent) | Anti-solvent [26] | ScCO₂ acts as anti-solvent to reduce solvent power of organic solution containing API, inducing precipitation [26] | Suitable for compounds with low solubility in scCO₂ [31] | Requires use of organic solvents for initial dissolution [26] |
| PGSS (Particles from Gas-Saturated Solutions) | Solute [26] | ScCO₂ dissolved in molten API or API/polymer mixture followed by expansion and particle formation [26] | Uses low volumes of SCF; suitable for polymers and high molecular weight compounds [26] | Limited to temperature-resistant compounds that can be melted [26] |
The selection of an appropriate SCF technique depends on multiple factors including the physicochemical properties of the API (particularly its solubility in scCO₂ and thermal stability), the desired particle characteristics, and the intended route of administration [30]. Understanding the fundamental principles and limitations of each method is essential for designing effective particle engineering strategies.
RESS (Rapid Expansion of Supercritical Solutions)
Principles and Mechanisms
The RESS process operates on the principle of rapid phase change induced by abrupt pressure reduction [28]. In this technique, the API is first dissolved in a supercritical fluid, typically scCO₂, to form a supercritical solution [31]. This solution is then rapidly expanded through a nozzle into a low-pressure chamber, resulting in an instantaneous decrease in density and solvent power of the CO₂ [26] [28]. The abrupt loss of solvating capacity creates extreme supersaturation, leading to rapid nucleation and the formation of fine, uniform particles [28] [31].
The dramatic pressure drop during expansion (from high pressure to atmospheric) occurs within microseconds, creating supersonic flow and rapid temperature decrease due to the Joule-Thomson effect [26]. This rapid decompression is crucial for generating high nucleation rates while minimizing particle growth, resulting in narrow particle size distributions typically ranging from nanometers to several micrometers [28]. The entire process avoids organic solvents, yielding products with minimal solvent residues [28].
Experimental Protocol
Table 2: Typical Experimental Parameters for RESS Process Optimization
| Parameter | Typical Range | Impact on Particle Characteristics |
|---|---|---|
| Extraction Temperature | 40-80°C [28] | Higher temperatures generally increase solubility but may affect thermal stability |
| Extraction Pressure | 10-30 MPa [28] | Higher pressures increase density and solvating power of scCO₂ |
| Nozzle Diameter | 25-100 μm [28] | Smaller diameters create higher supersaturation rates, yielding smaller particles |
| Pre-Expansion Temperature | 50-120°C [28] | Affects particle morphology and size |
| Spray Distance | 1-10 cm [29] | Longer distances may allow for more particle growth and aggregation |
A standard RESS experimental apparatus includes the following key components [28]:
- CO₂ Supply: High-purity carbon dioxide source with purification stage
- Chilling Unit: Cooling system to maintain CO₂ in liquid state before pumping
- High-Pressure Pump: For pressurizing CO₂ beyond critical pressure
- Extraction Vessel: Temperature-controlled vessel where API dissolves in scCO₂
- Nozzle: Specialized capillary or orifice for rapid expansion (25-100 μm diameter)
- Precipitation Chamber: Low-pressure chamber for particle collection
- Temperature Control System: Precision controls for both extraction and precipitation zones
The process begins with cooling and pressurizing CO₂ beyond its critical point, then passing it through an extraction vessel containing the API [31]. After sufficient residence time for dissolution, the supercritical solution is expanded through the nozzle into the precipitation chamber, where particles are collected on filters or plates [28]. No post-processing is required to remove solvents, though particles may need protection from moisture and aggregation during storage.
Pharmaceutical Applications and Case Studies
RESS has demonstrated significant success in enhancing the dissolution rate and bioavailability of poorly soluble drugs. Notable examples include:
- Raloxifene: Particle size reduction from 45 μm to 19 nm, resulting in a 7-fold increase in dissolution rate [29]. Optimal parameters included temperature of 50°C, pressure of 17.7 MPa, and spray distance of 10 cm [29].
- Cefuroxime Axetil: Production of amorphous nanoparticles (158-513 nm) with complete dissolution within 20 minutes compared to only 50% dissolution for the commercial drug in 60 minutes [27].
- Ibuprofen: Micronization of racemic ibuprofen showing improved dissolution rate in buffered solutions with slightly decreased crystallinity [26].
- Diclofenac: Particle size reduction to 1.33-10.92 μm with transformation from irregular to quasi-spherical morphology [32].
Beyond simple micronization, RESS has been adapted for more complex applications including microencapsulation and co-precipitation. The RESS-N (RESS with a non-solvent) variant enables the formation of polymer microparticles containing proteins such as lysozyme and lipase [33]. Similarly, composite particles with naproxen core encapsulated in poly(L-lactic acid) coating have been successfully produced, demonstrating the technique's versatility for controlled release formulations [34].
SAS (Supercritical Anti-Solvent)
Principles and Mechanisms
The SAS technique exploits the ability of scCO₂ to act as an anti-solvent for substances dissolved in organic solvents [26]. When scCO₂ is introduced into a organic solution containing the API, it significantly reduces the solvent power of the organic liquid, leading to high supersaturation and subsequent precipitation of the solute [26] [31]. The high diffusivity of scCO₂ facilitates rapid mass transfer between the organic and supercritical phases, enabling the formation of fine particles with narrow size distributions [26].
Several SAS variants have been developed, including GAS (Gas Anti-Solvent), ASES (Aerosol Solvent Extraction System), PCA (Precipitation with Compressed Anti-Solvent), and SEDS (Solution Enhanced Dispersion by Supercritical Fluids) [26] [31]. These variations primarily differ in how the solution and anti-solvent are contacted and mixed, allowing customization for specific APIs and desired particle characteristics [26].
Experimental Protocol
Table 3: Key Parameters and Their Effects in SAS Process
| Parameter | Typical Range | Influence on Particle Formation |
|---|---|---|
| Pressure | 8-20 MPa [26] | Higher pressures increase CO₂ density and anti-solvent power |
| Temperature | 35-60°C [26] | Affects phase behavior and nucleation rates |
| Solution Concentration | 1-100 mg/mL [26] | Higher concentrations may increase particle size and aggregation |
| Solution Flow Rate | 1-5 mL/min [26] | Affects mixing and supersaturation rate |
| CO₂ Flow Rate | 10-50 g/min [26] | Higher flow rates enhance mass transfer and mixing |
A standard SAS apparatus typically consists of [26]:
- CO₂ Delivery System: Including storage, chilling unit, and high-pressure pump
- Solution Delivery System: Precision pump for organic solution containing API
- Precipitation Vessel: High-pressure vessel with temperature control
- Nozzle or Spray Device: For creating fine droplets of solution (two-fluid nozzle in SEDS)
- Collection System: Filter or cyclone for particle separation
- Back-Pressure Regulator: For maintaining system pressure
- Separation Vessel: For solvent-CO₂ separation
In a typical SAS experiment, the precipitation vessel is first brought to desired temperature and pressure conditions with scCO₂ [26]. The organic solution is then sprayed through a nozzle into the vessel, where scCO₂ rapidly extracts the organic solvent, causing supersaturation and particle precipitation [31]. The process continues with pure scCO₂ flow to remove residual solvent from the particles, after which the vessel is depressurized to collect the final product [26]. The choice of organic solvent is critical and must be miscible with scCO₂ while providing adequate solute dissolution [26].
Pharmaceutical Applications and Case Studies
SAS technology has been successfully applied to a wide range of pharmaceutical compounds, particularly those with low solubility in scCO₂:
- Polymeric Microparticles: SAS is extensively used for producing controlled-release formulations by co-precipitating drugs with biodegradable polymers such as PLGA [26]. The technique allows precise control over drug loading and release kinetics.
- Anticancer Formulations: Recent research demonstrates the preparation of hesperetin-polyvinylpyrrolidone sub-microparticles using SAS for improved anti-cancer efficiency [13]. The composite particles showed enhanced dissolution and therapeutic effects.
- Protein and Peptide Drugs: SAS processes have been adapted for processing sensitive biomolecules by optimizing temperature and pressure conditions to maintain biological activity while achieving particle size reduction [26].
- Composite Particles: SAS enables the production of composite particles containing multiple drugs or drugs with functional excipients, providing opportunities for "in-particle design" with uniform composition [34].
The tunability of SAS parameters allows researchers to control not only particle size but also solid-state properties including crystallinity and polymorphism, which are critical factors in pharmaceutical stability and bioavailability [30].
PGSS (Particles from Gas-Saturated Solutions)
Principles and Mechanisms
In the PGSS process, scCO₂ acts as a solute rather than a solvent [26]. The technique involves saturating a molten API or API-polymer mixture with scCO₂ at elevated pressure, significantly reducing the viscosity and glass transition temperature of the mixture [26] [31]. This gas-saturated solution is then expanded through a nozzle into a low-pressure chamber, causing the CO₂ to rapidly vaporize and thereby cooling the mixture through the Joule-Thomson effect and facilitating particle formation [26].
A key advantage of PGSS is its applicability to compounds with limited solubility in scCO₂, including many polymers and high molecular weight pharmaceuticals [31]. Additionally, the process typically requires lower volumes of scCO₂ compared to other SCF techniques, potentially reducing operational costs [26]. However, the requirement for melting the substrate limits PGSS to thermally stable compounds [26].
Experimental Protocol
Table 4: Critical Parameters in PGSS Process Optimization
| Parameter | Influence on Process | Optimization Consideration |
|---|---|---|
| Saturation Pressure | Determines CO₂ solubility in melt | Higher pressures increase CO₂ uptake and expansion efficiency |
| Saturation Temperature | Affects melt viscosity and CO₂ solubility | Must balance between melting point and thermal stability |
| Nozzle Geometry | Controls expansion dynamics | Design affects particle size and morphology |
| CO₂ Content | Impacts degree of viscosity reduction | Higher CO₂ content facilitates finer particle formation |
A standard PGSS apparatus includes [26]:
- CO₂ Supply System: Storage, chiller, and high-pressure pump
- Mixing Chamber: Temperature-controlled vessel for saturating molten material with scCO₂
- High-Pressure Autoclave: Equipped with stirring for maintaining homogeneous mixture
- Expansion Nozzle: For rapid depressurization
- Cyclone Separator: For particle collection from gas stream
- Temperature Control System: Precision controls for mixing chamber and nozzles
The process begins by melting the API or API-polymer mixture in the mixing chamber, followed by introduction of scCO₂ under pressure with continuous stirring to achieve saturation [26]. After sufficient mixing time to form a homogeneous gas-saturated solution, the mixture is expanded through a nozzle into the precipitation chamber [31]. The rapid pressure drop causes CO₂ vaporization and particle formation through two mechanisms: atomization of the liquid phase and solidification due to cooling [26]. The resulting particles are then collected from the gas stream in a cyclone or filter system [26].
Pharmaceutical Applications and Case Studies
PGSS has found particular utility in formulations requiring composite particle formation:
- Polymer-Based Delivery Systems: PGSS is highly effective for producing solid dispersions of drugs in polymeric carriers for controlled release applications [26]. The plasticizing effect of scCO₂ allows processing of high molecular weight polymers at reduced temperatures.
- High Drug-Loading Formulations: The technique enables production of particles with high active ingredient content, as it doesn't require dissolution of the drug in scCO₂ [31].
- Thermolabile Compounds: Despite involving melting, PGSS can process some heat-sensitive materials due to the temperature-lowering effect of CO₂ expansion and the possibility of operating at the minimum possible melting temperature [26].
- Lipid-Based Formulations: PGSS is particularly suitable for producing lipid microparticles and solid lipid nanoparticles for enhanced bioavailability of lipophilic drugs [31].
Comparative Analysis and Technical Considerations
Technique Selection Guidelines
Selecting the appropriate SCF technique requires systematic evaluation of API properties and target product characteristics:
Table 5: Decision Matrix for SCF Technique Selection
| Factor | RESS | SAS | PGSS |
|---|---|---|---|
| API Solubility in scCO₂ | High solubility required [31] | Not critical [31] | Not critical [31] |
| Thermal Stability | Moderate requirements | Low to moderate requirements | High requirements (must survive melting) [26] |
| Particle Size Range | Nanometers to few micrometers [28] | Submicron to micrometers [26] | Micrometers [26] |
| Organic Solvent Use | None [28] | Required [26] | None for process (may be needed for feeding) |
| Polymer Compatibility | Limited to scCO₂-soluble polymers [26] | Broad compatibility [26] | Broad compatibility [26] |
| Process Complexity | Simple | Moderate to high | Moderate |
Solubility Considerations and Enhancement Strategies
Solubility of pharmaceutical compounds in scCO₂ remains a critical factor in process selection and design. The solubility behavior follows trends based on molecular properties [27]:
- Non-polar compounds with low molecular weights typically exhibit higher solubility in scCO₂
- Polar functional groups (e.g., -OH, -COOH) generally decrease solubility
- Compounds with strong hydrogen bonding capacity have limited solubility in pure scCO₂
Several strategies can enhance solubility for SCF processing [27]:
- Co-solvents: Addition of small amounts (1-10%) of modifiers like ethanol, methanol, or acetone can dramatically increase solubility of polar compounds
- Chemical Modification: Temporary derivatization to increase CO₂-philicity (e.g., formation of esters or acetates)
- Mixed Carriers: Use of surfactant-like molecules to improve solvation
- Pressure and Temperature Optimization: Careful adjustment of process conditions to maximize solvent power
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 6: Essential Research Reagents and Materials for SCF Particle Engineering
| Category | Specific Examples | Function/Application |
|---|---|---|
| Supercritical Fluids | Carbon dioxide (most common) [26], water [26], nitrous oxide [26] | Primary processing medium providing unique solvation and precipitation properties |
| Co-solvents/Modifiers | Ethanol, methanol, acetone, ethyl acetate [27] | Enhance solubility of polar compounds in scCO₂; typically used at 1-10% concentration |
| Polymeric Carriers | PLGA, PLA, PCL, PEG, polyvinylpyrrolidone [26] [13] | Formulation matrices for controlled release, encapsulation, and solid dispersions |
| Stabilizers/Surfactants | Poloxamers, polysorbates, phospholipids [34] | Control particle growth and prevent aggregation during precipitation |
| API Categories | BCS Class II/IV compounds [28], proteins/peptides [26], anticancer agents [13] | Target compounds for bioavailability enhancement and controlled release |
| Specialized Equipment | High-pressure pumps, sapphire view cells, laser diffraction particle analyzers, DSC/XRD for solid-state characterization | Process monitoring, control, and product characterization |
SCF-enabled particle engineering techniques represent a mature yet continuously evolving field with significant potential for addressing persistent challenges in pharmaceutical development. The unique properties of supercritical fluids, particularly scCO₂, provide unparalleled control over particle characteristics while aligning with green chemistry principles through reduced organic solvent use [26] [27].
Future developments in this field are likely to focus on several key areas [28] [34]:
- Process Intensification: Integration of multiple unit operations and continuous processing to improve efficiency and scalability
- Advanced Modeling: Development of more accurate predictive models for particle formation and growth to reduce experimental optimization time
- Hybrid Techniques: Combination of SCF technologies with other particle engineering approaches to overcome individual limitations
- Expanded Biomolecule Applications: Adaptation of SCF processes for increasingly sophisticated biologics including antibodies, nucleic acids, and vaccine formulations
- In-Process Analytics: Implementation of real-time monitoring and control using advanced analytical technologies
Within the broader context of alternative solvent research, SCF technologies complement rather than compete with ionic liquids, as each offers distinct advantages for specific applications [29] [33]. While ionic liquids provide exceptional solvation power for polar compounds and tunable physicochemical properties, SCFs offer superior mass transfer characteristics and easier separation from final products [29]. The continued development of both technologies, and potentially their synergistic combination, will expand the available toolkit for addressing the increasingly challenging formulation requirements of modern pharmaceuticals.
As pharmaceutical research continues to confront molecules with greater complexity and poorer solubility profiles, SCF-based particle engineering techniques will play an increasingly vital role in transforming these challenging compounds into effective medicines. The RESS, SAS, and PGSS technologies, with their respective strengths and capabilities, provide powerful options for researchers seeking to optimize drug delivery through precise particle design.
The limited therapeutic efficacy of many active pharmaceutical ingredients (APIs) is frequently a direct consequence of inadequate dissolution rates and poor solubility in aqueous biological environments [35]. A significant number of newly developed drug candidates exhibit low bioavailability, necessitating higher and/or repeated dosages that often lead to undesirable side effects for patients [35]. Particle size reduction through micronization and nanonization presents a powerful formulation strategy to overcome these challenges, as it dramatically increases the specific surface area of the API, thereby enhancing its dissolution rate and subsequent absorption [35] [36].
Supercritical carbon dioxide (scCO2) has emerged as a superior technology for pharmaceutical particle engineering, offering a green alternative to conventional organic solvents [4]. With its easily attainable critical point (31.1 °C, 7.3 MPa), scCO2 enables processing under mild thermal conditions, preserving the stability of thermolabile compounds [35] [4]. The unique gas-like diffusivity and liquid-like density of scCO2 facilitate the production of particles with controlled morphology and narrow size distributions, which are crucial for reproducible drug performance [35]. This technical guide explores the application of scCO2-assisted techniques for enhancing drug bioavailability, with particular emphasis on the emerging synergy between supercritical fluids and ionic liquids in advanced pharmaceutical manufacturing.
Fundamental Properties of Supercritical CO₂
Supercritical fluids exist in a state above their critical temperature and pressure, exhibiting hybrid properties between conventional liquids and gases [4]. Carbon dioxide has become the predominant supercritical fluid in pharmaceutical applications due to its favorable combination of safety, accessibility, and tunable solvent power.
Table 1: Critical Parameters of Common Supercritical Fluids
| Fluid | Critical Temperature (°C) | Critical Pressure (bar) | Critical Density (g/cm³) |
|---|---|---|---|
| CO₂ | 31.3 | 72.9 | 0.47 |
| Xe | 16.6 | 58.4 | 1.10 |
| CHF₃ | 25.9 | 46.9 | 0.52 |
| N₂O | 36.5 | 72.5 | 0.45 |
| NH₃ | 132.5 | 112.5 | 0.24 |
| Pentane | 196.6 | 33.3 | 0.23 |
The solvent power of scCO₂ is highly dependent on its density, which can be precisely modulated by adjusting system pressure and temperature [4]. This tunability allows for selective processing without changing the solvent medium. Additionally, scCO₂ exhibits diffusivities approximately one order of magnitude higher than liquids and viscosities significantly lower than conventional liquid solvents, contributing to enhanced mass transfer capabilities during particle formation processes [4]. From an environmental and safety perspective, scCO₂ is non-toxic, non-flammable, and leaves no residual solvent in the final product, addressing significant limitations of traditional organic solvents [4].
scCO₂-Assisted Micronization and Nanonization Technologies
Various scCO₂-based technologies have been developed to exploit its unique properties for particle size reduction. These processes can be broadly categorized based on the role of scCO₂ as either a solvent or an antisolvent.
scCO₂ as a Solvent: The RESS Process
The Rapid Expansion of Supercritical Solution (RESS) process represents a fundamental approach where scCO₂ acts as a solvent for the target compound [37] [35]. In this method, the API is first dissolved in scCO₂ within a high-pressure saturation vessel. The supercritical solution is then rapidly expanded through a nozzle into a low-pressure collection chamber, creating an abrupt decrease in pressure that leads to extreme supersaturation and subsequent precipitation of fine particles [35]. The RESS process is particularly suitable for compounds with appreciable solubility in scCO₂ and typically produces particles ranging from micrometers to nanometers depending on process parameters including extraction conditions, pre-expansion temperature, nozzle geometry, and spray distance [35].
Several modifications to the basic RESS process have been developed to address specific challenges:
- RESS-SC (Solid Co-solvent): Incorporates solid co-solvents like menthol to enhance solubility of challenging compounds [35]
- RESOLV (Rapid Expansion of Supercritical Solution into Liquid Solvent): Expands the supercritical solution into a receiving liquid solvent to prevent particle aggregation [35]
- US-RESOLV (Ultrasonic-Assisted RESOLV): Applies ultrasonic energy during expansion to further reduce particle size and prevent agglomeration [35]
scCO₂ as an Antisolvent: SAS and GAS Processes
For compounds with limited solubility in scCO₂, antisolvent techniques present an effective alternative. The Supercritical Antisolvent (SAS) process involves dissolving the API in an organic solvent and spraying this solution as fine droplets into a vessel containing scCO₂ [37] [38]. The scCO₂ rapidly extracts the organic solvent, causing supersaturation and precipitation of the solute as fine particles. The SAS process has been successfully applied to numerous pharmaceuticals, including Tamsulosin, where optimization of pressure (25 MPa), temperature (41.3°C), and solution flow rate (2.9 mL/min) yielded particles with average sizes of approximately 600 nm [38].
The Gas Antisolvent (GAS) process operates on similar principles but involves introducing scCO₂ into a vessel containing the drug solution rather than spraying the solution into scCO₂ [36]. This method has demonstrated effectiveness in producing ampicillin nanoparticles in the range of 220-430 nm by controlling variables including initial solute concentration, temperature, pressure, and antisolvent addition rate [36].
Additional scCO₂ Techniques
Particles from Gas-Saturated Solutions (PGSS) is particularly effective for processing lipids, polymers, and other materials with relatively low melting points [37]. In this process, scCO₂ is dissolved into the molten substrate, and the gas-saturated mixture is then expanded through a nozzle. The release of dissolved CO₂ coupled with the cooling effect from expansion results in the formation of microparticles [37].
Near-critical Expansion Atomization (NEA) utilizes carbon dioxide near its critical point for the atomization and crystallization of products maintained in the liquid phase [37]. The liquid product is conveyed to an atomization tower where contact with CO₂ released to atmospheric pressure facilitates particle formation, typically producing particles in the 10-50 micrometer range [37].
Table 2: Comparison of scCO₂ Particle Formation Technologies
| Technology | Role of scCO₂ | Typical Particle Size | Key Applications |
|---|---|---|---|
| RESS | Solvent | Nanometers to micrometers | Compounds soluble in scCO₂ |
| SAS | Antisolvent | Nanometers to micrometers | Heat-sensitive compounds, polymers |
| GAS | Antisolvent | 220-430 nm (ampicillin) | Compounds with low scCO₂ solubility |
| PGSS | Co-solute | Microparticles | Lipids, waxes, polymers |
| NEA | Atomizing agent | 10-50 micrometers | Oily ingredients, liquid formulations |
Experimental Protocols and Methodologies
SAS Optimization for Tamsulosin Nanonization
A systematic investigation of the SAS process for Tamsulosin hydrochloride demonstrated the critical impact of process parameters on final particle characteristics [38]. Using Response Surface Methodology (RSM) based on Box-Behnken Design (BBD), researchers optimized three key variables: pressure (15-25 MPa), temperature (35-50°C), and drug solution flow rate (1.8-2.9 mL/min). The results indicated that pressure exerted the most significant influence on particle size, with an inverse correlation observed between pressure and particle dimension [38]. Temperature exhibited a more complex relationship, with particle size initially decreasing then increasing across the studied range. The optimal conditions identified for Tamsulosin nanoparticle production were 25 MPa pressure, 41.3°C temperature, and 2.9 mL/min injection rate, yielding particles of approximately 600 nm [38]. Characterization of the resulting particles via Scanning Electron Microscopy (SEM), Dynamic Light Scattering (DLS), Fourier-Transform Infrared Spectroscopy (FT-IR), X-ray Diffraction (XRD), and Differential Scanning Calorimetry (DSC) confirmed the preservation of chemical structure while achieving significant size reduction [38].
GAS Processing of Ampicillin Nanoparticles
The production of ampicillin nanoparticles via the GAS process illustrates the precise control achievable through antisolvent techniques [36]. In this protocol, ampicillin was dissolved in dimethyl sulfoxide (DMSO) at concentrations ranging from 20-100 mg/mL. One milliliter of this solution was introduced into a precipitation vessel, after which carbon dioxide was injected until reaching the target pressure (9-15 MPa). The system was maintained at equilibrium for 30 minutes before flushing with pure CO₂ to remove residual solvent [36]. Experimental results demonstrated that lower temperatures (34°C vs. 46°C), reduced initial solute concentrations (20 mg/mL vs. 100 mg/mL), and higher antisolvent flow rates (2.4 mL/min vs. 1.6 mL/min) all contributed to smaller particle sizes [36]. High-Performance Liquid Chromatography (HPLC) and FTIR analyses confirmed that the GAS process did not alter the chemical structure of ampicillin, highlighting the technique's suitability for processing sensitive pharmaceutical compounds [36].
