Green vs. Conventional Nanoparticle Synthesis: A Sustainable Paradigm for Biomedical Applications
This article provides a comprehensive analysis of green synthesis methods for nanoparticles as sustainable alternatives to conventional chemical and physical approaches.
Green vs. Conventional Nanoparticle Synthesis: A Sustainable Paradigm for Biomedical Applications
Abstract
This article provides a comprehensive analysis of green synthesis methods for nanoparticles as sustainable alternatives to conventional chemical and physical approaches. Tailored for researchers and drug development professionals, it explores the foundational principles of green synthesis using plant extracts and microorganisms, detailing the mechanistic roles of phytochemicals. The scope extends to methodological advances, key applications in drug delivery and antimicrobial therapy, and critical troubleshooting of reproducibility and scalability challenges. A rigorous comparative validation assesses the biomedical efficacy and environmental impact of nanoparticles from different synthesis routes, concluding with future directions for clinical translation and the integration of AI-driven design.
The Rise of Green Nanosynthesis: Principles, Drivers, and Historical Context
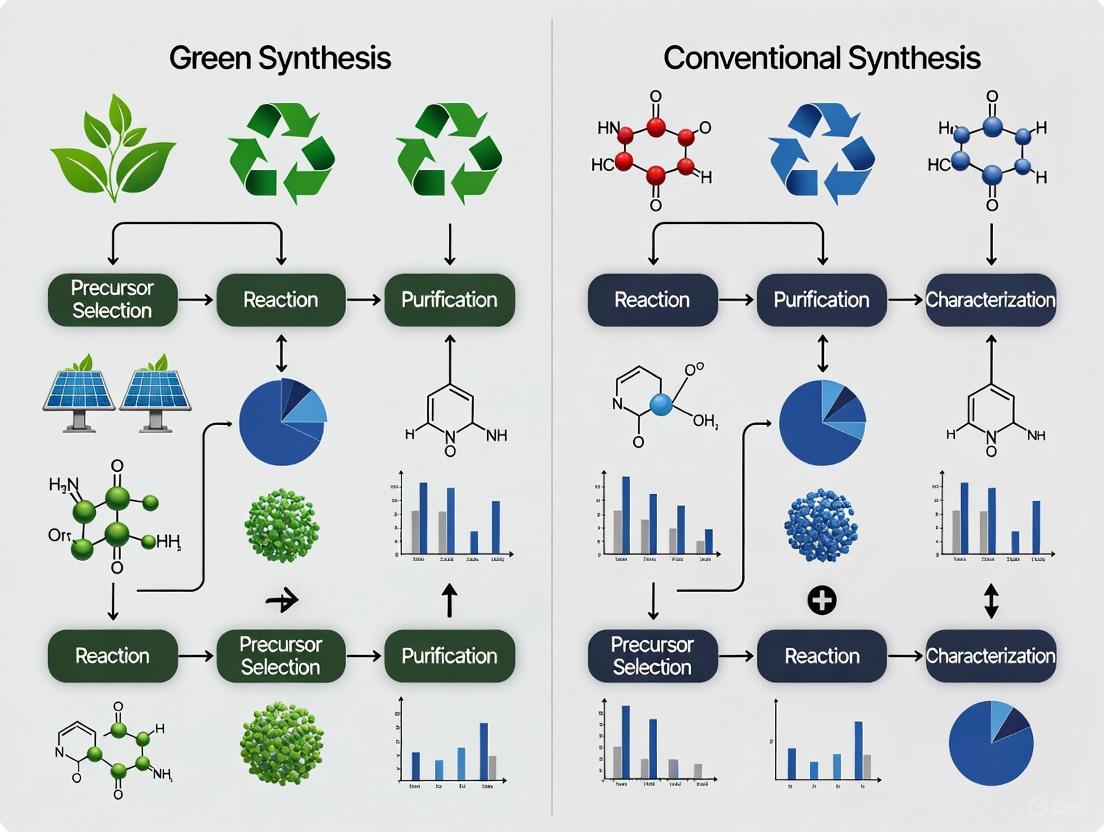
The synthesis of nanoparticles is a foundational process in nanotechnology, enabling advancements across medicine, electronics, and agriculture. Traditional synthesis methods have predominantly relied on physical and chemical approaches that often involve high energy inputs, hazardous reagents, and the generation of toxic byproducts [1] [2]. In response to these environmental and safety concerns, green synthesis has emerged as a sustainable alternative that aligns with the principles of green chemistry [3]. This approach utilizes biological resources such as plant extracts, microorganisms, and enzymes as reducing and stabilizing agents, offering a pathway to produce nanoparticles that is both eco-friendly and biologically superior [4] [5]. The core distinction lies in the replacement of synthetic chemicals with natural phytochemicals, leading to nanoparticles with enhanced biocompatibility and functionality while minimizing environmental impact [6] [3]. This guide provides a comprehensive comparison between green and conventional nanoparticle synthesis methods, focusing on their underlying principles, experimental outcomes, and practical applications for researchers and drug development professionals.
Core Principles and Comparative Framework
Green synthesis operates according to well-defined principles that differentiate it from conventional approaches. The Twelve Principles of Green Chemistry provide a foundational framework, emphasizing waste prevention, safer chemicals, energy efficiency, and the use of renewable feedstocks [3]. In practical terms, this translates to using plant metabolites instead of synthetic reductants, water as a solvent instead of toxic organic solvents, and ambient temperature reactions instead of energy-intensive processes [5].
A critical analysis of the term "green" itself is warranted. While biosynthesis is frequently characterized as environmentally friendly in comparison to chemical synthesis, this designation should be supported by comprehensive life cycle assessments rather than assumed [7]. Some studies suggest that certain green synthesis methods may involve substantial energy inputs or lower product yields compared to optimized chemical approaches, highlighting the need for rigorous comparative analysis [7].
The table below summarizes the fundamental distinctions between green and conventional synthesis approaches:
Table 1: Fundamental Comparison Between Green and Conventional Nanoparticle Synthesis
| Parameter | Green Synthesis | Chemical Synthesis | Physical Synthesis |
|---|---|---|---|
| Reducing Agents | Plant phytochemicals (e.g., flavonoids, terpenoids) [8] [5] | Synthetic chemicals (e.g., sodium borohydride, citrate) [2] | Physical forces (e.g., laser ablation, thermal evaporation) [2] |
| Stabilizing/Capping Agents | Natural biomolecules from extract [8] [5] | Synthetic polymers and surfactants (e.g., PVP, CTAB) [2] | Often requires additional stabilizing agents [2] |
| Solvent Medium | Typically water or other green solvents [3] | Often organic solvents [9] | Varies by method |
| Energy Requirement | Often ambient conditions or moderate heating [5] | Varies, can require high temperatures [2] | Generally high energy input (e.g., laser, thermal) [2] |
| Environmental Impact | Lower; biodegradable byproducts, sustainable resources [4] [3] | Higher; hazardous chemicals and waste [9] [2] | Lower chemical waste, but high energy consumption [2] |
| Scalability | Promising but challenges in standardization and reproducibility [5] [3] | Well-established and scalable [1] | Technically complex and costly to scale [1] |
Experimental Comparison: Green vs. Conventional Silver Nanoparticles
Methodology and Experimental Protocol
A comparative study investigating the efficacy of silver nanoparticles (AgNPs) for enhancing heat stress resilience in potato seeds provides robust experimental data [8]. The protocol for this comparative analysis is as follows:
- Green Synthesis of AgNPs: Silver nanoparticles were fabricated using a leaf extract from Azadirachta indica (neem). The extract, rich in terpenoids and flavonoids, served as both reducing and capping agent. A silver salt solution was mixed with the extract under controlled conditions [8].
- Chemical Synthesis of AgNPs: For comparison, AgNPs were synthesized chemically using sodium citrate as the reducing agent [8].
- Nanoparticle Characterization: Both nanoparticle types were characterized using UV-visible spectroscopy, X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), and dynamic light scattering (DLS) for size and zeta potential analysis [8].
- Nanopriming and Germination Assay: Potato seeds were primed with optimal concentrations (5 mg L⁻¹) of green or chemical AgNPs, as well as with water (hydropriming) as a control. Germination rates were monitored under normal and elevated temperature (32.2 °C) conditions. Seed uptake of nanoparticles was quantified using ICP-MS [8].
Quantitative Results and Performance Data
The experimental results demonstrated clear performance differences between the green and chemically synthesized nanoparticles.
Table 2: Experimental Characterization of Green vs. Chemical AgNPs [8]
| Characterization Metric | Green-Synthesized AgNPs | Chemically-Synthesized AgNPs |
|---|---|---|
| Crystallite Size (XRD) | 9.7 nm | 20.6 nm |
| Zeta Potential (DLS) | -55.2 mV | -35.7 mV |
| Colloidal Stability | Higher (due to higher zeta potential) | Lower |
| Germination Rate (Day 12) | 50% higher than hydroprimed control | 19% lower than green AgNPs |
| Metal Uptake in Seeds (ICP-MS) | 144 ppm | 105 ppm |
| Seed Mass Increase (Water Uptake) | 82% | Data not specified |
The superior physicochemical properties of green AgNPs directly translated to enhanced biological performance. Under heat stress, seeds primed with green AgNPs maintained a 10% higher germination rate compared to the hydroprimed control and exhibited more vigorous seedling growth [8]. The study attributed this enhanced efficacy to the smaller size and higher colloidal stability of the green AgNPs, which facilitated greater uptake and improved interaction with seed tissue [8].
Workflow and Property Relationships
The synthesis pathway directly determines the physicochemical properties of the resulting nanoparticles, which in turn dictates their performance in application. The following diagram illustrates this critical relationship and the general workflow for plant-mediated green synthesis.
The properties highlighted in the diagram are critically influenced by the choice of biological agent. For instance, the use of neem leaf extract resulted in smaller, more stable nanoparticles due to the effective capping action of its terpenoids and flavonoids [8]. This relationship underscores the importance of carefully selecting the biological resource based on its phytochemical profile to achieve desired nanoparticle characteristics.
The Scientist's Toolkit: Essential Reagents and Materials
Successful execution of green synthesis experiments requires specific reagents and materials. The following table details key components for plant-mediated synthesis, a common and accessible method.
Table 3: Essential Research Reagent Solutions for Green Synthesis
| Reagent/Material | Function in Synthesis | Example from Literature |
|---|---|---|
| Plant Leaf Extract | Serves as a source of natural reducing, capping, and stabilizing agents (e.g., flavonoids, phenols, terpenoids). | Azadirachta indica (Neem) leaf extract for AgNP synthesis [8]. Terminalia catappa for iron NP synthesis [6]. |
| Metal Salt Precursor | Provides the metal ions (e.g., Ag⁺, Fe³⁺, Zn²⁺) that are reduced to form nanoparticles (Ag⁰, Fe⁰, ZnO). | Silver nitrate (AgNO₃) for AgNPs [8]. Ferric chloride (FeCl₃·6H₂O) for iron NPs [6]. |
| Distilled Water | Acts as the primary, non-toxic solvent for preparing plant extracts and reaction mixtures. | Used universally as the solvent medium in green synthesis protocols [8] [6]. |
| Laboratory Centrifuge | Essential for purifying and concentrating synthesized nanoparticles from the reaction mixture. | Used to pellet nanoparticles after synthesis at speeds of 5000 rpm [6]. |
| Characterization Tools | A suite of instruments to confirm nanoparticle formation, size, shape, stability, and crystal structure. | UV-Vis Spectrophotometer, DLS/Zetasizer, SEM, TEM, XRD, FTIR [8] [6]. |
A significant challenge in the toolkit is the standardization of plant extracts. Variability in plant composition due to seasonality, geography, and cultivation practices can affect synthesis reproducibility [5]. Therefore, rigorous characterization and standardization of the starting plant material are crucial for consistent results.
The comparative data clearly establishes that green synthesis is not merely an alternative but often a superior strategy for producing functional nanoparticles. The core principles of sustainability and green chemistry are complemented by tangible performance benefits, including smaller particle size, enhanced stability, and improved biocompatibility, as demonstrated in agricultural applications [8] [6].
For researchers and drug development professionals, the future of green synthesis lies in addressing key challenges. Scalability and reproducibility remain primary hurdles, necessitating the development of standardized protocols and a deeper understanding of reaction kinetics [5] [3]. Furthermore, claims of environmental friendliness must be substantiated through comprehensive cradle-to-gate life cycle assessments that rigorously compare green routes with conventional ones across all environmental impact categories [7]. Continued innovation in utilizing renewable resources like agricultural waste and the development of hybrid synthesis methods will further solidify the role of green synthesis in fostering a sustainable and technologically advanced future [3].
The rapid development of nanotechnology has transformed industries from medicine to electronics, yet the conventional methods for synthesizing nanoparticles (NPs) present significant environmental and economic challenges. Traditional physical and chemical synthesis approaches are resource-intensive, relying on toxic chemicals and high energy consumption while creating substantial hazardous waste [10]. These methods have raised serious concerns about their long-term sustainability and environmental impact [10]. In response, green synthesis has emerged as a sustainable alternative that aligns with green chemistry principles and circular economy approaches [10]. This comprehensive analysis compares the performance of conventional and green nanoparticle synthesis methods, examining their relative toxicity, energy consumption, and economic feasibility through experimental data and life cycle assessment. The transition to sustainable synthesis methodologies addresses urgent problems of dwindling resources and environmental decline while presenting fresh possibilities for green technology innovation [10].
Comparative Analysis: Conventional vs. Green Synthesis
Quantitative Performance Comparison
Table 1: Direct comparison of conventional versus green synthesis methods across key performance metrics
| Performance Metric | Conventional Methods | Green Synthesis Methods | Experimental Support |
|---|---|---|---|
| Energy Consumption | High energy requirements | 30-40% reduction in energy use [10] | Life cycle assessment studies [10] |
| Production Costs | High operational costs | Up to 40% cost savings [10] [11] | Economic analysis of production processes [10] [11] |
| Production Output | Standard yield | Up to 50% increase in output [10] [11] | Scalability studies [11] |
| Environmental Toxicity | Uses hazardous chemicals, toxic solvents | Utilizes eco-friendly, biodegradable materials | Toxicity assays and environmental impact studies [4] [10] |
| Byproduct Management | Generates harmful waste | Minimal waste, valorization of byproducts | Circular economy principles application [10] |
| Synthesis Conditions | Harsh conditions (high T/P) | Ambient temperature and pressure [12] | Comparative synthesis experiments [12] |
Toxicity and Environmental Impact Assessment
Conventional nanoparticle synthesis employs toxic chemicals, hazardous substances, and toxic solvents that lead to harmful byproducts for the environment [4]. These methods typically involve metal precursors, reducing agents like citrate, ascorbate, or borohydride, and stabilizing agents such as polyvinylpyrrolidone and polyethylene glycol, all of which pose environmental and health concerns due to their toxicity, volatility, and use of non-renewable resources [10] [11]. The hazardous waste generated requires specialized disposal procedures and creates long-term environmental contamination risks [13].
In contrast, green synthesis utilizes eco-friendly, biodegradable materials and follows the principles of green chemistry, emphasizing non-toxic, resource-saving inputs [10]. Plant-based synthesis, for instance, uses natural phytochemicals including polyphenols, flavonoids, proteins, and alkaloids as reducing and stabilizing agents, eliminating the need for hazardous chemicals [14] [12]. Studies have confirmed that plant-derived nanoparticles are less likely to cause harmful side effects in humans compared to chemically synthesized counterparts [14]. Life cycle assessment (LCA) studies provide a systematic framework for evaluating the environmental impacts of both approaches, with green synthesis demonstrating significant advantages in reducing ecological footprints [10].
Energy Consumption Analysis
Traditional nanoparticle synthesis is energy-intensive, relying on processes such as high-temperature calcination, prolonged refluxing, hydrothermal reactions, and physical methods like laser ablation and arc discharge that require substantial energy inputs [10]. These energy demands contribute significantly to operational costs and environmental impacts through greenhouse gas emissions.
Green synthesis methods reduce energy consumption by 30-40% through several innovative approaches [10]. Microwave-assisted synthesis has emerged as a particularly efficient method, enabling rapid and uniform heating that significantly reduces reaction times and energy usage compared to conventional heating [10]. Additional low-energy methods include sol-gel processes, room-temperature synthesis, and mechanochemical approaches that eliminate or minimize energy requirements [10]. The implementation of sustainable synthesis reduces energy consumption by 30-40% while increasing production output by 50%, resulting in substantial cost savings [10].
Economic Viability and Production Efficiency
The economic advantages of green synthesis extend beyond energy savings to encompass multiple aspects of production efficiency. Green methods achieve cost savings of up to 40% compared to conventional approaches, making nanoparticle production more accessible and sustainable [11]. These savings derive from several factors: reduced expenses for specialized equipment, lower energy requirements, the utilization of inexpensive and renewable biological resources, and the elimination of costly toxic waste management procedures [4] [10].
Production output increases of up to 50% have been documented with green synthesis methods, enhancing their economic attractiveness [11]. This improved efficiency stems from simplified processes, faster reaction times, and the scalability of biological approaches. The use of renewable feedstocks such as plant extracts, agricultural waste, and microbial biomass further reduces material costs and enhances sustainability [10] [11]. The valorization of agro-industrial bio-waste has pioneered a novel method for converting agricultural residues and industrial byproducts into valuable nanomaterials, creating economic value from waste materials [11].
Experimental Protocols and Methodologies
Conventional Synthesis Workflow
Table 2: Detailed experimental protocol for conventional chemical synthesis of nanoparticles
| Experimental Step | Protocol Details | Purpose & Rationale |
|---|---|---|
| Precursor Preparation | Dissolve metal salts (e.g., AgNO3, HAuCl4) in deionized water or organic solvents | Provide metal ion source for nanoparticle formation |
| Reducing Agent Addition | Add chemical reducing agents (e.g., sodium citrate, sodium borohydride) under controlled conditions | Reduce metal ions to zerovalent atomic form |
| Stabilization | Introduce stabilizing agents (e.g., PVP, PEG, surfactants) | Control particle growth and prevent aggregation |
| Reaction Control | Maintain specific temperature, pH, and stirring conditions | Regulate nucleation and growth kinetics |
| Purification | Centrifugation, washing with solvents, dialysis | Remove unreacted precursors and byproducts |
| Characterization | UV-Vis, TEM, DLS, XRD, FTIR analysis | Verify size, shape, composition, and properties |
The experimental protocol for conventional synthesis typically involves reducing silver nitrate or other metal precursors in the presence of a stabilizing and reducing agent in aqueous solution [11]. Factors like the starting concentration of the metal salt solution, the stabilizing agent's concentration, and the molar ratio of metal salt to reducing agent significantly influence the size and properties of the resulting nanoparticles [11].
Green Synthesis Experimental Approach
Table 3: Standardized protocol for plant-mediated green synthesis of nanoparticles
| Experimental Step | Protocol Details | Purpose & Rationale |
|---|---|---|
| Plant Material Selection | Select and taxonomically identify plant species; collect specific parts (leaves, roots, seeds) | Source of bioactive reducing and stabilizing compounds |
| Extract Preparation | Wash, dry, grind plant material; prepare aqueous or ethanolic extract; filter to remove solids | Extract phytochemicals responsible for reduction |
| Metal Solution Preparation | Dissolve appropriate metal salt (e.g., AgNO3, HAuCl4, ZnAc) in deionized water | Provide metal ion precursor for nanoparticle formation |
| Reaction Initiation | Mix plant extract with metal solution at specific ratio, temperature, and pH | Initiate reduction of metal ions by phytochemicals |
| Color Change Observation | Monitor visual color change indicating nanoparticle formation | Preliminary confirmation of nanoparticle synthesis |
| Purification | Centrifugation, washing, and optional drying of nanoparticles | Remove biological debris and unreacted components |
| Characterization | Comprehensive analysis using UV-Vis, FTIR, TEM, SEM, DLS, XRD | Confirm nanoparticle properties and biomolecular capping |
A standardized method for the synthesis of nanoparticles utilizing plant extracts involves a systematic approach where a specific plant material is carefully selected and taxonomically identified, and the desired plant extract obtained [12]. Subsequently to the selection of plant parts, an extraction process using an appropriate solvent, followed by filtration/chromatography to eliminate any impurities, is carried out [12]. The continuous stirring of the reaction mixture offers better results in the form of uniform-sized nanoparticles, as is visually indicated by a noticeable change in colour [12].
Visualization of Synthesis Workflows
Conventional Synthesis Methodology
Green Synthesis Methodology
Research Reagent Solutions
Table 4: Essential research reagents and materials for nanoparticle synthesis experiments
| Reagent/Material | Function in Synthesis | Conventional Examples | Green Alternatives |
|---|---|---|---|
| Metal Precursors | Source of metal ions for nanoparticle formation | AgNO3, HAuCl4, ZnCl2, FeCl3 | Same metal salts with green extraction |
| Reducing Agents | Convert metal ions to elemental form | NaBH4, citrate, ascorbate, H2 gas | Plant polyphenols, flavonoids, terpenoids |
| Stabilizing Agents | Prevent aggregation and control growth | PVP, PEG, surfactants | Proteins, polysaccharides, alkaloids |
| Solvents | Reaction medium for synthesis | Toluene, DCM, hexane, DMF | Water, ethanol, water-ethanol mixtures |
| Biological Materials | Source of reducing/capping agents | Not applicable | Plant extracts, microorganisms, algae |
| Characterization Tools | Analyze NP properties | UV-Vis, TEM, SEM, FTIR, DLS | Same analytical techniques |
The selection of appropriate reagents is critical for both conventional and green synthesis approaches. For green synthesis, the three most important requirements are a green or environmentally friendly solvent (the most widely used are water, ethanol, and their mixtures), a suitable non-toxic reducing agent, and a safe substance for stabilization [14]. Plant extracts serve as ideal sources for reducing and stabilizing agents due to their diverse phytochemical composition including polysaccharides, vitamins, amino acids, proteins, phenolics, saponins, alkaloids, and terpenes [14]. These biomolecules collectively facilitate the reduction of metal ions and stabilization of the resulting nanoparticles through natural processes.
The comprehensive comparison between conventional and green synthesis methods demonstrates compelling environmental and economic advantages for sustainable approaches. Green synthesis methods significantly reduce toxicity by eliminating hazardous chemicals, decrease energy consumption by 30-40%, lower costs by up to 40%, and increase production output by 50% while maintaining nanoparticle quality and functionality [10] [11]. The experimental protocols for green synthesis leverage biological materials including plants, microorganisms, and waste products as sources of reducing and stabilizing agents, operating under ambient conditions that minimize environmental impact [11] [14] [12]. As nanotechnology continues to expand across industries, the adoption of green synthesis principles addresses critical sustainability challenges while offering economic benefits. Future research directions should focus on optimizing reaction parameters, enhancing reproducibility, scaling up production, and further evaluating the environmental footprint through comprehensive life cycle assessments. The integration of artificial intelligence and machine learning approaches presents promising opportunities for advancing green nanotechnology through improved prediction and control of synthesis parameters [10].
The evolution of nanoparticle synthesis represents a significant paradigm shift from traditional physical and chemical methods toward sustainable, bio-inspired approaches. This transition is driven by the need for environmentally friendly, cost-effective, and biocompatible nanomaterials for advanced applications in medicine, electronics, and environmental remediation [12] [15]. Conventional synthesis methods often involve harsh conditions, high energy inputs, and toxic chemicals, raising concerns about environmental impact and biocompatibility [16] [17]. In response, green synthesis utilizing biological resources such as plant extracts, fungi, bacteria, and algae has emerged as a promising alternative that aligns with green chemistry principles [4] [5]. This comprehensive analysis compares the historical development of these methodologies, their underlying mechanisms, experimental protocols, and performance metrics, providing researchers with objective data to guide synthesis strategy selection.
The fundamental distinction between approaches lies in their philosophical and practical foundations. Traditional physical and chemical methods typically follow a "top-down" (physical) or "bottom-up" (chemical) approach, often requiring external reducing agents, stabilizers, and high energy inputs [16] [1]. In contrast, bio-inspired synthesis harnesses the innate capabilities of biological systems to reduce metal ions and stabilize nanoparticles through naturally occurring biomolecules [12] [5]. This shift not only addresses environmental concerns but also opens new possibilities for creating nanoparticles with enhanced biocompatibility and novel functionalities for biomedical applications including drug delivery, biosensing, and wound healing [5] [17].
Historical Timeline and Methodological Evolution
The development of nanoparticle synthesis techniques has progressed through distinct eras, marked by key technological innovations. The timeline below illustrates the major methodological advancements that have shaped the field.