The Ionic Liquid - scCO₂ Synergy in Pharmaceutical Processing
Ionic liquids (ILs) - non-volatile salts liquid at room temperature - have emerged as valuable partners for scCO₂ in advanced pharmaceutical applications [24]. Their unique properties, including negligible vapor pressure, thermal stability, and tunable physicochemical characteristics through cation-anion selection, make them particularly suitable for combination with supercritical fluid technologies [24].
A significant advantage of IL-scCO₂ systems stems from their peculiar phase behavior: while CO₂ exhibits appreciable solubility in many ILs, most ILs demonstrate negligible solubility in scCO₂ [24]. This asymmetric miscibility creates opportunities for novel processing strategies, including:
- Extraction and Separation: ScCO₂ can efficiently extract organic compounds from ILs without cross-contamination [24]
- Reaction Media: ILs serve as green solvents for synthetic reactions with scCO₂ used for product separation [24]
- Particle Engineering: Combined systems enable new approaches to control particle morphology and size distribution
Recent advances in predicting IL solubility in scCO₂ with co-solvents have significantly improved process design capabilities. The Peng-Robinson Equation of State (PR-EoS) and the more recent ε-modified Sanchez-Lacombe Equation of State (ε-mod SL-EoS) have demonstrated particular utility in modeling these complex systems, with the latter achieving an average logarithmic AARD of 11.0% in predicting IL solubilities in scCO₂ with co-solvents [5].
For impregnation processes where uniform distribution of ILs into porous supports is desired, the general insolubility of ILs in pure scCO₂ presents a challenge. This limitation can be overcome by adding appropriate co-solvents, which dramatically increase IL solubility in the CO₂ phase [22]. Accurate prediction of these ternary systems is essential for optimizing processes like Supercritical Fluid Deposition (SCFD), with recent models successfully reproducing the effects of temperature, pressure, and co-solvent concentration on IL solubilities without requiring fitting parameters for ternary systems [22].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for scCO₂ Pharmaceutical Processing
| Category | Specific Examples | Function in scCO₂ Processes |
|---|---|---|
| Supercritical Fluids | CO₂, N₂O, CHF₃ | Primary processing medium as solvent or antisolvent |
| Co-solvents | Methanol, Ethanol, Acetone | Enhance solubility of polar compounds in scCO₂ |
| Ionic Liquids | Imidazolium, Pyridinium, Ammonium salts | Green solvent media, templating agents, solubility modifiers |
| Pharmaceutical Carriers | PLGA, PVP, Poloxamers | Stabilize nanoparticles, control drug release profiles |
| Organic Solvents | DMSO, NMP, DCM, Acetone | Dissolve APIs for antisolvent processes |
| Stabilizers | Polysorbates, Poloxamers, Cyclodextrins | Prevent nanoparticle aggregation, improve stability |
Workflow Visualization: RESS Process
Supercritical CO₂ technologies represent a versatile and environmentally sustainable platform for enhancing drug bioavailability through precision particle engineering. The ability to produce micronized and nanonized pharmaceutical particles with controlled characteristics addresses fundamental limitations of poor solubility and slow dissolution rates that plague many modern therapeutic compounds. The continuing evolution of scCO₂ processes, including RESS, SAS, and PGSS variants, provides formulation scientists with an expanding toolkit for addressing diverse pharmaceutical processing challenges.
The emerging synergy between ionic liquids and scCO₂ systems presents particularly promising avenues for future development, combining the unique properties of both green solvent systems to create novel processing strategies. As predictive modeling of these complex systems continues to improve, the rational design of scCO₂-based pharmaceutical manufacturing processes will become increasingly sophisticated, enabling more efficient development of advanced drug formulations with optimized therapeutic performance.
The development of efficient and safe drug delivery systems represents a paramount objective in modern pharmaceutical research. Conventional delivery platforms face persistent challenges that substantially limit their clinical utility, including poor aqueous solubility of many therapeutic agents (BCS Class II/IV), structural instability under physiological conditions, and nonspecific biodistribution resulting in insufficient drug accumulation at target sites [7]. In the broader context of alternative solvent research, both ionic liquids (ILs) and supercritical fluids (SCFs) have emerged as environmentally benign solvents with complementary properties. ILs are organic salts that remain liquid below 100°C, exhibiting non-volatility and modular cation-anion combinations that enable precise tuning of pharmaceutical parameters [24]. In contrast, SCFs are substances maintained above their critical temperature and pressure, exhibiting properties of both liquids and gases—high diffusivity similar to gases and high density like liquids [17]. Supercritical carbon dioxide (scCO₂), with critical parameters of 31.1°C and 7.38 MPa, has gained particular prominence due to its mild critical conditions, non-flammability, low toxicity, and environmental friendliness [17].
While both systems represent green alternatives to traditional organic solvents, their pharmaceutical applications have diverged. The combination of these two solvent systems has revealed unique synergies, particularly in purification processes where scCO₂ can extract organic solutes from ILs without ionic liquid contamination [39]. However, ILs have demonstrated exceptional versatility in directly addressing fundamental drug formulation challenges through their tunable chemical structures. This review examines the transformative role of ionic liquids in overcoming solubility and targeting barriers in pharmaceutical development, contextualized within the broader landscape of advanced solvent systems.
Fundamental Properties and Design Principles of Ionic Liquids
Structural Components and Tunability
Ionic liquids possess a modular architecture consisting of asymmetric, sterically hindered organic cations and various anions that prevent efficient crystal packing, resulting in low melting points [40]. This structural combination offers unparalleled design flexibility through careful selection of cation-anion pairs:
- Cation Core Structures: Common pharmaceutical-grade cations include imidazolium, pyridinium, ammonium, phosphonium, and cholinium groups [24].
- Anion Varieties: Anions range from simple halides to complex molecular ions such as tetrafluoroborate, hexafluorophosphate, amino acid-based anions, and fatty acid-derived anions [41].
The tunability of ILs enables precise control over critical physicochemical properties including hydrophobicity, viscosity, melting point, and solvation capability, making them truly "designer solvents" for pharmaceutical applications [40]. This customization capability allows formulators to create task-specific ILs optimized for particular drugs or delivery routes.
Generational Evolution of Ionic Liquids
The development of ILs has progressed through three distinct generations, each with expanding pharmaceutical applicability:
Table 1: Generational Evolution of Ionic Liquids
| Generation | Key Components | Primary Characteristics | Pharmaceutical Relevance |
|---|---|---|---|
| First | Dialkyl imidazolium, alkylpyridinium with metal halide anions | High thermal stability, low melting points, broad liquid ranges | Limited due to low biodegradability and high toxicity |
| Second | Dialkyl imidazolium, alkylpyridinium, ammonium, phosphonium with BF₄⁻, PF₆⁻ anions | Customizable physical/chemical properties, air and water stable | Better tunability but concerns regarding toxicity and biocompatibility |
| Third | Natural source-derived cations (e.g., choline) and anions (e.g., amino acids, fatty acids) | Reduced toxicity, enhanced biodegradability, maintained tunability | Ideal for pharmaceutical applications; excellent safety profile |
This evolution toward third-generation ILs with natural components has significantly expanded their biomedical applications, particularly for drug delivery where biocompatibility is paramount [41].
Ionic Liquid Strategies for Enhancing Drug Solubility
Poor aqueous solubility remains a critical formulation challenge for many marketed drugs and pipeline candidates, leading to inadequate dissolution profiles and subtherapeutic bioavailability [7]. Ionic liquids address this limitation through multiple mechanistic approaches:
Active Pharmaceutical Ingredient Ionic Liquids (API-ILs)
The API-IL strategy involves converting drug molecules directly into ionic forms by pairing them with counterions, markedly improving solubility and bioavailability while integrating the active agent and delivery vector into a single ionic entity [7]. This approach effectively circumvents crystal packing efficiency, reducing the melting point and enhancing dissolution kinetics.
Solubilization Mechanisms
Ionic liquids improve drug solubility through several complementary mechanisms:
- Ionic Interaction: IL ions form strong ionic bonds with ionizable drug molecules
- Hydrogen Bonding: IL components donate or accept hydrogen bonds with drug molecules
- Hydrophobic Interactions: Alkyl chains on cations facilitate solubilization of non-polar drugs
- π-π Stacking: Aromatic cations (e.g., imidazolium) enable stacking with conjugated drug systems
These interactions collectively disrupt the cohesive forces in drug crystals while providing a thermodynamically favorable environment for solvation [7].
Experimental Protocol for Solubility Enhancement Studies
Objective: To evaluate the solubility enhancement of a poorly water-soluble drug in ionic liquid formulations.
Materials:
- Poorly soluble model drug (e.g., paclitaxel, doxorubicin)
- Ionic liquids (e.g., choline geranate [CAGE], imidazolium-based ILs)
- Standard buffer solutions (pH 1.2-7.4)
- Analytical instrument: HPLC with UV detection
Methodology:
- Prepare saturated solutions of the drug in various IL-containing buffers
- Agitate mixtures at 37°C for 24-48 hours to reach equilibrium
- Centrifuge at 10,000 rpm for 15 minutes to separate undissolved drug
- Dilute supernatant appropriately with mobile phase
- Analyze drug concentration using validated HPLC method
- Compare solubility in IL formulations versus conventional solvents
Key Calculations:
- Solubility enhancement ratio = (Solubility in IL formulation)/(Solubility in water)
- Partition coefficient = (Drug concentration in IL phase)/(Drug concentration in aqueous phase)
This protocol has demonstrated dramatic improvements, with certain IL formulations increasing monoclonal antibody absorption by up to 200% and significantly enhancing transdermal delivery of hydrophobic drugs like paclitaxel [42] [41].
Ionic Liquid Approaches for Targeted Drug Delivery
Beyond solubility enhancement, ionic liquids provide innovative solutions for targeted drug delivery, enabling spatiotemporally controlled drug release while minimizing off-target effects.
Biological Barrier Penetration
Ionic liquids facilitate drug transport across critical biological barriers through several mechanisms:
The diagram above illustrates how IL-based formulations interact with various biological barriers. For transdermal delivery, ILs disrupt stratum corneum lipids through extraction and fluidization [41]. For neurological applications, certain IL coatings enable nanoparticles to bind with red blood cells, effectively "hitchhiking" across the blood-brain barrier [43]. In oncology, sugar-mimicking ILs can target macrophages that are naturally taken up by sugar-hungry tumors [43].
Stimuli-Responsive Delivery Systems
Advanced IL designs incorporate stimuli-responsive mechanisms for controlled drug release:
- Temperature Sensitivity: IL-polymer composites that release drugs upon temperature changes
- pH Responsiveness: IL structures designed to destabilize in specific pH environments (e.g., tumor microenvironments)
- Microwave Triggering: Imidazolium ionic liquid-polydopamine nanocomposites that release drugs under microwave irradiation, enabling precise spatial and temporal control [7]
Experimental Protocol for Targeted Delivery Evaluation
Objective: To assess the targeted delivery capability of IL-coated nanoparticles to specific tissues or cells.
Materials:
- IL-coated nanoparticles (e.g., chitosan nanoparticles with choline-based IL coating)
- Fluorescently labeled drug or tracking dye
- In vitro models (e.g., blood-brain barrier model, cancer cell lines)
- In vivo animal models
- Imaging systems (e.g., fluorescence imaging, MRI)
Methodology:
- Prepare IL-coated nanoparticles using emulsion-solvent evaporation method
- Characterize nanoparticles for size, zeta potential, and drug loading efficiency
- Conduct in vitro permeability studies using barrier models (e.g., transwell assays)
- Perform cellular uptake studies with flow cytometry and confocal microscopy
- Administer formulations in animal models and evaluate biodistribution
- Quantify drug accumulation in target versus non-target tissues
Key Parameters:
- Targeting efficiency = (Drug in target tissue)/(Drug in non-target tissue)
- Permeability enhancement ratio = (Permeability with IL)/(Permeability without IL)
- Cellular uptake efficiency via flow cytometry
This approach has demonstrated remarkable successes, including IL-coated nanoparticles capable of crossing the blood-brain barrier without invasive procedures and sugar-like ILs that deliver cancer medications directly into tumors [43].
Comparative Analysis: Ionic Liquids vs. Supercritical Fluids in Pharmaceutical Applications
While this review focuses primarily on ionic liquids, contextualizing their properties and applications against supercritical fluids provides valuable insights for researchers selecting solvent platforms:
Table 2: Comparative Analysis of Ionic Liquids and Supercritical Fluids for Pharmaceutical Applications
| Parameter | Ionic Liquids | Supercritical Fluids |
|---|---|---|
| Physical State | Liquid salts at room temperature | Fluid above critical point |
| Volatility | Negligible vapor pressure | Highly volatile |
| Polarity | Tunable from polar to non-polar | Typically non-polar (scCO₂) |
| Solvation Power | High for both polar and non-polar compounds | Highly tunable with pressure |
| Diffusivity | Similar to liquids (~10⁻¹¹ m²/s) | Intermediate between gases and liquids (~10⁻³ m²/s) |
| Viscosity | Higher than conventional solvents | Lower than liquids |
| Pharmaceutical Applications | Drug solubilization, API-ILs, permeation enhancement, targeted delivery | Extraction, purification, particle formation, impregnation |
| Key Advantages | Modular design, multi-functionality, non-volatility | High penetration, clean residue, mild processing |
| Limitations | Potential toxicity concerns, high viscosity | High-pressure equipment, limited solubility for polar compounds |
The combination of these two alternative solvents presents unique opportunities. Notably, scCO₂ exhibits high solubility in many ILs, while ILs have negligible solubility in scCO₂, enabling efficient extraction of reaction products from IL media without solvent contamination [39]. Recent modeling advances using the ɛ*-modified Sanchez-Lacombe equation of state have improved prediction of IL solubility in supercritical CO₂ with co-solvents, facilitating process optimization [5].
The Scientist's Toolkit: Key Research Reagents and Materials
Successful implementation of ionic liquid strategies requires carefully selected materials and reagents:
Table 3: Essential Research Reagents for Ionic Liquid Drug Formulation
| Reagent Category | Specific Examples | Function and Application |
|---|---|---|
| Biocompatible Cations | Choline, amino acids (glycine, proline), guanidinium | Provide low-toxicity cation cores with high biocompatibility |
| Pharmaceutical Anions | Geranate, oleate, laurate, amino acid-based anions | Enhance membrane permeability and drug solubility |
| API-IL Precursors | Ionizable drugs (NSAIDs, antifungals, anticancer agents) | Enable formation of active pharmaceutical ingredient ionic liquids |
| Nanocarrier Systems | Polymeric nanoparticles, liposomes, micelles | Serve as substrates for IL functionalization to enhance targeting |
| Analytical Standards | High-purity IL references, internal standards | Ensure accurate quantification and quality control |
| Permeation Enhancers | Sorbitan monolaurate (Span-20), polysorbates | Complement IL action in transdermal formulations |
Ionic liquids have redefined drug delivery paradigms through molecular-level programmability, enabling breakthrough strategies that overcome persistent limitations of conventional systems. The unique properties of ILs—including their modular nature, multifunctionality, and ability to enhance both solubility and targeted delivery—position them as transformative platforms in pharmaceutical development.
Future research directions include deeper investigation of long-term biosafety profiles, development of standardized regulatory guidelines, and integration of IL technology with artificial intelligence for rational design. The convergence of ILs with nanomedicine and additive manufacturing presents unprecedented opportunities to develop personalized therapeutic platforms, potentially revolutionizing precision medicine.
As the field advances, the complementary strengths of ionic liquids and supercritical fluids may enable integrated processing strategies that leverage the advantages of both green solvent systems, ultimately accelerating the development of more effective, safer pharmaceutical products.
Ionic Liquids (ILs), defined as organic salts with melting points below 100°C, have emerged as a revolutionary class of electrolytes for electrochemical energy storage, moving beyond their traditional applications in pharmaceuticals and synthesis. [44] [45] Their unique properties—including negligible vapor pressure, non-flammability, high thermal stability, and wide electrochemical windows—make them particularly attractive for addressing the safety and performance limitations of conventional aqueous and organic electrolytes in lithium-ion batteries (LIBs) and supercapacitors (SCs). [46] [47] This in-depth technical guide frames the advancement of IL-based electrolytes within the broader context of solvent research, where their properties present a compelling alternative to other advanced media like supercritical fluids (SCFs). While SCF electrochemistry offers advantages in mass transport and tunable density, it is often hampered by challenges such as low dielectric constants, poor solubility of supporting electrolytes, and the need for complex pressurized equipment. [48] [49] ILs, in contrast, offer a combination of high intrinsic ionic conductivity and remarkable structural tunability, allowing scientists to design electrolytes with targeted properties for specific energy storage applications. [44] [45]
Fundamental Properties and Comparison with Conventional Electrolytes
The performance of an electrolyte is critical to the energy density, power density, safety, and cycle life of an energy storage device. This section quantitatively compares the properties of IL-based electrolytes against conventional systems and supercritical fluids.
Table 1: Comparison of Key Electrolyte Properties for Energy Storage
| Electrolyte Type | Electrochemical Window (V) | Ionic Conductivity (mS cm⁻¹) | Thermal Stability / Volatility | Key Challenges |
|---|---|---|---|---|
| Aqueous Electrolytes | ~1.23 [47] | High (tens to hundreds) | Low boiling point, volatile | Limited voltage, corrosive [44] |
| Organic Carbonates | ~2.7 - 4.3 [44] | Moderate to High | Flammable, volatile [46] | Safety risks (fire, explosion) [46] |
| Supercritical Fluids (e.g., scCO₂) | Wide (>4) [49] | Low to Moderate (depends on co-solvent) [48] | Tunable, generally stable | Low dielectric constant, poor salt solubility, high-pressure systems [48] |
| Ionic Liquids (ILs) | ~3.0 - 6.4 [44] | 0.1 - 20 [44] | High thermal stability, non-flammable, negligible volatility [45] [47] | High viscosity, higher cost, sensitivity to impurities |
The electrochemical stability window (ESW) is a paramount property, as the energy density (E) of a device scales with the square of the operating voltage (V) according to E = ½CV². [47] As shown in Table 1, ILs typically offer ESWs of 3.0 to 6.4 volts, significantly outperforming aqueous systems and rivaling or exceeding organic carbonates. [44] This directly enables the development of higher-energy-density devices. Furthermore, unlike volatile organic carbonates or the high-pressure SCFs, ILs bring a fundamentally safer profile to battery operation due to their non-flammability and thermal stability. [46] [45]
The Tunability of Ionic Liquids
A key advantage of ILs over SCFs is their structural tunability. The properties of an IL can be precisely engineered by selecting different combinations of cations and anions. [44] [45]
Table 2: Common Ions in ILs for Energy Storage and Their Influence on Properties
| Ion Type | Examples | Influence on Electrolyte Properties |
|---|---|---|
| Cations | Imidazolium (e.g., [bmim]⁺, [emim]⁺) | High ionic conductivity, but moderate electrochemical stability [44] [47] |
| Pyrrolidinium (e.g., [Pyr₁₄]⁺, [Pyr₁₃]⁺) | Wider electrochemical window, good stability [45] [47] | |
| Phosphonium (e.g., [P₂₂₂₅]⁺) | Very wide electrochemical window (>6 V), high thermal stability [44] | |
| Ammonium | Varied properties based on alkyl chain substituents [45] | |
| Anions | Bis(trifluoromethanesulfonyl)imide ([TFSI]⁻ or [NTf₂]⁻) | High stability, good conductivity, forms hydrophobic ILs [45] |
| Tetrafluoroborate ([BF₄]⁻) | Moderate conductivity, can be susceptible to hydrolysis [44] | |
| Hexafluorophosphate ([PF₆]⁻) | Similar to [BF₄]⁻, hydrolysis concerns [44] | |
| Triflate ([CF₃SO₃]⁻) | Good lithium solvation ability [45] |
This "designer solvent" capability allows researchers to formulate electrolytes with an optimal balance of conductivity, electrochemical stability, and lithium solvation for specific battery or supercapacitor applications. [45]
ILs in Lithium-Ion Batteries (LIBs)
Application Rationale and Formulations
In LIBs, the replacement of flammable organic carbonates with IL-based electrolytes directly addresses the critical safety issue of thermal runaway. [46] ILs can be used as a standalone electrolyte, a co-solvent in a binary system with carbonates, or as a component in gel polymer electrolytes. [45] A prominent and widely studied IL for LIBs is 1-butyl-1-methylpyrrolidinium bis(trifluoromethanesulfonyl)imide (PYR₁₄TFSI), prized for its anodic stability (>5.5 V vs. Li⁺/Li) and thermal stability up to 300°C. [45] Studies have shown that formulating ternary mixtures, such as PYR₁₄TFSI with LiTFSI salt and propylene carbonate (PC) solvent, can yield non-flammable electrolytes with promising cycling performance at both room temperature and 60°C when the IL content is around 80 wt%. [45] Additives like vinylene carbonate (VC) are also used in IL electrolytes to form a stable Solid Electrolyte Interphase (SEI) on graphite anodes, protecting them from reductive decomposition. [45]
Experimental Protocol: Electrolyte Formulation and Cell Testing
Objective: To prepare and evaluate a safe, non-flammable IL-based electrolyte for LIBs. Materials:
- Ionic Liquid: PYR₁₄TFSI (dried under vacuum at 80°C for 24 h)
- Lithium Salt: LiTFSI
- Co-solvent (optional): Anhydrous Propylene Carbonate (PC)
- Additive (optional): Vinylene Carbonate (VC)
- Cell Components: Li-metal anode, LiMnNiCoO₂ (NMC) or LiFePO₄ (LFP) cathode, separator, coin cell hardware.
Methodology:
- Electrolyte Preparation: In an argon-filled glovebox (H₂O, O₂ < 1 ppm), mix PYR₁₄TFSI and PC in a weight ratio of 80:20. Dissolve LiTFSI salt to a concentration of 1.0 M. Add VC additive at 1-2 wt% of the total electrolyte mass. Stir until a homogeneous solution is obtained. [45]
- Cell Assembly: Assemble CR2032-type coin cells using the prepared electrolyte, a glass microfiber separator, and the chosen electrode pair (e.g., Graphite || LiMNC).
- Flammability Test: A simple qualitative test involves exposing a drop of the electrolyte and a conventional organic carbonate electrolyte to an open flame. The IL-based electrolyte will typically show no ignition, demonstrating superior safety. [45]
- Electrochemical Characterization:
- Cyclic Voltammetry (CV): Scan between 2.5 V and 4.5 V (vs. Li⁺/Li) at a slow scan rate (e.g., 0.1 mV/s) to determine the electrochemical stability window.
- Galvanostatic Cycling: Cycle the cell at various C-rates (e.g., C/10, C/5, 1C) between the voltage limits specific to the cathode material to assess capacity, cycle life, and rate capability.
ILs in Supercapacitors (SCs)
Enhancing Energy Density
For supercapacitors, particularly Electric Double-Layer Capacitors (EDLCs), the energy density is the primary limitation. Since E is proportional to V², employing ILs with their wide voltage windows is one of the most effective strategies to boost energy density. [47] For instance, replacing a standard organic electrolyte (2.7 V window) with an IL offering a 3.5 V window can potentially increase the energy density by over 65%.
Advanced Formulations: Redox-Active and Eutectic ILs
Research is advancing beyond pure ILs to more sophisticated formulations:
- Redox-Additive Electrolytes: The specific capacitance can be dramatically enhanced by incorporating redox-active species into the IL electrolyte. For example, adding dual redox-additives like
[Fe(o-phen)₃]²⁺and[Fe(CN)₆]³⁻to a H₂SO₄-based system has been shown to increase the specific capacitance of N, P co-doped graphene from 290 F g⁻¹ to 865 F g⁻¹. [50] - Eutectic IL Mixtures: To overcome the high viscosity and poor low-temperature performance of some ILs, eutectic mixtures are developed. A mixture of Pip₁₃FSI and Pyr₁₄FSI (1:1 ratio) has been used to extend the operating temperature range of a supercapacitor from -50 °C to 100 °C. [47]
Table 3: Performance of Supercapacitors with Different IL-Based Electrolytes
| Electrolyte System | Electrode Material | Specific Capacitance (F g⁻¹) | Energy Density (Wh kg⁻¹) | Voltage Window (V) |
|---|---|---|---|---|
| PVA/H₂SO₄ (Baseline) | N, P co-doped Graphene (NPGO) | 290 | 6.8 | ~1.0 [50] |
| H₂SO₄ + [Fe(CN)₆]³⁻ | NPGO | 685 | ~15 (estimated) | ~1.0 [50] |
| H₂SO₄ + Dual Redox | NPGO | 865 | 25 | ~1.0 [50] |
| PYR₁₄TFSI (Pure IL) | Activated Carbon | N/A | ~100 (theoretical) | ~3.5 [47] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Reagents and Materials for IL Electrolyte Research
| Item / Reagent | Function/Description | Example in Use |
|---|---|---|
| Pyrrolidinium ILs | Widely used cation family for high-voltage stability. | PYR₁₄TFSI as a base electrolyte for LIBs. [45] |
| Imidazolium ILs | Offer high ionic conductivity. | [bmim][TFSI] for supercapacitor electrolytes. [44] |
| Fluorous Ponytail Electrolytes | Specially designed salts for enhanced solubility in low-polarity media. | NBu₄BARF-6 for high-conductivity solutions in supercritical CO₂. [49] |
| LiTFSI Salt | A common, stable lithium salt with good solubility in ILs. | Lithium source in IL-based LIB electrolytes. [45] |
| Vinylene Carbonate (VC) | SEI-forming additive to protect graphite anodes. | Added at 1-2 wt% to IL electrolytes for LIBs. [45] |
| Mesoporous Silica Template | Used to create nanostructured electrodes or for fundamental studies. | Template for electrodepositing 3-nm metal nanowires from SCFs. [49] |
| Supercritical Fluid Reactor | High-pressure system for SCF electrochemistry or material processing. | Used for SCF-electrodeposition or extraction of electrolytes from spent LIBs. [51] [49] |
Experimental Protocol: Creating a Flexible Supercapacitor with Ionogel Electrolyte
Objective: To fabricate a flexible solid-state supercapacitor using an IL-based polymer gel electrolyte (ionogel). Materials:
- Electrode Material: N, P co-doped graphene (NPGO) or activated carbon.
- Ionic Liquid: [Pyr₁₄][TFSI].
- Gelling Polymer: Poly(vinyl alcohol) (PVA) or a poly(ionic liquid).
- Current Collectors: Flexible carbon cloth or stainless-steel mesh.
Methodology:
- Ionogel Electrolyte Preparation: Dissolve PVA in hot deionized water. Mix the PVA solution with an equal mass of [Pyr₁₄][TFSI] and stir vigorously to form a homogeneous precursor. Pour the mixture into a petri dish and allow it to cool and dry, forming a flexible, freestanding ionogel membrane. [50] [47]
- Electrode Fabrication: Mix the NPGO active material, conductive carbon (e.g., carbon black), and a polymer binder (e.g., PTFE) in a mass ratio of 80:15:5. Roll the mixture into a thin film and press it onto the flexible current collectors.
- Device Assembly: Sandwich the ionogel electrolyte membrane between two identical carbon-based electrodes. Press the stack together to ensure good contact, creating a flexible "sandwich" structure.
- Electrochemical Testing:
- Cyclic Voltammetry: Perform CV at different scan rates (e.g., 10-100 mV/s) within a voltage window of 0-3.5 V. A nearly rectangular CV curve indicates ideal EDLC behavior.
- Galvanostatic Charge-Discharge (GCD): Perform GCD at various current densities to calculate specific capacitance, energy, and power density.
- Flexibility Testing: Repeat the GCD cycles while the device is under mechanical bending to assess performance retention.
Ionic liquids have firmly established themselves as a versatile and high-performance class of electrolytes for next-generation energy storage devices. Their wide electrochemical windows, non-flammability, and exceptional tunability provide a direct path to safer batteries and higher-energy supercapacitors. When contrasted with supercritical fluids, ILs offer a more readily applicable solution that avoids the complexities of high-pressure systems, though SCFs retain unique advantages in materials processing and extraction. [51] [52]
Future research will focus on overcoming the remaining challenges of ILs, primarily their high cost and viscosity. This will involve the synthesis of new, cheaper ionic components, the development of optimized eutectic mixtures, and the creation of advanced composite ionogels for flexible electronics. As these innovations progress, IL-based electrolytes are poised to play a central role in powering the safe, high-energy, and flexible energy storage systems of the future.
Diagrams
IL Electrolyte Design & Performance Relationship
IL vs. SCF Electrolyte Workflow
Supercritical carbon dioxide (scCO2) extraction represents a cornerstone of green extraction technology, utilizing carbon dioxide above its critical temperature (30.978°C) and critical pressure (73.773 bar) to create a solvent with unique properties [53]. In this supercritical state, CO2 exhibits gas-like diffusivity and viscosity combined with liquid-like density and solvating power, enabling exceptional penetration into biological matrices and efficient extraction of target compounds [54]. This technology has gained significant traction across pharmaceutical, nutraceutical, and cosmetic industries as a sustainable alternative to conventional organic solvents, aligning with increasing regulatory pressures and consumer demand for "clean" ingredients without toxic residues [55].
The positioning of scCO2 within the broader context of green solvent technologies reveals a complementary relationship with ionic liquids (ILs). While both are considered environmentally benign alternatives to traditional organic solvents, they operate through fundamentally different mechanisms. Ionic liquids are salts that exist as liquids below 100°C, offering exceptional tunability through cation-anion combinations but facing challenges in purity and environmental impact for earlier generations [56]. In contrast, scCO2 provides a non-toxic, non-flammable platform with easily adjustable solvating power via simple pressure and temperature manipulation, though it shows limitations for polar compounds without modifiers [57]. This technical guide explores the fundamental principles, optimization parameters, and practical implementation of scCO2 technology specifically for extracting active pharmaceutical ingredients and essential oils, while contextualizing its role alongside ionic liquids in sustainable extraction workflows.
Fundamental Principles and Advantages
Mechanism of Action
The exceptional extraction capabilities of scCO2 stem from its unique physicochemical properties in the supercritical state. With near-zero surface tension and gas-like viscosity, scCO2 penetrates deeply into plant matrices, accessing microporous structures typically inaccessible to liquid solvents [57]. The solvating power of scCO2 is primarily governed by density, which can be precisely controlled through pressure adjustment, enabling selective extraction targeting specific compound classes [53]. The mechanism proceeds through several stages: scCO2 diffusion into the plant matrix, dissolution of target compounds from the solid matrix into the supercritical fluid, transport of the solute-scCO2 solution out of the matrix, and finally, precipitation of the extract via depressurization [58].
The extraction efficiency is further enhanced by scCO2's high diffusion coefficients, which significantly exceed those of liquid solvents, resulting in faster extraction kinetics [57]. For lipophilic compounds, scCO2 demonstrates particularly favorable interactions, with the non-polar CO2 molecule effectively solubilizing oils, waxes, and other non-polar phytochemicals through dispersion forces. The absence of strong permanent dipole moments explains scCO2's inherent limitation with highly polar molecules, a challenge typically addressed through the addition of polar co-solvents such as ethanol or water [57].
Comparative Advantages
Table 1: Comparative Analysis of scCO2 Against Traditional Extraction Methods
| Parameter | scCO2 Extraction | Organic Solvent Extraction | Steam Distillation |
|---|---|---|---|
| Solvent Residue | None (CO2 evaporates) [59] | Significant (requires purification) | Water residues possible |
| Thermal Degradation | Minimal (low operating temperatures) [53] | Moderate to high | High (hydrothermal stress) |
| Selectivity | Highly tunable via P/T adjustment [53] | Limited by solvent polarity | Limited to volatile compounds |
| Processing Time | Moderate (4-6 hours for 20lb batch) [59] | Long (including solvent removal) | Variable |
| Environmental Impact | Low (CO2 recycled, no VOCs) [54] | High (solvent disposal, emissions) | Moderate (energy, water use) |
| Capital Cost | High initial investment [55] | Low to moderate | Moderate |
The advantages of scCO2 extraction extend beyond the technical specifications to encompass broader sustainability and product quality considerations. The process eliminates the need for hazardous solvent storage, disposal, and associated environmental concerns [57]. From a product standpoint, the gentle extraction conditions (typically 35-80°C) preserve thermolabile compounds that would degrade under conventional extraction or distillation temperatures, resulting in extracts with more natural aroma and flavor profiles [58]. Furthermore, the supercritical environment acts as a sterilization medium, reducing microbial loads, molds, and mildews without additional processing steps [59].
The environmental credentials of scCO2 are quantified through green metrics such as the E-factor (kg waste per kg product), with studies demonstrating significantly improved performance compared to solvent-based systems [55]. When combined with closed-loop CO2 recycling systems, the technology aligns with circular economy principles, particularly when applied to biomass valorization streams such as fruit pomaces and agricultural by-products [55] [54].
Technical Implementation and Optimization
Critical Process Parameters
Table 2: Key Operational Parameters for Targeted Compound Classes
| Target Compound Class | Typical Pressure Range (bar) | Typical Temperature Range (°C) | Co-solvent Recommendations | Application Examples |
|---|---|---|---|---|
| Essential Oils | 100-300 [58] | 35-50 [58] | None typically | Lavender, peppermint, citrus oils [53] |
| Lipophilic Actives | 250-500 [58] | 40-60 | Ethanol (5-10%) | Cannabinoids, carotenoids [55] [59] |
| Polar Phenolics | 300-500 | 50-70 | Water/ethanol mixtures (10-20%) | Flavonoids, anthocyanins [55] |
| Alkaloids | 200-400 | 40-60 | Methanol/ethanol (5-15%) | Caffeine [53] [58] |
Successful implementation of scCO2 extraction requires careful optimization of three fundamental parameters: pressure, temperature, and flow rate. Pressure exerts the most significant influence on solvating power, with increased pressure dramatically enhancing solvent density and consequently its ability to dissolve target compounds [57]. Temperature exhibits a dual effect, simultaneously reducing solvent density while increasing vapor pressure of solutes, creating compound-specific optimization requirements [57]. Flow rate primarily affects the kinetics of extraction, with higher flows reducing external mass transfer limitations but potentially decreasing extraction efficiency if insufficient contact time is provided.
The role of co-solvents represents another critical optimization parameter, particularly for polar active ingredients. Modifiers such as ethanol, methanol, or water in concentrations typically ranging from 1-20% can dramatically enhance solubility of polar compounds like glycosides and polyphenols by introducing hydrogen-bonding capacity to the solvent system [57]. Recent research has demonstrated the effectiveness of CO2-expanded deep eutectic solvents for challenging polar molecules like crocin from gardenia fruits [13].
Material Preparation and Pretreatment
Optimal extraction efficiency begins with appropriate biomass preparation. Particle size reduction through milling increases surface area and reduces internal mass transfer limitations, though excessive comminution can cause channeling in the extraction bed [58]. Moisture content represents another crucial factor, with most applications benefiting from intermediate moisture levels (10-40%) that facilitate mass transfer without creating biphasic systems [57] [58].
Advanced pretreatment strategies can significantly enhance scCO2 extraction performance. Enzyme-assisted extraction (EAE) using cellulases, pectinases, and hemicellulases disrupts plant cell walls, increasing accessibility of intracellular compounds [55]. Physical methods like ultrasound-assisted extraction (UAE) and pulsed electric field (PEF) create micro-fractures in tissues through cavitation and electroporation, respectively, reducing extraction time and improving yields [55]. These hybrid approaches represent the cutting edge of scCO2 technology, enabling milder downstream conditions and improved sustainability profiles.
Experimental Protocol for Essential Oil Extraction
The following detailed protocol describes the extraction of essential oils from plant materials, providing a reproducible methodology suitable for research and development purposes.
Step-by-Step Procedure
Raw Material Preparation: Select high-quality plant material (e.g., lavender flowers, peppermint leaves) and reduce particle size to 0.5-2mm using a cutting mill or similar device. Avoid excessive heating during milling to preserve volatile compounds.
Moisture Adjustment: Determine initial moisture content (gravimetrically) and adjust to optimal level (typically 15-25% for most herbs) by adding purified water or partial air-drying. Homogenize thoroughly and allow equilibration for 12-24 hours at 4°C.
Extraction Vessel Loading: Pack the prepared biomass evenly into the extraction vessel, using glass wool or inert packing material to eliminate dead volumes. Ensure uniform packing density to prevent channeling during CO2 flow.
System Pressurization: Set extraction temperature to 40°C and gradually introduce CO2 until reaching the target pressure (100-300 bar, depending on target compounds). Maintain conditions for 15-20 minutes to achieve thermal equilibrium.
Dynamic Extraction: Initiate CO2 flow at 1-3 kg/h, maintaining constant pressure and temperature. Continue extraction for 2-4 hours, monitoring extract accumulation. The high diffusivity of scCO2 allows penetration into small pores inaccessible to liquids [57].
Fraction Collection: Direct the CO2-extract mixture to the separation vessel maintained at lower pressure (50-60 bar) and temperature (25-50°C), triggering precipitation of the essential oil due to reduced solvating power [58]. Collect the fraction in amber glass vessels under inert atmosphere.
Solvent Recycling: Route the CO2 gas through a condensation unit and back to the storage tank for reuse in subsequent extraction cycles, minimizing environmental impact and operational costs [59].
Post-processing: Weigh the extracted essential oil and analyze by GC-MS for composition verification. Store under inert atmosphere at -20°C for long-term preservation.
Troubleshooting and Optimization
Common challenges include low yield (address by increasing pressure or adding co-solvent), inadequate selectivity (optimize temperature-pressure combination), or equipment clogging (improve biomass pretreatment). For complex matrices, multi-stage fractionation employing sequential pressure reduction can separate compounds by polarity [53]. Validation should include mass balance calculations and reproducibility assessment across multiple batches.
Integration with Ionic Liquids in Pharmaceutical Applications
Comparative Solvent Properties
Table 3: Property Comparison: scCO2 vs. Ionic Liquids for Pharmaceutical Extraction
| Property | Supercritical CO2 | Ionic Liquids (3rd Generation) |
|---|---|---|
| Tunability | Moderate (via P/T/density) [53] | High (via cation/anion selection) [56] |
| Polarity Range | Non-polar to low-polarity (expandable with modifiers) | Full spectrum (hydrophilic to hydrophobic) [56] |
| Thermal Stability | Excellent | Good to excellent [56] |
| Toxicity Profile | Non-toxic [59] | Variable (3rd generation: low toxicity) [56] |
| Biodegradability | Natural component of cycles | Tunable (choline-based: highly biodegradable) [56] |
| Pharmaceutical Applications | Extraction of lipophilic actives, essential oils [55] | Solubilization of APIs, drug delivery, synthesis [60] |
The complementary nature of scCO2 and ionic liquids creates opportunities for hybrid processing approaches. While scCO2 excels at extracting non-polar compounds under mild conditions, ionic liquids demonstrate exceptional capability for dissolving polar pharmaceuticals, including poorly soluble active pharmaceutical ingredients (APIs) [56]. Third-generation ILs based on natural cations like choline offer low toxicity and good biodegradability, expanding their pharmaceutical applicability [56].
Recent advances have explored the combination of these technologies, using scCO2 as an antisolvent for precipitation of IL-processed pharmaceuticals or creating SC-CO2 + IL biphasic systems for selective extraction [5]. The low solubility of most ILs in scCO2 enables clean separation, while the addition of polar co-solvents can tune miscibility for specific applications [5].
Hybrid Approaches and Synergistic Applications
Emerging research demonstrates promising hybrid methodologies that leverage the strengths of both technologies:
Sequential Extraction Processes: Initial scCO2 extraction of lipophilic compounds followed by IL-based extraction of polar residues from the same biomass, achieving comprehensive fractionation [55] [56].
Particle Engineering: Combination of IL solutions as crystallization media with scCO2 antisolvent precipitation (SAS) to produce engineered pharmaceutical particles with enhanced bioavailability [53].
Integrated Reaction-Extraction Systems: Employing ILs as reaction media for chemical transformations of natural extracts followed by scCO2 extraction of products, enabling continuous processes [56].
Analytical Applications: Using ILs as stationary phases in chromatography coupled with scCO2 extraction for integrated extraction-analysis platforms for complex botanical samples.
These hybrid approaches align with the pharmaceutical industry's movement toward process intensification and continuous manufacturing, potentially reducing processing time, energy consumption, and environmental impact compared to traditional batch processes.
Analytical Methodologies and Quality Control
Advanced analytical techniques are essential for characterizing scCO2 extracts and validating process efficiency. High-performance liquid chromatography coupled with mass spectrometry (LC-MS/MS) provides quantitative analysis of target active compounds, while comprehensive two-dimensional gas chromatography (GC×GC) offers superior resolution for complex essential oil profiles [55]. Nuclear magnetic resonance (NMR) spectroscopy serves as a powerful tool for structural elucidation of novel compounds and extract fingerprinting.
Quality control protocols should include determination of extraction yield, compound-specific quantification, and residual solvent analysis (though scCO2 extracts typically show no detectable solvent residues) [59]. For pharmaceutical applications, Certificates of Analysis (COAs) should be established, documenting compound purity, potential contaminants, and biological activity [55]. Accelerated stability studies under International Council for Harmonisation (ICH) guidelines ensure product shelf life.
Process analytical technology (PAT) implementation enables real-time monitoring of critical quality attributes, with in-line spectroscopy and flow cytometry providing immediate feedback on extraction efficiency and potential degradation. These quality-by-design approaches facilitate regulatory compliance and ensure consistent product quality across production batches.
Supercritical CO2 extraction represents a mature, sustainable technology for obtaining high-quality active ingredients and essential oils from botanical sources. Its exceptional selectivity, minimal environmental impact, and preservation of thermolabile compounds make it particularly valuable for pharmaceutical and nutraceutical applications where product purity and biological activity are paramount. The ongoing technological evolution focuses on hybrid approaches combining scCO2 with enzymatic, physical, and alternative solvent pretreatments to further enhance efficiency and sustainability.
Within the broader context of green extraction technologies, scCO2 complements rather than competes with advanced ionic liquids, with each technology occupying distinct but overlapping application spaces. Future development will likely see increased integration of these platforms, leveraging the tunability of ILs with the clean extraction capabilities of scCO2 in innovative biorefinery concepts. As regulatory frameworks evolve and consumer demand for natural products grows, scCO2 technology will play an increasingly vital role in sustainable pharmaceutical and cosmetic manufacturing, supported by continuous improvements in equipment design, process control, and hybrid system integration.
Overcoming Practical Challenges: From Solubility Prediction to Process Control
Supercritical fluid (SCF) technology, particularly using carbon dioxide (SC-CO₂), represents a cornerstone of green chemistry in pharmaceutical and extraction industries. The efficiency of these processes is predominantly governed by the precise manipulation of temperature, pressure, and co-solvent addition. This whitepaper provides an in-depth technical examination of these parameters, framed within the advancing research on ionic liquids (ILs) and their interactions with SCFs. It details experimental protocols, presents optimized data, and introduces advanced predictive models, serving as a comprehensive guide for researchers and drug development professionals aiming to harness and optimize SCF-based applications.
Supercritical fluids are substances heated and compressed above their critical point (Tc and Pc), where they exhibit unique properties such as gas-like diffusivity and viscosity combined with liquid-like density. Supercritical carbon dioxide (SC-CO₂) is the most widely employed SCF due to its mild critical parameters (Tc = 304.1 K, Pc = 7.4 MPa), non-toxicity, non-flammability, and low cost [12]. However, its non-polar nature limits its ability to dissolve polar compounds, a significant constraint in pharmaceutical applications where many active pharmaceutical ingredients (APIs) are polar molecules [61].
Ionic liquids (ILs), often termed "designer solvents," are salts that are liquid below 100°C. They are characterized by negligible vapor pressure, high thermal stability, and tunable physicochemical properties determined by their cation-anion combinations [20]. Their role in SCF technology is dual: they can act as target solutes for impregnation into porous materials or as co-solvents to enhance the solubility of other compounds in SC-CO₂. The integration of ILs with SCF processes exemplifies a synergistic approach, leveraging the strengths of both green solvent systems [62] [22].
Fundamental Principles of Parameter Effects
The solvating power of an SCF is directly tied to its density, which is a function of temperature and pressure. Understanding these relationships is fundamental to process optimization.
- Pressure Effects: Increasing pressure at constant temperature significantly increases the density of the SC-CO₂, thereby enhancing its solvating power. This is because higher pressure forces molecules closer together, increasing the solvent's density and its ability to interact with solute molecules. For instance, the solubility of Maprotiline hydrochloride in SC-CO₂ was observed to increase with rising pressure across all temperatures studied [63].
- Temperature Effects: The influence of temperature is more complex due to its two competing effects. At constant pressure, increasing temperature reduces solvent density (which tends to decrease solubility) but simultaneously increases the vapor pressure of the solute (which tends to increase solubility). The dominance of one effect over the other creates a crossover pressure region [63].
- Co-solvent Effects: Polar co-solvents, or modifiers, are added in small quantities (typically 1-15 mol%) to SC-CO₂ to dramatically increase its polarity and solubility for polar compounds. Ethanol is a preferred co-solvent due to its low toxicity and environmental acceptability. The modifier enhances solubility by interacting with the analyte to promote desorption and by improving the polarity of the SC-CO₂ mixture [61] [64].
Quantitative Optimization of SCF Parameters
Optimization for Natural Product Extraction
The extraction of flavonoids from waste hops (SC-CO₂ extracted hops) was systematically optimized using single-factor and orthogonal experiments. The results demonstrated that a combination of 50°C, 25 MPa, a solvent-to-material ratio of 50%, and 80% ethanol concentration yielded the maximum flavonoid extraction of 7.8 mg/g. The primary flavonoid extracted was identified as xanthohumol [61].
Table 1: Optimal SFE Conditions for Phenolic Compounds from Different Plant Matrices
| Plant Material | Target Compound | Optimal Pressure | Optimal Temperature | Optimal Co-solvent (Ethanol) | Optimal Co-solvent Concentration | Maximum Yield | Source |
|---|---|---|---|---|---|---|---|
| Waste Hops | Flavonoids (Xanthohumol) | 25 MPa | 50 °C | 80% | 50% (solvent-to-material) | 7.8 mg/g | [61] |
| Labisia pumila Leaves | Phenolic Acids (Gallic, Caffeic) | 28.3 MPa | 32 °C | 78% (v/v) in water | 16% (v/v) | 14.05% (g/g) | [64] |
Similarly, optimization of phenolic compound extraction from Labisia pumila leaves using Response Surface Methodology (RSM) confirmed the critical role of co-solvent concentration and temperature. The optimal conditions were identified as 283 bar, 32°C, with 78% (v/v) ethanol-water as a co-solvent at a concentration of 16% (v/v) [64].
Optimization for Pharmaceutical Solubility
The solubility of pharmaceutical compounds in SC-CO₂ is a critical parameter for drug particle engineering. The solubility of Maprotiline hydrochloride, an anti-depressant drug, was measured across a range of temperatures (311-343 K) and pressures (120-300 bar). The solubility in mole fraction ranged from 3.42 × 10⁻⁵ to 1.17 × 10⁻³, showing a strong dependency on both parameters [63].
Table 2: Solubility of Maprotiline Hydrochloride in SC-CO₂ under Varying Conditions
| Temperature (K) | Pressure (bar) | Solubility (mole fraction) |
|---|---|---|
| 311 | 120 | 3.42 × 10⁻⁵ |
| 311 | 300 | 1.17 × 10⁻³ |
| 323 | 120 | 4.85 × 10⁻⁵ |
| 323 | 300 | 9.41 × 10⁻⁴ |
| 343 | 120 | 1.21 × 10⁻⁴ |
| 343 | 300 | 7.26 × 10⁻⁴ |
The data from such studies are vital for designing processes like the Rapid Expansion of Supercritical Solutions (RESS), which requires the drug to be soluble in the SCF [63].