Figure 1. Historical progression of nanoparticle synthesis methods, highlighting the transition from traditional to green approaches. Key milestones show the field's evolution toward more sustainable practices, particularly from the 2000s onward with the introduction of biological and plant-based methods.
The historical trajectory reveals a clear shift toward sustainable approaches. Traditional methods like physical vapor deposition (PVD) and chemical vapor deposition (CVD) enabled early atomic-level thin films, while the sol-gel process (1970s) facilitated metal oxide nanoparticle production [10]. The 1980s introduced laser ablation and arc discharge methods, followed by 1990s advancements in hydrothermal/solvothermal synthesis and the landmark discovery of carbon nanotubes [10]. A significant transition occurred in the 2000s with the emergence of microwave-assisted synthesis and plant-based green methods, gaining popularity for silver and gold nanoparticle production [10]. Recent decades have focused on solvent-free mechanochemical synthesis (2010s) and self-assembly techniques for complex nanostructures (2020s), reflecting the field's commitment to efficiency, precision, and sustainability [10].
Comparative Analysis of Synthesis Methodologies
Fundamental Synthesis Mechanisms
Nanoparticle fabrication strategies are fundamentally categorized into top-down and bottom-up approaches, each with distinct operational principles as visualized below.
Figure 2. Fundamental mechanisms of nanoparticle synthesis: top-down versus bottom-up approaches. Green synthesis primarily follows a bottom-up pathway using biological reduction agents, contrasting with traditional physical and chemical methods.
Top-down approaches involve the mechanical breakdown of bulk materials into nanoscale structures through methods like thermal/laser ablation, mechanical milling, chemical etching, and sputtering [16]. These methods typically require sophisticated equipment and significant energy input, often resulting in surface imperfections and broad size distributions [1].
Bottom-up approaches construct nanoparticles from atomic or molecular precursors via chemical reactions or biological reduction. Traditional chemical methods include chemical/electrochemical precipitation, spray pyrolysis, vapor deposition, and atomic/molecular condensation [16]. In contrast, bio-inspired bottom-up approaches utilize biological structures such as plant extracts, bacteria, fungi, algae, and yeasts as reducing and capping agents, creating nanoparticles through environmentally benign processes [16] [12].
Comprehensive Method Comparison
Table 1. Comparative analysis of traditional physical, chemical, and bio-inspired synthesis methods
| Parameter | Physical Methods | Chemical Methods | Bio-inspired Methods |
|---|---|---|---|
| General Approach | Top-down | Bottom-up | Bottom-up |
| Energy Consumption | High (high temperature/pressure) [16] | Moderate to high [16] | Low (ambient temperature/pressure) [12] [5] |
| Production Cost | High (sophisticated equipment) [16] | Moderate (chemical costs) [16] | Low (natural resources) [4] [5] |
| Environmental Impact | High energy footprint [16] | Toxic chemicals, hazardous byproducts [16] [12] | Eco-friendly, sustainable [16] [4] |
| Scalability | High for industrial scale [1] | Good scalability [1] | Challenges in standardization & scaling [18] [5] |
| Size Control | Moderate (broad distribution) [1] | Good control possible [1] | Variable (depends on biological system) [18] [12] |
| Shape Control | Limited [1] | Good with optimized parameters [1] | Moderate (biological template-dependent) [17] |
| Surface Chemistry | Often requires functionalization [1] | Tunable with capping agents [1] | Innately biocompatible [12] [5] |
| Biocompatibility | Variable (may require additional processing) [15] | Often limited (toxic residues) [12] | High (natural capping agents) [12] [5] |
| Key Examples | Laser ablation, mechanical milling, sputtering [16] | Chemical precipitation, sol-gel, hydrothermal [16] [1] | Plant extract synthesis, microbial synthesis [16] [12] |
Quantitative Performance Metrics
Table 2. Experimental performance data for different synthesis methods
| Performance Metric | Physical Methods | Chemical Methods | Bio-inspired Methods |
|---|---|---|---|
| Typical Yield | High mass production [1] | High yield [1] | Variable; plant extracts typically higher than microbial [16] [12] |
| Size Range (nm) | 10-100 [1] | 1-100 [1] | 1-100 (plant: 10-50 common) [19] [5] |
| Size Uniformity | Moderate to low [1] | Can be high with optimization [1] | Moderate; plant extracts generally better [16] [12] |
| Production Time | Minutes to hours [1] | Hours [1] | Minutes to hours (plant extracts faster) [12] |
| Energy Consumption | 30-50% higher than green methods [10] | 20-40% higher than green methods [10] | 30% reduction reported [10] |
| Antibacterial Efficacy | Variable | Variable | Strong (e.g., Ag NPs against E. coli, S. aureus) [16] [19] |
| Photocatalytic Activity | Material-dependent | Material-dependent | Excellent (e.g., SNPs degrade methylene blue) [19] |
| Thermal Conductivity Enhancement | Material-dependent | Material-dependent | Up to 170% in nanofluids [16] |
Experimental Protocols and Methodologies
Traditional Synthesis Procedures
Chemical Precipitation Method for Metal Oxide Nanoparticles:
- Precursor Preparation: Dissolve metal salt (e.g., zinc acetate for ZnO nanoparticles) in deionized water or organic solvent at concentration of 0.1-0.5 M [16]
- Precipitation: Add precipitating agent (e.g., sodium hydroxide, ammonium hydroxide) dropwise under constant stirring at 50-80°C until pH reaches 8-12 [16]
- Aging and Washing: Age precipitate for 1-24 hours, then wash repeatedly with deionized water and ethanol to remove impurities [16]
- Drying and Calcination: Dry at 80-120°C for 2-12 hours, followed by calcination at 300-600°C for 1-4 hours to obtain crystalline nanoparticles [16]
Laser Ablation for Metallic Nanoparticles:
- Target Preparation: Purify and polish bulk metal target (e.g., silver, gold) [1]
- Ablation Process: Immerse target in liquid medium (water, organic solvent), irradiate with pulsed laser (typical parameters: 1064 nm, 10 ns pulse duration, 10-100 mJ/pulse) [1]
- Collection: Centrifuge resulting colloidal solution to separate nanoparticles by size, resuspend in desired solvent [1]
Bio-inspired Synthesis Protocols
Plant-Mediated Synthesis of Silver Nanoparticles (SNPs):
- Plant Extract Preparation: Wash plant leaves (e.g., Ocimum sanctum, Azadirachta indica), dry, and grind to powder. Boil 1-10 g of powder in 100 mL deionized water for 10-30 minutes, filter through Whatman No. 1 filter paper [19]
- Reaction Mixture: Add 1-10 mL plant extract to 90 mL of 1 mM aqueous silver nitrate (AgNO₃) solution under constant stirring at 25-80°C [19] [5]
- Synthesis Monitoring: Observe color change from pale yellow to reddish-brown indicating SNP formation, typically within minutes to hours [19]
- Purification: Centrifuge at 10,000-15,000 rpm for 15-30 minutes, wash pellet with deionized water, redisperse via ultrasonication, repeat 2-3 times [19]
- Characterization: Analyze surface plasmon resonance using UV-Vis spectroscopy (peak at ~400-450 nm), size distribution via DLS, morphology by TEM/SEM [19] [5]
Microbial Synthesis of Gold Nanoparticles:
- Culture Preparation: Grow bacterial strain (e.g., Pseudomonas aeruginosa, Escherichia coli) or fungal culture in appropriate medium to late log phase [16] [12]
- Biomass Collection: Centrifuge culture, wash biomass with sterile deionized water [12]
- Exposure to Metal ions: Resuspend biomass in 1 mM chloroauric acid (HAuCl₄) solution, incubate at 25-37°C with shaking for 24-48 hours [12]
- Visual Confirmation: Color change to pink/purple indicates gold nanoparticle formation [12]
- Extraction: Centrifuge to separate biomass, collect nanoparticles from supernatant, purify via repeated centrifugation [12]
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3. Key research reagents and materials for nanoparticle synthesis
| Reagent/Material | Function | Traditional Synthesis | Bio-inspired Synthesis |
|---|---|---|---|
| Metal Precursors | Source of metallic elements | Metal salts (AgNO₃, HAuCl₄, ZnSO₄) [16] | Metal salts (AgNO₃, HAuCl₄, ZnSO₄) [16] |
| Reducing Agents | Convert metal ions to elemental form | Sodium borohydride, citrate, hydrazine [16] | Plant phytochemicals (polyphenols, flavonoids) [12] [5] |
| Stabilizing/Capping Agents | Control growth, prevent aggregation | Polymers (PVP), surfactants, thiols [16] | Natural biomolecules (proteins, polysaccharides) [12] [5] |
| Solvents | Reaction medium | Water, organic solvents (toluene, ethanol) [16] | Water, green solvents [10] |
| Biological Materials | Source of reducing/capping agents | Not typically used | Plant extracts, microorganisms, algae [16] [12] |
| Energy Sources | Drive reactions | High temperature, pressure, radiation [16] | Ambient conditions, microwave [12] [10] |
Applications and Performance Comparison
Biomedical Applications
Green-synthesized nanoparticles demonstrate exceptional performance in biomedical applications due to their innate biocompatibility and functionalization. Silver nanoparticles (SNPs) synthesized using plant extracts like Ocimum sanctum (tulsi), Curcuma longa (turmeric), and Azadirachta indica (neem) exhibit potent antibacterial efficacy against multidrug-resistant pathogens including Bacillus subtilis, Bacillus cereus, Pseudomonas aeruginosa, Staphylococcus aureus, and Escherichia coli [19]. These bio-inspired SNPs typically range from 10-50 nm with enhanced rigidity and tunable surface plasmon resonance, making them ideal for wound healing applications [19] [5].
In cancer therapeutics, gold nanoparticles synthesized through plant-mediated approaches show remarkable potential for drug delivery, photothermal therapy, and bioimaging due to their inert nature, comparable size to biological molecules, and tunable optical properties [5] [17]. Their high surface area and reactivity enhance production yields for biomedical applications while maintaining biocompatibility [5].
Environmental and Energy Applications
Green-synthesized nanoparticles demonstrate superior performance in environmental remediation and energy applications. Zinc oxide nanoparticles prepared through green methods exhibit significant antimicrobial and nematicidal activities against plant pathogens including Erwinia amylovora, Aspergillus flavus, Aspergillus niger, Fusarium oxysporum, and Alternaria alternate, making them valuable for agricultural applications [16].
In energy systems, nanofluids containing graphene and boron nitride nanoparticles synthesized through green methods with dry ice assistance show remarkable thermal conductivity enhancements up to 170% at 5 vol% concentration, demonstrating potential for improving efficiency in thermal management systems [16]. Cobalt ferrite/reduced graphene oxide (CoFe₂O₄/rGO) nanocomposites synthesized using gallic acid as a green agent enhance thermal performance in heat transfer systems, with studies reporting 27.8% enhancement in Nusselt number at 0.9 wt% nanoparticle concentration and Re = 1713 under laminar flow conditions [16].
The historical progression from traditional physical/chemical methods to bio-inspired synthesis represents a paradigm shift toward sustainable nanotechnology. While conventional approaches offer precision and scalability for industrial applications, green synthesis methods provide compelling advantages in environmental compatibility, reduced energy consumption, cost-effectiveness, and enhanced biocompatibility for medical applications [16] [10]. The integration of artificial intelligence and machine learning approaches promises to address current challenges in standardization and scalability of green synthesis methods [10]. Future research directions include optimizing bioreactor systems for microbial synthesis, standardizing plant extract compositions, developing circular economy approaches for nanomaterial production, and advancing hybrid methods that combine the precision of traditional approaches with the sustainability of bio-inspired synthesis [18] [10]. As the field evolves, the complementary strengths of both traditional and green approaches will likely converge to create innovative, sustainable nanotechnology solutions that address global challenges in healthcare, energy, and environmental protection.
The synthesis of nanoparticles (NPs) has remarkable importance across various medical, environmental, and industrial fields. Conventional physical and chemical synthesis methods often involve hazardous chemicals, toxic substances, and energy-intensive processes that lead to harmful byproducts [4] [20]. In contrast, green synthesis provides an environmentally responsible, economical, and safe alternative that encourages resource efficiency and reduced waste production [4]. This comparison guide objectively evaluates three key renewable resources—plant extracts, agricultural waste, and microorganisms—as biofactories for nanoparticle synthesis, with a specific focus on their performance in producing silver nanoparticles (AgNPs) and other metallic NPs. The content is framed within a broader thesis comparing sustainable methods against conventional approaches, providing researchers and drug development professionals with experimental data and protocols to inform their work.
Comparative Analysis of Biofactory Performance
The following tables summarize quantitative data on the synthesis conditions, nanoparticle characteristics, and application performance for the three primary bioresource categories.
Table 1: Synthesis Conditions and Nanoparticle Characteristics by Bioresource Type
| Bioresource Category | Example Sources | Optimal Size Range (nm) | Common Morphologies | Key Synthesis Parameters | Stability (Zeta Potential mV) | Synthesis Time |
|---|---|---|---|---|---|---|
| Plant Extracts | Neem, Tulsi, Turmeric leaves [20] [8] | 10-50 [20] | Spherical, rods, triangular, cubic [21] | pH, temperature, plant extract concentration [20] | -55.2 (Neem-mediated AgNPs) [8] | Few hours to 48 hours [22] |
| Agricultural Waste | Empty fruit bunches, banana peel, rice husks [23] [24] | 12.78-19.10 (EFB-AgNPs) [23] | Spherical, crystalline structures [23] | Extraction method, waste concentration, temperature | Not specified | Varies by waste type |
| Microorganisms | Bacteria (E. coli, Bacillus), Fungi (Fusarium, Aspergillus) [22] | 1-100 (AgNPs) [21] | Spherical, wire-like, star-shaped [21] | pH, temperature, reaction time, microbial strain [22] | High (fungal NPs generally more stable) [22] | 48-144 hours [22] |
Table 2: Application Performance of Nanoparticles from Different Bioresources
| Bioresource Category | Antibacterial Efficacy | Antioxidant Activity | Environmental Remediation | Agricultural Enhancement | Biomedical Applications |
|---|---|---|---|---|---|
| Plant Extracts | Broad-spectrum against multidrug-resistant pathogens [20] | High DPPH scavenging [23] | Excellent photocatalytic dye degradation [20] | 19% increased germination rate vs chemical AgNPs [8] | Drug delivery, wound healing [21] |
| Agricultural Waste | 40-80% inhibition against E. coli, S. aureus; 100% inhibition against Aspergillus species [23] | 81.49% DPPH scavenging at 160 μg/ml [23] | Dye degradation, heavy metal removal [24] | Improved seed germination, growth promotion [24] | α-amylase inhibition (74.64% at 100 μg/ml) [23] |
| Microorganisms | Effective against multidrug-resistant bacterial strains [22] | Reactive oxygen species (ROS) formation [22] | Water remediation, dye degradation [22] | Improved soil quality, plant growth promotion [22] | Anticancer activity, apoptosis induction [22] |
Experimental Protocols and Methodologies
Plant Extract-Mediated Synthesis
Protocol for Neem Leaf-Mediated AgNP Synthesis [8]
Preparation of Plant Extract: Collect fresh Azadirachta indica (neem) leaves, wash thoroughly with distilled water, and air dry. Boil 10 g of finely cut leaves in 100 mL of distilled water at 60-80°C for 10-15 minutes. Filter the mixture through Whatman No. 1 filter paper to obtain a clear extract.
Synthesis Reaction: Add 1 mM silver nitrate (AgNO₃) aqueous solution to the plant extract in a 1:9 ratio (extract:AgNO₃). Stir the reaction mixture continuously at room temperature for several hours until the color changes from pale yellow to brownish-red, indicating AgNP formation.
Purification: Centrifuge the resulting suspension at 15,000 rpm for 20 minutes. Discard the supernatant and resuspend the pellet in distilled water. Repeat this process three times to remove unwanted biological materials.
Characterization: Analyze the synthesized AgNPs using UV-Vis spectroscopy (surface plasmon resonance peak at 420-450 nm), XRD for crystallinity, TEM for size and morphology, and DLS for size distribution and zeta potential.
Agricultural Waste-Mediated Synthesis
Protocol for Empty Fruit Bunch (EFB) AgNP Synthesis [23]
Waste Processing: Collect empty fruit bunches from palm oil production and wash thoroughly. Dry the biomass at 60°C for 24 hours and grind to a fine powder.
Extract Preparation: Mix 10 g of EFB powder with 100 mL of distilled water and heat at 70°C for 1 hour. Filter the mixture through Whatman filter paper No. 1.
Nanoparticle Synthesis: Add the EFB extract to 1 mM AgNO₃ solution in a 1:4 ratio (extract:AgNO₃). Incubate the mixture at 80°C for 2 hours with continuous stirring until a dark brown color appears.
Characterization: Confirm AgNP formation through UV-Vis spectroscopy (peak at 477 nm), TEM analysis (size range 12.78-19.10 nm), and FTIR (identification of hydroxyl and amine groups as stabilizing agents).
Microorganism-Mediated Synthesis
Protocol for Fungal-Mediated AgNP Synthesis [22]
Microbial Cultivation: Grow fungal strains (e.g., Fusarium oxysporum) in appropriate liquid media (e.g., Malt Extract Glucose or Sabouraud Dextrose Broth) at 28°C for 72-96 hours under shaking conditions.
Biomass Separation: Filter the culture through Whatman filter paper No. 1 to separate the biomass from the culture filtrate. Wash the biomass with sterile distilled water to remove media components.
NP Synthesis: For extracellular synthesis, incubate the culture filtrate with 1 mM AgNO₃ solution in the dark at 28°C with continuous shaking. For intracellular synthesis, incubate the biomass with AgNO₃ solution under similar conditions.
Purification and Characterization: For extracellular synthesis, purify AgNPs by centrifugation at 15,000 rpm for 20 minutes. For intracellular synthesis, disrupt cells using sonication or French press before purification. Characterize using UV-Vis spectroscopy, TEM, and XRD.
Visualization of Synthesis Pathways and Workflows
Green Nanoparticle Synthesis Pathways
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents for Green Nanoparticle Synthesis Research
| Reagent/Material | Function in Synthesis | Specific Examples |
|---|---|---|
| Silver Nitrate (AgNO₃) | Primary precursor for silver nanoparticle formation | 1 mM aqueous solution for AgNP synthesis [23] [8] |
| Plant Extracts | Source of reducing and stabilizing phytochemicals | Neem, tulsi, turmeric leaf extracts [20] [8] |
| Agricultural Waste Biomass | Sustainable source of bioactive compounds | Empty fruit bunches, fruit peels, rice husks [23] [24] |
| Microbial Cultures | Biological factories for nanoparticle synthesis | Bacteria (E. coli, Bacillus), Fungi (Fusarium) [22] |
| Culture Media | Support microbial growth for synthesis | Malt Extract Glucose, Sabouraud Dextrose Broth [22] |
| Centrifuge | Separation and purification of nanoparticles | 15,000 rpm for 20 minutes for AgNP purification [23] [22] |
| Characterization Equipment | Size, morphology, and composition analysis | UV-Vis, TEM, XRD, FTIR, DLS [23] [21] [8] |
This comparison guide demonstrates that plant extracts, agricultural waste, and microorganisms each offer distinct advantages as biofactories for nanoparticle synthesis. Plant extracts provide rapid synthesis with excellent size control and diverse applications, particularly in biomedical fields [20] [21]. Agricultural waste offers superior sustainability by transforming environmental liabilities into valuable nanomaterials with significant biological activity [23] [24]. Microorganisms enable precise tuning of nanoparticle properties through genetic manipulation, with fungal systems particularly suited for scalable production [22].
While green synthesis methods show tremendous promise, challenges remain in standardization, scalability, and comprehensive toxicity profiling [20] [22]. Future research should focus on optimizing synthesis parameters for improved reproducibility, conducting thorough life cycle assessments, and exploring hybrid approaches that combine multiple bioresources to leverage their complementary advantages. These sustainable approaches represent a paradigm shift in nanoparticle synthesis that aligns with the principles of green chemistry and circular economy, offering researchers and drug development professionals environmentally responsible alternatives to conventional methods.
The synthesis of nanoparticles, particularly silver nanoparticles (AgNPs), is a cornerstone of modern nanotechnology with applications spanning biomedicine, catalysis, and environmental remediation. Traditional synthesis methods often rely on physical approaches requiring high temperature and pressure or chemical methods using toxic solvents and stabilizing agents, raising concerns about environmental impact and biocompatibility [25] [5]. In response, green synthesis has emerged as an environmentally friendly, safe, and cost-effective alternative. This method leverages biological resources, primarily plant extracts, to reduce metal ions and stabilize the resulting nanoparticles [26] [5]. The core of this process hinges on phytochemicals—bioactive compounds found in plants—which act as both reducing and stabilizing agents, eliminating the need for external, potentially hazardous chemicals [19] [5]. This guide objectively compares the performance of phytochemical-mediated synthesis against conventional methods, focusing on the mechanisms driven by key phytochemical classes such as phenolics, terpenoids, and proteins.
Comparative Mechanisms of Phytochemicals in Synthesis
In green synthesis, phytochemicals perform two critical functions: the reduction of metal ions (e.g., Ag⁺ to Ag⁰) and the stabilization of the newly formed nanoparticles to prevent aggregation. The specific role and efficacy depend on the phytochemical class involved. The table below provides a comparative overview of the primary phytochemical classes and their distinct contributions to nanoparticle formation and stability.
Table 1: Comparative Mechanisms of Key Phytochemical Classes in Nanoparticle Synthesis
| Phytochemical Class | Primary Role in Synthesis | Key Functional Groups | Impact on Nanoparticle Characteristics | Representative Sources |
|---|---|---|---|---|
| Phenolics & Polyphenols | Potent reduction; Strong stabilization via capping | Hydroxyl (-OH) groups on aromatic rings [27] | Smaller size; High stability; Spherical morphology [26] | Walnut green husk [25], A. australe [27], Tulsi, Turmeric [19] |
| Terpenoids | Reduction and stabilization | Carbonyl and hydroxyl groups [5] | Contributes to stability and bioactivity [5] | Essential oils [28] |
| Proteins & Amino Acids | Moderate reduction; Primary stabilization via electrostatic repulsion | Amino (-NH₂), carboxyl (-COOH), and thiol (-SH) groups [26] | Controls size and prevents aggregation; Polypeptides act as capping ligands [26] | Various plant extracts [26] |
| Flavonoids | Powerful reduction via tautomerization | Enol-to-keto tautomerization in flavonoid structure [26] | Rapid formation; High yield of nanoparticles [26] | Fruits, vegetables, tea [29] |
The overall biosynthesis process, from plant extract preparation to nanoparticle formation, follows a well-defined workflow that integrates the actions of these phytochemicals.
Figure 1: Generalized Workflow for the Green Synthesis of Metal Nanoparticles Using Plant Extracts. The process begins with the preparation of a phytochemical-rich extract, which is then mixed with a metal salt precursor to initiate a multi-stage reaction leading to stable nanoparticles [26].
Experimental Data and Performance Comparison
Quantitative Evidence from Experimental Studies
Robust experimental data underscores the efficacy of phytochemical-driven synthesis. The following table summarizes key performance metrics from published studies, highlighting the resulting nanoparticle properties and their enhanced bioactivity.
Table 2: Experimental Performance of Green-Synthesized Silver Nanoparticles (AgNPs)
| Plant Source / Synthesis Method | Average NP Size (nm) | Key Phytochemicals Involved | Reported Bioactivity (Compared to Controls) | Reference |
|---|---|---|---|---|
| Walnut Green Husk | 31.4 | Phenolic compounds | 70% cytotoxicity vs. MCF-7 cancer cells (40% for extract; 56% for commercial AgNPs). Significant antioxidant activity. Antibacterial activity (MIC = 5–30 µg/mL). | [25] |
| Acanthospermum australe | 14 ± 2 | Flavonoids, Phenolic compounds | Enhanced antimicrobial activity against skin pathogens. Lower cytotoxicity on human peripheral blood mononuclear cells. | [27] |
| Bacterium (S. flexneri) | 50 | Bacterial metabolites (as comparison) | Potent antioxidant and antifungal activity. Minimal toxicity against normal human cells. | [30] |
| Conventional Chemical Synthesis | Varies | Synthetic reductants (e.g., NaBH₄), Polymers | Often higher cytotoxicity in normal cells. Lacks bioactive, plant-derived capping layer. | [25] |
Detailed Experimental Protocol
To ensure reproducibility, a typical protocol for the green synthesis of silver nanoparticles using plant extracts is outlined below, based on methodologies from multiple studies [25] [27].