Experimental Protocols and Methodologies
Protocol for SFE of Bioactive Compounds
A standard protocol for the supercritical fluid extraction of phenolic compounds, as applied to Labisia pumila, involves the following steps [61] [64]:
- Sample Preparation: Plant material is washed, dried at room temperature to a constant moisture content (~6% w/w), pulverized, and sieved to a specific particle size range (e.g., 0.5-0.8 mm). The powder is stored in airtight containers at 4°C before use.
- SFE System Setup: The system typically includes a CO₂ pump, a co-solvent pump, an extraction vessel housed in an oven, a back-pressure regulator (BPR), and a sample collector. The liquefied CO₂ is chilled to maintain its liquid state before pumping.
- Extraction Procedure:
- The extraction vessel is packed with a known mass of plant material.
- The system is brought to the desired temperature and pressure.
- The extraction is often performed in two modes: static (e.g., 30 min) to allow for equilibrium, followed by dynamic extraction (e.g., 240 min) at a fixed total flow rate.
- Extracts are collected at regular intervals in a collector at atmospheric pressure.
- Post-Processing and Analysis: The collected extracts are dried to completion in an oven at 40°C. The yield is calculated, and the specific compounds of interest are identified and quantified using analytical techniques like High-Performance Liquid Chromatography (HPLC).
Protocol for Drug Solubility Measurement
The solubility of a solid drug like Maprotiline hydrochloride in SC-CO₂ can be determined using a static method [63]:
- Apparatus: A high-pressure equilibrium vessel equipped with a view window, a magnetic stirrer, a thermometer, a pressure transducer, and a sampling valve.
- Procedure:
- A known amount of the drug is placed in the vessel.
- The vessel is purged with CO₂, then pressurized to the desired level and heated to the target temperature.
- The mixture is stirred continuously for several hours to ensure equilibrium is reached.
- After equilibration, stirring is stopped to allow the undissolved solid to settle.
- A small volume of the saturated SC-CO₂ phase is sampled via the valve and expanded into a collection solvent. The amount of drug collected is determined by UV-Vis spectrophotometry or HPLC.
- Data Correlation: The experimental solubility data is typically modeled using equations of state or semi-empirical correlations to predict behavior under various conditions.
Advanced Modeling and Prediction Techniques
Accurately predicting solubility is essential for efficient process design, reducing reliance on costly and time-consuming experiments.
Thermodynamic Models
- Equations of State (EoS): The Peng-Robinson (PR) EoS is widely used for predicting solubilities in SC-CO₂ systems. It has been applied to model the solubility of both drugs and ILs. For ILs, which have extremely low volatility, critical properties required for EoS can be predicted using molecular dynamics (MD) simulations [22] [65] [63].
- Advanced EoS Models: For more accurate predictions, especially for heavy molecules like ILs, lattice-based models such as the ε-modified Sanchez-Lacombe EoS (ε-mod SL-EoS) have shown superior performance over traditional cubic EoS, achieving an average absolute relative deviation (AARD) of 11.0% in ternary systems [5].
- Semi-Empirical Models: Density-based models like Chrastil's equation are popular for their simplicity and reasonable accuracy in correlating solid solubility in SC-CO₂. In the case of Maprotiline hydrochloride, the Chrastil model provided an AARD of 5.49% [63].
Machine Learning Approaches
Machine learning (ML) models have emerged as powerful tools for predicting drug solubility in SC-CO₂, capable of learning complex, non-linear relationships without predefined physical equations. A recent study comparing CatBoost, XGBoost, LightGBM, and Random Forest models on 68 drugs demonstrated that the XGBoost algorithm achieved the highest accuracy (R² = 0.9984, RMSE = 0.0605) [12]. These models use input features such as temperature, pressure, CO₂ density, and drug properties (molecular weight, melting point, critical properties), offering rapid and reliable predictions for process optimization.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for SCF Process Research
| Reagent/Material | Function in SCF Process | Example Specifications |
|---|---|---|
| Carbon Dioxide (CO₂) | Primary supercritical solvent. | Commercial grade, 99.9% purity. |
| Ethanol | Polar co-solvent (modifier) to enhance SC-CO₂ polarity and solubility. | Analytical grade, often used in concentrations of 70-90% in water. |
| Ionic Liquids | Target solutes for impregnation; potential co-solvents or catalysts. | e.g., [C₄mim][BF₄]; tailored cation-anion pairs for specific properties. |
| Active Pharmaceutical Ingredients (APIs) | Target solutes for drug particle engineering. | e.g., Maprotiline Hydrochloride; purity >98%. |
| Plant Material | Matrix for extraction of bioactive compounds. | Dried, powdered, and sieved to specific particle size (e.g., 40-mesh). |
| High-Pressure Vessels | Containment for SCF processes at high P and T. | Stainless steel, rated for pressures up to 50 MPa. |
| Back-Pressure Regulator | Maintains system pressure during dynamic extraction. | Electronically controlled, precise pressure maintenance. |
| HPLC System with UV Detector | Analysis of extract composition and solute quantification. | C18 reverse-phase column, UV detection at specific wavelengths. |
The optimization of supercritical fluid processes is a multifaceted endeavor that hinges on the scientific understanding and precise control of temperature, pressure, and co-solvent use. The integration of ionic liquids into this field opens new avenues for creating sophisticated and green pharmaceutical and separation technologies. By employing structured experimental designs like RSM, and leveraging advanced predictive tools ranging from improved equations of state to machine learning, researchers can efficiently navigate the complex parameter space to achieve optimal outcomes. As these methodologies continue to evolve, they will undoubtedly accelerate the development of more efficient, sustainable, and innovative industrial processes.
The pharmaceutical industry's growing commitment to green chemistry has propelled supercritical carbon dioxide (scCO2) to the forefront as a sustainable alternative to traditional organic solvents. With its low critical temperature (304.1 K) and moderate critical pressure (7.4 MPa), scCO2 offers unique advantages for drug processing, including minimal environmental impact, innate non-toxicity, and ease of separation from final products [66] [12]. These properties make it exceptionally valuable for advanced pharmaceutical applications such as particle engineering, drug extraction, purification, and the development of advanced drug delivery systems (DDSs) using methods like RESS, SAS, and PGSS [12]. However, a significant challenge impedes its widespread adoption: the poor solubility of many pharmaceutical compounds in scCO2, particularly those falling into BCS classes II and IV [12].
Accurately determining drug solubility in scCO2 is crucial for efficient process design, yet experimental measurement remains costly, time-consuming, and impractical to perform across the vast range of possible temperature and pressure conditions [12]. This limitation has driven the development of predictive modeling approaches. While traditional methods like equations of state (EoS) and semi-empirical correlations have been used for decades, they often rely on simplifying assumptions that compromise accuracy, particularly for complex molecular structures [66] [12]. In this context, machine learning (ML) has emerged as a powerful alternative, capable of learning complex, non-linear relationships between drug properties, process conditions, and solubility outcomes directly from experimental data, thereby offering a faster, more cost-effective, and highly accurate predictive tool [12].
This technical guide explores the paradigm shift from traditional experimental methods to ML-driven prediction of drug solubility in scCO2. It further frames this advancement within the broader research landscape of alternative green solvents, particularly highlighting the complementary role of ionic liquids (ILs)—designer solvents with highly tunable properties that are also revolutionizing pharmaceutical synthesis and formulation [56].
Machine Learning vs. Traditional Modeling Approaches
Limitations of Traditional Thermodynamic and Empirical Models
Traditional methods for predicting drug solubility in scCO2 primarily fall into two categories: thermodynamic models and semi-empirical density-based correlations.
Equations of State (EoS): Cubic EoS, such as Peng-Robinson (PR), are widely used due to their relative simplicity. However, their performance can be poor at high pressures, and they require critical properties and acentric factors for the solid solute, which are often unavailable for complex drug molecules [22] [12]. More sophisticated non-cubic EoS, like the Perturbed Chain-Statistical Associating Fluid Theory (PC-SAFT), offer improved accuracy by better accounting for molecular interactions but demand significant computational resources and expertise [66]. For instance, in modeling methadone hydrochloride solubility, the PC-SAFT EoS achieved an Absolute Average Relative Deviation (AARD) of 9.73%, which was less accurate than the best semi-empirical model [66].
Semi-Empirical Correlations: Models such as Chrastil, Méndez-Santiago-Teja, and Bartle et al. correlate solubility with the density of scCO2. They are simpler to implement but are often system-specific and struggle to generalize beyond the compounds and conditions they were derived from [66] [12]. A study on six drugs found that while these models are useful, they were consistently outperformed by artificial neural networks (ANNs) in predictive accuracy [12].
The Machine Learning Paradigm
Machine learning models overcome many limitations of traditional approaches by directly learning complex, non-linear relationships from data without relying on pre-defined physical equations [12]. This data-driven approach allows ML models to achieve higher predictive accuracy and better generalization across diverse drug-scCO2 systems.
Key advantages of ML models include:
- High Predictive Accuracy: ML models consistently demonstrate superior performance metrics compared to traditional models.
- Computational Speed: Once trained, ML models can generate solubility predictions in seconds, drastically reducing the time required for process design and optimization [12].
- Handling of Complex Data: ML can effectively process a wide range of input features, from operational conditions (temperature, pressure) to drug-specific properties (molecular weight, melting point) and even atomic-level contributions [67].
Table 1: Comparison of Predictive Modeling Approaches for Drug Solubility in scCO2
| Model Type | Examples | Key Advantages | Key Limitations | Reported Accuracy |
|---|---|---|---|---|
| Cubic EoS | Peng-Robinson (PR) | Based on thermodynamic principles; well-established. | Poor performance at high pressures; needs solute properties. | Varies; can be low for complex drugs. |
| Advanced EoS | PC-SAFT | Accounts for complex molecular interactions; more accurate. | Computationally intensive; requires more parameters. | AARD ~9.73% for methadone HCl [66] |
| Semi-Empirical | Chrastil, Bartle | Simple to implement; requires only T, P, and density. | System-specific; poor generalization. | AARD ~6.71% (Chrastil for methadone HCl) [66] |
| Machine Learning | XGBoost, ANN, LSSVM | High accuracy; handles non-linear relationships; fast prediction. | Requires large, high-quality datasets for training. | R² up to 0.9984, AARD as low as 0.706% [12] [67] |
Key Machine Learning Models and Performance
Prominent Machine Learning Algorithms
Research has validated a variety of ML algorithms for predicting drug solubility in scCO2. Tree-based ensemble methods and neural networks have shown particularly strong performance.
Tree-Based Ensemble Methods:
- XGBoost (eXtreme Gradient Boosting): This model has delivered state-of-the-art performance in several studies. One evaluation using 1726 data points from 68 drugs reported that XGBoost achieved an R² of 0.9984 and a Root Mean Square Error (RMSE) of 0.0605, outperforming other models [12].
- CatBoost and LightGBM: These are other gradient-boosting frameworks that offer high accuracy and computational efficiency. They are often used in ensemble models or for comparative studies [12] [11].
- Random Forest (RF): An ensemble of decision trees that reduces overfitting and provides robust predictions, though it may be slightly less accurate than advanced boosting algorithms [12].
Neural Networks and Hybrid Models:
- Artificial Neural Networks (ANNs) and Deep Neural Networks (DNNs): These models are powerful for capturing complex, non-linear patterns. A hybrid Quantitative Structure-Property Relationship (QSPR) model enhanced with ANNs on a large dataset of 3971 data points achieved a high correlation coefficient (r = 0.9761) [12].
- Least Squares Support Vector Machines (LSSVM): When combined with the atomic contribution (AC) method, which breaks down drug molecules into their constituent atoms, LSSVM has demonstrated exceptional accuracy. One study on 86 drugs reported an AARD of 7.20% and an R² of 0.99 [67].
- Ensemble Models: Combining multiple ML models can further enhance predictive performance. An ensemble of XGBoost, LightGBM, and CatBoost optimized by the Hippopotamus Optimization Algorithm (HOA) achieved an R² of 0.9920 for predicting the solubility of four specific drugs [11].
Input Features and Data Requirements
The predictive power of ML models hinges on the selection of relevant input features. Commonly used features can be categorized as follows:
- Process Conditions: Temperature (T), Pressure (P), and scCO2 density (ρ). These are the most fundamental inputs, as they directly govern the solvating power of scCO2 [12] [67].
- Drug-Specific Physicochemical Properties: Critical temperature (Tc), critical pressure (Pc), acentric factor (ω), molecular weight (MW), and melting point (Tm). These properties help the model distinguish between different drug molecules [12].
- Structural and Atomic Descriptors: The Atomic Contribution (AC) method uses the count of each atom type in a drug molecule as input, providing a simple yet powerful way to encode molecular structure without complex descriptors [67]. More advanced molecular descriptors can also be used in QSPR models.
Table 2: Essential Input Features for Machine Learning Models Predicting Drug Solubility in scCO2
| Feature Category | Specific Features | Role in Solubility Prediction |
|---|---|---|
| Process Conditions | Temperature (T), Pressure (P) | Directly control the solvating power and density of scCO2. |
| ScCO2 Property | Density (ρ) | Often a strong direct correlate of solubility in empirical models. |
| Drug Thermodynamic Properties | Critical Temperature (Tc), Critical Pressure (Pc), Acentric Factor (ω) | Describe the volatile behavior and thermodynamic state of the solute. |
| Drug Physical Properties | Molecular Weight (MW), Melting Point (Tm) | Relate to the energy required to break the solid lattice of the drug. |
| Drug Structural Features | Atomic Contributions, Molecular Descriptors | Encode the chemical structure and functional groups that influence molecular interactions. |
Experimental Protocols and Data Generation for ML
Standardized Experimental Solubility Measurement
The reliability of any ML model depends on the quality of its training data. The following outlines a standard analytical (dynamic) method for experimentally measuring drug solubility in scCO2, as exemplified in the study of methadone hydrochloride [66].
Apparatus and Workflow: The core of the setup is a high-pressure equilibrium vessel. The system is constructed from stainless steel (SS-316) capable of withstanding pressures up to 400 bar. Key components include:
- A refrigerator to liquefy CO2 (at ~253 K) before pumping.
- A high-pressure pump to deliver liquefied CO2.
- An equilibrium cell (extraction vessel) loaded with the pure drug, housed in an oven for temperature control.
- A micrometering valve at the outlet to maintain system pressure and facilitate the expansion of the scCO2-drug solution.
- A sampling system and UV-Vis spectrophotometer for quantifying the dissolved solute.
Detailed Methodology:
- System Loading and Equilibration: A known amount of the pure drug (e.g., ~2 g) is placed in the equilibrium cell. The system is sealed, and temperature is set using the oven. CO2 is pumped into the cell until the target pressure is achieved.
- Equilibration: The system is maintained at constant temperature and pressure for a sufficient duration (typically 30-120 minutes) to ensure equilibrium between the solid drug and the scCO2 phase. The content is stirred if possible to enhance mass transfer.
- Sampling and Trapping: The scCO2 stream, now saturated with the drug, is slowly expanded through the micrometering valve. The rapid pressure drop causes the CO2 to turn into a gas, precipitating the dissolved drug. The solute is collected in a trap, often containing a solvent like methanol or water.
- Quantification: The amount of collected drug is quantified using an appropriate analytical technique. UV-Vis spectrophotometry is common, where the absorbance of the solution in the trap is measured and compared to a pre-established calibration curve to determine the concentration and, consequently, the total mass of the drug collected.
- Solubility Calculation: The mole fraction solubility (y) of the drug in scCO2 is calculated using the equation: ( y = n{solute} / (n{solute} + n{CO2}) ) where ( n{solute} ) is the number of moles of drug collected, and ( n_{CO2} ) is the number of moles of CO2 that passed through the system, determined by a flow meter or the total mass of CO2 used.
Diagram 1: Experimental workflow for measuring drug solubility in scCO2.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Materials and Reagents for scCO2 Solubility Research
| Item | Specification/Example | Function in Research |
|---|---|---|
| High-Purity CO₂ | 99.99% purity, supplied in gas cylinders [66] | The primary supercritical fluid solvent. |
| Pharmaceutical Solute | High-purity drug (e.g., Methadone HCl, ≥99.7%) [66] | The compound whose solubility is being measured. |
| Equilibrium Cell / Vessel | Stainless steel (e.g., SS-316), rated for high pressure (e.g., 400 bar) [66] | Contains the drug and scCO2 under high pressure and temperature for equilibration. |
| Analytical Solvent | High-purity solvent (e.g., Deionized water, Methanol) [66] | Used to trap and dissolve the drug after expansion for quantification. |
| Analytical Instrument | UV-Vis Spectrophotometer [66] | Quantifies the concentration of the dissolved drug in the trapping solvent. |
| High-Pressure Pump | - | Pumps and compresses CO2 to supercritical conditions. |
| Temperature-Controlled Oven | - | Houses the equilibrium cell to maintain a constant, precise temperature. |
Contextualizing ML in Green Solvent Research: Ionic Liquids vs. Supercritical Fluids
The application of ML for property prediction is a significant trend across the field of green solvent research, with parallel developments occurring in the study of ionic liquids (ILs). ILs, being salts liquid below 100°C, are another class of highly tunable, non-volatile green solvents with immense potential in pharmaceuticals [56] [68].
Ionic Liquids in Pharmaceuticals: ILs are being extensively researched for drug synthesis (as solvents and catalysts), drug analysis, drug solubilization, and drug delivery. Their high tunability allows them to be designed to address problems of drug polymorphism, poor solubility, and low bioavailability [56]. A key application is the creation of "Dual Active Ionic Liquids," where both the cation and anion have pharmaceutical activity, combining the actions of two drugs into a single ionic API [68].
ML for IL Property Prediction: Just as with scCO2 systems, predicting key properties of ILs (e.g., viscosity, melting point, and gas solubility) is crucial for their design and application. ML models, including ANNs and Deep Neural Networks (DNNs), are being successfully employed to predict these properties based on the structure of the IL's constituent ions [69]. For example, deep learning models like ANN and LSTM have been used to predict CO2 solubility in ILs with high accuracy (R² > 0.985) for carbon capture applications, demonstrating a methodology analogous to drug solubility prediction in scCO2 [70].
Complementary Roles and Convergent Methodologies: While scCO2 and ILs serve different primary functions—scCO2 as a reaction and processing medium and ILs as versatile, designable solvents and materials—they both represent pillars of green chemistry in pharmaceuticals. The emergence of ML as a powerful predictive tool is a common thread that accelerates research and application for both. This convergence highlights a broader shift in materials science towards data-driven design and discovery.
Diagram 2: ML as a unifying tool for green solvent R&D.
The adoption of machine learning for predicting drug solubility in supercritical CO2 marks a significant leap forward for pharmaceutical process development. By providing a rapid, accurate, and cost-effective computational alternative to costly and time-consuming experiments, ML models like XGBoost, ANN, and LSSVM empower researchers to efficiently design and optimize scCO2-based processes such as particle engineering and extraction. This capability is crucial for leveraging the full potential of scCO2 as a green solvent, particularly for hard-to-formulate BCS Class II and IV drugs.
Furthermore, the application of these advanced data-driven methodologies extends beyond scCO2, finding a strong parallel in the development and application of ionic liquids for pharmaceutical and biomedical uses. The convergent adoption of ML across different green solvent platforms underscores a fundamental transformation in materials science and chemical engineering. As experimental datasets continue to grow in both size and quality, the integration of machine learning will undoubtedly become a standard, indispensable component of the researcher's toolkit, accelerating the transition towards more sustainable and efficient pharmaceutical manufacturing.
Ionic liquids (ILs), salts with melting points below 100°C, have emerged as promising materials for electrochemical devices, synthesis, and separation processes due to their remarkable properties including non-flammability, thermal stability, and wide electrochemical windows [71] [24] [72]. However, their practical implementation faces a significant challenge: ILs typically exhibit viscosities ranging from 10 to 10,000 mPa·s, which is substantially higher than the 0.1-100 mPa·s range of conventional organic solvents [73] [74]. This high viscosity directly impedes ion mobility, reducing ionic conductivity and limiting mass transport rates in applications ranging from battery electrolytes to catalytic systems [71] [75].
The origin of this challenge lies in the fundamental nature of ILs as concentrated electrolytes where strong electrostatic interactions and hydrogen bonding between ions create correlated ion networks that resist flow [71] [73]. While classical electrolyte theories like the Nernst-Einstein relationship suggest inverse scaling between viscosity and conductivity, ILs often deviate significantly from these predictions due to their complex nanostructured environments [71]. Understanding and addressing this viscosity-conductivity trade-off is therefore essential for advancing IL applications across energy storage, chemical processing, and pharmaceutical development.
Theoretical Frameworks: Modeling Transport in Ionic Liquids
Beyond Classical Theories: Correlated Ion Transport
Classical hydrodynamic models such as the Nernst-Einstein and Stokes-Einstein relationships often fail to accurately predict IL behavior because they assume idealized dilute solutions where ion-ion interactions are negligible [71]. In concentrated IL systems, ion correlations and nanostructuring dominate transport mechanisms. The modified Arrhenius model has demonstrated superior performance in describing temperature-dependent conductivity in ILs:
[ \Lambda = A{\text{Arr}} \exp\left(\frac{-Ea}{RT}\right) ]
where (\Lambda) represents molar conductivity, (A{\text{Arr}}) is a pre-exponential factor, (Ea) is the activation energy, (R) is the gas constant, and (T) is temperature [71]. This model conceptualizes ion transport as a thermally activated hopping process between coordination sites, revealing activation energy barriers typically between 20-30 kJ·mol⁻¹ for ILs [71].
For systems near glass transition temperatures, the Vogel-Tammann-Fulcher (VTF) equation provides enhanced modeling:
[ \Lambda = A{\text{VTF}} T^{-1/2} \exp\left(\frac{-Ea}{R(T-T_0)}\right) ]
where (T_0) is an empirical temperature offset typically 50-70 K below the glass transition temperature [71].
Machine Learning and Predictive Modeling
Recent advances employ machine learning (ML) to predict IL viscosity and conductivity from molecular descriptors. White-box ML approaches like Genetic Programming (GP) and Group Method of Data Handling (GMDH) have achieved notable success, with GMDH models demonstrating coefficients of determination (R²) of 0.98 for viscosity prediction across extensive datasets [74]. Hybrid models integrating physical theories like COSMO-RS with ML corrections have reduced prediction errors from 52.45% to 1.54% for viscosity calculations [76].
Table 1: Performance Metrics for Viscosity Prediction Models
| Model Type | Data Points | Temperature Range (K) | AARD (%) | Key Features |
|---|---|---|---|---|
| GMDH (7 inputs) | 2813 | 253.15-573 | 8.14 | Temperature, pressure, molecular weight, critical properties [74] |
| COSMO-RS/ML Hybrid | - | - | 1.54 | Quantum chemical descriptors, solvation-free energy [76] |
| Group Contribution | 500 | 293-393 | 7.7 | Molecular structure, temperature [73] |
| QSPR (nonlinear) | 1502 | 253.15-395.2 | 6.58 | Structural descriptors, temperature, pressure [74] |
Experimental Approaches and Methodologies
Viscosity and Conductivity Measurement Protocols
Accurate characterization of IL transport properties requires standardized methodologies. Rotational viscometry remains the fundamental technique for viscosity measurement, employing concentric cylinder or cone-and-plate geometries to determine dynamic viscosity across temperature ranges of 253.15-573 K and pressures from 0.06-298.9 MPa [73] [74]. Temperature control within ±0.1 K is critical due to the strong temperature dependence of IL viscosity.
For conductivity characterization, impedance spectroscopy with platinum or gold electrodes in a two-electrode configuration provides reliable measurement of ionic conductivity. The standard protocol involves applying a small amplitude AC signal (typically 10 mV) across frequencies from 1 Hz to 1 MHz, with temperature stabilization periods of 20-30 minutes at each measurement point [71] [77]. Sample preparation must严格控制水分含量, as even minor water contamination significantly affects both viscosity and conductivity measurements [73].
Structural Characterization Techniques
Understanding the relationship between macroscopic transport properties and microscopic structure requires complementary characterization methods. Broadband Dielectric Spectroscopy (BDS) probes dipole reorientation and charge transport processes across wide frequency (10⁻²-10⁹ Hz) and temperature ranges, revealing fundamental insights into the interplay between glassy dynamics and conductivity [77]. Differential Scanning Calorimetry (DSC) determines thermal transitions including glass transition temperatures (Tg) that strongly influence transport mechanisms, with scan rates typically 10 K/min under nitrogen atmosphere [77].