Preparation of Plant Extract:
- Plant Material: Wash and dry the selected plant part (e.g., leaves, husks). Comminute into a fine powder.
- Aqueous Extraction: Boil a specific weight (e.g., 10-50 g) of plant material in sterile deionized water (e.g., 100-1000 mL) for 5-10 minutes. Allow to steep at room temperature for approximately 1 hour.
- Filtration and Concentration: Filter the mixture sequentially through Whatman filter paper and a 0.22 µm membrane filter. The filtrate can be concentrated using a rotary evaporator and lyophilized to obtain a dry powder for storage.
Green Synthesis of Nanoparticles:
- Reaction Mixture: Reconstitute the lyophilized extract (or use the aqueous filtrate directly) in deionized water. Adjust the pH to an optimal level (often pH 8-10 using NaOH) to enhance reduction efficiency.
- Reduction Reaction: Gradually add a predetermined concentration of silver nitrate solution (e.g., 1-6 mM) to the extract solution under constant stirring (e.g., 1000 rpm) in dark conditions.
- Incubation and Observation: Incubate the reaction mixture at a mild temperature (e.g., 37-40°C) for several hours to a day. The formation of nanoparticles is indicated by a color change (e.g., to dark brown for AgNPs), which can be confirmed by UV-Vis spectroscopy with a characteristic Surface Plasmon Resonance (SPR) peak around 415-430 nm.
Purification and Characterization:
- Purification: Centrifuge the reaction mixture at high speed (e.g., 13,000 rpm for 15 minutes). Wash the pellet with deionized water to remove excess extract residues and lyophilize the final product.
- Characterization: Employ a suite of techniques:
- UV-Vis Spectroscopy: To confirm synthesis and monitor stability.
- Dynamic Light Scattering (DLS): To determine hydrodynamic size distribution and polydispersity index (PDI).
- Zeta Potential: To measure surface charge and colloidal stability.
- Transmission Electron Microscopy (TEM): To visualize nanoparticle size, morphology, and dispersion.
- Fourier-Transform Infrared Spectroscopy (FTIR): To identify the functional groups of phytochemicals bound to the nanoparticle surface, confirming their role in capping and stabilization.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful research in green nanoparticle synthesis requires specific reagents and instrumentation. The following table details essential items and their functions.
Table 3: Research Reagent Solutions for Green Nanoparticle Synthesis
| Reagent / Material | Function in Research | Specific Example / Note |
|---|---|---|
| Plant Material | Source of reducing and stabilizing phytochemicals (phenolics, terpenoids, proteins). | Select based on known phytochemical profile; standardize source and preparation. |
| Metal Salts | Precursor for nanoparticle formation. | Silver nitrate (AgNO₃) is most common; also chloroauric acid (HAuCl₄) for gold. |
| pH Adjusters | To optimize the synthesis reaction for size and yield. | NaOH or HCl solutions. |
| Solvents | For extraction and purification. | Sterile deionized water is standard for green synthesis. |
| Characterization Consumables | For sample preparation and analysis. | Carbon-coated copper grids for TEM; quartz cuvettes for UV-Vis. |
| Centrifuge | For purifying and concentrating synthesized nanoparticles. | Requires high speeds (e.g., 13,000+ rpm). |
| Spectrophotometer | To confirm nanoparticle synthesis via Surface Plasmon Resonance. | UV-Vis spectrometer (300-700 nm range). |
| Microscopy | To determine precise size, shape, and morphology. | Transmission Electron Microscope (TEM) is ideal. |
| Zetasizer | To measure hydrodynamic size, PDI, and zeta potential. | Dynamic Light Scattering (DLS) instrument. |
| FTIR Spectrometer | To identify phytochemical functional groups on nanoparticle surfaces. | Confirms capping and stabilization mechanism. |
Molecular Pathways and Mechanistic Insights
The reduction and stabilization of metal nanoparticles by phytochemicals involve specific molecular interactions. Phenolic compounds and flavonoids are particularly effective reductants due to their chemical structures.
Figure 2: Molecular Reduction Pathways of Silver Ions by Flavonoids and Phenolics. Flavonoids reduce metal ions through enol-to-keto tautomerization, generating the electrons needed for reduction. Phenolic compounds act via oxidation and deprotonation of their hydroxyl groups. The resulting phytochemical oxidation products then adsorb onto the nascent nanoparticle surface, providing steric or electrostatic stabilization [26] [27].
The superior performance of green-synthesized nanoparticles often stems from this bio-capping layer. For instance, the enhanced cytotoxicity of walnut-synthesized AgNPs against MCF-7 cancer cells, compared to both the plain extract and commercially synthesized AgNPs, is attributed to a synergistic effect between the silver core and the bioactive phytochemical coating [25]. This coating not only stabilizes the nanoparticle but can also contribute directly to its biological activity.
The evidence demonstrates that phytochemicals like phenolics, terpenoids, and proteins provide a powerful, nature-derived mechanism for the reduction and stabilization of metal nanoparticles. When compared to conventional synthesis, this green approach offers a compelling combination of enhanced biocompatibility, unique bioactivity derived from the capping agents, and a more environmentally sustainable profile. The experimental data confirms that green-synthesized nanoparticles can outperform their chemically synthesized counterparts in specific biological applications, such as targeted anticancer and antimicrobial activity, while exhibiting reduced toxicity to normal cells.
Despite these advantages, challenges remain for the widespread adoption and standardization of green synthesis. Key issues include the batch-to-batch variability of plant extracts due to geographical, seasonal, and cultivation differences, and the need for a more precise understanding of reaction kinetics to better control nanoparticle size and shape [5]. Future research should focus on the standardization of plant extracts, detailed investigations into the structure-activity relationships of specific phytochemicals, and rigorous, large-scale toxicity studies to pave the way for clinical translation. Integrating green synthesis with personalized nanomedicine represents a promising frontier in biomedical research.
Methodologies in Action: Green Synthesis Protocols and Their Biomedical Breakthroughs
The synthesis of metal and metal oxide nanoparticles (NPs) has traditionally relied on physical and chemical methods, which often involve high energy consumption, toxic reagents, and environmentally hazardous byproducts [31] [32]. In contrast, plant-mediated green synthesis has emerged as a sustainable alternative that leverages the natural reducing and stabilizing capabilities of plant phytochemicals [33] [34]. This approach aligns with the principles of green chemistry by using biological resources as replacements for synthetic chemicals, operating under ambient conditions, and minimizing waste generation [8] [20]. The biochemical diversity present in various plant parts—including leaves, stems, and seeds—provides a versatile toolkit for producing nanoparticles with tailored properties for specialized applications in biomedicine, agriculture, and environmental remediation [34] [32].
This guide objectively compares plant-based protocols against conventional methods, presenting experimental data on synthesis efficiency, nanoparticle characteristics, and functional performance. By providing detailed methodologies and comparative analysis, we aim to equip researchers with the practical knowledge needed to implement these sustainable synthesis approaches in their own work, particularly within the context of drug development and biomedical research where biocompatibility is paramount [35] [34].
Comparative Analysis: Plant-Mediated vs. Conventional Synthesis
Table 1: Overall comparison between plant-mediated and conventional synthesis methods
| Parameter | Plant-Mediated Green Synthesis | Chemical Synthesis | Physical Synthesis |
|---|---|---|---|
| Reducing/Stabilizing Agents | Plant phytochemicals (polyphenols, flavonoids, terpenoids) [34] [20] | Synthetic chemicals (citrate, borohydride) [20] | High energy (laser, thermal) [20] |
| Typical Conditions | Ambient temperature/pressure, aqueous medium [34] [32] | May require elevated temperature, organic solvents [31] | High energy input (ultraviolet, laser ablation) [31] |
| Environmental Impact | Low; biodegradable reagents [34] [8] | High; toxic chemicals and byproducts [31] [32] | Moderate; high energy consumption [31] |
| Biocompatibility | High; natural capping agents [35] [34] | Variable; potential cytotoxic residues [34] | Generally good; no chemical solvents |
| Size Control | Moderate (broader distribution) [33] | Good (precise control) [33] | Fair [31] |
| Shape Control | Moderate [33] | Good [33] | Limited [31] |
| Scalability | Promising for large-scale production [34] | Well-established [33] | Cost and energy-intensive [31] |
| Cost | Low (abundant resources) [34] [31] | Moderate to high (reagent costs) [31] | High (equipment, energy) [31] |
Table 2: Experimental performance data for silver nanoparticles (AgNPs) from different synthesis routes
| Synthesis Method | Source Material | Size Range (nm) | Zeta Potential (mV) | Germination Rate (%) | Antimicrobial Efficacy | Reference |
|---|---|---|---|---|---|---|
| Green Synthesis | Azadirachta indica (Neem) leaf | 22-30 [31] | -55.2 [8] | 70% (Tomato) [31] | High (broad spectrum) [20] | |
| Green Synthesis | Azadirachta indica (Neem) leaf | 9.7 [8] | -55.2 [8] | 19% increase over chemical AgNPs (Potato) [8] | Not specified | |
| Chemical Synthesis | Sodium citrate | 20.6 [8] | -35.7 [8] | Baseline [8] | High (but potential toxicity) [34] | |
| Physical Synthesis | Laser ablation | 10-100 [31] | Not specified | Not specified | Good [31] |
Plant-Mediated Synthesis Protocols
Fundamental Mechanisms
Plant-mediated synthesis operates on the principle of using phytochemicals as natural reducing and stabilizing agents [34]. The process involves bioreduction, where secondary metabolites such as polyphenols, flavonoids, terpenoids, and alkaloids donate electrons to convert metal ions (Ag⁺, Zn²⁺, Cu²⁺, etc.) to their zero-valent metallic or oxide states [34] [32]. Subsequently, these same phytochemicals form a protective capping layer around the nascent nanoparticles, preventing aggregation and ensuring stability [33] [20]. This dual functionality eliminates the need for external stabilizing agents typically required in conventional synthesis methods [34].
The synthesis can be performed through either intracellular or extracellular approaches, with plant extract-based extracellular synthesis being predominantly preferred for its simplicity and efficiency [32]. This method involves simply mixing aqueous plant extracts with metal salt solutions under controlled conditions, facilitating rapid nanoparticle formation without the complexities of maintaining cell cultures [31] [32].
Detailed Experimental Protocols
Protocol 1: Leaf-Mediated Synthesis of Silver Nanoparticles
Source: Azadirachta indica (Neem) leaves [31] [8]
Materials and Reagents:
- Fresh neem leaves (20 g)
- Silver nitrate (AgNO₃) solution (1-10 mM)
- Distilled deionized water
- Ethanol (for extraction, optional)
Procedure:
- Plant Extract Preparation: Thoroughly wash neem leaves with distilled water to remove surface contaminants. Air-dry at room temperature. Macerate 20 g of leaves in 200 mL of distilled water. Heat the mixture at 70°C for 30 minutes to facilitate extraction. Filter the resulting extract through Whatman No. 1 filter paper to obtain a clear solution [31].
- Nanoparticle Synthesis: Mix 10 mL of neem leaf extract with 10 mL of 1 mM AgNO₃ solution in a 1:1 ratio. Incubate the reaction mixture at 70°C for 3 hours under continuous stirring. Observe color change from pale yellow to reddish-brown, indicating silver nanoparticle formation [31].
- Purification: Centrifuge the nanoparticle suspension at 12,000 rpm for 20 minutes. Discard the supernatant and resuspend the pellet in distilled water. Repeat this process twice to remove unreacted phytochemicals and silver ions [31].
- Characterization: Analyze the synthesized nanoparticles using UV-Vis spectroscopy (absorption peak at 420-440 nm), SEM (size and morphology), FTIR (identifying functional groups involved in capping), and XRD (crystallinity) [31].
Critical Parameters:
- Temperature: 70°C optimal for rapid synthesis [31]
- pH: Neutral to slightly alkaline pH favors smaller particle size [31]
- Reaction Time: 3 hours for complete reduction [31]
- Extract to Salt Ratio: 1:1 volume ratio for optimal size control [31]
Protocol 2: Seed Priming with Green-Synthesized Nanoparticles
Source: Solanum tuberosum (Potato) seeds with neem-synthesized AgNPs [8]
Materials and Reagents:
- Green-synthesized silver nanoparticles (5 mg L⁻¹ concentration)
- Potato seeds
- Distilled water
Procedure:
- Nanoparticle Synthesis: Prepare neem-mediated AgNPs as described in Protocol 1 [8].
- Seed Priming: Prepare a nanopriming solution with optimal concentration of 5 mg L⁻¹ of green-synthesized AgNPs. Immerse potato seeds in the nanopriming solution for a specified duration (typically 12-24 hours) [8].
- Germination Test: Sow primed seeds under controlled conditions, including elevated temperature (32.2°C) to simulate heat stress. Monitor germination rates and seedling growth parameters [8].
- Analysis: Evaluate germination percentage, mean germination time, radicle emergence, and biochemical markers (chlorophyll, protein content) compared to hydroprimed controls and chemically-synthesized AgNP treatments [8].
Results: Green-synthesized AgNPs increased germination by 19% compared to chemical AgNPs and by 50% over hydroprimed controls under heat stress conditions [8].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key reagents and materials for plant-mediated nanoparticle synthesis
| Reagent/Material | Function | Examples/Specifications |
|---|---|---|
| Plant Materials | Source of reducing and stabilizing agents | Leaves (Neem, Tulsi), stems, seeds; fresh or properly dried [31] [20] |
| Metal Salts | Precursor for nanoparticle formation | AgNO₃, ZnSO₄, HAuCl₄, CuSO₄; high purity (≥99%) [31] [8] |
| Extraction Solvent | Medium for phytochemical extraction | Distilled deionized water, ethanol (food grade) [31] |
| Filtration System | Clarification of plant extracts | Whatman filter paper (No. 1), syringe filters (0.22 µm) [31] |
| pH Adjusters | Optimization of synthesis conditions | NaOH, HCl; analytical grade [31] |
| Centrifuge | Purification of nanoparticles | Capable of 12,000-15,000 rpm [31] |
Advantages, Limitations, and Research Gaps
Advantages of Plant-Mediated Synthesis
The quantitative data reveals significant advantages of plant-mediated synthesis. In agricultural applications, green-synthesized silver nanoparticles demonstrated superior performance compared to their chemically-synthesized counterparts, increasing potato seed germination by 19% under heat stress conditions [8]. This enhanced efficacy is attributed to their smaller size (9.7 nm for green vs. 20.6 nm for chemical AgNPs) and improved stability (zeta potential of -55.2 mV vs. -35.7 mV), which facilitates better cellular uptake and interaction [8].
The economic and environmental benefits are equally compelling. Green synthesis eliminates the need for toxic chemical reagents, reduces energy requirements by operating at ambient temperatures, and utilizes renewable plant resources [34] [31]. The presence of natural capping agents on green-synthesized nanoparticles enhances their biocompatibility, making them particularly suitable for biomedical applications such as drug delivery, wound healing, and antimicrobial treatments [35] [34].
Limitations and Research Challenges
Despite these advantages, plant-mediated synthesis faces several challenges that require further research. Batch-to-batch variability remains a significant issue due to differences in plant phytochemical composition influenced by geographical location, seasonal variations, and cultivation practices [33] [34]. This variability complicates the reproduction of identical nanoparticle characteristics across different batches.
Compared to chemical methods, green synthesis typically offers less precise control over size and shape distributions [33]. While chemical synthesis can produce highly uniform nanoparticles with narrow size distributions, plant-mediated approaches often result in broader polydispersity [33]. The complexity of plant extracts containing numerous phytochemicals also makes it difficult to pinpoint exact reduction mechanisms and reproduce specific surface properties [34].
Plant-mediated synthesis represents a transformative approach to nanoparticle production that effectively balances performance with sustainability. The experimental data demonstrates that green-synthesized nanoparticles can surpass conventionally produced counterparts in key performance metrics, particularly in biological applications where their natural capping layers enhance biocompatibility and functionality [35] [34] [8].
Future research should focus on standardizing plant extracts through quantitative phytochemical profiling, implementing advanced process control technologies including machine learning for parameter optimization, and developing comprehensive toxicity profiles for specific application domains [33] [34] [36]. The integration of hybrid approaches that combine the precision of chemical methods with the sustainability of biological resources may offer a promising middle ground [36].
As the field progresses, plant-mediated synthesis is poised to play an increasingly vital role in sustainable nanotechnology, particularly for pharmaceutical and biomedical applications where the combination of efficacy, safety, and environmental responsibility is paramount. The protocols and data presented here provide a foundation for researchers to further develop and optimize these green synthesis approaches for their specific applications.
The growing demand for sustainable nanotechnology has positioned microbial synthesis as a formidable alternative to conventional chemical and physical methods for nanoparticle (NP) production. Conventional methods often involve hazardous chemicals, toxic substances, and high energy consumption, leading to harmful environmental byproducts [4] [37]. In contrast, microbial synthesis provides an environmentally responsible, economical, and safe way to produce nanoparticles with outstanding mechanical, chemical, biological, thermal, and physical qualities [4]. Microbes including bacteria, fungi, and algae act as sophisticated nano-factories, capable of transporting metals from their environment and converting them into elemental nanoparticle forms through intracellular or extracellular pathways [37] [38]. This biological approach aligns with the principles of green chemistry, promoting resource efficiency, energy conservation, and reduced waste production [4]. The resulting nanoparticles have remarkable applications across biomedical research, biosensors, pharmaceuticals, drug delivery, environmental remediation, and agriculture [4] [37] [39]. This review provides a comprehensive comparison of intracellular and extracellular synthesis mechanisms across different microbial systems, supported by experimental data and protocols, within the broader context of sustainable nanotechnology.
Fundamental Synthesis Mechanisms: Intracellular vs. Extracellular Pathways
Microbes synthesize nanoparticles through two primary mechanisms: intracellular and extracellular. The choice of mechanism significantly influences the nanoparticle characteristics, purification processes, and potential applications.
Intracellular Synthesis
In intracellular synthesis, metal ions from the surrounding environment are first trapped and absorbed by the microbial cells. These ions are then reduced within the cell wall, cytoplasm, or periplasmic space by enzymes acting as nucleation sites, which provide electrons for metal reduction [37] [38]. This process leads to the formation of nanoclusters that eventually diffuse from the cell wall into the solution [37]. The intracellular route often requires additional processing steps, such as cell disruption and purification, to recover the synthesized nanoparticles [22]. A classic example is Pseudomonas stutzeri AG259, which was observed to accumulate silver nanoparticles intracellularly in the periplasmic space [37].
Extracellular Synthesis
Extracellular synthesis is generally preferred for its simpler downstream processing [22]. In this method, microbes are cultured in suitable media, and the supernatant containing microbial enzymes and metabolites is separated from the biomass via centrifugation [37]. This cell-free supernatant is then allowed to react with metal salt solutions. The bioreduction of metal ions occurs outside the cells, resulting in the formation of nanoparticles in the solution [37] [22]. Fungi are particularly well-suited for extracellular synthesis, often secreting larger quantities of reducing enzymes and metabolites compared to bacteria, which facilitates easier harvesting of nanoparticles without complex purification [40] [22].
The following diagram illustrates the key stages of both intracellular and extracellular microbial synthesis pathways.
Comparative Analysis of Microbial Systems
The efficacy of nanoparticle synthesis varies significantly across different microorganisms. Each class—bacteria, fungi, and algae—offers distinct advantages and limitations in terms of synthesis speed, nanoparticle stability, scalability, and potential for genetic manipulation. The following table provides a detailed comparison of these microbial systems based on critical performance parameters.
Table 1: Performance Comparison of Microbial Systems for Nanoparticle Synthesis
| Parameter | Bacteria | Fungi | Algae |
|---|---|---|---|
| Preferred Synthesis Route | Both intra- and extracellular [37] | Predominantly extracellular [22] | Both intra- and extracellular [38] |
| Typical Synthesis Duration | Faster (e.g., 24-48 hours) [40] [22] | Slower (e.g., 72-120 hours) [22] | Variable (24-96 hours) [38] |
| NP Stability | Moderate | Typically higher stability [40] | Moderate to High |
| Scalability Potential | High (simple culture requirements) [40] | High (well-suited for mass production) [22] | Moderate (influenced by growth conditions) |
| Genetic Manipulation Potential | Greater potential for precise control [40] | More complex | Emerging |
| Key Advantages | Rapid growth, simple culture, genetic tractability [40] | High metabolite production, excellent NP stability [40] [22] | CO₂ sequestration, cost-effective biomass [38] |
| Major Limitations | Potential pathogenicity, lower NP stability compared to fungi | Slower growth and synthesis kinetics [22] | Seasonal variation, polydispersity potential [38] |
Bacteria-Mediated Synthesis
Bacteria are highly efficient nanofactories due to their rapid growth, simple culture requirements, and well-understood genetics. Various species of bacteria, including Escherichia coli, Lactobacillus sp., Bacillus cereus, and Pseudomonas sp., have been extensively explored for the synthesis of metal and metal oxide nanoparticles such as silver (Ag), gold (Au), and zinc oxide (ZnO) [37]. Their inherent ability to withstand abiotic stresses and reduce heavy metals makes them particularly suitable for nanoparticle synthesis [37]. The genetic machinery of bacteria can be altered through genetic engineering to fabricate nanoparticles with specific physical, chemical, and biological properties, enhancing their versatility [37].
Fungi-Mediated Synthesis (Mycosynthesis)
Fungi, through mycosynthesis, are a powerful platform for extracellular nanoparticle production. They secrete large quantities of enzymes (e.g., reductases) and metabolites that act as reducing and capping agents, leading to the formation of highly stable nanoparticles [40] [22]. This extracellular mechanism simplifies downstream processing, making it advantageous for scalable production [22]. Fungal systems often produce nanoparticles with enhanced stability compared to bacterial counterparts, which is a critical factor for storage and application [40]. Furthermore, fungi are generally easier to cultivate in laboratory settings than some fastidious bacteria and possess high growth rates, supporting mass production [22].
Algae-Mediated Synthesis
Algae, both macro- and microalgae, represent a sustainable and eco-friendly resource for nanoparticle synthesis. They are abundant in nature, require simple cultivation conditions, and can accumulate and reduce metal ions from their environment through intracellular and extracellular pathways [38]. Their metabolic processes, driven by photosynthesis, can be harnessed for the biogenic production of nanoparticles. However, similar to plant-based synthesis, seasonal variations can potentially lead to differences in the phytochemical profile of extracts, which may affect the yield and polydispersity of the resulting nanoparticles [38].
Experimental Protocols and Methodologies
Reproducible synthesis of nanoparticles via microbial routes requires standardized protocols. Below are detailed methodologies for intracellular and extracellular synthesis, applicable to bacteria and fungi.
General Protocol for Extracellular Synthesis
This protocol is commonly used for fungi and bacteria that secrete reducing agents into the culture medium [37] [22].
- Microbial Cultivation: Inoculate the desired microbial strain (e.g., Fusarium oxysporum for fungi or Pseudomonas aeruginosa for bacteria) into an appropriate liquid growth medium (e.g., Potato Dextrose Broth for fungi, Luria-Bertani Broth for bacteria). Incubate under optimal conditions (e.g., 25-30°C for fungi, 37°C for bacteria) with agitation for 24-72 hours to achieve robust growth [37].
- Biomass Separation: Culture broth is centrifuged at high speed (e.g., 8,000-10,000 rpm for 15-20 minutes) to separate the microbial biomass from the cell-free supernatant (filtrate) containing extracellular enzymes and metabolites [37].
- Bioreduction Reaction: The supernatant is reacted with an aqueous solution of the target metal salt (e.g., 1-10 mM AgNO₃ for silver nanoparticles) in a separate vessel. The mixture is incubated in the dark under constant agitation. The formation of nanoparticles is often indicated by a visual color change (e.g., pale yellow to brown for AgNPs) [37] [22].
- Nanoparticle Harvesting: The synthesized nanoparticles are recovered from the solution via high-speed centrifugation (e.g., 12,000-15,000 rpm for 30-60 minutes). The pellet is washed multiple times with distilled water or ethanol to remove any residual biomolecules and then re-dispersed in a suitable solvent or dried into a powder for further use [22].
General Protocol for Intracellular Synthesis
This method is used when nanoparticles are formed within the microbial cells [37].
- Biomass Preparation and Washing: Microbial cultures are grown as described above. The biomass is harvested by centrifugation and thoroughly washed with sterile distilled water to remove any components of the growth medium [37].
- Metal Ion Exposure: The clean microbial biomass is suspended in sterile distilled water or a mild buffer and then exposed to an aqueous solution of the metal salt. The concentration of the metal ion solution is a critical parameter that requires optimization [37] [39].
- Incubation and Intracellular Reduction: The reaction mixture is incubated under optimal conditions for a specified period, which can range from 24 to 120 hours. During this time, metal ions are transported into the cells and reduced by intracellular enzymes [37].
- Cell Lysis and Nanoparticle Purification: After incubation, the biomass is subjected to cell disruption techniques such as sonication or repeated freeze-thaw cycles. The released nanoparticles are then separated from cellular debris via centrifugation, filtration, or density gradient centrifugation [37] [22].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful microbial synthesis of nanoparticles relies on a specific set of reagents, materials, and instrumentation. The following table lists the key components of a research toolkit for this field.
Table 2: Essential Research Reagent Solutions and Materials
| Category | Item | Function/Application | Experimental Example |
|---|---|---|---|
| Microbial Strains | Bacillus spp., Pseudomonas spp. | Model bacteria for AgNP, AuNP synthesis [37] [38] | Bacillus brevis for antimicrobial AgNPs [38] |
| Fusarium oxysporum, Aspergillus niger | Model fungi for extracellular synthesis [22] | F. oxysporum for stable, extracellular AgNPs [22] | |
| Marine thraustochytrids, Shewanella spp. | Algae/bacteria for metal reduction and synthesis [41] [38] | Shewanella for metal oxide NPs via respiration [38] | |
| Chemical Reagents | Metal Salts (AgNO₃, HAuCl₄, ZnSO₄) | Precursor for nanoparticle synthesis [37] [39] | 1mM AgNO₃ solution for AgNP synthesis [37] |
| Culture Media (LB, PDB, Nutrient Broth) | Supports microbial growth and metabolite production [37] | Potato Dextrose Broth for fungal culture [37] | |
| Biocompatible Capping Agents (Starch, Sodium Alginate) | Stabilizes NPs, prevents aggregation [39] | Starch for stabilizing bimetallic Fe/Pd NPs [39] | |
| Laboratory Equipment | Centrifuge | Separates biomass from supernatant and harvests NPs [37] | Centrifugation at 10,000 rpm for 15 min [37] |
| UV-Vis Spectrophotometer | Confirms NP synthesis via Surface Plasmon Resonance [37] | Detection of AgNP peak at ~420 nm [37] | |
| Electron Microscopy (SEM, TEM) | Determines NP size, shape, and morphology [37] [39] | TEM analysis showing spherical NPs of 20 nm [39] | |
| X-ray Diffraction (XRD) | Analyzes crystallinity and phase of NPs [37] | XRD confirming crystalline nature of Fe₃O₄ NPs [39] |
Microbial synthesis represents a paradigm shift in nanotechnology, offering a sustainable and eco-friendly alternative to conventional methods. As this comparative guide illustrates, bacteria, fungi, and algae each provide unique advantages as nanofactories, operating through distinct intracellular and extracellular pathways. The choice of microbial system and synthesis route directly impacts the characteristics, yield, and application potential of the resulting nanoparticles. While challenges in reproducibility, scalability, and precise control over nanoparticle properties remain, the integration of synthetic biology, data-driven design, and advanced modeling holds immense promise for the future. By leveraging the intricate biochemical machinery of microorganisms, researchers can continue to develop next-generation nanoparticles that align with the principles of green chemistry, ultimately driving innovation in biomedicine, environmental remediation, and beyond.
The synthesis of functional nanomaterials is a cornerstone of advancements in energy storage, catalysis, and biomedicine. However, conventional synthesis methods often pose significant environmental risks due to their high energy demands, lengthy reaction times, and use of hazardous chemicals [3]. In response, the principles of green chemistry have catalyzed a shift toward more sustainable production techniques [42]. This guide objectively compares two prominent strategies—Microwave-Assisted Synthesis and the Sol-Gel Method—focusing on their efficiency, environmental footprint, and performance in producing advanced materials.
Microwave-Assisted Synthesis utilizes microwave radiation to heat reaction mixtures volumetrically and almost instantaneously, leading to dramatically faster reaction times, higher product yields, and unique material morphologies [43] [44]. In contrast, the Sol-Gel Method is a versatile chemical technique for fabricating metal oxides through the transition of a solution ("sol") into a solid ("gel") network, allowing for exceptional control over composition and structure at low temperatures [3] [45]. When these methods are combined into Microwave-Assisted Sol-Gel synthesis, they synergize to offer superior energy efficiency and material quality [46] [45].
Fundamental Principles and Comparative Mechanism
Understanding the distinct heating mechanisms of these methods is key to appreciating their efficiency profiles. The following diagram illustrates the fundamental workflow and energy input in conventional versus microwave-assisted heating.
The core difference lies in energy transfer. Conventional heating relies on conduction and convection from an external source, creating a temperature gradient from the hot vessel surface to the cooler core. This is inherently slow and can lead to inhomogeneous reaction conditions [45]. In contrast, microwave heating delivers energy directly to the molecules within the entire reaction volume via dipole rotation and ionic conduction. This eliminates the thermal gradient, enabling rapid and uniform heating, which is a primary source of its efficiency [47] [44].
The Sol-Gel process, whether conventional or microwave-assisted, follows a specific chemical pathway for metal oxide formation, as shown below.
The Sol-Gel pathway begins with molecular precursors undergoing hydrolysis and condensation reactions to form a metal-oxygen-metal (M-O-M) network. This network grows into a wet gel, which is then aged, dried, and thermally treated to yield the final metal oxide material [3] [45]. The low-temperature nature of the initial steps is intrinsically less energy-intensive than solid-state synthesis.
Comparative Performance Data
Quantitative data from recent research demonstrates the clear advantages of microwave-assisted techniques in head-to-head comparisons with conventional methods.
Table 1: Performance Comparison of Synthesis Methods for Energy Materials
| Material Synthesized | Synthesis Method | Key Synthesis Conditions | Reaction Time | Performance Outcome | Source |
|---|---|---|---|---|---|
| LiNi₀.₅Mn₁.₅O₄ (LNMO) | Microwave Sol-Gel | ~20 min for thermal treatment | 20 minutes | Capacity retention of 84.13% after 1500 cycles; superior rate performance. | [46] |
| Conventional Sol-Gel | Several hours for thermal treatment | Hours | Lower cycling stability and rate capability compared to MW-SG. | [46] | |
| LiFePO₄ | Microwave-Hydrothermal (MH) | -- | ~30 min | Phase-pure product achievable, but poorer electrochemical performance. | [43] |
| Microwave-Solvothermal (MS) | Glycol solvent | ~30 min | Specific discharge capacity: 162 mAh g⁻¹ at 0.1C. | [43] | |
| Mn-doped ZnO | Microwave Sol-Gel | 140-850 W microwave power | Hours to Minutes | Narrow particle size distribution, high purity, uniform morphology. | [45] |
| Conventional Sol-Gel | Conventional furnace heating | Days to Hours | Broader particle size distribution, longer processing times. | [45] | |
| TaC Nanorods | Microwave Carbothermal | 1300 °C | 20 minutes | High-quality nanorods with enhanced EM wave absorption. | [48] |
| Conventional Carbothermal | High temperature | Several hours | Typically lower quality and poor efficiency. | [48] |
Table 2: Qualitative Advantages and Limitations of Each Method
| Aspect | Microwave-Assisted Synthesis | Conventional Sol-Gel |
|---|---|---|
| Heating Mechanism | Volumetric, direct dipole interaction. | Superficial, via conduction/convection. |
| Reaction Speed | Extremely fast (minutes). | Slow (hours to days). |
| Energy Efficiency | Very High. | Low to Moderate. |
| Product Homogeneity | Excellent; uniform nucleation. | Can be inhomogeneous; gradient-driven. |
| Morphology Control | High; can yield unique nanostructures. | Good, but requires careful process control. |
| Scalability | Challenging; requires reactor design. | Well-established for industrial scale. |
| Process Control | Rapid and precise. | Slower response. |
Detailed Experimental Protocols
To ensure reproducibility, detailed methodologies from key studies are outlined below.
Protocol: Microwave-Assisted Sol-Gel Synthesis of LiNi₀.₅Mn₁.₅O₄ (LNMO)
This protocol, derived from Su et al. [46], highlights the acceleration of phase evolution.
- Precursor Solution: Lithium acetate (LiCH₃COO), nickel acetate tetrahydrate (Ni(CH₃COO)₂·4H₂O), and manganese acetate tetrahydrate (Mn(CH₃COO)₂·4H₂O) were dissolved in deionized water in a molar ratio of Li:Ni:Mn = 1.05:0.5:1.5.
- Chelating Agent: A separate citric acid (C₆H₈O₇) solution was prepared in deionized water, with a mole ratio of total transition metals (Ni+Mn) to citric acid of 1:1.
- Gel Formation: The citric acid solution was added dropwise to the metal precursor solution under stirring. The mixture was then evaporated to form a viscous gel.
- Microwave Treatment: The gel was subjected to microwave irradiation for approximately 20 minutes for the thermal treatment and crystallization step, which is dramatically faster than conventional methods.
- Final Product: The resulting powder was calcined to obtain the final LNMO cathode material with a disordered spinel structure, fewer impurities, and lower Mn³⁺ content.
Protocol: Comparative Synthesis of Mn-doped ZnO Nanopowders
This protocol, based on the work by various researchers [45], compares conventional and microwave-assisted sol-gel routes.
- Common Precursors: Both methods used a solution of zinc acetate dihydrate and manganese acetate in absolute ethanol, with triethanolamine.
- Conventional Sol-Gel (SG) Path: The solution was gelled, dried, and then thermally treated using a conventional furnace. The entire process, including gelation and calcination, could take days.
- Microwave-Assisted (MW) Path: The precursor solution was exposed to microwave irradiation (power ranging from 140 W to 850 W) to induce gelation and subsequent crystallization. This method reduced the reaction time to hours or even minutes.
- Outcome: The MW-synthesized powders exhibited more uniform properties and enhanced photocatalytic performance for mineralizing oxalic acid under solar light compared to the SG samples.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Microwave-Assisted and Sol-Gel Synthesis
| Reagent Category | Specific Examples | Function in Synthesis |
|---|---|---|
| Metal Precursors | Acetates (e.g., Zn(CH₃COO)₂, FeSO₄·7H₂O), Alkoxides (e.g., Ti(OiPr)₄), Chlorides (e.g., FeCl₂), Ta₂O₅ | Source of metal cations for the formation of the target metal oxide or ceramic framework. |
| Solvents | Water (Ultrapure), Ethylene Glycol, Absolute Ethanol | Medium for dissolving precursors and facilitating reactions. Choice affects reaction kinetics and mechanism (e.g., glycol in solvothermal). |
| Reducing/ Capping Agents | Ascorbic Acid, Citric Acid, Plant Extracts (e.g., Neem, Turmeric), Sucrose | Reduce metal ions to lower oxidation states or elemental form. Also act as surfactants to control nanoparticle growth and prevent agglomeration. |
| Chelating Agents/ Gelling Agents | Citric Acid, Triethanolamine (TEA) | Complex with metal ions to control hydrolysis rates, promote homogeneity, and aid in gel formation. |
| Structure-Directing Agents | NaCl (Molten Salt), Glyoxylic Acid, Sodium Carbonate | Control morphology (e.g., creating nanorods), induce gelation in specific systems (e.g., iron aerogels), or act as a catalyst. |
| Dopant Sources | Manganese Acetate, Nickel Acetate | Introduce specific foreign elements into a host material to tailor its electrical, optical, or magnetic properties. |
The comparative analysis presented in this guide unequivocally demonstrates that microwave-assisted synthesis techniques offer significant advantages in energy efficiency, reaction speed, and product quality over conventional methods. The integration of microwave heating with the versatile sol-gel process creates a powerful synergy, enabling the rapid and sustainable production of high-performance nanomaterials for applications ranging from high-power batteries to photocatalysts. While scalability remains a focus for ongoing research, the experimental data and protocols provided underscore the transformative potential of microwave-assisted and sol-gel methods in reducing the environmental footprint of nanomaterial synthesis, aligning perfectly with the core principles of green chemistry.
The synthesis of nanoparticles for biomedical applications primarily follows two divergent paths: conventional (chemical/physical) methods and green synthesis. Conventional methods often rely on toxic chemical reducing agents and high energy consumption, leading to concerns about biocompatibility and environmental impact [49]. In contrast, green synthesis utilizes biological materials like plant extracts, fungi, or bacteria as reducing and stabilizing agents, offering an eco-friendly, cost-effective, and biocompatible alternative [4]. This guide provides an objective comparison of nanoparticles produced via these routes, focusing on their performance in targeted drug delivery, wound healing, and as antimicrobial agents, with supporting experimental data for researchers and drug development professionals.
Performance Comparison: Green vs. Conventional Nanoparticles
The following tables summarize key experimental findings comparing the performance and properties of green-synthesized and conventional nanoparticles across various biomedical applications.
Table 1: Comparative Analysis of Synthesis Methods and Biocompatibility
| Comparison Parameter | Green Synthesis | Conventional Synthesis |
|---|---|---|
| Reducing/Stabilizing Agents | Plant extracts (e.g., Ehretia rigida, Hibiscus rosa sinensis), fungi, bacteria [49] [50] | Chemical agents (e.g., citrate, borohydride) [49] |
| Environmental Impact | Eco-friendly, sustainable, uses renewable resources [4] | Often involves hazardous chemicals and toxic byproducts [49] [4] |
| Energy Consumption | Lower (often occurs at ambient temperature/pressure) [4] | High for physical methods (e.g., evaporation-condensation) [49] |
| Typical Cytotoxicity | Generally lower, enhanced biocompatibility [51] [50] | Often higher due to chemical reagent contamination [49] |
| Scalability Challenge | Geographic/seasonal variation in plant resources [49] | High cost and complex purification processes [49] |
Table 2: Experimental Efficacy in Antimicrobial and Wound Healing Applications
| Application & Nanoparticle Type | Experimental Model | Key Performance Results | Comparative Efficacy |
|---|---|---|---|
| Antimicrobial: Green AgNPs from Hibiscus rosa sinensis (HRS) [52] | In vitro against Gram-positive and Gram-negative bacteria | - Strong antibacterial activity, especially against Gram-positive bacteria- Effectively disrupted bacterial biofilms- Size: 10-50 nm | Green AgNPs showed broad-spectrum antibacterial and antibiofilm activity, with minimal toxicity to normal cells. |
| Antimicrobial: Conventional AgNPs [49] | In vitro against multidrug-resistant (MDR) bacteria | - Broad-spectrum activity via membrane disruption, ROS generation- Potential cytotoxicity and host cell damage | High antimicrobial efficacy but with greater concerns regarding cytotoxicity and bacterial resistance. |
| Wound Healing: Green AgNPs from Ehretia rigida (Er) [50] | In vitro scratch assay on skin fibroblasts (KMST-6) and keratinocytes (HaCaT) | - Promoted cell migration and wound closure- Negligible cytotoxicity at concentrations < 25 µg/mL- IC50 for protein denaturation: 532.9 µg/mL | Green AgNPs significantly enhanced wound closure compared to plant extract alone, with high biocompatibility. |
| Wound Healing: Green AgNPs from Glycyrrhiza glabra (Licorice) [53] | In vivo excision and incision rat model | - Promoted complete wound contraction and healing in 21 days- Histopathology showed complete epithelialization, regenerated hair follicles, and higher collagen | The green synthesis successfully capped therapeutic plant biomolecules on AgNPs, enhancing wound healing. |
Experimental Protocols for Key Applications
Protocol: Green Synthesis of Silver Nanoparticles (AgNPs) using Plant Extract
This is a generalized protocol based on the method used for synthesizing Ehretia rigida and Hibiscus rosa sinensis AgNPs [52] [50].
- Plant Extract Preparation: Wash, dry, and grind plant leaves. Prepare an aqueous extract (e.g., 6.25 mg/mL) in deionized water. The solution may be adjusted to an alkaline pH (e.g., 11) to enhance reduction efficiency.
- Silver Ion Reduction: Mix the plant extract with a silver nitrate solution (e.g., 2 mM) at a defined ratio (e.g., 1:10 plant extract to AgNO3 solution).
- Reaction and Synthesis: Stir the mixture at a moderate speed (e.g., 750 rpm) at an elevated temperature (e.g., 50°C) for several hours (e.g., 4 hours). A color change (to brownish) indicates nanoparticle formation.
- Purification: Centrifuge the resulting nanoparticle suspension at high speed (e.g., 15,000 rpm) to pellet the AgNPs, then re-disperse them in sterile deionized water. Repeat this process 2-3 times.
- Characterization: Characterize the synthesized nanoparticles using:
- UV-Vis Spectrophotometry: Confirm synthesis by detecting the Surface Plasmon Resonance (SPR) peak (~428 nm for HRS-AgNPs [52]).
- Dynamic Light Scattering (DLS): Determine hydrodynamic size distribution and polydispersity index.
- Transmission Electron Microscopy (TEM): Analyze the core size, shape, and morphology.
- Fourier-Transform Infrared (FTIR) Spectroscopy: Identify functional groups from phytochemicals (e.g., flavonoids, phenols) responsible for capping and stabilization.
The workflow below visualizes this synthesis and characterization process:
Protocol: In Vitro Wound Healing Scratch Assay
This protocol is used to evaluate the potential of nanoparticles, like Ehretia rigida AgNPs, to promote cell migration and wound closure [50].
- Cell Seeding: Seed appropriate cell lines (e.g., human keratinocytes HaCaT or skin fibroblasts KMST-6) in a multi-well plate until they form a 90-100% confluent monolayer.
- Scratch Creation: Use a sterile pipette tip (e.g., 200 µL) to create a uniform scratch ("wound") through the center of each cell monolayer.
- Washing: Gently wash the wells with phosphate-buffered saline (PBS) to remove detached cells and debris.
- Treatment Application: Treat the cells with different concentrations of the test material (e.g., green AgNPs, plant extract alone, control media). It is critical to use non-cytotoxic concentrations, determined via a prior cytotoxicity assay (e.g., WST-1 assay).
- Imaging and Analysis: Capture images of the scratch at time zero (immediately after creating it) and at regular intervals thereafter (e.g., 6, 12, 24, 48 hours) using an inverted microscope. Use image analysis software to measure the remaining scratch area at each time point.
- Data Calculation: Calculate the percentage of wound closure using the formula:
% Wound Closure = [(Area at T0 - Area at Tx) / Area at T0] * 100, where T0 is time zero and Tx is a given time point.
Protocol: Assessing Anti-inflammatory Activity via Protein Denaturation Inhibition
This method, used in studies on Ehretia rigida AgNPs, evaluates a compound's ability to inhibit the denaturation of proteins, a mechanism implicated in inflammation [50].
- Reaction Preparation: Prepare a 1% w/v solution of Bovine Serum Albumin (BSA) in a suitable buffer (e.g., Tris-HCl, pH 6.5).
- Sample Addition: Incubate the BSA solution with varying concentrations of the test sample (e.g., green AgNPs, plant extract, standard drug like diclofenac sodium) and a control (buffer only) at 37°C for 30 minutes.
- Thermal Denaturation: Heat the reaction mixtures at a high temperature (e.g., 70°C) for 20 minutes to induce denaturation.
- Measurement: After cooling to room temperature, measure the turbidity of the solution at 660 nm. Turbidity increases with protein denaturation.
- Calculation: Calculate the percentage inhibition of protein denaturation:
% Inhibition = [(Absorbance of Control - Absorbance of Sample) / Absorbance of Control] * 100. Determine the IC50 value (concentration that inhibits 50% of denaturation) for quantitative comparison.
Signaling Pathways and Mechanistic Insights
Antimicrobial Mechanisms of Silver Nanoparticles (AgNPs)
Green and conventional AgNPs share broad-spectrum antimicrobial mechanisms, but the bioactive capping from plant extracts may enhance the efficacy and biocompatibility of green AgNPs [52] [49]. The diagram below illustrates their multi-target mechanism of action against bacterial cells.
The primary antimicrobial mechanisms include [52] [49]:
- Membrane Disruption: AgNPs attach to the bacterial cell wall and membrane, causing structural damage, pit formation, and increased permeability, leading to leakage of cellular content.
- Reactive Oxygen Species (ROS) Generation: AgNPs catalyze the production of ROS (e.g., superoxide radicals, hydrogen peroxide), inducing oxidative stress that damages lipids, proteins, and DNA.
- Interaction with Cellular Components: AgNPs and released silver ions (Ag+) can interact with sulfur-containing proteins and phosphorus-containing DNA, inhibiting enzymatic activity, respiration, and cell division.
Nanoparticle-Mediated Wound Healing Mechanisms
Nanoparticles, particularly green AgNPs, promote wound healing through a combination of antimicrobial, anti-inflammatory, and pro-proliferative effects that act across the different phases of the healing process [53] [51] [50].
The diagram below maps these multifaceted contributions to the standard phases of wound healing.
Key mechanistic contributions include:
- During Inflammation: Green AgNPs exert potent antimicrobial effects, preventing biofilm formation and infection. Their anti-inflammatory properties, such as inhibiting protein denaturation, help control excessive inflammation [50].
- During Proliferation: Studies on green AgNPs from Glycyrrhiza glabra and Ehretia rigida show they directly promote fibroblast and keratinocyte migration and proliferation, accelerating wound contraction and re-epithelialization. They also enhance the deposition of collagen, a critical structural protein [53] [50].
- During Remodeling: By providing a conducive environment for healing and facilitating matrix reorganization, nanoparticles contribute to the formation of stronger and more organized tissue [51].
The Scientist's Toolkit: Essential Research Reagents and Materials
This table lists key reagents and their functions for conducting experiments in green nanoparticle synthesis and evaluation, based on the cited protocols.
Table 3: Essential Research Reagents and Materials
| Reagent/Material | Function/Application | Example from Research |
|---|---|---|
| Plant Extract | Serves as a reducing and capping agent in green synthesis, providing phytochemicals like flavonoids and phenols. | Ehretia rigida leaf extract [50], Hibiscus rosa sinensis flower extract [52] |
| Silver Nitrate (AgNO3) | Precursor salt providing Ag+ ions for the synthesis of silver nanoparticles. | Used in most cited AgNP synthesis protocols [52] [50] |
| Cell Lines for Cytotoxicity/Wound Healing | In vitro models to assess biocompatibility and therapeutic efficacy. | Human keratinocytes (HaCaT), skin fibroblasts (KMST-6) [50] |
| Cell Viability Assay Kits | Quantitative measurement of cell health and proliferation after treatment with nanoparticles. | WST-1 assay [50], MTT assay [52] |
| Bovine Serum Albumin (BSA) | Substrate for evaluating anti-inflammatory activity via the protein denaturation inhibition assay. | Used to test anti-inflammatory potential of Er-AgNPs [50] |
| Bacterial Strains | Models for evaluating antimicrobial and antibiofilm efficacy. | S. aureus, E. faecalis, E. coli, P. aeruginosa [52] |
| Dynamic Light Scattering (DLS) & Zeta Potential Analyzer | Instrumentation for characterizing nanoparticle hydrodynamic size, distribution, and surface charge stability. | Used for characterizing Er-AgNPs and HRS-AgNPs [52] [50] |
| Transmission Electron Microscope (TEM) | High-resolution imaging to determine the core size, shape, and morphology of nanoparticles. | Used for characterizing Er-AgNPs and HRS-AgNPs [52] [50] |
The experimental data and comparisons presented indicate that green-synthesized nanoparticles, particularly metallic ones like AgNPs, are not merely an eco-friendly alternative but often outperform their conventional counterparts in biomedical applications due to their enhanced biocompatibility and the synergistic therapeutic action of the metallic core and bioactive phytochemical capping. They demonstrate significant promise as effective antimicrobial agents against drug-resistant strains, promoters of wound healing, and potential vehicles for targeted drug delivery [52] [50] [54]. However, challenges in standardization, scalability of synthesis, and the need for more comprehensive in vivo toxicity profiles remain critical hurdles to clinical translation [55] [49]. Future research should focus on optimizing synthesis protocols for reproducibility, exploring a wider range of plant resources, and conducting detailed preclinical studies to fully harness the potential of green nanotechnology in modern medicine.
The increasing prevalence of emerging contaminants in wastewater, including pharmaceuticals, heavy metals, and organic dyes, poses significant environmental and health challenges worldwide [56]. Traditional water treatment methods often fail to effectively remove these pollutants due to critical limitations in addressing modern contaminants [56]. In this context, nanotechnology has emerged as a promising solution, with nanoparticles demonstrating unique properties that enhance their ability to adsorb, degrade, or transform pollutants [56]. The synthesis of these nanoparticles itself has become a critical focus, with green synthesis methods now offering an environmentally responsible, economical, and safe alternative to conventional physical and chemical approaches [4]. This guide provides a comprehensive comparison of green-synthesized metal nanoparticles against conventionally synthesized alternatives, focusing on their performance in pollutant degradation and heavy metal reduction, with supporting experimental data and methodologies to aid researcher selection and application.