Diagram 1: Viscosity Prediction Modeling Workflow. This diagram illustrates the integration of input parameters, machine learning approaches, and theoretical foundations for predicting ionic liquid viscosity.
Strategic Mitigation of High Viscosity
Molecular Design and Functionalization
Strategic molecular engineering presents the most direct approach to modulating IL viscosity. Ether functionalization has emerged as a particularly effective strategy, with compounds like tributyl(methoxymethyl)phosphonium hexafluorophosphate ([P₄₄₄₁O₁][PF₆]) demonstrating significantly reduced viscosity while maintaining beneficial electrochemical properties [75]. The oxygen atoms in ether groups facilitate weaker coordination with lithium ions and reduce cationic-anionic interactions, leading to viscosity reductions of 30-60% compared to their alkyl analogs [75].
Cation and anion selection fundamentally determines transport properties. Small, symmetric ions typically yield lower viscosities, while asymmetric cations with delocalized charge reduce melting points and viscosity [72] [73]. Imidazolium-based cations generally provide favorable viscosity profiles, with anions such as bis(trifluoromethylsulfonyl)imide ([Tf₂N]⁻) and tetrafluoroborate ([BF₄]⁻) offering balanced transport characteristics [72] [73].
Table 2: Transport Properties of Selected Ionic Liquids at 298K
| Ionic Liquid | Viscosity (mPa·s) | Molar Conductivity (S·cm²·mol⁻¹) | Activation Energy (kJ·mol⁻¹) |
|---|---|---|---|
| [C₄mim][Tf₂N] | 70 | 4.5 | 22.5 [73] |
| [C₄mim][BF₄] | 109 | 3.8 | 24.1 [71] |
| [P₄₄₄₁O₁][PF₆] | 45* | 6.2* | 19.8* [75] |
| [EtOHVIM][HSO₄] | 285 | 1.3 | 27.3 [77] |
| [C₆mim][Tf₂N] | 70 | 4.5 | 22.5 [73] |
*Estimated values from experimental data
System Optimization Strategies
Temperature manipulation represents the simplest approach to viscosity reduction, with most ILs exhibiting exponential viscosity decreases with increasing temperature. The temperature range of 323-363 K often provides optimal balance between viscosity reduction and thermal stability [73] [74].
Dilution with co-solvents significantly enhances transport properties while maintaining beneficial IL characteristics. Locally concentrated ionic liquid electrolytes (LCILEs) incorporating 20-30% ether solvents like 1,2-dimethoxyethane (DME) demonstrate substantially reduced viscosity while preserving anion-rich solvation structures necessary for forming stable electrode interphases [75]. Similarly, binary IL mixtures enable fine-tuning of viscosity and conductivity beyond what is achievable with single-component systems.
The Researcher's Toolkit: Essential Materials and Methods
Table 3: Research Reagent Solutions for Ionic Liquid Transport Studies
| Reagent/Material | Function/Application | Key Characteristics |
|---|---|---|
| 1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([C₄mim][Tf₂N]) | Reference IL for transport studies | Low viscosity (70 mPa·s), high conductivity, hydrophobic [73] |
| Tributyl(methoxymethyl)phosphonium hexafluorophosphate ([P₄₄₄₁O₁][PF₆]) | Ether-functionalized IL for enhanced transport | Low viscosity, flame-retardant, anion-rich solvation [75] |
| 1-Hydroxyethyl-3-vinylimidazolium hydrogen sulfate ([EtOHVIM][HSO₄]) | Monomeric IL for fundamental studies | Low glass transition temperature, high thermal stability [77] |
| 1,2-Dimethoxyethane (DME) | Co-solvent for LCILE formulations | Low viscosity, good lithium salt dissociation, ether functionality [75] |
| Grubbs 2nd Generation Catalyst | Metathesis catalyst for IL synthesis | High activity, functional group tolerance [72] |
The challenge of high viscosity in ionic liquids necessitates multifaceted approaches combining molecular design, computational prediction, and system engineering. The emerging paradigm emphasizes functionalization strategies like ether incorporation that selectively modulate ion-ion interactions while maintaining electrochemical performance [75]. Advanced machine learning frameworks now enable accurate prediction of transport properties from molecular descriptors, accelerating the design of task-specific ILs [76] [74]. Furthermore, the development of hybrid electrolyte systems such as LCILEs demonstrates how strategic combination of ILs with molecular solvents can achieve optimal transport characteristics while preserving the safety and stability benefits of ionic liquids [75].
As research progresses, the integration of data science with fundamental physical models promises to unravel the complex relationships between molecular structure, nanostructuring, and bulk transport properties [71]. This integrated approach will ultimately enable the rational design of next-generation ionic liquids that overcome the historical limitations of high viscosity while unlocking their full potential as advanced materials for energy storage, separation processes, and pharmaceutical applications.
The performance of electrochemical devices is intrinsically linked to the purity of their components, particularly the electrolytes. For ionic liquids (ILs)—a class of low-temperature molten salts—and supercritical fluids (SCFs), managing purity is not merely a procedural step but a fundamental requirement for achieving reliable and reproducible results. These neoteric solvents are central to a green chemistry revolution, offering unique properties like negligible vapor pressure, wide electrochemical stability windows, and tunable physicochemical characteristics [24] [44]. Their application spans from energy storage in batteries and supercapacitors to advanced synthesis and drug development [78] [79]. However, their "designer solvent" nature makes them exceptionally susceptible to performance degradation from even trace-level impurities. Effective impurity management is therefore the cornerstone of leveraging their full potential in high-stakes electrochemical applications, forming a critical branch of research into their properties compared to supercritical fluids.
The Impact of Impurities on Electrochemical Performance
Impurities in ionic liquids can originate from their synthesis, handling, or storage, and they profoundly impact key physicochemical and electrochemical properties.
- Water: The most common impurity, absorbed from the atmosphere due to the hygroscopic nature of many ILs.
- Halides (e.g., Chloride): Often leftover from metathesis reactions during synthesis [80].
- Organic Solvents and Excess Reagents: Residual from synthesis or purification steps, such as unreacted starting materials or solvents like dichloromethane and acetonitrile [80].
- Metal Ions: Can leach from processing equipment or be present in precursor salts.
Consequences of Impurities on Key Properties
The presence of these impurities can lead to:
- Altered Physicochemical Properties: Even trace amounts of water and chloride can significantly influence the viscosity and density of ionic liquids, thereby affecting mass transport and reaction kinetics [80].
- Narrowed Electrochemical Stability Window (ESW): Impurities often reduce the practical ESW of an IL by facilitating undesired side reactions, such as the hydrolysis of anions like
[PF6]−to produce hazardous HF [44]. This narrowing limits the operating voltage of devices like supercapacitors and batteries. - Increased Corrosivity: Halide impurities can accelerate the corrosion of electrodes and current collectors, compromising device integrity [80].
- Degradation of Device Performance and Lifespan: In energy storage devices, impurities contribute to increased internal resistance, reduced capacitance, and faster capacity fading over charge-discharge cycles [79]. For instance, the stability of electroactive polymers in electrochromic devices is severely hampered by impure electrolytes [80].
Table 1: Common Impurities in Ionic Liquids and Their Primary Impacts on Electrochemical Systems
| Impurity Type | Typical Sources | Primary Impact on Electrochemical Performance |
|---|---|---|
| Water (H₂O) | Atmospheric absorption, incomplete drying | Reduces electrochemical stability window; promotes hydrolysis of sensitive anions (e.g., [BF₄]⁻, [PF₆]⁻) [44] [80] |
| Halides (e.g., Cl⁻) | Incomplete metathesis reaction during synthesis | Increases viscosity and corrosion; degrades electrode materials [80] |
| Residual Organic Solvents | Synthesis/purification processes | Can alter viscosity, polarity, and reduce thermal stability; introduces flammable components |
| Metal Ions | Precursor salts, equipment leaching | Can catalyze decomposition reactions; interfere with redox processes |
Analytical Techniques for Purity Assessment
Rigorous characterization is essential to quantify impurity levels and certify an ionic liquid's suitability for electrochemical applications. A combination of techniques provides a comprehensive purity profile.
Core Analytical Methods
- Karl Fischer (KF) Titration: The standard method for determining water content with high precision, typically capable of detecting parts-per-million (ppm) levels [80].
- Ion Chromatography (IC): Effectively quantifies anionic impurities such as chloride, bromide, and fluoride, as well as cationic impurities from metal ions or residual precursors [80].
- Nuclear Magnetic Resonance (NMR) Spectroscopy: Particularly
¹Hand¹³CNMR, is indispensable for identifying and quantifying organic impurities and verifying the chemical structure of the IL ions [80]. - Electrochemical Methods:
- Cyclic Voltammetry (CV): Used to determine the practical electrochemical stability window (ESW) of the IL. A clean CV trace, free of extraneous peaks, indicates high purity. The ESW is typically measured from the cathodic to the anodic limit, defined by a predetermined current density threshold [81] [44].
- Electrochemical Impedance Spectroscopy (EIS): Measures the ionic conductivity of the IL. Impurities that increase viscosity or form ion aggregates can lead to a measurable drop in conductivity [44].
Table 2: Key Analytical Techniques for Assessing Ionic Liquid Purity
| Technique | Primary Measured Parameter(s) | Information on Impurities | Typical Detection Limits |
|---|---|---|---|
| Karl Fischer Titration | Water content | Quantifies total H₂O concentration | ppm level [80] |
| Ion Chromatography (IC) | Ionic species concentration | Quantifies halides (Cl⁻, Br⁻), other anions, metal cations | ppm to ppb level |
| NMR Spectroscopy | Chemical environment of nuclei | Identifies and quantifies organic molecules, solvent residues, structural defects | ~1 mol% |
| Cyclic Voltammetry (CV) | Electrochemical Stability Window (ESW) | Reveals redox-active impurities and practical voltage limits [81] | N/A |
| Inductively Coupled Plasma Mass Spectrometry (ICP-MS) | Elemental composition | Quantifies trace metal ion content | ppb to ppt level |
Purification Protocols and Experimental Methodologies
Achieving high-purity ionic liquids requires meticulous purification protocols. The following methods are standard in research and industrial settings.
Standard Purification Workflow
A generalized, robust workflow for purifying hydrophobic ionic liquids like those with [NTf2]− or [BF4]− anions is outlined below.
Diagram 1: IL Purification Workflow
Materials:
- Crude Ionic Liquid: The sample to be purified.
- Ultra-Pure Solvents: High-purity dichloromethane (DCM), ethyl acetate, and water.
- Activated Adsorbents: Activated carbon, neutral alumina, and molecular sieves.
- Equipment: Schlenk line or vacuum oven, filtration apparatus (e.g., fritted funnel), rotary evaporator, and an argon-filled glovebox.
Detailed Protocol:
- Dissolution and Initial Filtration: Dissolve the crude IL in a minimal amount of a high-purity, volatile solvent like DCM. Filter the solution through a fine-porosity fritted glass filter to remove any particulate matter or insoluble salts.
- Liquid-Liquid Extraction (for halide removal): Vigorously stir the IL solution with ultra-pure water in a separatory funnel. Halide salts are often more soluble in the aqueous phase and will partition into it. Separate the aqueous layer and repeat the process 3-5 times until halide levels are undetectable by ion chromatography [80].
- Adsorbent Stirring: To remove organic impurities and residual water, stir the IL solution for 24-48 hours with activated adsorbents. A common combination is activated carbon (to remove colored organic impurities) and neutral alumina. The adsorbents must be activated according to manufacturer protocols prior to use [80].
- Removal of Volatiles: After filtration to remove the adsorbents, concentrate the solution using a rotary evaporator to remove the bulk solvent. Subsequently, place the IL under high vacuum (e.g., < 10⁻² mbar) on a Schlenk line for several hours to remove trace solvents and water.
- Final Drying and Filtration: For the highest purity standards, especially for electrochemical applications, transfer the IL to an argon-filled glovebox. Perform a final high-temperature vacuum drying (e.g., 60-70 °C under dynamic vacuum for 12-24 hours) [80]. Optionally, pass the hot IL through a micro-filter (e.g., 0.2 µm PTFE) inside the glovebox to remove any particulates.
Purity Validation Experiment
After purification, the ionic liquid's quality must be validated experimentally.
Objective: To determine the electrochemical stability window (ESW) and ionic conductivity of the purified ionic liquid.
Materials and Reagents:
- Purified Ionic Liquid: e.g.,
[P2225][NTf2]or[bmim][BF4]. - Electrochemical Cell: A sealed, three-electrode cell compatible with air-sensitive materials.
- Electrodes:
- Working Electrode: Pt, Au, or glassy carbon micro-disc electrode (for CV).
- Counter Electrode: Pt wire.
- Quasi-Reference Electrode: Pt or Ag wire (potential calibrated against a known redox couple) [81].
- Equipment: Potentiostat, conductivity meter, and glovebox.
Methodology:
- Cell Preparation: All cell assembly and electrolyte transfer should be conducted inside an argon-filled glovebox (O₂ and H₂O < 1 ppm).
- Cyclic Voltammetry (CV):
- Fill the electrochemical cell with the purified IL.
- Record CV curves at a slow scan rate (e.g., 10-50 mV/s) across a wide potential range.
- The anodic and cathodic limits are defined as the potentials where the current density reaches a predefined threshold (e.g., ±0.1 mA/cm²) [81]. The ESW is the voltage difference between these two limits.
- Ionic Conductivity Measurement:
- Use a calibrated conductivity cell with a known cell constant.
- Immerse the cell in the purified IL and measure the resistance, typically via EIS. The conductivity (κ) is calculated as κ = G • kcell, where G is the conductance and kcell is the cell constant.
The Scientist's Toolkit: Essential Reagent Solutions
Successful experimentation with ionic liquids requires a suite of specialized reagents and materials.
Table 3: Essential Research Reagent Solutions for IL Purity Management
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| Activated Neutral Alumina | Adsorbent for removing polar impurities, acids, and water. | Must be activated by heating (e.g., 300-400 °C) before use to remove physisorbed water [80]. |
| Activated Charcoal (Carbon) | Adsorbent for removing colored, non-polar organic impurities. | Used in conjunction with alumina for broad-spectrum impurity removal. |
| Molecular Sieves (3Å or 4Å) | Selective adsorbent for water removal from dried ILs. | Must be activated by heating under vacuum prior to use. |
| High-Purity Solvents (DCM, Acetonitrile) | Medium for dissolution, washing, and extraction. | Must be of the highest available purity (e.g., 99.9+%, water < 10 ppm) to avoid re-introducing impurities. |
| Karl Fischer Reagent | Titrant for the quantitative determination of water content. | Essential for verifying the success of the drying process [80]. |
| Electrode Polishing Kit | For maintaining reproducible electrode surfaces for electrochemistry. | Includes alumina or diamond slurries of varying particle sizes (e.g., 1.0, 0.3, 0.05 µm). |
Performance Implications in Electrochemical Devices
The direct correlation between IL purity and device performance is unequivocal. High-purity ILs enable wider operational voltage windows, which is critical because the storable energy in a capacitor scales with the square of the voltage (E = ½CV²) [44]. For instance, phosphonium-based ILs, when purified, can exhibit ESWs exceeding 6.0 V, making them superior electrolytes for high-energy-density supercapacitors [44] [80].
In lithium-ion batteries, replacing flammable organic carbonates with purified ILs enhances safety by eliminating volatile and flammable components [79]. The non-flammability and thermal stability of ILs prevent thermal runaway, a significant safety concern in large-scale battery packs [79]. Furthermore, high-purity ILs have been shown to form more stable solid-electrolyte interphases (SEI) on lithium metal anodes, improving cycling efficiency and lifespan [79]. The exceptional stability afforded by pure ILs is also demonstrated in electrochromic devices, where polyaniline-based displays showed no significant loss in electroactivity after one million cycles when using a pure IL electrolyte [80]. Managing purity is thus the key to unlocking the superior performance, safety, and longevity promised by ionic liquid electrolytes.
Diagram 2: Purity to Performance Pathway
Navigating Material Compatibility and Equipment Selection for Scalable Processes
The transition from laboratory research to industrial-scale production presents significant challenges in process chemistry, particularly when employing advanced solvents like ionic liquids (ILs) and supercritical fluids (SCFs). These substances offer unique advantages for drug development and advanced materials synthesis, but their implementation requires careful consideration of material compatibility and equipment selection. This guide provides researchers and scientists with a systematic framework for addressing these critical aspects, enabling scalable and reproducible processes while maintaining safety and efficiency.
Fundamental Properties of Ionic Liquids and Supercritical Fluids
Ionic Liquids: Tunable Solvents for Advanced Applications
Ionic liquids are salts that exist in liquid state below 100°C, composed entirely of ions whose properties can be finely tuned by selecting different cation-anion combinations. Their non-volatility, thermal stability, and designer solvent capabilities make them valuable for various applications including perovskite solar cells, wearable medical devices, seawater desalination, and tunable nanoreactors [82]. The structure-tunable nature of ILs allows researchers to tailor their chemical and physical properties for specific process requirements, creating task-specific solvents for specialized applications [24].
Supercritical Fluids: Hybrid Properties for Green Processing
Supercritical fluids exist beyond their critical temperature and pressure, exhibiting properties intermediate between gases and liquids. This unique state provides high diffusivity and low viscosity characteristic of gases, combined with the density-dependent solvent power of liquids [83] [4]. Supercritical carbon dioxide (scCO₂) has emerged as a particularly valuable SCF due to its accessible critical parameters (31.3°C, 72.9 bar), non-toxicity, and environmental benefits [4]. The density of SCFs can be precisely modulated by adjusting pressure and temperature, allowing fine control of solvent properties without changing the processing medium [4].
Table 1: Comparative Properties of Common Supercritical Fluids [4]
| Fluid | Critical Temperature (°C) | Critical Pressure (bar) | Critical Density (g/cm³) | Key Applications |
|---|---|---|---|---|
| CO₂ | 31.3 | 72.9 | 0.47 | Extraction, particle design, chromatography |
| NH₃ | 132.5 | 112.5 | 0.24 | Specialized reactions, waste treatment |
| Pentane | 196.6 | 33.3 | 0.23 | Lipid processing, organic synthesis |
| Water | 374.0 | 221.0 | 0.32 | Oxidation, hydrolysis, waste destruction |
Table 2: Characteristic Properties of Common Ionic Liquid Cations and Anions [82] [24]
| Cation Type | Anion Type | Key Characteristics | Typical Applications |
|---|---|---|---|
| Imidazolium | BF₄⁻, PF₆⁻ | Moderate hydrophilicity, good stability | Catalysis, electrochemistry |
| Phosphonium | NTf₂⁻ | High thermal stability, low viscosity | High-temperature processes |
| Pyridinium | Cl⁻, Br⁻ | Biodegradable options available | Pharmaceutical synthesis |
| Ammonium | DCA⁻ | Variable hydrophobicity | Separation processes |
Material Compatibility Assessment
Understanding Material-Solvent Interactions
Material compatibility forms the foundation for successful process scale-up, particularly with aggressive solvents like ILs and SCFs. Incompatible materials can lead to equipment failure, product contamination, and safety hazards. For hydraulic systems using ionic liquids, seal material compatibility has emerged as a critical concern, with different elastomers exhibiting varying resistance to specific IL formulations [84].
The manufacturing perspective emphasizes that material properties directly impact production costs and lead times. When material compatibility is not properly considered, machinability, formability, and thermal processing requirements can increase production time by 300% or more [85]. Material hardness exceeding 35 HRC typically requires specialized tooling and extended cycle times, while softer materials may present challenges with surface finish and dimensional stability due to deflection during processing [85].
Experimental Protocol for Seal and Material Compatibility Testing
Standardized testing methodologies are essential for evaluating material compatibility with ILs and SCFs. The following protocol provides a systematic approach for researchers:
Sample Preparation: Prepare standardized specimens (typically 25mm diameter, 3mm thickness) of seal materials including nitrile rubber (NBR), fluorocarbon rubber (FPM/FKM), ethylene propylene diene monomer (EPDM), and polytetrafluoroethylene (PTFE) [84].
Baseline Measurements: Measure and record initial properties including:
- Hardness (Shore A scale)
- Volume and dimensions
- Tensile strength
- Elongation at break
Exposure Conditions: Immerse samples in the ionic liquid or supercritical fluid under investigation. Testing should include:
- Standard conditions: 70 hours at 100°C
- Extended conditions: 1000 hours at elevated temperatures relevant to process conditions
- Control samples maintained in ambient conditions
Post-Exposure Analysis: Following exposure and conditioning, evaluate:
- Change in volume (swell or shrinkage)
- Hardness change (Shore A points)
- Tensile strength retention
- Elongation change
- Visual inspection for cracking, blistering, or discoloration
Acceptance Criteria: Determine material suitability based on standardized percentages:
- Volume change: ±10% maximum for static seals, ±5% for dynamic seals
- Hardness change: ±10 points maximum
- Tensile strength change: ±30% maximum
- Elongation change: -50% to +20% maximum [84]
Material Compatibility Data and Guidelines
Table 3: Seal Material Compatibility with Ionic Liquids [84]
| Seal Material | Volume Change (%) | Hardness Change (Points) | Tensile Strength Change (%) | Compatibility Rating |
|---|---|---|---|---|
| Nitrile (NBR) | +5 to +8 | -2 to -5 | -10 to -15 | Good to Excellent |
| Fluorocarbon (FKM) | +1 to +3 | 0 to -2 | -5 to -8 | Excellent |
| EPDM | +8 to +12 | -3 to -7 | -15 to -25 | Fair to Good |
| PTFE | +0.5 to +1.5 | 0 | -2 to -5 | Excellent |
| Polyurethane | +10 to +15 | -5 to -10 | -20 to -30 | Poor to Fair |
Equipment Selection Framework
Systematic Approach to Equipment Selection
Equipment selection for processes involving ILs and SCFs requires careful consideration of multiple factors beyond basic functionality. The decision-making process should incorporate material compatibility, process requirements, and scalability. The following diagram illustrates the systematic approach to equipment selection:
Equipment Selection Process Flow
GMP-Compatible Equipment Considerations
For pharmaceutical applications, equipment must meet Good Manufacturing Practice (GMP) standards to ensure product quality and patient safety. GMP-compatible equipment selection requires:
- Documentation and Validation: Comprehensive equipment qualification including Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) [86].
- Material Standards: Use of 316L stainless steel or higher-grade materials for product contact surfaces, with electropolished finishes for cleanability.
- Design Features: Clean-in-place (CIP) and sterilize-in-place (SIP) capabilities, minimal dead legs, and sanitary fittings.
- Control Systems: Validated software with audit trails, electronic records compliance (21 CFR Part 11), and alarm management systems.
- Change Control: Formal procedures for managing equipment modifications to maintain validated state.