Performance Comparison: Green vs Conventional Nanoparticles
The following tables summarize experimental data comparing the performance of green-synthesized and conventional nanoparticles in environmental remediation applications, based on recent research findings.
Table 1: Performance Comparison in Pollutant Degradation
| Nanoparticle Type | Synthesis Method | Target Pollutant | Degradation Efficiency | Time Required | Key Experimental Conditions |
|---|---|---|---|---|---|
| Iron NPs [57] | Green (Eucalyptus extract) | Acetaminophen | 95% | 30 min | Catalytic ozonation, pH 7, 25°C |
| Iron NPs [57] | Green (Mulberry extract) | Sulfamethoxazole | 90% | 30 min | Catalytic ozonation, pH 7, 25°C |
| Iron NPs [57] | Conventional (Ammonia) | Acetaminophen | 85% | 30 min | Catalytic ozonation, pH 7, 25°C |
| Silver NPs [56] | Green (Ficus carica leaf) | Organic pollutants | Significant degradation | Varies | Photocatalytic, antibacterial activity |
| TiO₂ NPs [56] | Green (Annona muricata L. extract) | Organic pollutants | Promising efficiency | Varies | Photocatalytic |
| Silver NPs [58] | Bacterial (G. stearothermophilus) | Congo Red dye | 100% | 20 min | Catalytic reduction |
| Silver NPs [58] | Bacterial (G. stearothermophilus) | 4-nitrophenol | 100% | 35 min | Catalytic reduction |
Table 2: Performance Comparison in Heavy Metal Removal
| Nanoparticle Type | Synthesis Method | Target Heavy Metal | Removal Efficiency | Adsorption Capacity (mg/g) | Key Experimental Conditions |
|---|---|---|---|---|---|
| Iron Oxide NPs [59] | Mycosynthesis (A. niger) | Copper (Cu²⁺) | 92.47% | Not specified | Synthetic wastewater, optimized conditions |
| Iron Oxide NPs [59] | Mycosynthesis (A. niger) | Manganese (Mn²⁺) | 84.76% | Not specified | Synthetic wastewater, optimized conditions |
| Iron Oxide NPs [59] | Mycosynthesis (A. niger) | Chromium (Cr³⁺) | 80.79% | Not specified | Synthetic wastewater, optimized conditions |
| Iron Oxide NPs [59] | Mycosynthesis (A. niger) | Zinc (Zn²⁺) | 70.28% | Not specified | Synthetic wastewater, optimized conditions |
| Iron NPs [60] | Green (Leaves extracts) | Chromium (Cr(VI)) | Not specified | 34.0 | Adsorption |
| nZVI [60] | Conventional (Borohydride) | Chromium (Cr(III)) | Not specified | 453.5 | Adsorption |
| nZVI [60] | Conventional (Borohydride) | Chromium (Cr(VI)) | Not specified | 372.0 | Adsorption |
| ZnO NPs [60] | Green (Using casein) | Cadmium (Cd(II)) | Not specified | 156.74 | Adsorption |
| ZnO NPs [60] | Green (Using casein) | Lead (Pd(II)) | Not specified | 194.93 | Adsorption |
Table 3: Antibacterial Performance of Silver Nanoparticles
| Synthesis Method | Source | Size Range (nm) | Antibacterial Efficacy | Key Findings |
|---|---|---|---|---|
| Bacterial [58] | G. stearothermophilus GF16 | 16-17 | Complete inhibition at 100 μg/mL | Broad-spectrum activity against S. aureus, P. aeruginosa, S. Typhimurium |
| Plant-based [20] | Ocimum sanctum, Curcuma longa | 10-50 | Enhanced antibacterial efficacy | Effective against multidrug-resistant pathogens |
| Chemical [60] | Loaded on activated carbon | 12-16 | Complete inhibition of bioaerosols | Spherical morphology |
Key Advantages of Green Synthesis Approaches
Green synthesis methods demonstrate significant advantages over conventional approaches across multiple parameters that are critical for sustainable environmental applications.
Environmental Sustainability and Safety
Green synthesis eliminates the need for hazardous chemicals commonly used in conventional methods, utilizing biological materials instead [4]. This approach produces ecologically friendly substances and byproducts, utilizes minimum energy, and promotes resource efficiency [4]. The process aligns with green chemistry principles, reducing toxic waste accumulation and environmental impact [5].
Enhanced Performance Characteristics
Experimental evidence indicates that green-synthesized nanoparticles often exhibit superior performance in environmental applications. The biological capping agents present on green-synthesized nanoparticles provide natural functionalization that can enhance reactivity and selectivity toward specific pollutants [58]. Studies have demonstrated improved catalytic activity, adsorption capacity, and pollutant degradation efficiency compared to conventionally synthesized counterparts [57].
Economic Viability
Green synthesis methods are generally more cost-effective due to reduced energy requirements and the elimination of expensive chemical reagents [4]. The use of readily available biological materials such as plant extracts or microorganisms significantly lowers production costs while maintaining high product yield and quality [5].
Detailed Experimental Protocols
Materials and Methods:
- Fungal Strain: Aspergillus niger AUMC 16028, isolated from industrial wastewater effluents
- Culture Medium: Potato dextrose agar (PDA) for isolation and maintenance
- Optimization: Box-Behnken design (BBD) and Plackett-Burman design (PBD) for parameter optimization
- Characterization: FT-IR, XRD, EDAX, SEM, TEM
Experimental Procedure:
- Isolate fungal strains from metal-polluted wastewater samples on PDA plates
- Identify the most effective fungal strain through molecular analysis
- Prepare cell-free extract from the selected fungal strain
- Optimize synthesis conditions (8 g/L yeast extract, 40°C reaction temperature, 6-day culture period)
- Characterize the synthesized iron oxide nanoparticles using specified techniques
- Evaluate heavy metal removal efficiency using synthetic and industrial wastewater
Performance Validation: The myco-synthesized iron oxide nanoparticles demonstrated high removal efficiencies for multiple heavy metals: copper (92.47%), iron (72.77%), manganese (84.76%), zinc (70.28%), and chromium (80.79%) in synthetic wastewater [59].
Materials and Methods:
- Plant Sources: Eucalyptus and mulberry leaves
- Chemical Precursors: Ferric chloride solution (Fe²⁺ and Fe³⁺ in 2:1 ratio)
- Control: Conventionally synthesized nanoparticles using ammonia
- Target Contaminants: Malachite green, methylene blue, sulfamethoxazole, atrazine, acetaminophen
Experimental Procedure:
- Prepare plant extracts by washing and processing leaves
- Mix plant extract with iron precursor solution
- Adjust pH and maintain temperature for nanoparticle formation
- Characterize nanoparticles using XRD, SEM, and other techniques
- Evaluate catalytic performance in ozonation processes
- Compare efficiency with conventionally synthesized nanoparticles
Performance Validation: Green-synthesized iron nanoparticles demonstrated superior catalytic activity compared to conventional counterparts, with Eucalyptus-synthesized nanoparticles achieving 95% acetaminophen degradation and Mulberry-synthesized nanoparticles achieving 90% sulfamethoxazole degradation under identical conditions [57].
Materials and Methods:
- Bacterial Strain: Geobacillus stearothermophilus GF16 from hydrothermal volcanic area
- Culture Conditions: LB medium, 60°C, 180 rpm for 16-24 hours
- Characterization: UV-Vis spectroscopy, DLS, FT-IR, SEM, TEM
Experimental Procedure:
- Culture G. stearothermophilus GF16 in LB medium under optimized conditions
- Harvest cells by centrifugation and collect supernatant
- Mix supernatant with silver nitrate solution for nanoparticle synthesis
- Optimize parameters including precursor concentration, temperature, pH, and reaction time
- Characterize nanoparticles for size, morphology, and stability
- Evaluate catalytic activity in dye degradation
Performance Validation: The biosynthesized silver nanoparticles demonstrated complete degradation of Congo Red within 20 minutes and 4-nitrophenol within 35 minutes, showcasing exceptional catalytic activity [58].
Synthesis Pathways and Mechanisms
The following diagrams illustrate key synthesis pathways and mechanisms for green-synthesized nanoparticles in environmental applications.
Green Nanoparticle Synthesis and Application Workflow
Pollutant Degradation Mechanisms
The Researcher's Toolkit: Essential Materials and Reagents
Table 4: Essential Research Reagents for Green Nanoparticle Synthesis
| Reagent/Material | Function | Example Specifications | Alternative Options |
|---|---|---|---|
| Plant Materials | Source of reducing and stabilizing agents | Leaves: Eucalyptus, Mulberry, Neem; Fruits: Turmeric | Various plant parts based on phytochemical content [20] |
| Microbial Strains | Biological nanofactories | Aspergillus niger, Geobacillus stearothermophilus | Other fungi, bacteria, yeast based on metal resistance [59] [58] |
| Metal Salts | Nanoparticle precursors | FeCl₃, AgNO₃, HAuCl₄ | Various water-soluble metal salts depending on target NP [57] |
| Culture Media | Microbial growth support | LB medium, Potato Dextrose Agar | Specific media based on microbial requirements [59] |
| pH Adjusters | Reaction condition control | NaOH, HCl solutions | Other alkaline/acid solutions for pH optimization [59] |
| Characterization Tools | NP analysis | UV-Vis, FT-IR, XRD, SEM, TEM | Various analytical techniques based on required data [57] |
The comprehensive comparison presented in this guide demonstrates that green-synthesized nanoparticles offer a competitive and often superior alternative to conventionally synthesized nanoparticles for environmental applications. The experimental data reveals comparable and in some cases enhanced performance in pollutant degradation and heavy metal removal, coupled with significant advantages in environmental sustainability, safety, and economic viability. The detailed protocols and reagent information provide researchers with practical guidance for implementing these green synthesis approaches in their own laboratories. As the field continues to advance, green synthesis methods are poised to play an increasingly critical role in developing sustainable nanotechnology solutions for pressing environmental challenges.
Overcoming Green Synthesis Hurdles: Scalability, Reproducibility, and Characterization
Green synthesis of nanoparticles using plant extracts has emerged as a sustainable alternative to conventional chemical and physical methods, offering benefits such as reduced environmental toxicity, lower energy requirements, and enhanced biocompatibility [61] [62]. However, a significant challenge hindering its standardization and widespread adoption is the limited reproducibility of synthesis protocols, primarily driven by biological variability in plant sources [63]. Unlike conventional methods where precursors are chemically defined, green synthesis relies on phytochemical complexes whose composition fluctuates based on plant genetics, geographical location, seasonal changes, and extraction methodologies [64] [65]. This article analyzes how these variables impact the critical quality attributes (CQAs) of nanoparticles and provides a comparative assessment of strategies to mitigate these challenges, framing the discussion within the broader context of green versus conventional synthesis for pharmaceutical and biomedical applications.
Comparative Analysis of Key Variables Affecting Reproducibility
The reproducibility of green-synthesized nanoparticles is influenced by multiple interdependent factors related to the biological source material. The table below summarizes the impact of these key variables on nanoparticle characteristics and compares the associated challenges with conventional synthesis methods.
Table 1: Impact of Biological Variables on Green Nanoparticle Synthesis Reproducibility
| Variable | Impact on Nanoparticle Characteristics | Key Challenges for Reproducibility | Conventional Synthesis Analog & Comparison |
|---|---|---|---|
| Plant Source & Geography | Size, shape, stability, and biological activity [63] [34]. | Phytochemical diversity across species and geographical locations [61]. | Precursor Purity: Chemically defined precursors ensure consistency, unlike variable plant extracts [64]. |
| Seasonal Variation | Concentration of reducing/stabilizing agents, leading to variations in yield, size distribution, and stability [34]. | Fluctuations in secondary metabolite profiles due to harvest time [63]. | Process Control: Synthetic reactions are buffered from seasonal biological variation [62]. |
| Extraction Method | Efficiency in extracting active phytochemicals, directly affecting reduction rate and capping efficiency [31]. | Lack of standardized protocols for extraction (e.g., solvent, temperature, duration) [63] [62]. | Standardized Protocols: Well-established, optimized steps with tight control over parameters [64]. |
| Extract Characterization | Unknown composition leads to unpredictable outcomes and poor control over NP morphology [63]. | Incomplete profiling of phytochemicals responsible for reduction and capping [63]. | Defined Mechanisms: Reaction pathways and reducing agents are precisely known and controlled [61]. |
Experimental Data and Protocols: Quantifying Variability
Impact of Extraction Methodology
A pivotal study optimizing the synthesis of Silver Nanoparticles (AgNPs) using Azadirachta indica (neem) leaf extract demonstrates how extraction and synthesis parameters directly influence nanoparticle properties [31]. The researchers followed a detailed protocol and quantified the impact of several factors.
Experimental Protocol Overview [31]:
- Extract Preparation: Neem leaves were washed, dried, and ground. The extract was prepared using different solvents and methods (e.g., boiling, maceration).
- Synthesis: 10 mL of leaf extract was reacted with 10 mL of 1 mM silver nitrate under varying conditions.
- Characterization: UV-vis spectroscopy (absorption peak at 420-440 nm), FTIR (identification of functional groups), SEM (size and morphology), and XRD (crystallinity).
Table 2: Impact of Synthesis Parameters on AgNP Characteristics [31]
| Parameter | Condition Varied | Observed Impact on AgNPs | Optimal Condition Found |
|---|---|---|---|
| Temperature | Room Temp, 30°C, 40°C, 70°C | No synthesis at lower temps; highest yield and stability at 70°C | 70 °C |
| Reaction Time | Up to 3 hours | Intensity of SPR peak increased with time, indicating higher yield | 3 hours |
| pH | Acidic, Neutral, Basic | Affected reaction kinetics, size, and stability of nanoparticles | Neutral |
| Extract Volume/Concentration | Different volumes and dilutions | Higher concentrations accelerated reduction but could cause aggregation | 10 mL extract with 10 mL of 1 mM AgNO₃ |
Comparative Performance: Green vs. Chemical Nanoparticles
Experimental data from a study on AgNPs for agricultural applications provides a direct comparison of critical quality attributes between green and chemically synthesized counterparts.
Table 3: Direct Comparison of Green vs. Chemically Synthesized AgNPs [8]
| Quality Attribute | Green-Synthesized AgNPs (Neem Extract) | Chemically Synthesized AgNPs (Sodium Citrate) |
|---|---|---|
| Crystallite Size | 9.7 nm | 20.6 nm |
| Zeta Potential | -55.2 mV | -35.7 mV |
| Colloidal Stability | Higher (due to higher zeta potential) | Lower |
| Silver Uptake in Seeds | 144 ppm | 105 ppm |
| Germination Rate Increase | 50% over control | 31% over control |
| Cost & Environmental Impact | Lower cost, eco-friendly, biodegradable capping [8] | Higher cost, energy-intensive, toxic byproducts [62] |
The superior performance of green AgNPs in this application is attributed to their smaller size and higher stability, facilitated by the natural capping agents in the neem extract [8]. This highlights the potential benefit of green synthesis, contingent on overcoming reproducibility issues.
The Scientist's Toolkit: Essential Reagents and Materials
To conduct reproducible research in plant-mediated nanoparticle synthesis, specific reagents and analytical tools are essential. The following table details key solutions and their functions.
Table 4: Essential Research Reagent Solutions for Green Nanoparticle Synthesis
| Reagent / Material | Function in Synthesis | Critical Considerations |
|---|---|---|
| Plant Material | Source of reducing and capping agents (e.g., flavonoids, polyphenols) [34] [65]. | Document species, genotype, geographical origin, harvest time, and plant part used [63]. |
| Extraction Solvent | Medium for extracting active phytochemicals (e.g., water, ethanol, methanol). | Solvent polarity impacts the profile of extracted compounds; requires standardization [31]. |
| Metal Salt Precursor | Source of metal ions for nanoparticle formation (e.g., AgNO₃, HAuCl₄) [31]. | Purity and concentration must be consistent; a known variable versus chemical synthesis. |
| Characterization Suite | UV-vis, FTIR, SEM/TEM, DLS/Zeta Potential, XRD [32]. | Non-negotiable for batch-to-batch comparison and linking NP properties to biological activity [63] [31]. |
| Buffer Solutions | To control and standardize the pH during synthesis [31]. | pH is a critical process parameter that significantly influences NP size and shape [31]. |
Workflow and Strategic Approaches for Enhanced Reproducibility
The following diagram illustrates the interconnected challenges and strategic solutions for ensuring reproducibility in green nanoparticle synthesis.
The strategies outlined in Figure 1, such as protocol harmonization and the integration of advanced analytical tools like LC-MS and NMR for comprehensive phytochemical profiling, are crucial for transitioning from empirical trials to a predictable science [63]. Furthermore, the application of Artificial Intelligence (AI) can significantly enhance consistency by predicting optimal plant sources and synthesis parameters, thereby reducing reliance on trial-and-error approaches [66].
The variability inherent in plant sources—driven by geography, seasonality, and extraction techniques—presents a formidable challenge to the reproducibility of green-synthesized nanoparticles. However, as comparative data shows, overcoming this challenge is worthwhile, given the potential for green nanoparticles to outperform their chemically synthesized counterparts in terms of size, stability, and functional efficacy in applications like drug delivery and agriculture [8]. The path forward requires a concerted shift from empirical methods to a rigorous, data-driven framework. By adopting standardized protocols, leveraging advanced characterization, and fostering open science, researchers can mitigate the impact of biological variability. This will enable the full realization of green synthesis as a robust, scalable, and sustainable platform for producing nanomaterials for pharmaceutical and biomedical applications, firmly establishing its value proposition against conventional methods.
The synthesis of nanoparticles via green methods, which utilize biological resources like plant extracts or microorganisms, has emerged as a remarkable alternative to conventional chemical and physical approaches. Green synthesis is celebrated for being economical, safe, and environmentally responsible, as it often avoids toxic chemicals, utilizes sustainable resources, and encourages resource efficiency [4]. In the context of drug development and biomedical applications, this method offers the added advantage of producing nanoparticles with inherent biocompatibility, making them highly attractive for therapeutic applications, including targeted drug delivery and wound healing [5].
However, a significant chasm exists between demonstrating these promising properties in a laboratory setting and achieving industrial-scale production. The very factors that make green synthesis appealing at the benchtop—such as the use of complex biological extracts—introduce unique challenges in reproducibility, process control, and cost-effectiveness when scaling up. This guide objectively compares the performance of green-synthesized nanoparticles with their conventional counterparts and examines the specific scalability challenges that must be overcome to bridge this critical gap.
Experimental Protocols for Performance Comparison
To objectively compare nanoparticle performance, researchers typically follow standardized synthesis and evaluation protocols. The methodologies below are representative of those used to generate the comparative data in the following sections.
Protocol for Green Synthesis of Metal Oxide Nanoparticles
This protocol is adapted from the green synthesis of Bismuth Oxide (Bi₂O₃) nanoparticles using olive leaf extract [67].
- Preparation of Plant Extract: 40 g of freshly chopped olive leaves are added to 500 mL of distilled water. The mixture is heated to 95°C and maintained at that temperature for 60 minutes with constant stirring. The resulting extract is cooled, filtered to remove solid debris, and stored at 4°C for future use.
- Synthesis of Nanoparticles: 50 mL of a 0.1 M solution of Bismuth Nitrate Pentahydrate (Bi(NO₃)₃·5H₂O) is prepared using acetic acid as a solvent. This precursor solution is added dropwise to 400 mL of the olive leaf extract at 60°C under vigorous stirring. The reaction is allowed to proceed for 3 hours.
- Recovery and Processing: The resulting precipitate is collected via filtration, washed thoroughly with distilled water, and dried in an oven at 70°C for 4 hours. The dried powder is then calcined in a muffle furnace at 600°C for 2 hours to obtain the final crystalline Bi₂O₃ nanoparticles.
Protocol for Conventional Chemical Synthesis of Iron Nanoparticles
This protocol for conventional (chemical) Iron Nanoparticle (C-Fe-NP) synthesis is derived from a comparative study with green methods [57].
- Solution Preparation: A 250 mL solution containing Fe²⁺ and Fe³⁺ ions in a 2:1 molar ratio is prepared. To this, 500 μL of oleic acid is added as a stabilizer.
- Reduction and Precipitation: The solution is placed under mechanical stirring at 1000 rpm. Ammonium hydroxide (a strong chemical reductant) is added dropwise until the pH of the solution reaches 11, initiating the precipitation of iron nanoparticles.
- Ageing and Washing: The reaction mixture is heated to 85°C and maintained with stirring for 30 minutes. The nanoparticles are then collected by filtration and washed repeatedly with ultrapure water until a neutral pH is achieved in the filtrate.
Standardized Catalytic Performance Test
A common method to evaluate the functional performance of synthesized nanoparticles is through catalytic degradation tests [57].
- Reactor Setup: A specified concentration of the target contaminant (e.g., 250 μg/L of the drug Sulfamethoxazole) is prepared in a reaction vessel.
- Reaction Initiation: The catalyst (the nanoparticles to be tested) is added to the solution at a defined concentration (e.g., 0.5 g/L). The reaction is initiated by introducing ozone at a constant rate (e.g., 3.5 mg/min).
- Monitoring: Samples are taken from the reactor at regular time intervals. The concentration of the remaining contaminant is analyzed using techniques like High-Performance Liquid Chromatography (HPLC) to determine the degradation efficiency and kinetics.
Comparative Performance Data: Green vs. Conventional Nanoparticles
The following tables summarize experimental data from comparative studies, highlighting the performance of green-synthesized nanoparticles against conventional ones in key application areas.
Table 1: Comparative Catalytic Performance in Pollutant Degradation [57]
| Nanoparticle Type | Contaminant | Degradation Efficiency (%) | Reaction Time (min) |
|---|---|---|---|
| Conventional Fe-NP (C-Fe-NP) | Sulfamethoxazole | 65.3 | 30 |
| Green Fe-NP (E-Fe-NP, Eucalyptus) | Sulfamethoxazole | 98.5 | 30 |
| Green Fe-NP (M-Fe-NP, Mulberry) | Sulfamethoxazole | 92.1 | 30 |
| Conventional Fe-NP (C-Fe-NP) | Acetaminophen | 58.0 | 30 |
| Green Fe-NP (E-Fe-NP, Eucalyptus) | Acetaminophen | 95.0 | 30 |
Table 2: Properties of Unsaturated Polyester Nanocomposites for Radiation Shielding [67]
| Filter Type (in Polyester) | Filter Loading (wt%) | Linear Attenuation Coefficient, LAC (cm⁻¹ at 59 keV) | Temperature for 10% Weight Loss (°C) | Compressive Strength (MPa) |
|---|---|---|---|---|
| None (Pure Polyester) | 0 | 0.250 | 247 | 10.00 |
| Green-Synthesized Bi₂O₃ NPs | 20 | 1.668 | 326 | 4.94 |
Table 3: Advantages and Disadvantages of Synthesis Approaches
| Aspect | Green Synthesis | Conventional Chemical Synthesis |
|---|---|---|
| Environmental Impact | Lower inherent toxicity, biodegradable byproducts [4] | Use of hazardous chemicals and toxic substances [4] |
| Energy Consumption | Can be high (e.g., prolonged reaction times, elevated temps) [7] | Can be low (e.g., room temperature reactions possible) [7] |
| Process Control | Low; complex bio-extracts lead to variability [5] | High; precise control over reaction parameters [68] |
| Scalability | Challenging due to biological variability and process control [69] | Established, but concerns over waste and cost remain [69] |
| Product Yield | Often lower and less reported [7] | Typically high and reproducible [7] |
| Biocompatibility | Generally enhanced due to bio-derived capping agents [5] | Often requires post-synthesis functionalization |
Analysis of Scalability Challenges and Industrial Pathways
The transition from lab to industry requires moving from batch processes producing grams of material to continuous processes producing kilograms or tons, while maintaining consistent quality. The data in Table 3 highlights the core tension: green synthesis excels in sustainability and biocompatibility but faces significant hurdles in process control and scalability.
Key Scalability Challenges for Green Synthesis
- Biological Variability: The chemical composition of plant extracts can vary with season, geography, and plant age, leading to inconsistencies in nanoparticle size, shape, and properties [5]. This lack of standardization is a major barrier to reproducibility in large-scale batches.
- Process Intensification: Many green synthesis protocols require long reaction times and elevated temperatures, which are energy-intensive at scale [7]. Furthermore, downstream processes like purification, separation, and drying of nanoparticles from complex biological mixtures present significant engineering challenges [69].
- Economic Viability and Yield: While often cited as cost-effective, the economic picture at scale is complex. Lower reported product yields compared to conventional methods (Table 3) mean more raw material is needed per unit of output, potentially increasing costs and environmental impact [7]. A comprehensive lifecycle assessment is necessary for a true eco-efficiency evaluation.
Promising Industrial Scale-Up Techniques
To address these challenges, the industry is adapting and developing several scalable production technologies, which can be categorized into top-down and bottom-up approaches [69].
- Top-Down Methods: These involve breaking down bulk material into nanoparticles. Mechanical Milling is a well-established, scalable technique, though it can introduce impurities and lacks precise control over particle shape [69]. High-Pressure Homogenization is another scalable top-down method, valuable for producing nanoemulsions and nanocrystals [70].
- Bottom-Up Methods: These construct nanoparticles from atomic or molecular precursors. Spray Drying is an economical and scalable technique for converting nanoparticle suspensions into dry, stable powders, which is crucial for storage, transport, and product formulation [70]. Microreactor Technology offers a path for continuous flow synthesis, providing superior control over temperature and mixing, which can lead to more uniform nanoparticles and is highly amenable to process automation and scaling [70].
Diagram 1: The pathway from laboratory-scale synthesis to industrial production, mapping key challenges against potential scale-up solutions.
The Scientist's Toolkit: Essential Reagents and Materials
The experimental protocols and scalable synthesis methods rely on a core set of reagents and equipment. The following table details key solutions and their functions in green nanoparticle research and development.
Table 4: Research Reagent Solutions for Green Nanoparticle Synthesis
| Reagent / Material | Function in Research & Development | Example in Use |
|---|---|---|
| Plant Extract | Serves as a source of phytochemicals (e.g., polyphenols, flavonoids) that act as reducing and stabilizing/capping agents. | Olive leaf extract for synthesizing Bi₂O₃ NPs [67]. |
| Metal Salt Precursors | The source of metal ions that are reduced to form the nanoparticle core (e.g., Ag⁺, Au³⁺, Fe²⁺/³⁺, Bi³⁺). | Bismuth Nitrate Pentahydrate, Silver Nitrate, Gold Chloride [67] [20]. |
| Stabilizing Agents (Chemical) | In conventional synthesis, these prevent aggregation of nanoparticles (e.g., polymers, surfactants). | Oleic acid, Polyvinylpyrrolidone (PVP) [57]. |
| Reducing Agents (Chemical) | In conventional synthesis, these forcefully donate electrons to reduce metal ions (e.g., borohydride, hydrazine). | Sodium Borohydride, Ammonium Hydroxide, Hydrazine [7] [57]. |
| Microreactor Systems | Continuous flow devices for scalable, reproducible synthesis with superior control over reaction parameters. | Used for producing metallic, semiconductor, and organic NPs with consistent quality [70]. |
| Spray Dryer | Industrial equipment for rapid conversion of nanoparticle suspensions into dry, free-flowing powders. | Preserves nanomaterial structure during drying for pharmaceuticals and nutraceuticals [70]. |
| High-Pressure Homogenizer | Top-down equipment using high pressure to create nanoemulsions or reduce particle size. | Gold standard for scalable nanoemulsion preparation [70]. |
The comparative data clearly demonstrates that green-synthesized nanoparticles can not only match but in some cases surpass the functional performance of conventional nanoparticles, particularly in catalytic and biomedical applications. However, their path to widespread industrial adoption is fraught with unique challenges centered on reproducibility, process control, and economic viability.
Bridging the gap from bench to production will require a concerted interdisciplinary effort. This includes the standardization of biological extracts, the adoption of continuous manufacturing technologies like microreactors, and the implementation of comprehensive lifecycle assessments to truly validate the environmental and economic benefits of green synthesis at scale. The future of industrial nanoparticle production will likely hinge on successfully integrating the ecological advantages of green chemistry with the rigorous demands of process engineering.
In the realm of nanotechnology, particularly for biomedical and drug development applications, the precise control over nanoparticle size and shape transcends mere optimization—it constitutes a fundamental requirement for functionality. Size dictates cellular uptake, biodistribution, and circulation time, while shape influences targeting efficiency, flow dynamics, and intracellular trafficking [71]. Traditional physical and chemical synthesis methods, though capable of producing nanoparticles, often involve hazardous chemicals, generate toxic byproducts, and present challenges in achieving uniform, reproducible results [72] [1]. Green synthesis, which utilizes biological resources like plant extracts as reducing and stabilizing agents, presents a sustainable, eco-friendly, and cost-effective alternative [72] [73].
This guide objectively compares the performance of green synthesis approaches against conventional methods, with a specific focus on how parameters such as pH, temperature, and reaction time serve as critical "optimization levers" for controlling nanoparticle characteristics. The ability to fine-tune these parameters in green synthesis protocols enables researchers to produce nanoparticles with tailored properties that rival, and in some aspects surpass, those produced through conventional routes, especially for sensitive applications like drug delivery [74] [75]. The following sections provide a detailed comparison supported by experimental data, delineate specific experimental protocols, and visualize the optimization pathways.
Comparative Analysis of Green vs. Conventional Synthesis
The pursuit of precise nanoparticles necessitates a clear understanding of how different synthesis methods perform. The table below provides a comparative overview of green and conventional synthesis approaches, highlighting their efficacy in controlling size and shape through key parameters.
Table 1: Comparison of Green and Conventional Synthesis Methods for Nanoparticle Control
| Synthesis Method | Control Over Size & Shape | Role of pH | Role of Temperature | Role of Reaction Time | Key Advantages | Key Limitations |
|---|---|---|---|---|---|---|
| Green Synthesis (Biological) | High, with optimization; can produce spheres, rods, hexagons [76] [73]. | Critical for stability & size; higher pH often yields smaller, more stable NPs (e.g., -64 mV at pH 11) [73]. | Higher temperature generally increases size & shape variety (e.g., spheres at 4°C vs. rods/hexagons at 60°C) [77] [76]. | Longer time can lead to larger particles and eventual aggregation [76]. | Eco-friendly, uses non-toxic reagents, biocompatible products, cost-effective [72] [75]. | Optimization can be complex; batch-to-batch variation possible [1]. |
| Chemical Reduction | Moderate to high; often spherical, but shapes can be tailored with capping agents [78]. | Important for reduction rate and nucleation; can influence final size. | Key driver for reaction kinetics and final particle size. | Determines the growth phase and final size distribution. | Well-established, high yield, good size control [72]. | Uses hazardous reducing agents (e.g., NaBH₄), toxic byproducts [72]. |
| Polyol Method | High for shape control (e.g., spheres, cubes, wires) [78]. | Less emphasized; reaction is driven by temperature and precursor concentration. | Primary factor for controlling shape and size; precise thermal control is required. | Affects the growth phase and crystallinity. | Excellent shape and size control, high monodispersity [78]. | Can involve high-boiling-point solvents, complex reaction setups. |
| Physical Methods (Laser Ablation, Milling) | Low to moderate; often polydisperse, limited shape control (mainly spheres) [72]. | Not a direct factor. | Can influence particle properties post-synthesis. | Determines yield; longer time typically increases yield. | No solvent contamination, simple concept [72]. | High energy consumption, broad size distribution, low yield [72] [1]. |
Quantitative Data on Optimization Parameters
Experimental data from green synthesis studies provides clear, quantitative evidence of how pH, temperature, and reaction time directly influence nanoparticle characteristics. The following tables consolidate key findings from recent research.
Table 2: Experimental Data on the Effect of pH in Green Synthesis
| Nanoparticle Type | Biological Source | pH Variation | Impact on Size | Impact on Shape | Impact on Stability (Zeta Potential) | Reference |
|---|---|---|---|---|---|---|
| Silver (Ag) NPs | Pistacia atlantica leaf | pH 7 to pH 11 | Size control not specified | Shape control not specified | Increased from -21.7 mV to -64.0 mV | [73] |
| Silver (Ag) NPs | Eucalyptus camaldulensis & Terminalia arjuna | Neutral pH (7) | Optimal for synthesis yield | Optimal for synthesis yield | Not specified | [74] |
Table 3: Experimental Data on the Effect of Temperature in Green Synthesis
| Nanoparticle Type | Biological Source | Temperature Variation | Impact on Size | Impact on Shape | Key Findings | Reference |
|---|---|---|---|---|---|---|
| Zinc Oxide (ZnO) NPs | Pineapple peel waste | 28°C (Room Temp) | 8 - 45 nm | Mixture of spherical and rod shapes | Lower temperature favored smaller, more mixed shapes. | [77] |
| 60°C | 73 - 123 nm | Flower rod shapes | Higher temperature produced larger, defined structures. | [77] | ||
| Silver (Ag) NPs | Curcumin (Solid-state) | 4°C | Smaller particles | Spherical | Smaller particles exhibited better catalytic performance. | [76] |
| 60°C | Larger particles (>100 nm) | Spherical, rods, hexagonal | Higher temperature and longer time produced larger particles and more shape diversity. | [76] | ||
| Silver (Ag) NPs | Rubus discolor leaf | 56.5°C (Optimal) | ~37 nm (average) | Spherical, well-dispersed | Optimized via RSM; temperature was a key factor. | [75] |
Table 4: Experimental Data on the Effect of Reaction Time in Green Synthesis
| Nanoparticle Type | Biological Source | Time Variation | Impact on Size | Impact on Shape | Key Findings | Reference |
|---|---|---|---|---|---|---|
| Silver (Ag) NPs | Curcumin (Solid-state) | 1 - 16 hours; 1 - 7 days | Size increased with time | Shape diversity increased with time | After 5-7 days at 60°C, particles exceeded nanoscale. | [76] |
| Silver (Ag) NPs | Eucalyptus camaldulensis & Terminalia arjuna | 60 minutes (Optimal) | Optimal for synthesis yield | Optimal for synthesis yield | Identified as optimal for maximum yield of stable, spherical NPs. | [74] |
Experimental Protocols for Green Synthesis Optimization
To ensure reproducibility and provide a practical guide for researchers, detailed methodologies from key studies are outlined below.
Protocol 1: Optimization of Silver Nanoparticles Using Plant Extracts
This protocol is adapted from studies using Eucalyptus camaldulensis and Terminalia arjuna extracts, which optimized conditions via a systematic approach [74].
- Step 1: Preparation of Plant Extract. Collect healthy leaves of E. camaldulensis and bark of T. arjuna. Wash thoroughly with distilled water to remove impurities. Dry the plant material in an oven at 60°C overnight. Grind the dried material into a coarse powder. To prepare the extract, boil 10 g of powder in 100 mL of distilled water for 15 minutes. Cool the mixture to room temperature and filter it using Whatman No. 1 filter paper. The clear filtrate can be stored at 4°C for further use.
- Step 2: Synthesis of Silver Nanoparticles. Prepare a 1 mM aqueous solution of silver nitrate (AgNO₃). Mix the plant extract with the AgNO₃ solution. The optimal conditions for synthesis, as determined by response surface methodology, are: a temperature of 75°C, a reaction time of 60 minutes, and a neutral pH [74].
- Step 3: Purification and Recovery. After the reaction is complete, cool the mixture to room temperature. Purify the synthesized nanoparticles by centrifugation at high speed (e.g., 15,000 rpm for 5-15 minutes). Discard the supernatant and re-disperse the pellet in distilled water. Repeat this washing process 2-3 times to remove any unbound biological compounds. Finally, dry the purified nanoparticle powder at 60°C for characterization.
Protocol 2: Temperature-Dependent Synthesis of Zinc Oxide Nanoparticles
This protocol, derived from research using pineapple peel waste, highlights the direct influence of synthesis temperature on the size and morphology of ZnO nanoparticles [77].
- Step 1: Preparation of Fruit Peel Extract. Wash fresh pineapple peels and dry them in an oven at 60°C overnight. Grind the dried peels into a powder. Add 10 g of this powder to 100 mL of distilled water and boil for 15 minutes. Filter the cooled mixture to obtain a clear extract.
- Step 2: Synthesis of ZnO Nanoparticles at Different Temperatures. Add 50 mL of a 0.01 M solution of zinc nitrate hexahydrate to 1 mL of the pineapple peel extract. Adjust the pH of the solution to 12 using 5 M sodium hydroxide (NaOH). Divide this mixture into two parts. Stir one part continuously at 60°C on a hotplate for 2 hours, and keep the other part at room temperature (e.g., 28°C) with stirring for the same duration. A white precipitate indicates the formation of ZnO nanoparticles.
- Step 3: Purification. Centrifuge the precipitates at 15,000 rpm for 5 minutes to collect the nanoparticles. Wash the pellets with distilled water or ethanol and dry them for further characterization using techniques like TEM, XRD, and FTIR.
Pathways and Workflows for Nanoparticle Optimization
The following diagram synthesizes the information from the cited studies to illustrate the logical relationship between synthesis parameters and the final properties of green-synthesized nanoparticles. This serves as a visual guide for the optimization process.
The Scientist's Toolkit: Essential Research Reagents and Materials
Success in green nanoparticle synthesis relies on a specific set of reagents and equipment. The following table details the essential components of a research toolkit for this field.
Table 5: Essential Research Reagent Solutions for Green Synthesis
| Reagent / Material | Function in Synthesis | Specific Examples from Literature |
|---|---|---|
| Metal Salt Precursors | Source of metal ions for reduction into nanoparticles (NPs). | Silver nitrate (AgNO₃) for Ag NPs [74] [75]; Zinc nitrate (Zn(NO₃)₂) for ZnO NPs [77]. |
| Biological Reducing Agents | Provide phytochemicals (e.g., phenolics, flavonoids) that reduce metal ions to their zero-valent state. | Leaf extracts of Pistacia atlantica [73], Eucalyptus camaldulensis [74], Rubus discolor [75]; fruit peel extract of pineapple [77]. |
| pH Modifiers | Adjust the pH of the reaction mixture, which critically influences reduction rate, nucleation, and nanoparticle stability. | Sodium hydroxide (NaOH) to maintain basic conditions (pH 12) [77]; acids or buffers for precise pH control. |
| Capping / Stabilizing Agents | Phytochemicals in the extract (e.g., proteins, terpenoids) that adsorb to nanoparticle surfaces, preventing aggregation and controlling growth. | Compounds in Pistacia extracts stabilize NPs [73]; Polyvinylpyrrolidone (PVP) is a common chemical capping agent [78]. |
| Solvents | Medium for the reaction; water is predominantly used in green synthesis. | Deionized/Distilled water [77] [74]; ethanol for washing and purification [76]. |
| Purification Equipment | Separate synthesized nanoparticles from the reaction mixture and unbound compounds. | Laboratory centrifuge [77] [74] [75]; filters (e.g., Whatman filter paper) [77]. |
The experimental data and protocols presented in this guide unequivocally demonstrate that green synthesis is a powerful and viable methodology for producing nanoparticles with controlled size and shape. Parameters such as pH, temperature, and reaction time are potent and accessible levers that researchers can manipulate to tailor nanoparticles for specific applications, particularly in drug development. While conventional methods offer a historical baseline of control, green synthesis excels in providing a sustainable pathway without compromising on the ability to fine-tune critical nanoparticle characteristics. The reproducibility and scalability of these green approaches, supported by statistical optimization methods like Response Surface Methodology (RSM), continue to strengthen their position in the nanotechnology landscape, offering a promising tool for scientists and drug development professionals dedicated to advancing nanomedicine.
The synthesis of nanoparticles, whether through conventional chemical routes or sustainable green methods, inevitably generates organic impurities and byproducts. These unwanted compounds can significantly alter the physicochemical properties of the nanoparticles, affecting their performance in applications ranging from drug delivery to environmental remediation. Conventional synthesis methods often employ toxic reducing agents like sodium borohydride and stabilizing agents like polyvinylpyrrolidone, which leave residual impurities that require complex removal strategies [61] [64]. In contrast, green synthesis approaches utilize biological extracts containing phytochemicals that serve as natural reducing and capping agents, resulting in different, though not necessarily absent, impurity profiles [5] [79].
The purification and analysis techniques employed must therefore be tailored to the specific synthesis method and the nature of the impurities involved. This comparison guide objectively examines the key differences in impurity management between these two paradigms, supported by experimental data and detailed methodologies to assist researchers in selecting appropriate protocols for their specific applications.
Comparative Analysis: Conventional vs. Green Synthesis Impurities
Nature and Origin of Synthesis Byproducts
Conventional Chemical Synthesis typically generates impurities from:
- Unreacted precursors: Metal salts and reducing agents that haven't fully participated in the redox reaction [64]
- Chemical stabilizers: Compounds like citrate, cetyltrimethylammonium bromide (CTAB), or polymers added to control nanoparticle growth and prevent aggregation [61]
- Toxic intermediates: Reactive byproducts formed during synthesis that may persist through initial processing stages [64]
Green Biogenic Synthesis produces distinct byproducts originating from:
- Phytochemical residues: Polyphenols, flavonoids, terpenoids, and other plant metabolites not incorporated into the capping layer [5] [79]
- Biological macromolecules: Proteins, enzymes, and polysaccharides from plant or microbial extracts that may adsorb to nanoparticle surfaces [3]
- Natural degradation products: Compounds formed through the oxidation or transformation of biological reducing agents during synthesis [80]
Quantitative Comparison of Purification Efficiency
Table 1: Efficiency of Purification Techniques for Different Nanoparticle Synthesis Methods
| Purification Technique | Synthesis Method | Removal Efficiency (%) | Key Impurities Targeted | Impact on Nanoparticle Properties |
|---|---|---|---|---|
| Dialysis | Green | >85% [79] | Small-molecule phytochemicals | Minimal effect on natural capping layer |
| Conventional | >90% [64] | Ionic byproducts, small organics | May destabilize uncapped nanoparticles | |
| Centrifugation | Green | 70-80% [5] | Macromolecular biological residues | Potential for aggregation at high G-force |
| Conventional | >95% [64] | Stabilizer aggregates, large impurities | Effective for concentration and washing | |
| Chromatography | Green | >90% [80] | Specific phytochemical classes | Can selectively preserve functional caps |
| Conventional | >98% [61] | Synthetic polymers, precise size selection | May require toxic solvents | |
| Ultrafiltration | Green | 75-85% [79] | Proteins, polysaccharides | Retains beneficial capping agents |
| Conventional | >95% [64] | Molecular weight-based separation | Excellent for size uniformity |
Table 2: Environmental and Operational Comparison of Purification Methods
| Parameter | Green Synthesis Purification | Conventional Synthesis Purification |
|---|---|---|
| Energy Consumption | 30-40% lower overall [10] | High energy requirements |
| Chemical Usage | Primarily aqueous solutions [79] | Often requires organic solvents |
| Hazardous Waste Generation | Minimal, mostly biodegradable [3] | Significant, requiring special disposal |
| Process Time | Generally shorter dialysis times [5] | Multiple washing cycles often needed |
| Scalability | Challenges in maintaining consistency [64] [80] | Well-established scale-up protocols |
Experimental Protocols for Impurity Removal and Analysis
Detailed Methodology: Purification of Plant-Mediated Silver Nanoparticles
Principle: This protocol describes the purification of silver nanoparticles synthesized using Ficus carica leaf extract, removing excess phytochemicals while preserving the functional capping layer [56] [79].
Materials and Reagents:
- Crude nanoparticle synthesis solution: Silver nanoparticles (0.1-10 nm) synthesized from Ficus carica leaf extract
- Dialysis membrane: Molecular weight cutoff 12-14 kDa
- Ultrapure water: 18.2 MΩ·cm resistivity
- Centrifuge: Capable of 15,000-20,000 × g
- Amicon ultrafiltration units: 10 kDa molecular weight cutoff
Procedure:
- Primary Separation: Subject the crude synthesis mixture to centrifugation at 12,000 × g for 20 minutes at 4°C to pellet large aggregates and insoluble plant material [79].
- Dialysis Purification:
- Transfer the supernatant to a pre-treated dialysis membrane
- Dialyze against ultrapure water (4°C) with constant stirring
- Change water every 4 hours for the first 12 hours, then every 12 hours for 2-3 days
- Monitor conductivity until it stabilizes below 5 μS/cm, indicating removal of ionic impurities
- Concentration and Storage:
- Concentrate the dialyzed suspension using ultrafiltration (10 kDa cutoff) at 3,000 × g
- Resuspend in the desired buffer for specific applications
- Store at 4°C protected from light
Quality Control Assessment:
- UV-Vis Spectroscopy: Monitor surface plasmon resonance peak at ~420 nm for silver nanoparticles; sharpening indicates improved size homogeneity
- Fourier Transform Infrared Spectroscopy: Verify removal of small-molecule phytochemicals while preserving functional capping layer
- Dynamic Light Scattering: Measure hydrodynamic diameter and polydispersity index; values <0.3 indicate successful purification
Detailed Methodology: Analysis of Organic Impurities
Principle: Comprehensive characterization of organic impurities and capping agents on purified nanoparticles using spectroscopic and chromatographic techniques [80] [79].
Materials and Reagents:
- Purified nanoparticle suspension: From Protocol 3.1
- High-Performance Liquid Chromatography system: With C18 column and diode array detector
- FTIR Spectrometer: With ATR attachment
- Ultrapure solvents: Methanol, acetonitrile, water (HPLC grade)
Procedure:
- Sample Preparation for HPLC:
- Acidify 1 mL of purified nanoparticle suspension to pH 3.0 with 0.1% formic acid
- Extract three times with equal volumes of ethyl acetate
- Combine organic layers and evaporate under nitrogen stream
- Reconstitute in 100 μL methanol for HPLC analysis [79]
- Chromatographic Separation:
- Column: C18 reverse phase (250 × 4.6 mm, 5 μm)
- Mobile phase: A) 0.1% formic acid in water, B) 0.1% formic acid in acetonitrile
- Gradient: 5-95% B over 30 minutes
- Flow rate: 1.0 mL/min
- Detection: 200-600 nm using diode array detector
- FTIR Analysis:
- Deposit concentrated nanoparticle suspension on ATR crystal
- Dry under nitrogen stream
- Acquire spectra from 4000-500 cm⁻¹ at 4 cm⁻¹ resolution
- Compare with spectra of pure plant extract to identify surface-bound compounds
Data Interpretation:
- HPLC: Identify and quantify specific phytochemical impurities by comparing retention times and UV-Vis spectra with authentic standards
- FTIR: Characterize functional groups present in the capping layer; typical bands include O-H (3200-3600 cm⁻¹), C=O (1650-1750 cm⁻¹), and C-O (1000-1300 cm⁻¹) stretches
Visualization of Purification Workflows
Purification Workflow for Green Nanoparticles
Impurity Analysis Pathway
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Reagents for Nanoparticle Purification and Analysis
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Dialysis Membranes | Removal of small molecular weight impurities | MWCO 12-14 kDa optimal for retaining nanoparticles while removing phytochemicals [79] |
| Ultrafiltration Devices | Concentration and size-based separation | 10 kDa MWCO effectively concentrates nanoparticles with minimal loss [79] |
| Chromatography Columns | Separation of specific impurity classes | C18 reverse-phase for phytochemical analysis; size exclusion for nanoparticle purification [80] |
| FTIR Standards | Reference for capping agent identification | Library of plant metabolite spectra essential for green synthesis characterization [79] |
| HPLC Solvents and Standards | Quantification of organic impurities | HPLC-grade solvents with phytochemical standards for accurate quantification [79] |
The purification and analysis of nanoparticles require fundamentally different approaches depending on the synthesis methodology employed. Conventional synthesis generates impurities that are often well-characterized but frequently toxic, requiring rigorous removal protocols that can be energy and resource-intensive [64]. Green synthesis, while producing generally safer byproducts, presents unique challenges in standardizing purification processes due to the variability in biological source materials [5] [80].
The experimental data presented demonstrates that green synthesis routes typically require less intensive purification to achieve biocompatibility, though careful analysis is still essential to understand the nature of the surface capping and its implications for downstream applications. As the field advances, the development of standardized protocols for impurity characterization across different synthesis methods will be crucial for enabling comparative assessments and regulatory approval, particularly for biomedical applications where purity and surface chemistry directly impact safety and efficacy [80] [79].
The synthesis of nanoparticles with precise characteristics is a cornerstone of advancements in medicine, energy, and electronics. Traditional methods often rely on laborious, trial-and-error experimentation. However, the convergence of artificial intelligence (AI) and machine learning (ML) with green synthesis principles is creating a new paradigm for predictive and optimized nanomaterial design [4] [81]. This guide compares AI-driven green synthesis against conventional methods, providing an objective analysis of their performance through experimental data and detailed protocols.