Specialized Equipment for Ionic Liquid and Supercritical Fluid Processes
Process equipment for ILs and SCFs requires specific design considerations:
High-Pressure Reactor Systems:
- Pressure ratings: 100-500 bar for SCF applications
- Corrosion-resistant materials: Hastelloy, Inconel, or titanium for aggressive ILs
- Magnetic drive agitators to prevent seal leakage
- Advanced sampling systems for representative sampling without pressure release
Extraction and Separation Equipment:
- Counter-current columns for SCF extraction
- Microfiltration systems for IL recovery and recycling
- Pressure let-down systems for particle formation
- Integrated phase separation equipment
Scale-Up Methodologies and Process Intensification
Pilot Plant Scale-Up Strategies
The transition from laboratory to commercial scale requires systematic scale-up approaches. Successful pilot plant operations bridge the gap between bench-scale research and full-scale production, allowing for process validation and optimization [87]. Key scale-up considerations include:
- Scale-Up Ratios: Conservative scale-up factors (typically 10x) between successive stages to minimize risk
- Process Parameters: Maintaining similar reaction conditions, mixing intensity, and heat transfer characteristics across scales
- Mass and Heat Transfer: Addressing potential limitations in larger equipment through enhanced mixing and heat exchange surfaces
Experimental Protocol for Scalability Assessment
A structured approach to scalability assessment ensures successful technology transfer:
Laboratory Process Characterization:
- Determine key process parameters and their acceptable ranges
- Identify critical quality attributes (CQAs) and critical process parameters (CPPs)
- Establish process robustness through design of experiments (DoE)
Pilot-Scale Demonstration:
- Implement process in appropriately sized equipment (typically 10-100L)
- Verify mass and heat transfer characteristics
- Confirm product quality meets specifications
- Optimize process economics
Engineering Design Basis Development:
- Generate data for commercial equipment design
- Establish operating and control philosophies
- Develop process flow diagrams (PFDs) and piping and instrumentation diagrams (P&IDs)
Addressing Scale-Up Challenges
Common scale-up challenges and mitigation strategies include:
- Heat Management: Implementation of advanced cooling and heating systems to maintain optimal reaction conditions at larger scales [87]
- Mixing Efficiency: Optimization of reactor design and impeller selection to ensure uniform mixing and efficient mass transfer
- Residence Time Distribution: Maintenance of consistent residence times despite increased equipment volumes
- Raw Material Consistency: Ensuring consistent quality and supply of ionic liquids and other specialized reagents
Research Reagent Solutions and Essential Materials
Table 4: Essential Research Reagents and Materials for IL and SCF Processes
| Reagent/Material | Function | Key Considerations | Compatible Materials |
|---|---|---|---|
| Imidazolium-based ILs | Reaction medium, catalyst | Hydrophilicity/hydrophobicity tuning | Glass, 316SS, PTFE |
| Phosphonium-based ILs | High-temperature processes | Thermal stability >300°C | Hastelloy, Inconel |
| scCO₂ | Extraction, particle formation | Critical parameters: 31.3°C, 72.9 bar | 316SS, PTFE, PEEK |
| Co-solvents (ethanol, methanol) | Modifiers for SCF | Polarity adjustment for solubility | Glass, 316SS, PTFE |
| Seal materials (FKM, PTFE) | Equipment integrity | Chemical resistance, temperature rating | Various (see Table 3) |
| High-pressure fittings | Equipment assembly | Pressure rating, sealing mechanism | 316SS, Hastelloy |
| Pressure relief devices | Safety systems | Set pressure, corrosion resistance | 316SS, Monel |
Integrated Process Design and Economic Considerations
Lifecycle Assessment and Sustainability
Process design incorporating ILs and SCFs should consider environmental impact and sustainability:
- Solvent Recovery and Recycling: Development of efficient recovery systems for ionic liquids, which despite their non-volatility, can be lost through mechanical carryover or degradation
- Energy Integration: Optimization of energy-intensive operations, particularly SCF compression and decompression cycles
- Waste Minimization: Implementation of processes that minimize waste generation, leveraging the recyclability of ILs and the non-toxic nature of scCO₂
Economic Evaluation Framework
The implementation of IL and SCF technologies requires thorough economic analysis:
- Capital Cost Considerations: High-pressure equipment, specialized materials of construction, and safety systems
- Operating Costs: Ionic liquid replacement, energy consumption for SCF maintenance, and maintenance requirements
- Value Proposition: Enhanced product quality, environmental benefits, and regulatory advantages
Successful implementation of ionic liquids and supercritical fluids in scalable processes requires integrated consideration of material compatibility, equipment selection, and scale-up methodology. By adopting a systematic approach to compatibility testing, equipment specification, and process design, researchers and development professionals can overcome the challenges associated with these advanced solvents. The frameworks and protocols presented in this guide provide a foundation for developing robust, scalable processes that leverage the unique properties of ILs and SCFs while ensuring operational reliability, safety, and economic viability.
Selecting the Right Tool: A Data-Driven Comparison for Research Goals
The pursuit of sustainable industrial processes, particularly in sectors like pharmaceuticals and environmental remediation, has catalyzed the search for alternative solvents that align with the principles of green chemistry. Conventional organic solvents, characterized by high volatility, toxicity, and environmental persistence, pose significant health and ecological risks. Within this context, ionic liquids (ILs) and supercritical fluids (SCFs), notably supercritical carbon dioxide (scCO₂), have emerged as two of the most promising classes of environmentally benign solvents. This whitepaper provides a direct performance comparison of ILs and SCFs, evaluating their solvation power, process efficiency, and environmental impact to guide researchers and drug development professionals in solvent selection for advanced applications. Framed within broader research on their properties, this analysis draws upon recent scientific advancements to offer a technically rigorous guide.
Fundamental Properties and Solvation Mechanisms
Ionic Liquids (ILs)
Ionic liquids are classically defined as salts that are liquid below 100°C, often existing as liquids at room temperature (Room-Temperature Ionic Liquids, or RTILs) [88]. They are composed entirely of ions—typically bulky, asymmetric organic cations and organic or inorganic anions [88] [20]. This ionic structure confers a unique set of properties, including negligible vapor pressure, high thermal stability, and high ionic conductivity [88] [89]. A paramount advantage of ILs is their structural designability; by selecting different cation-anion combinations, properties such as polarity, hydrophobicity, and viscosity can be finely tuned for specific applications, earning them the moniker "designer solvents" [88] [20].
Their solvation power is derived from strong Coulombic forces, hydrogen bonding, and van der Waals interactions. The ability to dissolve a wide range of materials, from polar compounds to metals and polymers, makes them exceptionally versatile [88] [20].
Supercritical Fluids (SCFs)
A supercritical fluid is a state of matter achieved when a substance is heated and compressed above its critical temperature (T𝑐) and critical pressure (P𝑐) [4]. Beyond this critical point, the substance exhibits properties intermediate between those of a gas and a liquid. Supercritical carbon dioxide (scCO₂), with a critical temperature of 31.3°C and a critical pressure of 72.9 bar, is the most widely used SCF [4].
- Liquid-like Density: Enables scCO₂ to dissolve a wide range of compounds. Its density, and consequently its solvent power, can be precisely modulated by adjusting the pressure and temperature [4].
- Gas-like Viscosity and Diffusivity: Results in low viscosity and high diffusivity, which significantly enhance mass transfer processes compared to liquid solvents [4].
- Environmental Profile: scCO₂ is non-toxic, non-flammable, and readily available. Its use often avoids the generation of hazardous waste associated with organic solvents [4] [90].
The following diagram illustrates the phase behavior of a pure substance and the pathway to achieving the supercritical state, with scCO₂'s relatively accessible critical point being a key reason for its widespread adoption.
Direct Performance Comparison
The table below provides a quantitative and qualitative comparison of ionic liquids and supercritical fluids across key performance metrics.
Table 1: Direct Performance Comparison of Ionic Liquids vs. Supercritical Fluids
| Performance Metric | Ionic Liquids (ILs) | Supercritical Fluids (SCFs), notably scCO₂ |
|---|---|---|
| Solvation Power & Tunability | Highly tunable via cation/anion selection. Can dissolve polar, non-polar, polymeric, and metallic compounds [88] [20]. | Tunable with pressure/temperature. High power for non-polar solutes; requires chelating agents for metal ions [4] [90]. |
| Process Efficiency | High viscosity can limit mass transfer. Excellent thermal stability allows for high-temperature operations [88] [20]. | High efficiency due to low viscosity and high diffusivity, enhancing mass transfer [4]. |
| Volatility & Flammability | Negligible vapor pressure, non-flammable [88] [89]. | Non-flammable, though will expand to gas state upon depressurization [4]. |
| Toxicity & Biodegradability | Variable; some are toxic and poorly biodegradable ("green credential" requires verification) [89]. | Generally recognized as safe (GRAS) for food/pharma applications; scCO₂ is non-toxic [4]. |
| Environmental Impact | Potential for reuse, but synthesis can be energy-intensive. Environmental persistence is a concern [89]. | Low environmental impact; scCO₂ is obtained from waste streams, leaves no solvent residues [4] [90]. |
| Key Applications | Polymer synthesis, catalysis, electrolytes, biomass processing, drug delivery [88] [20]. | Extraction (e.g., caffeine, hops), nanoparticle formation, chemical reactions, environmental remediation [4] [90]. |
Experimental Protocols and Methodologies
Protocol: Supercritical CO₂ Chelation Extraction of Heavy Metals
This protocol, adapted from research on treating drilling fluid waste, details the application of scCO₂ for extracting heavy metal ions, a process that overcomes the innate non-polarity of CO₂ through chelation chemistry [90].
Principle: Charged metal ions are complexed by chelating agents to form neutral, stable complexes that are soluble in scCO₂, enabling their extraction from solid matrices [90].
Key Reagents & Materials:
- CO₂ Gas Source (high purity, ≥99.95%)
- Chelating Agent (e.g., Ethylenediaminetetraacetic acid - EDTA, or Citric Acid)
- Contaminated Solid Matrix (e.g., drilling fluid waste, soil)
- High-Pressure Extraction Reactor (e.g., 1000 ml capacity, rated to 400 bar)
- Temperature Control System (heating jacket with ±1 K accuracy)
- Pressure Measurement System (transducer with 0.1 bar accuracy)
- Magnetically Coupled Stirrer (for efficient mixing at high pressure)
Procedure:
- Sample Preparation: The solid waste matrix is homogenized and mixed with the selected chelating agent (e.g., EDTA).
- Reactor Loading: The mixture is loaded into the extraction reactor.
- Pressurization & Heating: The reactor is sealed and pressurized with CO₂ to the target pressure (e.g., 220 bar) using a compressor. Simultaneously, the system is heated to the target temperature (e.g., 348.15 K / 75°C).
- Dynamic Extraction: The system is maintained at the target pressure and temperature for the extraction duration (e.g., 70 minutes) with continuous stirring to ensure contact between the scCO₂, chelating agent, and metal ions.
- Separation & Collection: The scCO₂ stream, now containing the metal-chelate complexes, is depressurized through a separator. The pressure drop reduces the solvent power of CO₂, causing the complexes to precipitate for collection.
- Analysis: The extracted metals are quantified in the collected fraction and the residual solid matrix (e.g., via ICP-MS) to determine extraction efficiency.
The workflow for this chelation extraction process is summarized below:
Protocol: Ionic Liquid-Assisted Analytical Determination of Pharmaceuticals
This protocol outlines the use of ILs in the sample preparation and separation of pharmaceuticals from complex biological and environmental matrices, improving selectivity and reducing the use of organic solvents [89].
Principle: ILs can function as high-performance extractants or modifiers in chromatographic systems due to their tunable hydrophobicity/hydrophilicity and unique interactions with analytes, leading to enhanced extraction efficiency and improved chromatographic peak shape [89].
Key Reagents & Materials:
- Ionic Liquid (e.g., imidazolium or phosphonium-based, selected for specific analyte-matrix interaction)
- Biological/Environmental Sample (e.g., urine, wastewater)
- Organic Solvent (minimal volume for back-extraction or mobile phase modification)
- Centrifuge
- HPLC System with Detector (e.g., UV or MS)
- SPME Fiber or HPLC Column (can be IL-modified)
Procedure:
- IL Selection: Choose an IL with cations/anions that provide optimal affinity for the target pharmaceutical(s). For example, hydrophobic ILs may be selected for extracting non-polar drugs.
- Sample Preparation (Extraction):
- Dispersive Liquid-Liquid Microextraction (DLLME): A small volume of IL is rapidly dispersed into the aqueous sample solution, forming a fine cloud of droplets that extract the analytes.
- Solid-Phase Microextraction (SPME): An IL-based polymeric coating on a fiber is exposed to the sample headspace or directly immersed in the solution to adsorb analytes.
- Phase Separation: After extraction, the IL phase is separated from the sample matrix. In DLLME, this is often achieved by centrifugation.
- Analysis (Chromatography): The IL-rich extract can be injected directly or after dilution into an HPLC or GC system. ILs can also be added as mobile phase additives in HPLC to mask residual silanol groups on silica-based stationary phases, thereby reducing peak tailing and improving separation efficiency [89].
- Detection & Quantification: The target pharmaceuticals are detected and quantified using appropriate detectors (e.g., UV, fluorescence, or mass spectrometry).
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagents and Materials for IL and SCF Research
| Item | Function/Application | Examples / Notes |
|---|---|---|
| Ionic Liquids (Research Grade) | Tunable solvents for synthesis, extraction, and as electrolytes. | Imidazolium (e.g., [C₄mim][BF₄]), pyrrolidinium, phosphonium salts. Selection depends on required polarity, viscosity, and hydrophobicity [88] [89]. |
| Supercritical Fluid Extraction System | High-pressure apparatus for scCO₂-based extraction and reactions. | Consists of a CO₂ pump, a heated extraction vessel, pressure control valves, and a separator [90]. |
| Chelating Agents for scCO₂ | Enable extraction of metal ions by forming neutral complexes. | EDTA, Citric Acid, or crown ethers (for selectivity based on ionic radius) [90]. |
| High-Pressure View Cell | Allows visual observation of phase behavior and solubility in SCFs. | Critical for determining solubility parameters and monitoring reactions in scCO₂. |
| Polymeric Ionic Liquids (PILs) | ILs polymerized to create solid sorbents or advanced materials. | Used as SPME fiber coatings or membranes for enhanced extraction and separation [89] [20]. |
| Chiral Ionic Liquids | Facilitate enantioselective synthesis and separations. | Contain chiral cations or anions; relevant for pharmaceutical synthesis [89]. |
The direct comparison between ionic liquids and supercritical fluids reveals two powerful but distinct classes of green solvents. The choice between them is not a matter of superiority, but of application-specific suitability. Supercritical CO₂ excels in applications where low environmental impact, high diffusion rates, and facile post-process separation are paramount, such as in the extraction of natural products and environmental remediation of organic pollutants and heavy metals. Its tunability via pressure and temperature offers significant control. Ionic liquids, with their unparalleled structural tunability and exceptional solvation properties for a vast range of materials, are indispensable in catalysis, polymer science, and as sophisticated media in analytical chemistry and electrochemistry. However, their "green" status must be evaluated on a case-by-case basis, considering their synthesis and potential environmental persistence. The future of sustainable solvent technology likely lies in hybrid systems that leverage the unique advantages of both ILs and SCFs, for instance, using scCO₂ to extract products from an IL reaction medium, thereby combining the selectivity of ILs with the clean separation offered by scCO₂. This synergistic approach will be pivotal in advancing green manufacturing processes across the pharmaceutical and chemical industries.
In the pursuit of sustainable chemical processes, ionic liquids (ILs) and supercritical fluids (SCFs) have emerged as promising alternatives to conventional volatile organic solvents. Framed within broader research on their properties, this whitepaper provides a technical comparison of the safety and operational profiles of ILs and SCFs, focusing on the critical parameters of non-flammability, toxicity, and volatility. Understanding these profiles is paramount for researchers and drug development professionals selecting solvents for applications ranging from synthesis and extraction to particle formation and formulation. This document synthesizes current data, provides standardized experimental protocols for safety assessment, and offers a practical toolkit for their application in research environments.
Fundamental Properties and Safety Profiles
Defining Characteristics
Ionic Liquids (ILs) are commonly defined as salts with a melting point below 100 °C, often liquid at room temperature. They are composed of large, asymmetric organic cations (e.g., imidazolium, pyridinium, phosphonium) and organic or inorganic anions (e.g., tetrafluoroborate, hexafluorophosphate, chloride) [91]. Their key safety-related characteristic is their negligible vapor pressure, which renders them non-volatile and eliminates inhalation risks under normal handling conditions [20] [91].
Supercritical Fluids (SCFs) are substances maintained at temperatures and pressures above their critical point, where distinct liquid and gas phases do not exist. They exhibit properties intermediate between liquids and gases, such as liquid-like densities and gas-like diffusivities and viscosities [4]. Supercritical carbon dioxide (scCO₂) is the most widely used SCF, with a critical temperature of 31.3°C and a critical pressure of 72.9 bar [4]. scCO₂ is non-flammable and considered a safe, environmentally benign solvent due to its non-toxic nature [92] [4].
Comparative Safety Data
The following tables summarize the core safety and operational characteristics of ILs and SCFs, providing a direct comparison for researchers.
Table 1: Volatility and Flammability Profile Comparison
| Property | Ionic Liquids (ILs) | Supercritical CO₂ (scCO₂) | Conventional Organic Solvents |
|---|---|---|---|
| Vapor Pressure | Negligible [20] [91] | High in its gaseous state; not applicable in supercritical state [4] | High |
| Volatility | Non-volatile [93] | N/A (Supercritical fluid) | Volatile |
| Flammability | Generally non-flammable [93] | Non-flammable [4] | Often flammable |
| Inhalation Risk | Very low | Low (asphyxiant at high concentrations) | High |
Table 2: Toxicity and Environmental Impact Profile
| Aspect | Ionic Liquids (ILs) | Supercritical CO₂ (scCO₂) |
|---|---|---|
| General Toxicity | Ranges from low to high; highly structure-dependent [94] [91] | Non-toxic, Generally Recognized As Safe (GRAS) for food use [92] |
| Primary Concern | Cytotoxicity and environmental ecotoxicity [94] [91] | High-pressure operation, potential for asphyxiation |
| Environmental Fate | Potentially persistent; can be designed for biodegradability [91] | Benign; sourced from waste streams [4] |
| Solvent Residue | Potential for contamination if not purified | No residual solvent [92] |
Table 3: Quantitative Cytotoxicity of Common IL Cations (Representative Data) Data compiled from a comprehensive dataset of 1227 ILs [94]
| Cation Type | Example | Typical Cytotoxicity Range (IC₅₀/EC₅₀) | Key Structural Influence |
|---|---|---|---|
| Imidazolium | [C₄mim]⁺ | Varies widely (µM to mM) | Toxicity generally increases with alkyl chain length [91] |
| Phosphonium | [P₆₆₆₁₄]⁺ | Often more toxic than imidazolium analogues | Increased toxicity with chain length [91] |
| Ammonium | [N₁₈₈₈]⁺ | Varies widely (µM to mM) | Toxicity generally increases with alkyl chain length |
| Choline | [HOC₂H₄N(CH₃)₃]⁺ | Low toxicity | Considered a "green" IL precursor [91] |
| Amino Acid-derived | e.g., [C₂mim][Gly] | Designed for lower toxicity and biodegradability | Emerging class with improved safety profiles [91] |
Experimental Protocols for Safety Assessment
Protocol for Assessing IL Cytotoxicity
This protocol is adapted from methodologies used to create comprehensive IL cytotoxicity datasets [94].
1. Objective: To determine the half-maximal inhibitory, effective, or cytotoxic concentration (IC₅₀, EC₅₀, or CC₅₀) of an ionic liquid against a specific eukaryotic cell line.
2. Materials:
- Test Ionic Liquid: High-purity sample with known structure, empirical formula, and molecular weight.
- Cell Line: Relevant mammalian cell lines (e.g., HeLa, HepG2, CaCo-2).
- Assay Kit: Cell viability assay, such as MTT, MTS, or Alamar Blue.
- Equipment: Cell culture incubator (37°C, 5% CO₂), microplate reader, centrifuge, sterile tissue culture tools.
3. Methodology:
- Cell Seeding: Seed cells in a 96-well plate at a standardized density and allow to adhere for 24 hours.
- IL Exposure: Prepare a serial dilution of the IL in the cell culture medium. Remove the growth medium from the cells and add the IL-containing medium. Include wells with only medium (blank) and wells with untreated cells (control).
- Incubation: Incubate the cells for a defined period (typically 24-72 hours). The incubation time must be standardized and reported [94].
- Viability Assay: Following incubation, add the viability assay reagent according to the manufacturer's instructions. Incubate for the required time to allow formazan formation or fluorescence development.
- Measurement: Measure the absorbance or fluorescence using a microplate reader.
- Data Analysis: Calculate cell viability as a percentage of the untreated control. Plot viability (%) versus log(IL concentration) and use non-linear regression to calculate the IC₅₀/EC₅₀/CC₅₀ value. Report the value along with its range (e.g., standard error, confidence interval) [94].
4. Key Considerations:
- The chemical information (name, formula, CAS, SMILES) and experimental details (cell line, incubation time, assay type) must be meticulously recorded for reproducibility and database inclusion [94].
- ILs should not contain active pharmaceutical ingredients (APIs) for baseline toxicity assessment, as the API would dominate the cytotoxic effect [94].
Protocol for Phase Behavior Measurement in SCF Systems
1. Objective: To experimentally determine the pressure-temperature (P-T) phase diagram of a pure compound or mixture to establish its supercritical operating window.
2. Materials:
- High-Pressure View Cell: A vessel with sapphire windows capable of withstanding high pressures, equipped with temperature control and a pressure transducer.
- Pump: A high-pressure pump for fluid delivery (e.g., syringe pump).
- Materials: High-purity CO₂ or other SCF candidate; solute of interest for mixture studies.
3. Methodology:
- Loading: Charge the view cell with a known amount of the pure substance or mixture.
- Equilibration: Set the system to a specific temperature (T₁) using the thermostat. Gradually increase the pressure using the pump while continuously stirring the contents.
- Observation: Visually observe the phase changes through the sapphire windows. Record the pressure at which a single, homogeneous supercritical phase is formed (dew/bubble point for mixtures; critical point for pure substances).
- Data Point Collection: Repeat the pressure increase at multiple set temperatures (T₂, T₃, ..., Tₙ) to map out the phase boundary.
- Modeling (Optional): Fit the experimental data to an equation of state (e.g., Peng-Robinson) for modeling and prediction purposes, as performed in studies of CCUS fluids and asymmetric mixtures [13].
4. Key Considerations:
- Safety is paramount when working with high-pressure systems. Equipment must be rated for the intended pressures and operated within safe limits.
- For mixture studies, the composition must be precisely known and maintained.
Visualization of Workflows and Relationships
IL Cytotoxicity Assessment Workflow
The following diagram illustrates the key steps and decision points in evaluating the cytotoxicity of an ionic liquid.
SCF Phase Behavior Analysis
This diagram outlines the experimental process for determining the phase behavior of a supercritical fluid system.
The Scientist's Toolkit: Research Reagent Solutions
This section details essential materials and their functions for working with ILs and SCFs in a research context.
Table 4: Essential Research Reagents and Materials
| Item | Function/Application | Specific Examples & Notes |
|---|---|---|
| Common IL Cations | Building blocks for designing task-specific ILs [20]. | Imidazolium (e.g., 1-butyl-3-methylimidazolium), Pyridinium, Phosphonium, Choline (low toxicity) [91]. |
| Common IL Anions | Tune physicochemical properties like hydrophobicity, viscosity, and solvation [20]. | Halides (Cl⁻, Br⁻), [BF₄]⁻, [PF₆]⁻, [Tf₂N]⁻, Alkylsulfates, Amino acid-based (biodegradable) [91]. |
| Supercritical CO₂ | Primary solvent for SCF extraction and reaction processes; non-toxic, non-flammable [92] [4]. | Must be of high purity; critical point: 31.1°C, 73.8 bar [4]. |
| Co-solvents/Modifiers | Enhance the solvating power of scCO₂ for more polar compounds [92]. | Methanol, Ethanol, Water (typically 1-15% by volume). |
| Cell Lines for Toxicity | In vitro models for assessing cytotoxicity of ILs [94]. | Human cell lines like Caco-2 (colon adenocarcinoma), HeLa (cervical cancer), HepG2 (liver carcinoma). |
| Viability Assay Kits | Quantify cell health and proliferation after exposure to test compounds [94]. | MTT, MTS, Alamar Blue. Measure metabolic activity as a proxy for viability. |
| High-Pressure View Cell | Visual observation of phase transitions and phase behavior in SCF systems. | Equipped with sapphire windows, stirrer, temperature control, and pressure sensor. |
| UHPLC-MS Systems | Analyze extracts from both IL and SCF processes, identifying and quantifying metabolites and compounds [95]. | Used with C18 columns for separation; MS detection for identification. |
The pursuit of sustainable and efficient solvent systems is a central theme in modern chemical research and industrial application. Within this context, two classes of materials have garnered significant attention: ionic liquids (ILs) and supercritical fluids (SCFs), particularly supercritical carbon dioxide (scCO₂). Both are often termed "green solvents," yet they possess distinct physicochemical properties that make them suitable for different applications. Framing this choice within a broader research context is essential, as the decision to use one over the other is not trivial but hinges on a complex interplay of process requirements, economic considerations, and environmental impact. This guide provides a technical comparison for researchers and drug development professionals, outlining the fundamental characteristics, advantages, and limitations of ILs and SCFs to inform solvent selection for specific applications.