Performance Comparison: AI-Guided Green Synthesis vs. Conventional Methods
The integration of AI into green synthesis routes offers measurable advantages over conventional chemical and physical methods. The table below summarizes a quantitative comparison based on recent research.
Table 1: Performance Comparison of Nanoparticle Synthesis Methods
| Evaluation Parameter | AI-Guided Green Synthesis | Conventional Chemical Synthesis |
|---|---|---|
| Experimental Iterations | Reduced by over 80% through predictive modeling [82] | Relies heavily on trial-and-error |
| Production Cost | Approx. 20-50% lower; uses inexpensive biological precursors [83] [84] | High cost of chemical precursors and hazardous waste disposal [83] |
| Size Control Precision | High precision; ML models predict parameters for desired size [81] [85] | Moderate precision; requires careful manual control |
| Crystallite Size (Example: NiO NPs) | ~10.6 - 10.8 nm (controlled via ML-calibrated calcination) [85] | Often larger and less uniform without advanced control |
| Cytotoxicity (Example: NiO NPs) | Cell viability <40% in HepG2 cancer cells at 600°C calcination [85] | Varies; can be higher due to toxic capping agents |
| Environmental Impact | Minimal; uses plant extracts, biodegradable polymers, and reduced waste [4] [84] | High; involves hazardous chemicals and toxic byproducts [4] [6] |
| Scalability & Reproducibility | Enhanced by continuous flow systems and AI optimization [86] [82] | Challenges with batch-to-batch variation in large reactors [83] |
Experimental Protocols
Protocol 1: AI-Optimized Green Synthesis of Nickel Oxide (NiO) Nanoparticles for Anticancer Applications
This protocol details the synthesis and evaluation of phytogenic NiO nanoparticles, where machine learning was used to predict and correlate synthesis parameters with cytotoxicity [85].
- Objective: To synthesize NiO nanoparticles using a green reducing agent and to apply ML models to predict their cytotoxicity against liver cancer (HepG2) cells.
- Synthesis Methodology:
- Precursor Solution: 7.26 g of nickel nitrate hexahydrate (Ni(NO₃)₂·6H₂O) was dissolved in 50 mL of deionized water [85].
- Green Reducing Agent: 50 mL of egg white was dissolved in 30 mL of deionized water. Egg white proteins act as natural reducing and stabilizing agents [85].
- Reaction: The nickel nitrate solution was added dropwise to the egg white solution under stirring at 70°C [85].
- Calcination: The resulting mixture was dried and then calcined in a tube furnace at temperatures between 400°C and 600°C to form crystalline NiO nanoparticles [85].
- Characterization: X-ray diffraction (XRD) was used to determine the crystalline structure and crystallite size, which was calculated using the Scherrer equation [85].
- Bioassay: The cytotoxicity of the synthesized NiO NPs was tested against HepG2 cell lines using an MTT assay. Cells were treated with various concentrations of NPs (0–400 µg/mL) for 24 hours, and cell viability was calculated [85].
- Machine Learning Analysis: A decision tree model was implemented in Google Colab to predict cytotoxicity based on experimental input parameters (e.g., calcination temperature, NP concentration). A correlation matrix was generated to analyze parameter interdependence [85].
Protocol 2: Microfluidics and ML for Polymeric Nanoparticle (PNP) Synthesis
This protocol describes a scalable, continuous-flow approach for synthesizing polymeric nanoparticles, integrated with AI for process optimization [86] [82].
- Objective: To synthesize uniform polymeric nanoparticles using microfluidic technology and optimize the process parameters with machine learning.
- Synthesis Methodology:
- Microfluidic Device Setup: A capillary-based or chip-based droplet generator was used.
- Active Method: External energy sources (acoustic, thermal) were applied for precise mixing and droplet generation [86].
- Polymer Solution: Biopolymers like chitosan (dissolved in dilute acetic acid) or PLGA (dissolved in an organic solvent) were used as the organic phase [84].
- Aqueous Phase: A surfactant solution (e.g., polyvinyl alcohol) was used as the continuous phase [86].
- Flow Focusing: The organic phase was focused by the aqueous phase within the microchannel to form monodisperse droplets. Nanoparticles formed via solvent diffusion or evaporation [86].
- AI Integration: ML algorithms, such as Gaussian Processes or support vector machines, were used to model the relationship between input parameters (e.g., flow rates, polymer concentration, temperature) and output characteristics (e.g., particle size, polydispersity) [81] [86]. This model then guided the autonomous optimization of the synthesis process.
Workflow and Pathway Visualizations
AI-Optimized Green Synthesis Workflow
AI-Green NP Synthesis Workflow
ML Model Optimization Cycle
ML Parameter Optimization Cycle
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for AI-Guided Green NP Synthesis
| Reagent/Material | Function in Synthesis | Example from Research |
|---|---|---|
| Plant Extracts | Act as natural reducing and capping agents, replacing harsh chemicals. | Terminalia catappa for iron NPs [6], Tridax procumbens for zinc NPs [6] |
| Biopolymers | Form the matrix or shell of polymeric nanoparticles; are biodegradable and biocompatible. | Chitosan, alginate, cellulose [84] |
| Egg White | Rich protein content acts as a natural reducing and stabilizing agent for metal oxide NPs. | Used in the synthesis of NiO nanoparticles [85] |
| Metal Salts | Serve as the precursor source for the metal component of the nanoparticle. | Nickel nitrate (for NiO NPs) [85], Iron chloride (for Fe NPs) [6] |
| Microfluidic Reactors | Provide a controlled environment for continuous, scalable, and reproducible NP synthesis. | Capillary or chip-based droplet generators [86] |
| ML Algorithms | Predict optimal synthesis conditions and map input parameters to output NP characteristics. | Decision Trees, Random Forests, Gaussian Processes [81] [85] [82] |
Comparative Analysis and Validation: Performance, Safety, and Commercial Viability
The synthesis of palladium nanoparticles (PdNPs) represents a significant area of focus in nanotechnology due to their exceptional catalytic properties and burgeoning biomedical applications. Traditionally, PdNPs are produced through chemical or physical methods that often involve energy-intensive processes, hazardous solvents, or toxic stabilizing agents [87]. In contrast, biologically synthesized PdNPs (Bio-PdNPs) have emerged as a sustainable alternative, utilizing the inherent reducing capabilities of plants, bacteria, or fungi to convert palladium ions into nanoscale structures [88]. This review provides a direct performance comparison between Bio-PdNPs and their chemically synthesized counterparts (Chem-PdNPs) within the broader context of green nanotechnology. By examining catalytic efficiency in environmental remediation and potential efficacy in biomedical applications, this analysis aims to offer researchers and drug development professionals a comprehensive, evidence-based guide for selecting appropriate synthesis methodologies for specific applications.
Performance Comparison: Catalytic and Environmental Remediation
The catalytic performance of PdNPs is critically dependent on their physical characteristics, which are in turn influenced by the synthesis method. Bio-PdNPs often exhibit superior properties due to more favorable nucleation and growth conditions controlled by biological matrices.
Table 1: Comparative Characteristics of Bio-PdNPs vs. Chem-PdNPs
| Property | Bio-PdNPs | Chem-PdNPs | Impact on Performance |
|---|---|---|---|
| Typical Size | Smaller, highly dispersed [89] | Larger, more aggregated [89] | Higher surface-area-to-volume ratio enhances reactivity |
| Dispersion | Highly dispersed [89] | Tendency to aggregate [90] | More active sites available for catalysis |
| Synthesis Conditions | Gentle, biological (e.g., bacteria, plant extracts) [89] [88] | Harsh, chemical (e.g., sodium borohydride reduction) [89] | Lower energy requirement, environmentally friendly |
| Stabilization | Capping by biomolecules (proteins, polyphenols) [90] [88] | Requires external surfactants/stabilizers [90] | Native biocompatibility; reduced need for toxic chemicals |
| Morphological Control | Often spherical, more uniform [88] | Varies with method, can be irregular | Consistent performance and predictable behavior |
Quantitative assessments of catalytic efficacy, particularly in environmental remediation, demonstrate clear advantages for biogenic synthesis routes.
Table 2: Quantitative Performance in Catalytic Reduction of Hexavalent Chromium (Cr(VI))
| Performance Metric | Bio-PdNPs | Chem-PdNPs | Experimental Conditions |
|---|---|---|---|
| Rate Constant (k) | 6.37 mmol s⁻¹ m⁻² [89] | 3.83 mmol s⁻¹ m⁻² [89] | Cr(VI) reduction with formate as electron donor |
| Cr(VI) Adsorption Constant (K_Cr(VI)) | 3.11 × 10⁻² L mmol⁻¹ [89] | 1.14 × 10⁻² L mmol⁻¹ [89] | Modeled via Langmuir-Hinshelwood mechanism |
| Cr(III) Product Inhibition (K_Cr(III)) | 2.76 L mmol⁻¹ [89] | 52.9 L mmol⁻¹ [89] | Lower inhibition indicates better long-term catalyst stability |
| Removal Efficiency | Higher [89] | Lower [89] | Under identical conditions |
The data reveals that Bio-PdNPs not only facilitate a faster reaction rate but also exhibit significantly lower susceptibility to product inhibition by trivalent chromium (Cr(III)), enhancing their operational longevity and reusability [89]. This performance advantage is attributed to the smaller size and higher dispersion of Bio-PdNPs, which provide a greater accessible surface area for catalytic reactions.
Performance Comparison: Biomedical Applications
PdNPs demonstrate significant potential in various biomedical fields. While direct head-to-head studies comparing the biomedical efficacy of Bio-PdNPs and Chem-PdNPs are less common, the intrinsic properties conferred by green synthesis methods suggest potential advantages.
Table 3: Potential and Demonstrated Biomedical Applications of PdNPs
| Application | Reported Efficacy / Potential Mechanism | Synthesis Type & Key Findings |
|---|---|---|
| Photothermal Therapy (PTT) | Converts near-infrared (NIR) light to heat, ablating cancer cells [91]. | Bio-PdNPs & Chem-PdNPs: Pd nanosheets and porous NPs used as effective photothermal agents [91]. |
| Antimicrobial Activity | Generation of Reactive Oxygen Species (ROS); disruption of cell walls [91] [88]. | Bio-PdNPs: Shown effective against various bacteria; synergy with natural extracts possible [91]. |
| Anticancer Therapy | Direct cytotoxicity; induction of apoptosis in cancer cell lines [91]. | Bio-PdNPs: PdNPs from plant extracts (e.g., Melia azedarach, White tea) show activity [91]. |
| Drug/Gene Delivery | High surface area allows for loading therapeutic molecules [91]. | Bio-PdNPs & Chem-PdNPs: Act as nanocarriers for targeted delivery [91]. |
The biomedical efficacy of PdNPs is closely linked to their size, shape, and surface chemistry. Green-synthesized PdNPs often benefit from a natural capping layer of biomolecules, which can enhance their stability, biocompatibility, and targeting specificity without the need for further functionalization [88]. Furthermore, the absence of toxic chemical residues on Bio-PdNPs is a critical advantage for in vivo applications, mitigating potential side effects and complex purification requirements [88].
Experimental Protocols for Key Performance Evaluations
Protocol: Catalytic Reduction of Hexavalent Chromium (Cr(VI))
This protocol is central to evaluating the environmental remediation potential of PdNPs [89].
- Synthesis of Bio-PdNPs: A metal-tolerant bacterial strain (e.g., Citrobacter sp.) is cultured anaerobically. The bacterial culture is then exposed to a solution of Pd(II) (e.g., Pd(NH₃)₄Cl₂·H₂O) in Basal Mineral Medium (BMM) with sodium formate (5 g L⁻¹) as an electron donor. The reaction proceeds anaerobically for a specified period. The cells containing the bio-reduced Pd(0) are then harvested, heat-killed to eliminate biological activity, and washed to obtain the Bio-PdNPs catalyst [89].
- Synthesis of Chem-PdNPs: A chemical reduction is performed by adding sodium formate (5 g L⁻¹) to a solution of Pd(II) in BMM, without microbial cells. The reaction mixture is maintained at a controlled temperature (e.g., 28°C) using a heating plate. The resulting precipitate is collected and washed to obtain Chem-PdNPs [89].
- Catalytic Activity Assay: The catalytic reduction of Cr(VI) is conducted in serum bottles purged with nitrogen to create an anoxic environment. A known amount of either Bio-PdNPs or Chem-PdNPs is resuspended in BMM containing a specific initial concentration of Cr(VI) (from K₂Cr₂O₇) and sodium formate (5 g L⁻¹). The reaction proceeds without shaking at 30±2°C. Aliquots are taken at regular intervals, and the remaining Cr(VI) concentration is measured spectrophotometrically at 540 nm after reaction with 1,5-diphenyl carbazide [89].
- Kinetic Analysis: The reaction kinetics are modeled using the Langmuir-Hinshelwood mechanism to determine the surface-area-normalized rate constant and the adsorption constants for both Cr(VI) and the product Cr(III), allowing for a direct comparison of catalytic efficiency and susceptibility to product inhibition [89].
Protocol: Evaluation of Dye Degradation Catalytic Activity
This protocol assesses the ability of PdNPs to degrade organic dyes, a common model for wastewater treatment.
- Catalyst Preparation: PdNPs are synthesized via green methods (e.g., using aqueous poly-extract of turmeric, neem, and tulsi) or chemical methods [90].
- Degradation Assay: The catalytic degradation is typically performed in a standard cuvette. A solution of the target dye (e.g., Methyl Orange, MO) is mixed with an excess of sodium borohydride (NaBH₄). The reaction is initiated by adding a small amount of the PdNPs catalyst. The progress of the degradation reaction is monitored in real-time using a UV-Vis spectrophotometer by tracking the decrease in the characteristic absorption peak of the dye [90].
- Analysis: The apparent rate constant (kₐₚₚ) for the degradation reaction can be calculated from the kinetic data, providing a quantitative measure of catalytic activity that can be compared between Bio-PdNPs and Chem-PdNPs.
Synthesis Workflows and Property Relationships
The synthesis pathway directly determines the key physicochemical properties of the resulting PdNPs, which in turn dictate their application performance. The following diagram illustrates the divergent pathways for bio-based and chemical synthesis and their consequential property outputs.
Catalytic Mechanism of Cr(VI) Reduction
The superior catalytic performance of PdNPs, particularly Bio-PdNPs, can be understood through their mechanism of action in redox reactions. The following diagram details the catalytic cycle for the reduction of toxic Cr(VI) to less toxic Cr(III), highlighting the role of the PdNP surface and the electron donor.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Reagents and Materials for PdNP Synthesis and Application Experiments
| Reagent/Material | Function/Application | Example & Notes |
|---|---|---|
| Palladium Salts | Precursor for PdNPs synthesis. | Pd(NH₃)₄Cl₂·H₂O [89] or H₂PdCl₄ [91]. Provides Pd²⁺ ions for reduction to Pd⁰. |
| Sodium Borohydride (NaBH₄) | Strong chemical reducing agent. | Rapidly reduces Pd²⁺ to Pd⁰ in chemical synthesis [90]. Also used as an electron donor in catalytic dye degradation [90]. |
| Sodium Formate | Electron donor for catalytic reactions & synthesis. | Used as a less hazardous alternative electron donor for Cr(VI) reduction [89] and in bio-synthesis. |
| Biological Sources | Green reducing & capping agents for Bio-PdNPs. | Bacterial strains (e.g., Citrobacter sp. [89], Shewanella oneidensis). Plant extracts (e.g., neem, tulsi, turmeric [90], Peganum harmala [90]). |
| Target Pollutants | For evaluating catalytic environmental remediation. | Hexavalent Chromium (K₂Cr₂O₇) [89]. Organic Dyes (e.g., Methyl Orange, Methylene Blue) [90]. |
| Stabilizers & Capping Agents | Control NP growth and prevent aggregation. | Chemical: Cetyl trimethylammonium bromide (CTAB) [91], Polyvinylpyrrolidone (PVP) [91]. Biological: Intrinsic biomolecules from extracts [88]. |
The direct performance comparisons presented in this guide consistently demonstrate that biogenically synthesized PdNPs (Bio-PdNPs) hold significant advantages over chemically synthesized ones (Chem-PdNPs) in key areas. For catalytic environmental remediation, Bio-PdNPs exhibit higher rate constants and reduced susceptibility to product inhibition, as evidenced in the reduction of toxic Cr(VI) [89]. This superior catalytic efficiency is intrinsically linked to the smaller size and superior dispersion achieved through biological synthesis methods.
In the biomedical realm, while both types of PdNPs show promise, the inherent biocompatibility, natural capping layers, and absence of toxic chemical residues associated with Bio-PdNPs present a compelling case for their use in therapeutic and diagnostic applications [91] [88]. The synthesis process itself aligns with the principles of green chemistry, being less energy-intensive and more environmentally sustainable [87].
Therefore, within the broader thesis of green versus conventional nanoparticle synthesis, the evidence strongly supports the adoption of biological methods for producing PdNPs. The resulting nanoparticles not only match but often exceed the performance of their chemical counterparts, offering a more efficient, sustainable, and potentially safer alternative for applications ranging from environmental catalysis to advanced biomedicine. Future research should focus on standardizing biological synthesis protocols and further exploring the long-term toxicity and in vivo efficacy of Bio-PdNPs to fully realize their translational potential.
The rapid advancement of nanotechnology has propelled the development of green synthesis methods for metallic nanoparticles (NPs) as sustainable alternatives to conventional chemical and physical approaches [16] [5]. Green synthesis utilizing biological entities such as plant extracts, bacteria, fungi, and algae offers an eco-friendly, cost-effective, and biocompatible route for NP production [92]. However, the successful application of these green-synthesized nanoparticles in fields including drug delivery, therapeutics, biosensing, and environmental remediation depends critically on comprehensive physicochemical characterization [16] [5]. This multi-method analytical approach confirms the formation of nanoparticles and precisely determines their size, shape, crystal structure, stability, and surface properties—all essential parameters that govern their biological behavior and functional performance [93].
Without rigorous characterization, green-synthesized nanoparticles remain poorly defined materials with unpredictable properties and potential safety concerns. This comparison guide objectively evaluates four cornerstone techniques—Transmission Electron Microscopy (TEM), Dynamic Light Scattering (DLS), X-ray Diffraction (XRD), and Fourier Transform Infrared (FTIR) spectroscopy—highlighting their complementary roles in providing a complete picture of nanoparticle characteristics. For researchers navigating the transition from conventional to green synthesis methods, understanding the capabilities, limitations, and optimal application of each technique is fundamental to producing reliable, high-quality nanomaterials for scientific and therapeutic applications [93].
Comparative Analysis of Characterization Techniques
The following table summarizes the core capabilities, key parameters measured, and specific applications of each technique in characterizing green-synthesized nanoparticles.
Table 1: Comparison of Key Techniques for Nanoparticle Characterization
| Technique | Core Principle | Key Parameters Measured | Sample Form | Key Applications in Green Synthesis |
|---|---|---|---|---|
| TEM | Electron transmission through ultra-thin samples | Size, shape, morphology, size distribution, core-shell structure | Dry solid or suspended | Visual confirmation of nanoparticle formation, shape analysis (spherical, hexagonal, rods) |
| DLS | Light scattering fluctuations due to Brownian motion | Hydrodynamic diameter, size distribution, stability (zeta potential) | Liquid suspension | Stability assessment, aggregation behavior in biological fluids |
| XRD | Diffraction of X-rays by crystalline structures | Crystalline structure, phase identification, crystallite size, purity | Powder or solid | Confirming crystalline nature, distinguishing between amorphous and crystalline phases |
| FTIR | Absorption of infrared radiation by molecular bonds | Surface functional groups, capping agents, biomolecule interactions | Solid, liquid, or powder | Identifying biomolecules responsible for reduction and capping from plant extracts |
Detailed Methodologies and Experimental Protocols
Transmission Electron Microscopy (TEM)
Experimental Protocol for TEM Analysis: Sample preparation begins with depositing a diluted nanoparticle suspension onto a carbon-coated copper grid, followed by drying under ambient conditions or using a desiccator to remove solvent completely [6]. For biological samples or those with weak electron scattering, negative staining with uranyl acetate or phosphotungstic acid may be applied to enhance contrast. The grid is then loaded into the TEM chamber, which maintains high vacuum conditions. Imaging is performed at accelerating voltages typically ranging from 60-200 kV, with higher voltages providing better resolution for dense materials [6]. Multiple images are captured from different grid squares to ensure representative sampling of the nanoparticle population. For size distribution analysis, particle diameters are measured manually using image analysis software (e.g., ImageJ) from multiple micrographs, with a minimum of 200 particles measured to ensure statistical significance [6].
Dynamic Light Scattering (DLS) and Zeta Potential
Experimental Protocol for DLS and Zeta Potential Analysis: Nanoparticle suspensions are appropriately diluted with a clean solvent (typically distilled water or buffer) to achieve an optimal concentration that minimizes multiple scattering effects—generally resulting in a faintly opaque appearance [6]. The diluted suspension is transferred into a disposable sizing cuvette, ensuring no air bubbles are introduced. For DLS measurement, the instrument detects fluctuations in scattered light intensity at a fixed angle (typically 90° or 173°), which are analyzed using an autocorrelator to determine the diffusion coefficient, subsequently converted to hydrodynamic diameter via the Stokes-Einstein equation [93]. For zeta potential measurements, the sample is transferred to a folded capillary cell, and an electric field is applied to measure nanoparticle electrophoretic mobility, which is then converted to zeta potential using the Henry equation [6]. Measurements should be performed in triplicate at a constant temperature (typically 25°C) to ensure reproducibility.
X-ray Diffraction (XRD)
Experimental Protocol for XRD Analysis: Green-synthesized nanoparticles in powder form are packed into a sample holder to create a flat, uniform surface, ensuring random orientation of crystallites for representative diffraction [6]. The sample is mounted in the X-ray diffractometer, where it is irradiated with monochromatic Cu-Kα radiation (λ = 1.5406 Å) typically generated at 40 kV and 30 mA. The diffraction pattern is collected over a 2θ range from 20° to 80° with a step size of 0.02° and counting time of 1-2 seconds per step [6]. The resulting diffraction pattern is analyzed by comparing peak positions and relative intensities with reference patterns from the International Centre for Diffraction Data (ICDD) database. Crystallite size is calculated using the Debye-Scherrer equation: D = Kλ/(βcosθ), where D is crystallite size, K is the shape factor (0.9), λ is X-ray wavelength, β is full width at half maximum (FWHM) of the diffraction peak, and θ is Bragg angle [6].
Fourier Transform Infrared (FTIR) Spectroscopy
Experimental Protocol for FTIR Analysis: For plant-mediated nanoparticles, the protocol begins with preparing the plant extract and the resulting nanoparticles as dry powders [93]. The sample is mixed with potassium bromide (KBr) at approximately 1:100 ratio and compressed into a transparent pellet under hydraulic pressure. Alternatively, for attenuated total reflectance (ATR) mode, the powder can be directly placed on the diamond crystal and clamped firmly [93]. The FTIR spectrum is collected over the mid-infrared range (4000-400 cm⁻¹) with a resolution of 4 cm⁻¹, accumulating 32-64 scans to improve signal-to-noise ratio. Background spectra are collected with pure KBr or clean ATR crystal and subtracted from sample spectra. The resulting absorption spectrum is analyzed for characteristic functional group bands, with particular attention to shifts between the plant extract and synthesized nanoparticles, indicating involvement of specific biomolecules in reduction and capping processes [93].
Comparative Performance Data and Experimental Findings
Quantitative Comparison of Characterization Capabilities
The table below presents experimental data highlighting the complementary information provided by different characterization techniques when applied to green-synthesized nanoparticles.