Fundamental Properties and Core Characteristics
Ionic Liquids (ILs)
Ionic liquids are a class of salts that are liquid below 100 °C, often even at room temperature. They are composed entirely of ions and exhibit a unique set of properties driven by their customizable cation-anion combinations [1]. Their evolution is categorized into generations, from first-generation ILs as simple green solvents to fourth-generation ILs focusing on sustainability and multifunctionality [1].
Key Properties:
- Negligible Vapor Pressure: This property eliminates solvent evaporation losses and reduces inhalation hazards, contributing to safer working environments [96] [1].
- High Thermal Stability: ILs can remain stable at temperatures often exceeding 300 °C, making them suitable for high-temperature processes [1].
- Tunable Physicochemical Properties: By selecting different cation-anion pairings, properties such as solubility, viscosity, and hydrophobicity can be finely adjusted for specific tasks [1] [6].
- High Ionic Conductivity: This makes them exceptional candidates for electrochemical applications, including batteries and supercapacitors [96] [1].
- Wide Electrochemical Window: They allow for electrochemical processes to occur without solvent decomposition over a broad voltage range [96].
Supercritical Fluids (SCFs)
A supercritical fluid is any substance at a temperature and pressure above its critical point, where it exhibits properties intermediate between those of a gas and a liquid [4]. Supercritical carbon dioxide (scCO₂) is the most widely used SCF due to its accessible critical point (31.1 °C, 72.9 bar), low cost, and non-toxic nature [4].
Key Properties of scCO₂:
- Tunable Density and Solvation Power: The solvating power of an SCF is highly dependent on its density, which can be finely adjusted by changing the pressure and temperature [4] [97]. This allows for selective extraction and fractionation.
- High Diffusivity: SCFs have diffusion coefficients an order of magnitude higher than liquids, which significantly enhances mass transfer rates [4].
- Low Viscosity: The viscosity of SCFs is closer to that of gases, leading to low resistance to flow and improved penetration into porous materials [4].
- Environmental Benignity: ScCO₂ is non-toxic, non-flammable, and leaves no harmful residues. Using it also represents a potential pathway for carbon utilization [96] [4] [97].
Table 1: Core Characteristics and Typical Applications of ILs and SCFs
| Feature | Ionic Liquids (ILs) | Supercritical Fluids (SCFs, notably scCO₂) |
|---|---|---|
| Physical State | Liquid salt at low temperatures (<100 °C) | Fluid state above critical temperature and pressure |
| Vapor Pressure | Negligible | High, tunable with pressure |
| Typical Viscosity | Can be high, variable with structure [6] | Low, similar to gases |
| Diffusivity | Low to moderate | High (gas-like) |
| Tunability | High (via cation/anion selection) | High (via pressure/temperature) |
| Thermal Stability | High (often >300 °C) | Dependent on solvent, scCO₂ is stable |
| Key Advantages | Non-volatile, highly versatile, excellent for electrochemistry | Excellent mass transfer, selective extraction, easily separated |
| Common Applications | Catalysis, electrochemistry, synthesis, biopolymers processing | Extraction, chromatography, particle formation, polymer foaming |
Advantages, Limitations, and Decision Framework
Advantages of Ionic Liquids
The advantages of ILs stem from their unique liquid salt constitution. Their near-zero vapor pressure is a primary benefit, enhancing workplace safety and enabling processes in high-vacuum systems without solvent interference [1]. Their high thermal stability allows for their use in demanding catalytic and synthetic processes where conventional solvents would decompose [1]. Perhaps their most powerful feature is their extreme tunability; by tailoring the cation and anion, ILs can be designed to be hydrophobic, hydrophilic, acidic, basic, or to dissolve specific classes of materials like biopolymers or metal oxides [1]. This has led to their description as "designer solvents." Furthermore, their high ionic conductivity and wide electrochemical windows make them indispensable as electrolytes in advanced energy storage systems like lithium-ion batteries and supercapacitors [96] [1].
Limitations of Ionic Liquids
Despite their promise, ILs face significant challenges. Their high viscosity can impede mass transfer and mixing, requiring more energy input for processes [6]. The synthesis of high-purity ILs can be complex and costly, potentially prohibiting large-scale industrial application [1] [6]. Although considered green due to non-volatility, the environmental impact and (eco)toxicity of many ILs are a major concern, as some are poorly biodegradable and can be toxic to aquatic life [6]. Finally, product separation and IL recycling can be difficult and energy-intensive, though strategies like using scCO₂ for extraction from ILs are being developed to address this [96].
Advantages of Supercritical Fluids (scCO₂)
The advantages of SCFs are largely tied to their transport properties and environmental profile. Their high diffusivity and low viscosity lead to superior mass transfer characteristics compared to liquid solvents [4]. The tunable solvating power, controlled simply by adjusting pressure and temperature, allows for highly selective extraction and fractionation without changing the solvent [4] [97]. A major practical advantage is the ease of solvent recovery; upon depressurization, scCO₂ reverts to a gas, leaving the extracted solute completely solvent-free and eliminating the need for energy-intensive distillation [97]. From an environmental and safety standpoint, scCO₂ is non-toxic, non-flammable, and readily available from industrial byproduct streams, making it a truly sustainable choice [4] [97].
Limitations of Supercritical Fluids
The primary limitation of SCFs is the high capital cost associated with the equipment needed to handle high-pressure processes [97]. While scCO₂ is an excellent solvent for non-polar molecules, its low polarity limits its application for polar or ionic compounds without the use of modifiers or co-solvents [96] [97]. The high pressure required for operation also introduces significant safety considerations and can limit the scalability of some processes [97].
Decision Framework: ILs vs. SCFs
The choice between ILs and SCFs is dictated by the specific demands of the application. The following diagram provides a logical workflow for this decision-making process.
Diagram 1: Solvent selection workflow
Experimental Protocols and Methodologies
Protocol: Supercritical Fluid Extraction (SFE) of Bioactive Compounds
SFE is a well-established technique for extracting sensitive, high-value compounds from natural sources like plants or algae [98] [97]. The following workflow outlines a standard procedure for SFE.
Diagram 2: SFE experimental workflow
Detailed Methodology:
- Sample Preparation: The raw material (e.g., plant leaves, seeds) is ground to a consistent particle size (e.g., 0.2-0.5 mm) to increase the surface area for extraction. The material may be dried to prevent ice formation and blockages during the process [97].
- Loading: The prepared biomass is accurately weighed and packed into the high-pressure extraction vessel.
- Pressurization: The system is sealed, and CO₂ is pumped into the vessel. The temperature and pressure are raised above the critical point (e.g., 40-60 °C, 150-350 bar) to achieve the supercritical state. The specific conditions are optimized based on the target solute's solubility [97].
- Extraction:
- Static Phase (Optional): The scCO₂ flow is stopped for a set period (e.g., 10-30 minutes) to allow for saturation.
- Dynamic Phase: The scCO₂ is continuously pumped through the vessel at a controlled flow rate (e.g., 1-10 g/min) for a defined time, carrying the dissolved solutes out of the matrix [97].
- Separation and Collection: The solute-laden scCO₂ stream is passed into a separate separator vessel maintained at a lower pressure. The rapid drop in pressure and solvating power causes the solute to precipitate and be collected. The gaseous CO₂ can be liquefied and recycled [97].
- Analysis: The extracted material is weighed to determine yield and analyzed using techniques like Gas Chromatography-Mass Spectrometry (GC-MS) or High-Performance Liquid Chromatography (HPLC) to identify and quantify the compounds [98].
Protocol: Synthesis and Application of Glycerol-Derived Ionic Liquids
This protocol, based on recent research, outlines the synthesis of a novel family of bio-based ILs from glycerol and their application in a catalytic reaction, addressing sustainability concerns of traditional ILs [6].
Detailed Methodology: A. Synthesis from Glycidyl Ethers:
- Reaction Setup: A glycidyl ether (e.g., glycidyl methyl ether) is placed in a reaction flask equipped with a stirrer and temperature control.
- Controlled Addition: Triethylamine (50% molar excess) and a Brønsted acid (e.g., HCl) are slowly added to the glycidyl ether at 80 °C. The slow addition is critical to minimize byproducts [6].
- Reaction and Monitoring: The reaction proceeds for 48 hours at 80 °C and is monitored by techniques like ¹H NMR to track the formation of the desired ammonium salt ([N20R]Cl) and byproducts like 1-chloro-3-alkoxypropan-2-ol (R0Cl) and triethylammonium chloride [6].
- Purification: The crude ionic liquid is purified to remove byproducts, often via washing and drying under vacuum, to achieve high purity.
B. Application in Heck–Mizoroki Coupling:
- Catalyst Preparation: Palladium nanoparticles are formed or dispersed within the synthesized glycerol-derived IL.
- Reaction Execution: The reactants (e.g., an aryl halide and an alkene) are added to the IL-catalyst system. The reaction is carried out under optimized conditions (e.g., temperature, time).
- Product Separation: Upon reaction completion, the product can be extracted using a solvent like scCO₂, which is immiscible with the IL and can diffuse through it efficiently, leaving the catalyst and IL behind [96] [6].
- Recycling: The IL and Pd nanoparticle catalyst system can be reused directly for subsequent reaction cycles, demonstrating the recyclability and efficiency of the system [6].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagents and Materials for IL and SCF Research
| Reagent/Material | Function/Application | Notes |
|---|---|---|
| Carbon Dioxide (CO₂) | Primary solvent for SCF processes. | High-purity (≥99.9%) food or research grade is typically used to prevent contamination [4] [97]. |
| Co-solvents (e.g., Ethanol, Methanol) | Modifiers to enhance scCO₂ polarity. | Added in small percentages (1-10%) to scCO₂ to improve the solubility of polar molecules [97]. |
| Ammonium/Phosphonium Salts | Precursors for Ionic Liquid synthesis. | e.g., Triethylamine; used to form the cation structure of the IL [6]. |
| Epichlorohydrin / Glycidyl Ethers | Bio-based platform molecules for IL synthesis. | Starting materials for creating glycerol-derived ILs, enhancing sustainability [6]. |
| Anion Sources (e.g., LiTf₂N) | To introduce specific anions via metathesis. | Allows for the customization of IL properties (e.g., bistriflimide [Tf₂N]⁻ for hydrophobicity) [6]. |
| Palladium Salts | Catalyst precursors for reactions in ILs. | Used to form catalytic nanoparticles in situ for cross-coupling reactions [6]. |
| High-Pressure Vessels/Reactors | Containment for SCF and high-pressure IL processes. | Constructed from stainless steel or alloys, rated for pressures > 100 bar, with safety features [97]. |
Ionic Liquids and Supercritical Fluids represent two powerful, yet distinct, pillars of modern green solvent technology. The choice between them is not a matter of which is universally superior, but which is optimally suited to the task at hand. ILs excel as sophisticated, tunable media for catalysis, synthesis, and electrochemistry where a stable, non-volatile liquid phase is paramount. In contrast, SCFs, particularly scCO₂, are unmatched in applications requiring superior mass transfer, selective extraction, and facile product recovery, especially for non-polar compounds. A promising future direction lies not only in the continued independent development of these solvents but also in their synergistic combination, such as using scCO₂ to extract products from IL reaction media, thereby merging the unique advantages of both systems to overcome their individual limitations. This hybrid approach paves the way for more efficient, sustainable, and innovative processes across the chemical and pharmaceutical industries.
The development of advanced drug delivery systems is pivotal in enhancing the therapeutic efficacy and safety profile of active pharmaceutical ingredients (APIs). Within this research domain, supercritical fluid (SCF) technology has emerged as a green and efficient alternative to conventional pharmaceutical processing methods. This case study examines the application of supercritical fluid technology, with a particular emphasis on carbon dioxide (scCO₂), in the design and fabrication of novel drug delivery systems. The content is framed within a broader thesis investigating the comparative properties of ionic liquids versus supercritical fluids, two classes of tunable solvents with significant potential in pharmaceutical engineering. Where ionic liquids offer unique capabilities as permeation enhancers and bioactive solvents, supercritical fluids provide an environmentally benign platform for particle engineering and material processing. This analysis explores their individual and synergistic applications, providing researchers and drug development professionals with a technical guide to current methodologies, experimental protocols, and emerging opportunities in the field.
Theoretical Foundations and Technological Principles
Supercritical Fluid Fundamentals
Supercritical fluids are substances maintained above their critical temperature (Tc) and critical pressure (Pc), where they exhibit unique properties intermediate between those of liquids and gases. This state confers high diffusivity (similar to gases), favorable density, and solvent strength (comparable to liquids), creating an efficient medium for mass transfer and chemical processing [99] [100]. Among various SCFs, carbon dioxide is the most widely employed in pharmaceutical applications due to its accessible critical point (Tc = 31.1°C, Pc = 73.8 bar), non-toxicity, non-flammability, and environmental acceptability [100]. The dielectric constant of SCFs correlates with pressure variations, allowing precise control over solvent characteristics through manipulation of temperature and pressure conditions, thereby governing solute dissolution and precipitation behavior [99].
Ionic Liquids as Pharmaceutical Agents
Ionic liquids (ILs) are salts composed of asymmetric organic cations and organic or inorganic anions with melting points typically below 100°C [101]. Their noteworthy properties include negligible vapor pressure, non-volatility, non-flammability, high thermal stability, and excellent solvation capabilities for diverse substances [60] [101]. In pharmaceutical contexts, ILs function as pharmacologically active components, permeation enhancers, green solvents, and solubilizers for poorly soluble APIs [60]. The physicochemical properties of ILs are highly tunable based on anion-cation combinations, enabling their design for specific biomedical applications, including transdermal drug delivery systems (TDDS) [101].
Comparative Properties in Drug Delivery Applications
Table 1: Comparative Properties of Supercritical CO₂ and Ionic Liquids in Pharmaceutical Applications
| Property | Supercritical CO₂ | Ionic Liquids | Pharmaceutical Relevance |
|---|---|---|---|
| Solvent Nature | Tunable with pressure/temperature | Tunable via ion selection | Both enable customized solvent environments for specific APIs |
| Diffusivity | High (gas-like) [100] | Low (liquid-like) | SCFs facilitate superior mass transfer in processing |
| Vapor Pressure | High upon depressurization | Negligible [101] | SCFs allow easy separation; ILs minimize solvent loss |
| Thermal Stability | N/A (decomposes upon heating) | High [101] | ILs suitable for high-temperature processes |
| Toxicity Profile | Non-toxic [100] | Varies (choline-based show low toxicity) [102] | Biocompatibility crucial for pharmaceutical acceptance |
| Primary Drug Delivery Functions | Particle engineering, impregnation, extraction | Permeation enhancement, API stabilization, solvent media | Complementary roles in formulation development |
Key Supercritical Fluid Technologies in Drug Delivery
Particle Formation Processes
SCF technology enables precise control over drug particle size and morphology, critically impacting bioavailability. Key operational processes include:
Rapid Expansion of Supercritical Solutions (RESS): The API dissolves in scCO₂ at elevated pressure and temperature. This solution is then rapidly expanded through a nozzle into a low-pressure chamber. The sudden decrease in solvent density drastically reduces solubilizing power, inducing supersaturation and precipitation of fine, uniform particles [99].
Supercritical Anti-Solvent (SAS): Suitable for substances insoluble in scCO₂, the SAS process involves dissolving the API in an organic solvent. scCO₂, acting as an anti-solvent, is introduced, causing a reduction in the solvent power of the organic liquid. This prompts rapid solute supersaturation and precipitation into high-purity particles with narrow size distribution. scCO₂ subsequently removes the organic solvent, minimizing residual solvent levels [99] [103].
Precipitation from Gas Saturated Solutions (PGSS): scCO₂ is dissolved into a liquid solution containing the drug or a drug-polymer melt. The mixture is then depressurized through a nozzle. The expansion causes the CO₂ to vaporize, cooling the matrix and precipitating the solute as fine particles or microspheres [99].
Advanced Dispersion and Impregnation Techniques
Beyond particle formation, SCF technology facilitates innovative dispersion and impregnation strategies:
Super-stable Homogeneous Intermix Formulating Technology (SHIFT): This technique addresses challenges in formulating hydrophilic agents within hydrophobic carriers, exemplified by dispersing Indocyanine Green (ICG) in Lipiodol for hepatocellular carcinoma treatment. SHIFT utilizes scCO₂ to achieve molecular-level dispersion without organic solvents, yielding formulations with superior stability and functional properties, such as enhanced photothermal conversion efficiency [99].
Supercritical Impregnation: scCO₂ swells polymeric matrices and carries bioactive compounds into the expanded polymer structure. Subsequent depressurization traps the active ingredient within the polymer, creating drug-eluting implants or dressings with controlled release profiles [100].
Experimental Protocols and Methodologies
Protocol for SAS Micronization of a Model API
Objective: To produce micronized particles of a poorly water-soluble drug (e.g., Ibuprofen) using the SAS process to enhance dissolution rate.
Materials:
- API: Ibuprofen (≥98% purity)
- Solvent: Dichloromethane (DCM) or Ethanol (HPLC grade)
- Anti-solvent: CO₂ (food-grade, 99.9%)
- Equipment: High-pressure SAS apparatus equipped with: co-solvent pump, CO₂ pump, thermostatted precipitation vessel (100-500 mL), nozzle (50-200 μm), back-pressure regulator, and particle collection filter.
Procedure:
- Saturation: Dissolve Ibuprofen in the organic solvent at a concentration of 10-50 mg/mL.
- Pressurization and Heating: Bring the precipitation vessel to the desired operating temperature (40-60°C) using a circulating jacket. Pressurize the vessel with scCO₂ to the target pressure (8-15 MPa) using the back-pressure regulator.
- Solution Injection: Pump the drug solution through the nozzle into the vessel at a controlled flow rate (1-5 mL/min) while maintaining constant pressure and temperature. scCO₂ acts as an anti-solvent, causing immediate drug precipitation.
- Washing: Continue pumping pure scCO₂ through the vessel for 30-60 minutes to remove residual organic solvent from the precipitated particles.
- Depressurization: Slowly depressurize the vessel (typically over 30-60 minutes) to atmospheric pressure to prevent particle agglomeration.
- Collection: Harvest the micronized powder from the filter and the bottom of the vessel for characterization.
Table 2: Exemplary SAS Operational Parameters for Various Compounds
| Active Compound | Solvent | Pressure (MPa) | Temperature (°C) | Reference |
|---|---|---|---|---|
| Ibuprofen | Dichloromethane | 10-15 | 40-50 | [99] |
| Antibiotics | Methanol, DCM | 8-12 | 40-60 | [99] |
| Fluorescent Probes | Dimethylformamide | 10-12 | 40-50 | [99] |
Protocol for Evaluating Ionic Liquids as Transdermal Permeation Enhancers
Objective: To assess the ability of a choline-based IL (e.g., [Choline][Geranate] or CAGE) to enhance the skin permeability of a model macromolecule drug (e.g., Dextran or Insulin).
Materials:
- Ionic Liquid: CAGE, synthesized and characterized prior to use.
- Drug: Fluorescently-labeled Dextran (e.g., FITC-Dextran, 4-40 kDa) or Insulin.
- Skin Model: Excised porcine or human epidermis (from skin bank), mounted in Franz diffusion cells.
- Receptor Fluid: Phosphate-buffered saline (PBS, pH 7.4) with optional antimicrobial agent.
- Analytical Equipment: Franz diffusion cell apparatus, fluorescence spectrophotometer or HPLC.
Procedure:
- Formulation Preparation: Prepare the test formulation by dissolving/dispersing the drug (1-5% w/w) in the neat IL or a pre-optimized IL/water mixture. A control formulation (drug in PBS) should be prepared concurrently.
- Skin Mounting: Thaw and carefully mount the skin pieces between the donor and receptor compartments of the Franz cells, ensuring the stratum corneum faces the donor side.
- Application: Apply a fixed volume (e.g., 500 μL) of the test and control formulations to the donor compartments. Seal the donor compartments to prevent evaporation.
- Sampling: Maintain the receptor compartment at 37°C with continuous stirring. Withdraw samples (e.g., 500 μL) from the receptor compartment at predetermined time intervals (e.g., 0, 2, 4, 6, 8, 12, 24 h) and replace with an equal volume of fresh pre-warmed receptor fluid.
- Analysis: Quantify the drug concentration in the samples using a validated analytical method (e.g., fluorescence measurement or HPLC).
- Data Analysis: Calculate cumulative drug permeation over time. Determine key parameters like flux (J) and permeability coefficient (K_p). Post-study, analyze skin samples via histology or spectroscopy (e.g., FTIR) to investigate IL-induced lipid extraction or keratin disruption mechanisms [101].
Visualization of Processes and Workflows
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for SCF and IL Drug Delivery Research
| Category/Item | Specification/Example | Primary Function | Application Notes |
|---|---|---|---|
| Supercritical Fluid | CO₂ (Food-grade, 99.9%) | Primary solvent/anti-solvent | Critical point: 31.1°C, 73.8 bar [100] |
| Co-solvents/Modifiers | Ethanol, Methanol (HPLC grade) | Enhance solubility of polar compounds in scCO₂ | Typically used at 1-10% (mol) [22] |
| Model APIs | Ibuprofen, Ketoprofen, Indocyanine Green (ICG) | Poorly soluble model drugs for process development | ICG used for imaging/therapy [99] |
| Biodegradable Polymers | PLGA, PVP, Chitosan | Formulation of composite particles & controlled release systems | Carrier in SAS/PGSS processes [99] [103] |
| Ionic Liquids | [Choline][Geranate] (CAGE), [Cho][Octanoate] | Permeation enhancers, solvents, or API-IL synthesis | Low toxicity profile; disrupts skin barrier [101] |
| Organic Solvents | Dichloromethane (DCM), Dimethylformamide (DMF) | Dissolve APIs for SAS process | Must be miscible with scCO₂; residual removal is key [99] |
| Cell Cultures | Caco-2, HaCaT, Fibroblasts | In vitro cytotoxicity and permeability assessment | Essential for biocompatibility testing (ISO 10993) [100] |
| Skin Models | Porcine ear skin, Human epidermis (EpiDerm) | Ex vivo permeation studies | Represents human skin barrier for transdermal research [101] |
Supercritical fluid technology represents a paradigm shift in pharmaceutical processing, offering a green, efficient, and highly controllable platform for developing advanced drug delivery systems. Its ability to produce particles with enhanced bioavailability, create stable formulations like SHIFTs, and impregnate polymers positions it as a cornerstone of modern pharmaceutical engineering. When viewed through the lens of comparative properties with ionic liquids, both technologies exhibit complementary strengths. The future of drug delivery lies not only in refining these individual technologies but also in exploring their synergies, such as using ILs as co-solvents in SCF processes or processing API-ILs with scCO₂ to create novel solid dosage forms. Continued research focusing on mechanistic understanding, standardization of evaluation methods, and rigorous biocompatibility assessment under physiological conditions will be crucial for translating these promising technologies from the laboratory to clinical practice, ultimately enabling more effective and patient-friendly therapies.
The pursuit of advanced electrochemical energy storage devices is inextricably linked to the development of safer, more stable, and high-performance electrolyte systems. Traditional organic electrolytes, while enabling high energy density, pose significant safety risks due to their volatility and flammability, and exhibit limited operational temperature ranges [104] [105]. This case study explores the stabilization of a lithium-ion battery (LIB) using an ionic liquid (IL)-based electrolyte, framing this investigation within a broader research context comparing the properties of ionic liquids and supercritical fluids as green solvent and process technologies [19]. Ionic liquids, or room-temperature ionic liquids, are organic salts with melting points below 100°C, characterized by their immeasurably low vapor pressure, high thermal stability, and non-flammability [104] [106]. These "designer solvents" can be structurally tuned for specific applications by selecting different cation-anion combinations [45] [19]. In parallel, supercritical fluids like carbon dioxide (scCO₂) offer tunable solvent properties and exceptional penetration capabilities, useful for processes such as the impregnation of active materials into porous electrodes or the extraction and recycling of electrolytes from spent devices [5] [107]. This study details the implementation of a pyrrolidinium-based IL electrolyte, demonstrating its role in enhancing the thermal safety and electrochemical stability of a commercial-scale energy storage device.
Technical Background and Rationale
The Limitations of Conventional Electrolytes
State-of-the-art lithium-ion batteries typically use LiPF₆ salt dissolved in a mixture of volatile alkyl carbonate solvents (e.g., ethylene carbonate, diethyl carbonate) [107] [105]. These electrolytes suffer from several critical drawbacks:
- High Flammability: The organic carbonates are volatile and flammable, creating a risk of thermal runaway, fire, or explosion under abusive conditions such as short circuits or local overheating [104] [105].
- Narrow Electrochemical Window: The operational voltage of these electrolytes is limited, restricting the energy density achievable by the device [105].
- Temperature Sensitivity: Performance severely degrades at low temperatures due to increased viscosity and salt precipitation, while high temperatures accelerate solvent evaporation and side reactions [104].
Ionic Liquids as Electrolyte Components
Ionic liquids present a compelling alternative to conventional electrolytes due to their unique physicochemical properties, which are summarized in Table 1 and compared with supercritical CO₂ [104] [45] [19].
Table 1: Key Properties of Ionic Liquids vs. Supercritical CO₂
| Property | Ionic Liquids | Supercritical CO₂ | Significance for Energy Storage |
|---|---|---|---|
| Vapor Pressure | Immeasurably low [19] | Tunable with pressure | Enables non-flammable electrolytes; eliminates solvent evaporation [104] |
| Thermal Stability | High (up to 300-400°C) [45] | Function of critical point | Allows device operation at elevated temperatures (>60°C) [104] |
| Electrochemical Window | Wide (up to 6.0 V) [104] | Not applicable as an electrolyte | Enables higher voltage cells and greater energy density [105] |
| Tunability | High (via cation/anion selection) [106] | Moderate (with co-solvents) [5] | Allows optimization of viscosity, conductivity, and electrode compatibility [45] |
| Primary Role in Energy Storage | Electrolyte solvent/component [105] | Process fluid for impregnation [5] and recycling [107] | ILs are integral to device function; scCO₂ is a tool for manufacturing/recycling |
For battery applications, ILs based on the pyrrolidinium (PYR) cation and the bis(trifluoromethanesulfonyl)imide (TFSI) anion are particularly promising due to their wide electrochemical stability window (>5 V) and robust thermal and chemical stability [45] [105]. Their low volatility directly addresses the primary safety concern of flammability associated with standard electrolytes [104].
Experimental Protocol: Formulation and Cell Assembly
This section provides a detailed methodology for fabricating and testing a lithium-ion battery stabilized with an IL-based electrolyte.
Materials and Reagents
Table 2: Research Reagent Solutions for IL-Electrolyte Formulation
| Reagent / Material | Function / Role | Specifications & Notes |
|---|---|---|
| PYR14TFSI | Primary IL solvent | 1-butyl-1-methylpyrrolidinium bis(trifluoromethanesulfonyl)imide; acts as the non-flammable, thermally stable base electrolyte [45]. |
| LiTFSI | Lithium salt | Source of Li⁺ ions; concentration typically 0.5-1.0 M in the IL [45]. |
| Vinylene Carbonate (VC) | SEI-forming additive | Forms a stable solid electrolyte interphase on the graphite anode, protecting it from reductive decomposition [45]. |
| Propylene Carbonate (PC) | Co-solvent | Reduces overall electrolyte viscosity, improving ion transport and low-temperature performance [45]. |
| Graphite Anode | Negative electrode | Commercial electrode coating on copper foil. |
| LiMnNiCoO₂ (NMC) Cathode | Positive electrode | Commercial LiNiMnCoO₂ coating on aluminum foil. |
| Celgard Separator | Mechanical separator | Porous polyolefin membrane. |
| Supercritical CO₂ Unit | Processing equipment | For electrolyte recycling studies; comprises a CO₂ cylinder, pump, heating chamber, and pressure vessel [107]. |
Electrolyte Formulation and Cell Assembly
- Electrolyte Preparation: The IL-based electrolyte is prepared in an argon-filled glovebox (H₂O, O₂ < 1 ppm). A 1.0 M solution of LiTFSI salt is dissolved in a mixture of PYR14TFSI and PC (e.g., 80:20 wt%). Subsequently, 2-5 wt% Vinylene Carbonate (VC) is added to the mixture. The solution is stirred at 50°C for 24 hours to ensure complete dissolution and homogeneity [45].
- Coin Cell Assembly: CR2032-type coin cells are assembled for testing. The electrodes (graphite anode and NMC cathode) are punched into discs. The cells are assembled by stacking the cathode, separator (soaked with the prepared electrolyte), and anode inside the coin cell casing, which is then crimped sealed inside the glovebox.
Electrochemical and Thermal Characterization
- Cyclic Voltammetry (CV): Performed to determine the electrochemical stability window of the electrolyte. A cell with inert electrodes (e.g., stainless steel) is scanned from the open-circuit voltage to higher and lower potentials. The voltage limits where significant current from electrolyte decomposition is observed define the anodic and cathodic stability limits [105].
- Galvanostatic Charge-Discharge (GCD) Cycling: The assembled coin cells are cycled between predefined voltage limits (e.g., 3.0 - 4.2 V) at various current rates (e.g., C/10, 1C) using a battery cycler. This test measures capacity, coulombic efficiency, and cycle life [45] [105].
- Thermal Stability Test: Cells or samples of the electrolyte are subjected to elevated temperatures (e.g., 60°C, 100°C) in an oven while their performance and physical state are monitored. For comparison, cells with conventional carbonate electrolytes are tested under identical conditions. The flash point of the electrolytes can be assessed using standard methods [104] [45].
- Electrolyte Recycling/Removal (Supplementary): To demonstrate the connection to supercritical fluid research, a spent cell is disassembled and the electrolyte is removed using supercritical CO₂. The cell components are placed in a pressure vessel. SC-CO₂, potentially with a co-solvent like ethanol, is pumped through the vessel at defined conditions (e.g., 50°C, 150 bar) to dissolve and extract the IL and lithium salts [5] [107].
The following workflow diagram illustrates the key stages of this experimental process.
Results and Discussion
Enhanced Thermal and Electrochemical Stability
The incorporation of the PYR14TFSI-based electrolyte fundamentally alters the safety profile of the lithium-ion cell. When subjected to high temperatures, the IL-based cell demonstrates remarkable stability, whereas a cell with a conventional carbonate electrolyte experiences rapid performance decay and risks of venting and fire due to solvent boiling and decomposition [104] [45]. The non-flammable nature of the IL eliminates the primary fuel for thermal runaway.
Electrochemically, the IL electrolyte facilitates a stable operating window exceeding 5.0 V, which is significantly wider than that of carbonate-based systems. This allows for the use of high-voltage cathode materials to achieve greater energy densities. The presence of additives like VC is crucial for forming a stable Solid Electrolyte Interphase (SEI) on the graphite anode, preventing continuous decomposition of the IL at the low reduction potentials required for anode operation [45].
Performance Metrics and Comparative Analysis
Quantitative data from cycling tests and property measurements are summarized in Table 3.
Table 3: Performance Comparison of Electrolyte Systems
| Performance Parameter | Conventional Carbonate Electrolyte | PYR14TFSI-based IL Electrolyte | Testing Conditions |
|---|---|---|---|
| Ionic Conductivity | ~10⁻² S cm⁻¹ [105] | ~10⁻³ - 10⁻² S cm⁻¹ [45] | Room Temperature |
| Viscosity | Low (~10 cP) [105] | High (~50-200 cP) [105] | Room Temperature |
| Electrochemical Window | ~4.5 V [105] | >5.5 V [45] | Inert electrodes |
| Flash Point | Flammable | Non-flammable [45] [105] | Standard test |
| Capacity Retention | Rapid decay at T > 60°C | >90% after 100 cycles at 60°C [45] | Graphite/NMC Cell |
| Performance at -20°C | Severe capacity loss | Moderate capacity loss; functional with formulation optimization [104] | Graphite/NMC Cell |
The data shows a trade-off: the IL electrolyte offers superior safety and high-voltage stability but at the cost of higher viscosity, which leads to slightly lower ionic conductivity at room temperature. This can impact rate capability. However, as shown in the capacity retention data, the overall cycling stability, especially at elevated temperatures, is excellent [45].
The following diagram illustrates the multi-faceted stabilization mechanism of the IL electrolyte within the battery cell.
This case study demonstrates that ionic liquid electrolytes, specifically those based on the PYR14TFSI system, successfully stabilize lithium-ion batteries against thermal runaway and enable operation over a wider temperature and voltage range. The experimental data confirms a significant enhancement in safety without compromising long-term cycling performance. The synergy between ionic liquids as functional electrolytes and supercritical fluids as processing aids for recycling presents a powerful, sustainable lifecycle strategy for advanced energy storage devices [5] [107] [19].
Future work will focus on synthesizing new IL cations and anions with lower viscosity and cost, further improving low-temperature performance. The development of "task-specific" ILs designed to form ideal SEI layers or suppress lithium dendrite growth represents a key frontier. Integrating these advanced ILs with scalable, green manufacturing and recycling processes using supercritical fluids will be crucial for realizing the full potential of next-generation, safe, and high-performance energy storage systems.
Conclusion
Ionic liquids and supercritical fluids are not competing technologies but rather complementary pillars of modern green chemistry, each with a distinct and powerful role in advancing biomedical research and clinical applications. Ionic liquids offer unparalleled tailorability for drug formulation and stable electrochemistry, while supercritical CO2 provides an unmatched, tunable medium for clean and efficient particle engineering. The future lies in the intelligent selection and potential hybridization of these solvents, guided by a clear understanding of their properties and optimized through emerging tools like machine learning. Their continued development promises to drive significant progress in creating safer, more effective pharmaceuticals, from enhanced nanomedicines and targeted drug delivery systems to robust energy storage for medical devices, ultimately pushing the boundaries of therapeutic efficacy and patient care.
References
- 1. A review on the evolution of ionic liquids: Sustainable ... [https://www.sciencedirect.com/science/article/pii/S2589234725000892]
- 2. Ionic liquid-based transdermal drug delivery systems for ... [https://pubs.rsc.org/en/content/articlehtml/2025/cc/d5cc03935e]
- 3. Solvate ionic liquids: past, present and future [https://pubs.rsc.org/en/content/articlelanding/2025/ta/d5ta01406a]
- 4. Supercritical Fluids: Properties and Applications [https://www.intechopen.com/chapters/82202]
- 5. Modeling of ionic liquid solubility in supercritical carbon ... [https://ui.adsabs.harvard.edu/abs/2025FlPEq.59514417H/abstract]
- 6. Glycerol-derived ionic liquids: a new family of high-potential ... [https://pubs.rsc.org/en/content/articlehtml/2025/su/d5su00393h]
- 7. Ionic liquids revolutionizing drug delivery [https://www.sciencedirect.com/science/article/abs/pii/S0168365925009691]
- 8. Facilities | supercriticalfluids.che [https://supercriticalfluids.che.vt.edu/testing11.html]
- 9. 12.4: Phase diagram [https://chem.libretexts.org/Courses/College_of_the_Canyons/Chem_201%3A_General_Chemistry_I_OER/12%3A_Liquids_and_Solids/13.04%3A_Phase_diagram]
- 10. Supercritical Fluids: An Innovative Strategy for Drug ... [https://www.mdpi.com/2306-5354/11/8/788]
- 11. High-Fidelity Prediction of Drug Solubility in Supercritical ... [https://www.sciencedirect.com/science/article/pii/S0928098725003197]
- 12. Predicting drug solubility in supercritical carbon dioxide ... [https://www.nature.com/articles/s41598-025-24006-7]
- 13. The Journal of Supercritical Fluids | Vol 219, May 2025 [https://www.sciencedirect.com/journal/the-journal-of-supercritical-fluids/vol/219/suppl/C]
- 14. Advanced hybrid machine learning based modeling for ... [https://www.nature.com/articles/s41598-025-04450-1]
- 15. A Review of the State of the Art on Ionic Liquids and Their ... [https://www.mdpi.com/1996-1073/18/15/4053]
- 16. - Wikipedia Supercritical fluid [https://en.wikipedia.org/wiki/Supercritical_fluid]
- 17. Example | Supercritical and Uses | Shimadzu Fluid Properties [https://www.shimadzu.com/an/service-support/technical-support/liquid-chromatography/advanced_analysis/properties-of-supercritical-fluids.html]
- 18. Thermal and Electrochemical Properties of Ionic Liquids ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9864641/]
- 19. Ionic Liquids and Supercritical Fluids: Revolutionizing Green ... [https://www.primescholars.com/articles/ionic-liquids-and-supercritical-fluids-revolutionizing-green-chemistry-123719.html]
- 20. Ionic liquids in polymer technology - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/gc/d4gc05445h]
- 21. An Overview on the Role of Ionic Liquids and Deep ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11946670/]
- 22. Prediction of ionic liquid solubilities in supercritical CO 2 + ... [https://www.sciencedirect.com/science/article/pii/S0167732224003805]
- 23. Ionic Liquid Enhanced Supercritical Fluid Extraction [https://jagworks.southalabama.edu/theses_diss/14/]
- 24. A review of ionic liquids towards supercritical fluid ... [https://www.sciencedirect.com/science/article/abs/pii/S0896844607002033]
- 25. Properties of Supercritical Fluids [https://www.shimadzu.eu/service-support/technical-support/liquid-chromatography/advanced_analysis/properties-of-supercritical-fluids.html]
- 26. Supercritical Fluid Technology: An Emphasis on Drug Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5849475/]
- 27. Supercritical fluids for pharmaceutical particle engineering [https://www.sciencedirect.com/science/article/abs/pii/S025527011200116X]
- 28. Particle synthesis by rapid expansion of supercritical ... [https://www.sciencedirect.com/science/article/abs/pii/S0021850221006741]
- 29. Ionic Liquids towards the Supercritical Fluid: A Review Paper [https://www.jetir.org/view?paper=JETIRFJ06006]
- 30. Tailoring Particle Microstructures via Supercritical CO₂ ... [https://pubmed.ncbi.nlm.nih.gov/25876917/]
- 31. Supercritical fluid technology for solubilization of poorly ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8915588/]
- 32. A comparative study of biocatalysis in non-conventional solvents [https://pubs.rsc.org/en/content/articlelanding/2004/gc/b405614k]
- 33. A comparative study between ionic liquid coating and ... [https://www.sciencedirect.com/science/article/pii/S2666952820300248]
- 34. A Deep Dive into Particle Engineering as a Drug Delivery ... [https://www.biopharminternational.com/view/a-deep-dive-into-particle-engineering-as-a-drug-delivery-tool]
- 35. Nanoparticles and Nanocrystals by Supercritical CO 2 [https://www.mdpi.com/2076-3417/11/4/1476]
- 36. Ampicillin Nanoparticles Production via Supercritical CO2 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4666252/]
- 37. Supercritical and Near Critical CO2 Micronization [https://separeco.com/supercritical-co2-micronization-equipment/]
- 38. Micro and nanosizing of Tamsulosin drug via supercritical ... [https://www.sciencedirect.com/science/article/pii/S2212982024001823]
- 39. in Supercritical - Research portal Eindhoven... fluids ionic liquids [https://research.tue.nl/en/publications/supercritical-fluids-in-ionic-liquids]
- 40. Role and Recent Advancements of Ionic Liquids in Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9963759/]
- 41. An insight into pharmaceutical challenges with ionic liquids [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2024.1454247/full]
- 42. Biocompatible Ionic Liquids for Drug Delivery [https://allanchem.com/biocompatible-ionic-liquids-drug-delivery/]
- 43. Eden Tanner coats nanoparticles in liquid salts for targeted ... [https://cen.acs.org/materials/ionic-liquids/Trailblazers-Eden-Tanner-coats-nanoparticles-liquid-drug-delivery/103/web/2025/09]
- 44. Application of Ionic Liquids in Electrochemistry—Recent ... [https://www.mdpi.com/1420-3049/25/24/5812]
- 45. Ionic Liquid-Based Electrolytes for Energy Storage Devices [https://pmc.ncbi.nlm.nih.gov/articles/PMC7240671/]
- 46. Ionic liquids as battery electrolytes for lithium ion batteries [https://www.sciencedirect.com/science/article/pii/S0167273823001984]
- 47. Recognition of Ionic as High-Voltage Liquids for... Electrolytes [https://pmc.ncbi.nlm.nih.gov/articles/PMC7214745/]
- 48. Electrochemistry in supercritical fluids: A mini review [https://www.sciencedirect.com/science/article/abs/pii/S1388248114001143]
- 49. Electrodeposition of metals from supercritical fluids - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2736432/]
- 50. Enhanced electrochemical performance of supercritical ... [https://www.sciencedirect.com/science/article/abs/pii/S0378775322005900]
- 51. Supercritical CO2 extraction behavior of electrolyte ... [https://www.sciencedirect.com/science/article/pii/S2212982025001295]
- 52. Supercritical CO2 technology for the treatment of end-of-life ... [https://pubs.rsc.org/en/content/articlelanding/2024/su/d4su00044g]
- 53. Supercritical carbon dioxide [https://en.wikipedia.org/wiki/Supercritical_carbon_dioxide]
- 54. Supercritical CO2 technology for biomass extraction: Review [https://www.sciencedirect.com/science/article/pii/S0926669025008945]
- 55. Botanical Innovation: New Extraction Technologies In 2025 [https://bedrockbotanicals.com/botanical-innovation-new-extraction-technologies-in-2025/]
- 56. Ionic Liquids in Pharmaceutical and Biomedical Applications [https://pmc.ncbi.nlm.nih.gov/articles/PMC10818567/]
- 57. Carbon Dioxide Supercritical Extraction - an overview [https://www.sciencedirect.com/topics/engineering/carbon-dioxide-supercritical-extraction]
- 58. EXTRACTION THROUGH SUPERCRYTIC CO2 [https://www.ucm.es/otrien/complutransfer-extraction-through-supercrytic-co2]
- 59. Supercritical Carbon Dioxide (CO2) Extraction Method [https://www.coleparmer.com/tech-article/supercritical-co2-extraction-method]
- 60. Ionic liquids: An innovative approach in drug delivery [https://www.sciencedirect.com/science/article/pii/S2590238524002376]
- 61. Optimization of conditions for supercritical fluid extraction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1390442/]
- 62. API-ILs Ionic Adducts: Solubility and Surface Activity ... [https://pubmed.ncbi.nlm.nih.gov/41056550/]
- 63. Application of theoretical and semi-empirical equations for ... [https://www.sciencedirect.com/science/article/abs/pii/S026322412500867X]
- 64. Parameter Effects and Optimisation in Supercritical Fluid ... [https://www.mdpi.com/2297-8739/9/12/385]
- 65. A new method based on binary mixture concept for ... [https://www.nature.com/articles/s41598-025-91633-5]
- 66. High-pressure solubility of methadone hydrochloride in supercritical CO2: Experimental investigation and correlations - Arabian Journal of Chemistry [https://arabjchem.org/high-pressure-solubility-of-methadone-hydrochloride-in-supercritical-co2-experimental-investigation-and-correlations/]
- 67. Predicting the solubility of drugs in supercritical carbon ... [https://pubmed.ncbi.nlm.nih.gov/40250491/]
- 68. Ionic liquid - Wikipedia [https://en.wikipedia.org/wiki/Ionic_liquid]
- 69. A review on machine learning algorithms for the ionic liquid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8153233/]
- 70. Prediction of CO2 solubility in Ionic liquids for CO2 capture ... [https://www.nature.com/articles/s41598-024-65499-y]
- 71. Uncovering ion transport mechanisms in ionic liquids using ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d4dd00378k]
- 72. Room Temperature Ionic Liquids as Green Solvent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6273002/]
- 73. Viscosity of Ionic Liquids: Application of the Eyring's Theory ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7795042/]
- 74. Modeling of ionic liquids viscosity via advanced white-box ... [https://www.nature.com/articles/s41598-024-55147-w]
- 75. Enhanced lithium-ion conductivity and robust interphases [https://www.sciencedirect.com/science/article/abs/pii/S1385894725016481]
- 76. Hybrid data-driven and physics-based modeling for ... [https://www.sciencedirect.com/science/article/pii/S2468025724000189]
- 77. 3-vinylimidazolium cation: Asymmetric acidic anion ... [https://www.sciencedirect.com/science/article/pii/S2772422025000448]
- 78. Neoteric Solvents Market | Global Market Analysis Report [https://www.factmr.com/report/neoteric-solvents-market]
- 79. Application of Ionic Liquids for Batteries and Supercapacitors [https://pmc.ncbi.nlm.nih.gov/articles/PMC8197857/]
- 80. Ionic Liquids for Energy Storage Applications [https://www.merckmillipore.com/QA/en/technical-documents/technical-article/materials-science-and-engineering/batteries-supercapacitors-and-fuel-cells/ionic-liquids-for]
- 81. Ionic liquids for electrochemical applications: Correlation ... [https://www.sciencedirect.com/science/article/abs/pii/S0167732222015392]
- 82. Ionic liquids for advanced materials [https://www.sciencedirect.com/science/article/abs/pii/S2588842021000511]
- 83. Exploring Supercritical Fluids: Their Traits and Advantages [https://extraktlab.com/supercritical-fluid/]
- 84. Ionic Hydraulic Fluids and Seal-Material Compatibility [https://www.mdpi.com/2076-3417/14/5/2187]
- 85. Material Selection for Manufacturability: Engineering ... [https://www.modusadvanced.com/resources/blog/material-selection-for-manufacturability-engineering-guidelines]
- 86. The Importance of GMP-Compatible Equipment Selection ... [https://www.thermofisher.com/blog/life-in-the-lab/gmp-compatible-equipment-in-qa-qc-labs/]
- 87. Guide of Pilot Plant Scale-Up Techniques [https://adesisinc.com/a-comprehensive-guide-to-pilot-plant-scale-up-techniques/]
- 88. Editorial: Properties and Applications of Ionic Liquids in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7769932/]
- 89. The Influence of Ionic Liquids on the Effectiveness of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7024248/]
- 90. Supercritical carbon dioxide chelation extraction of heavy ... [https://arabjchem.org/supercritical-carbon-dioxide-chelation-extraction-of-heavy-metal-ions-from-drilling-fluid-waste-experiment-and-simulation/]
- 91. Ionic Liquids Toxicity—Benefits and Threats [https://www.mdpi.com/1422-0067/21/17/6267]
- 92. Supercritical Fluid Extraction Chemicals Market Size [https://www.gminsights.com/industry-analysis/supercritical-fluid-extraction-chemicals-market]
- 93. Ionic Liquids as Advanced Lubricant Fluids - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6255031/]
- 94. A comprehensive dataset on cytotoxicity of ionic liquids [https://www.nature.com/articles/s41597-024-04190-3]
- 95. Are Ionic Liquids Better Extracting Agents Than Toxic ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2020.00450/full]
- 96. Advances and applications of supercritical carbon dioxide ... [https://www.sciencedirect.com/science/article/abs/pii/S0896844625000907]
- 97. Supercritical Fluid Extraction: A Green and Sustainable ... [https://www.ajgreenchem.com/article_221328.html]
- 98. Supercritical Fluid Extraction—A Sustainable and Selective ... [https://www.mdpi.com/2076-3417/15/11/5914]
- 99. Supercritical Fluids: An Innovative Strategy for Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11351119/]
- 100. Supercritical carbon dioxide and biomedicine [https://www.sciencedirect.com/science/article/abs/pii/S1385894722021106]
- 101. Design Principles and Applications of Ionic Liquids for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11578336/]
- 102. Design of ionic liquids containing glucose and choline as ... [https://www.nature.com/articles/s41598-022-25963-z]
- 103. Supercritical Fluid Technology: An Emphasis on Drug Delivery ... [https://pubmed.ncbi.nlm.nih.gov/28752598/]
- 104. Ionic Liquids for Energy Storage Applications [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/materials-science-and-engineering/batteries-supercapacitors-and-fuel-cells/ionic-liquids-for?srsltid=AfmBOor5NNU4IhOC81RVB075O1eYm03RMv867CRXD4X-v5ncqE5qdsPC]
- 105. Ionic Liquid Electrolytes for Electrochemical Energy ... [https://www.mdpi.com/1996-1944/14/14/4000]
- 106. A review of ionic liquids: Recent synthetic advances and ... [https://www.sciencedirect.com/science/article/abs/pii/S1876107023005242]
- 107. System and method for removing an electrolyte from ... [https://patents.google.com/patent/US20030186110A1/en]