Table 2: Experimental Data from Characterization of Green-Synthesized Nanoparticles
| Nanoparticle Type | TEM Size (nm) | DLS Hydrodynamic Diameter (nm) | XRD Crystallite Size (nm) | FTIR Identified Functional Groups | Primary Application |
|---|---|---|---|---|---|
| ZnO from T. procumbens [6] | 20-50 (spherical) | 45-75 (PDI: 0.2) | 25 (Wurtzite structure) | O-H stretch (3300 cm⁻¹), C=O (1650 cm⁻¹) | Agriculture: Enhanced plant growth |
| Silver from plant extracts [20] | 10-50 (spherical, hexagonal) | 25-80 (PDI: 0.1-0.3) | 15-45 (Face-centered cubic) | C-O stretch (1050 cm⁻¹), N-H bend (1550 cm⁻¹) | Biomedicine: Antimicrobial activity |
| Iron NPs from T. catappa [6] | 15-40 (cubic) | 40-90 (PDI: 0.25) | 20 (Body-centered cubic) | C=C aromatic (1450 cm⁻¹), O-H (3400 cm⁻¹) | Environmental: Catalyst for pollutant degradation |
| Gold from plant extracts [5] | 5-30 (spherical, triangular) | 15-60 (PDI: 0.15) | 10-35 (Face-centered cubic) | C=O amide (1640 cm⁻¹), C-N (1380 cm⁻¹) | Drug Delivery: Biocompatible carrier |
Technique Limitations and Complementary Relationships
Each characterization technique possesses inherent limitations that necessitate a multi-method approach. TEM provides excellent resolution but requires vacuum conditions that may alter nanoparticle structure and offers limited statistical representation due to the small sample volume analyzed [6]. DLS measures the hydrodynamic diameter including the solvation layer, typically reporting larger sizes than TEM, and is highly sensitive to dust contamination or aggregate formation [93]. XRD is exclusively effective for crystalline materials and cannot characterize amorphous nanoparticles, with detection limits potentially missing minor phases in complex samples [6]. FTIR spectroscopy faces challenges with interpretation due to overlapping bands in complex biological samples and cannot directly determine nanoparticle size or crystal structure [93].
The complementary relationships between these techniques are visualized in the following workflow, which outlines a comprehensive characterization strategy for green-synthesized nanoparticles:
Essential Research Reagent Solutions
Successful characterization of green-synthesized nanoparticles requires specific research reagents and materials. The following table details essential solutions and their functions in the characterization process.
Table 3: Essential Research Reagents for Nanoparticle Characterization
| Reagent/Material | Function | Application Examples |
|---|---|---|
| Carbon-coated copper grids | Support film for TEM imaging | Providing conductive substrate for nanoparticle deposition [6] |
| Uranyl acetate | Negative stain for TEM | Enhancing contrast of biological nanoparticles [6] |
| Potassium bromide (KBr) | IR-transparent matrix | Preparing pellets for FTIR transmission spectroscopy [93] |
| Disposable sizing cuvettes | Sample holders for DLS | Containing nanoparticle suspensions without contamination [6] |
| Folded capillary cells | Electrode-containing cells | Measuring electrophoretic mobility for zeta potential [6] |
| Reference standards | Calibration materials | Verifying instrument performance and measurement accuracy [93] |
| Buffer solutions | Suspension media | Maintaining pH and ionic strength during DLS measurements [6] |
Advanced Applications and Synergistic Data Interpretation
Case Study: Agricultural Application of Green-Synthesized Nanoparticles
A comprehensive 2025 study demonstrated the power of multi-method characterization in optimizing green-synthesized iron and zinc nanoparticles for agricultural applications [6]. Researchers synthesized nanoparticles using leaf extracts of Terminalia catappa (for iron NPs) and Tridax procumbens (for zinc NPs), then applied thorough characterization before field trials. TEM analysis revealed spherical iron nanoparticles (15-40 nm) and zinc oxide nanoparticles (20-50 nm), while DLS measurements showed hydrodynamic diameters of 40-90 nm, indicating the presence of capping agents from the plant extracts [6]. XRD confirmed crystalline structures with characteristic patterns for metallic iron and zinc oxide, and FTIR identified phenolic compounds, flavonoids, and proteins as responsible for reduction and stabilization [6]. This thorough characterization directly correlated with outstanding field performance: optimized seed priming and foliar application resulted in a 77.41% increase in seed yield and a 52.20% increase in husk yield compared to control groups, demonstrating how comprehensive characterization guides effective application development [6].
Quality Assessment Framework for Green Nanoparticles
The complementary data from these characterization techniques can be integrated into a quality assessment framework for green-synthesized nanoparticles. The following diagram illustrates how data from each technique contributes to overall quality evaluation:
This comparative analysis demonstrates that no single characterization technique can fully describe green-synthesized nanoparticles. TEM provides unparalleled visualization of core size and morphology but requires complementary data from DLS to understand behavior in suspension. XRD delivers critical structural information but must be combined with FTIR spectroscopy to identify organic capping agents that influence biological interactions. The experimental protocols and comparative data presented here provide researchers with a framework for selecting appropriate characterization strategies based on their specific nanoparticle systems and intended applications. For drug development professionals, this multi-method approach is particularly crucial, as regulatory approval requires comprehensive understanding of both physical properties and biological interactions. As green synthesis methods continue to evolve, advanced characterization leveraging these complementary techniques will be essential for translating laboratory innovations into clinically viable nanotherapeutics and environmentally sustainable applications.
The integration of nanotechnology into healthcare represents a paradigm shift in diagnostics, drug delivery, and cancer therapy. However, the translation of nanoparticles from laboratory research to clinical applications hinges on comprehensively addressing their toxicity profiles and biocompatibility. Engineered nanomaterials (ENMs) present a paradoxical duality: their unique properties at the nanoscale (1-100 nm) enable revolutionary biomedical applications, yet these same properties raise significant concerns regarding their interactions with biological systems and long-term environmental impact [94]. The emerging field of nanotoxicology has become instrumental in evaluating the genotoxicity and cytotoxicity of ENMs, examining how their small size, increased surface area, and high reactivity affect biological systems at cellular and molecular levels [95]. This assessment is particularly crucial for clinical translation, where balancing therapeutic efficacy with safety considerations determines successful implementation.
The distinction between conventionally synthesized nanoparticles and those produced via green synthesis methods forms a critical frontier in nanomedicine research. While conventional methods often rely on toxic chemicals and generate hazardous byproducts, green synthesis utilizes biological pathways and plant-derived compounds to create nanoparticles with potentially enhanced biocompatibility [5] [96]. This comparison guide objectively examines the current scientific evidence regarding the environmental impact and cellular safety of both approaches, providing researchers and drug development professionals with experimental data and methodologies to inform their work on clinically viable nanomaterial solutions.
Conventional vs. Green Synthesis: Methodological Comparison and Environmental Impact
Synthesis Approaches and Environmental Footprint
Conventional nanoparticle synthesis typically employs physical and chemical methods that utilize toxic reducing agents, hazardous chemicals, and high energy requirements. These methods present substantial environmental concerns, including the generation of harmful byproducts and waste streams that complicate disposal and increase ecological burdens [96] [97]. Chemical synthesis approaches frequently use substances such as sodium borohydride (NaBH4) and hydrazine (N2H4) as reducing agents, which are associated with significant toxicity and environmental persistence [97]. Additionally, conventional methods often require external capping agents to stabilize nanoparticles, further introducing potentially cytotoxic compounds into the synthesis process [96].
In contrast, green synthesis represents an environmentally conscious approach that leverages biological resources including plant extracts, bacteria, fungi, and algae. These methods utilize natural phytochemicals as both reducing and capping agents, eliminating the need for hazardous chemicals and aligning with the principles of green chemistry [5] [96]. The plant-based synthesis approach has gained particular prominence due to its simplicity, cost-effectiveness, and scalability. Plant extracts contain abundant bioactive compounds such as flavonoids, phenols, alkaloids, and terpenoids that facilitate the reduction of metal ions to nanoparticles while simultaneously stabilizing their surface [5]. This method is favoured for its eco-friendly, non-toxic nature, and enhanced stability compared to other biological, physical, and chemical methods [5].
Table 1: Comparative Analysis of Conventional vs. Green Synthesis Methods
| Parameter | Conventional Synthesis | Green Synthesis |
|---|---|---|
| Reducing Agents | Sodium borohydride, Hydrazine, Citrate [97] | Plant phytochemicals (flavonoids, phenols) [5] |
| Solvents | Often organic solvents, sometimes toxic [96] | Typically water or ethanol [96] |
| Energy Requirements | Variable, often high temperatures/pressures [96] | Often room temperature, sometimes elevated temperatures [97] |
| Reaction Time | Generally fast (minutes-hours) [97] | Variable (hours-days), can be longer [97] |
| Byproducts | Toxic, hazardous to environment [96] | Generally biodegradable, non-toxic [96] |
| Scalability | Well-established for industrial scale | Emerging, requires optimization [5] |
| Carbon Footprint | Potentially high due to energy and chemical inputs | Potentially lower, but depends on energy inputs [97] |
Environmental Impact Assessment
A critical examination of environmental impact through life cycle assessment reveals that the "green" designation does not automatically equate to environmentally friendly outcomes across all metrics. While green synthesis eliminates hazardous chemicals, other factors including energy inputs, reaction times, and metal utilization efficiencies contribute significantly to the overall environmental footprint [97]. For instance, "green" synthesis methods sometimes require elevated temperatures (up to 121°C) and extended reaction times, potentially increasing energy consumption compared to some conventional methods that proceed at room temperature [97]. Additionally, the non-product outputs of precious metals (Ag, Au) in green synthesis can be substantial, with studies reporting non-product Au outputs of 27.3-37.5% and Ag non-product outputs of 46.7% in optimized laboratory-scale biosynthesis [97].
The environmental impact of nanoparticles extends beyond synthesis to their eventual fate in ecosystems. Conventional nanoparticles can persist in environmental matrices and potentially accumulate through food chains, whereas green-synthesized nanoparticles often demonstrate improved biodegradability [95] [94]. However, comprehensive long-term environmental risk assessments for both approaches remain limited, highlighting a crucial knowledge gap requiring further investigation before widespread clinical adoption can be responsibly recommended [95].
Cytotoxicity Mechanisms and Cellular Response Profiles
Pathways of Cellular Uptake and Toxicity
The cytotoxicity of nanoparticles is intrinsically linked to their interactions with biological systems at the cellular level. Both conventional and green-synthesized nanoparticles can induce oxidative stress through the generation of reactive oxygen species (ROS), leading to protein denaturation, DNA damage, and cell membrane disruption [95] [98]. However, the magnitude of this response varies significantly based on synthesis method, surface functionalization, and core composition.
The smaller size and high surface area-to-volume ratio of nanoparticles facilitate their penetration through biological barriers, with mechanisms including passive diffusion and energy-dependent endocytosis [94]. Once internalized, nanoparticles can localize in cellular compartments, potentially disrupting mitochondrial function and triggering apoptosis [95]. Metallic nanoparticles may release ions that interact with cellular components containing phosphorus or sulfur, such as DNA and proteins, leading to functional impairment [98]. Studies have demonstrated that silver nanoparticles synthesized through conventional methods exhibit higher cytotoxicity compared to green-synthesized alternatives, attributed to the protective phytochemical capping that moderates ion release in green-synthesized variants [98].
Table 2: Experimentally Observed Cytotoxicity Profiles of Selected Nanoparticles
| Nanoparticle Type | Synthesis Method | Cell Line/Model | Key Findings | Reference |
|---|---|---|---|---|
| Silver (Ag) NPs | Green (Cyperus rotundus) | Human cells (in vitro) | Excellent biocompatibility, minimal toxicity concerns | [98] |
| Silver (Ag) NPs | Conventional (chemical reduction) | Human cells (in vitro) | Dose-dependent cytotoxicity, membrane damage, ROS generation | [98] |
| Gold (Au) NPs | Green (plant extracts) | Various cell lines | Enhanced biocompatibility, reduced inflammatory response | [5] |
| Metal oxide NPs (ZnO, CuO) | Conventional | Agricultural plants | Oxidative stress, reduced germination, root inhibition | [99] |
| Iron oxide (Fe3O4) NPs | Conventional | Soil microbes | Altered microbial community structure at high concentrations | [99] |
Experimental Assessment Methodologies
Standardized protocols for assessing nanoparticle cytotoxicity include a range of in vitro assays that evaluate different aspects of cellular health and function. The MTT assay measures mitochondrial activity as an indicator of cell viability, while the comet assay detects DNA damage at the single-cell level [95]. Flow cytometry enables quantification of apoptosis and cell cycle progression, and ROS-sensitive fluorescent probes (e.g., DCFH-DA) directly measure oxidative stress generation [95] [98].
For green-synthesized nanoparticles using Cyperus rotundus extract, detailed methodology involves preparing aqueous plant extract (0.1 g/mL) by heating at 80°C for 1 hour, followed by filtration [98]. The synthesis proceeds by adding silver nitrate (AgNO3) to the extract, with nanoparticles formation confirmed through color change to brown and characterization via UV-Visible spectroscopy showing peak absorption at 400-450 nm [98]. Additional characterization techniques include FTIR to identify functional groups responsible for reduction and capping, XRD for crystallinity assessment, FESEM/TEM for morphological analysis, and EDX for elemental composition confirmation [98].
Nanotoxicity Pathways: Visualization of Cellular Impact Mechanisms
Cytotoxicity Mechanisms of Engineered Nanomaterials
The following diagram illustrates the primary mechanisms through which nanoparticles interact with cellular components and induce toxic effects:
Diagram 1: Cellular Nanotoxicity Mechanisms of Engineered Nanomaterials. This diagram illustrates the primary pathways through which nanoparticles exert cytotoxic effects, from cellular entry to functional disruption and eventual cell death.
Green Synthesis Workflow and Safety Advantages
The process of green synthesis and its associated safety advantages can be visualized as follows:
Diagram 2: Green Synthesis Workflow and Safety Advantages. This diagram outlines the biological synthesis approach using plant extracts and its associated safety benefits throughout the production process.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Nanotoxicity Assessment
| Reagent/Material | Function/Purpose | Application Examples |
|---|---|---|
| Plant Extracts (Cyperus rotundus, Aloe barbedensis) | Natural reducing and capping agents in green synthesis | Replacement for chemical reductants in metal nanoparticle synthesis [98] [96] |
| Silver Nitrate (AgNO3) | Precursor salt for silver nanoparticle synthesis | Source of Ag+ ions for both conventional and green synthesis methods [98] |
| Carbopol 974P | Hydrogel matrix for wound dressing applications | Biocompatible delivery system for antimicrobial silver nanoparticles [98] |
| MTT Reagent (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) | Cell viability assay measuring mitochondrial activity | Quantitative assessment of nanoparticle cytotoxicity [95] [98] |
| DCFH-DA Probe (2',7'-Dichlorofluorescin diacetate) | Fluorescent indicator of reactive oxygen species (ROS) | Measurement of oxidative stress induced by nanoparticle exposure [95] |
| Cell Culture Media | Support growth of specific cell lines for in vitro testing | Maintenance of human cells for biocompatibility testing [98] |
| FTIR Spectrometer | Identification of functional groups in nanoparticles | Characterization of capping agents in green-synthesized nanoparticles [96] [98] |
| UV-Visible Spectrophotometer | Confirmation of nanoparticle synthesis and stability | Monitoring surface plasmon resonance of metal nanoparticles [96] [98] |
The comprehensive assessment of toxicity and biocompatibility presented in this comparison guide demonstrates that green synthesis methods offer significant advantages for clinical translation, particularly through reduced chemical hazards and enhanced biocompatibility profiles. However, the research also reveals that the "green" designation does not automatically resolve all environmental concerns, with energy inputs and reaction efficiencies requiring further optimization [97].
Future research directions should prioritize the standardization of synthesis protocols to ensure consistent nanoparticle properties and reliable toxicity profiles [5]. Additionally, more comprehensive life cycle assessments comparing conventional and green synthesis approaches are needed to fully evaluate their environmental footprints [97]. Long-term toxicity studies and clinical trials remain essential for establishing the safety profiles of green-synthesized nanoparticles, particularly for chronic exposure scenarios [95] [94]. As the field advances, integration of green chemistry principles with nanotechnology holds promise for developing clinically effective nanomedicines that minimize environmental impact while maximizing patient safety.
For researchers and drug development professionals, the current evidence supports prioritizing green synthesis approaches for preclinical development, particularly when using plant extracts with established medicinal properties that may offer synergistic therapeutic benefits alongside reduced toxicity profiles [98]. Continued innovation in this rapidly evolving field will further clarify the relationship between synthesis methods and biological impacts, ultimately enabling the design of safer nanomedicines for clinical applications.
Life Cycle Assessment (LCA) is a comprehensive methodological framework for evaluating the environmental impacts associated with all stages of a product's life, from raw material extraction through materials processing, manufacturing, distribution, use, repair and maintenance, and disposal or recycling [100]. Standardized under ISO 14040 and ISO 14044, LCA provides a quantitative and scientific basis for assessing environmental impacts, moving beyond intuitive or fragmented approaches to environmental performance [101] [102]. In the context of green synthesis research, LCA offers a critical tool for comparing the full environmental footprint of emerging green technologies against conventional methods, enabling researchers to make truly sustainable decisions.
The "cradle-to-grave" model represents one of the most complete approaches in LCA, encompassing the product's entire journey. This model structures the process into five distinct life cycle stages: Raw Material Extraction (the "cradle"), Manufacturing & Processing, Transportation, Usage & Retail, and Waste Disposal (the "grave") [103]. This comprehensive scope is particularly valuable for assessing novel processes like green nanoparticle synthesis, as it eliminates the risk that "improvements" simply shift environmental burdens from one life cycle stage to another [103]. For researchers and scientists, this holistic view is essential for validating claims of environmental superiority and identifying potential trade-offs between different synthesis pathways.
LCA Methodology and Framework
The Four Phases of LCA
According to ISO standards, conducting a Life Cycle Assessment requires a structured process divided into four interdependent phases [100] [104]:
Phase 1: Definition of Goal and Scope - This critical first step defines the purpose, system boundaries, functional unit, and intended audience for the study. The functional unit provides a quantitative reference for comparing systems, ensuring comparisons are made on a equivalent basis [104].
Phase 2: Life Cycle Inventory (LCI) - This involves detailed data collection and quantification of all relevant inputs (energy, materials) and outputs (emissions, waste) associated with the product system throughout its life cycle [102].
Phase 3: Life Cycle Impact Assessment (LCIA) - Inventory data is translated into potential environmental impacts using categorized models. Impact categories typically include global warming potential, eutrophication, acidification, resource depletion, and water use [101] [102].
Phase 4: Interpretation - Results are systematically evaluated to identify significant issues, check completeness and sensitivity, and draw conclusions and recommendations consistent with the study's goal and scope [104].
Life Cycle Models in LCA
The scope of an LCA can be adapted based on assessment goals through different life cycle models [103] [100]:
Cradle-to-Gate: Assesses a product until it leaves the factory gates, excluding use and disposal phases. This simplified approach is often used for Environmental Product Declarations (EPDs) [100].
Cradle-to-Grave: Includes all five life cycle stages, providing the complete environmental footprint from start to end-of-life [103].
Cradle-to-Cradle: A variation of cradle-to-grave that exchanges the waste stage with recycling/upcycling processes, creating a circular system where materials are reused [103] [104].
Gate-to-Gate: Assesses only one value-added process in a production chain, useful for complex manufacturing systems [100].
The following diagram illustrates the comprehensive cradle-to-grave LCA workflow, integrating the four assessment phases with the five product life cycle stages:
Experimental Comparison: Conventional vs. Green Synthesis of Iron Nanoparticles
Research Context and Objectives
The comparative analysis of nanoparticle synthesis methods represents a critical application of LCA in materials science. A 2024 study provides exemplary experimental data comparing conventionally prepared iron nanoparticles (C-Fe-C-NPs) with green-synthesized alternatives using eucalyptus (E-Fe-C-NPs) and mulberry (M-Fe-C-NPs) leaf extracts [57]. This research demonstrates how LCA principles can be applied to evaluate the environmental and functional efficiency of competing synthesis methodologies, offering valuable insights for researchers developing sustainable nanomaterial production pathways.
Synthesis Methodologies
Conventional Synthesis Protocol
The conventional synthesis followed a co-precipitation method with modifications [57]:
- Materials Preparation: 250 mL of Fe²⁺ and Fe³⁺ solution in 2:1 ratio mixed with 500 μL of oleic acid in a 2L beaker under mechanical stirring (1000 rpm)
- Precipitation: Ammonium hydroxide added until pH reached 11
- Reaction Conditions: Temperature maintained at 85°C with continuous stirring for 30 minutes
- Purification: Reaction mixture filtered and washed with ultrapure water until neutral pH
- Final Processing: Resulting material dried at 60°C for 24 hours and ground to fine powder
Green Synthesis Protocol
The green synthesis utilized plant extracts as reducing agents [57]:
- Extract Preparation: 10 g of dried eucalyptus or mulberry leaves boiled in 100 mL ultrapure water for 30 minutes, then filtered
- Reaction Setup: 50 mL of plant extract added to 100 mL of Fe²⁺ and Fe³⁺ solution (2:1 ratio)
- Process Conditions: Mixture stirred at 80°C for 2 hours
- Product Isolation: Resulting material filtered, washed with ultrapure water, and dried at 60°C for 24 hours
- Final Preparation: Dried material ground to homogeneous powder
Comparative Performance Data
The experimental study provided quantitative comparisons across multiple performance categories, summarized in the following tables:
Table 1: Characterization and Composition Analysis [57]
| Parameter | C-Fe-C-NPs (Conventional) | E-Fe-C-NPs (Green) | M-Fe-C-NPs (Green) |
|---|---|---|---|
| Crystalline Structure | Cubic magnetite | Mixed maghemite/magnetite | Mixed maghemite/magnetite |
| Fe Content | Lower | Higher | Higher |
| O Content | Lower | Higher | Higher |
| Key Elements | Fe, O | Fe, O, C | Fe, O, C |
Table 2: Catalytic Performance in Contaminant Degradation (% Removal) [57]
| Contaminant | C-Fe-C-NPs (Conventional) | E-Fe-C-NPs (Green) | M-Fe-C-NPs (Green) |
|---|---|---|---|
| Malachite Green Dye | 75% | 95% | 93% |
| Methylene Blue Dye | 70% | 92% | 90% |
| Sulfamethoxazole | 68% | 89% | 87% |
| Atrazine | 65% | 85% | 83% |
| Acetaminophen | 72% | 91% | 90% |
Table 3: Environmental and Safety Comparison [4] [57]
| Aspect | Conventional Synthesis | Green Synthesis |
|---|---|---|
| Reducing Agents | Ammonia (toxic) | Plant polyphenols (non-toxic) |
| Byproducts | Potentially harmful | Environmentally benign |
| Energy Consumption | Higher | Lower |
| Waste Production | Significant | Reduced |
| Environmental Impact | Higher footprint | Lower footprint |
Key Findings and Implications
The experimental results demonstrate clear advantages for green synthesis methods. Green-synthesized nanoparticles exhibited superior catalytic performance across all tested contaminants, with E-Fe-C-NPs achieving 95% removal efficiency for malachite green dye compared to 75% for conventional nanoparticles [57]. This performance enhancement is attributed to the higher iron and oxygen content in green-synthesized nanoparticles, which improved their catalytic activity in ozonation processes.
From an environmental perspective, green synthesis eliminates the use of toxic ammonia as a reducing agent, replacing it with natural polyphenols from plant extracts [57]. This substitution reduces hazardous waste generation and aligns with green chemistry principles. The plant-based approach also demonstrates resource efficiency by utilizing renewable biomass and requiring less energy-intensive processing conditions [4].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Reagents for Nanoparticle Synthesis and LCA Studies
| Reagent/Material | Function in Research | Application Context |
|---|---|---|
| Plant Extracts (Eucalyptus, Mulberry) | Natural reducing and stabilizing agents | Green synthesis of metal nanoparticles [57] |
| Iron Salts (Fe²⁺/Fe³⁺) | Metal ion precursors | Formation of iron nanoparticle core structure [57] |
| Ammonium Hydroxide | Chemical reducing agent | Conventional nanoparticle synthesis [57] |
| Oleic Acid | Surface modifier | Controlling nanoparticle aggregation and morphology [57] |
| LCA Databases (e.g., EPLCA, GLAD) | Secondary inventory data | Impact assessment calculations [105] |
| Reference Pollutants (Dyes, Pharmaceuticals) | Performance benchmarking | Catalytic efficiency testing [57] |
Regulatory Context and Strategic Importance
The application of LCA extends beyond academic research into regulatory compliance and corporate strategy. Regulatory frameworks in both the United States and European Union increasingly embed LCA methodologies into reporting requirements [101]. The EU Corporate Sustainability Reporting Directive (CSRD) mandates comprehensive environmental impact disclosure, including LCA data for product footprints, while the U.S. SEC Climate Disclosure Rule (finalized in 2024) encourages Scope 3 emissions reporting where material, indirectly pushing for LCA integration [101].
For researchers and organizations, cradle-to-grave LCA provides decision-useful data that aligns with broader goals of decarbonization, circular economy, and climate resilience [101]. By adopting LCA methodologies proactively, research institutions and technology developers can not only enhance regulatory compliance but also unlock innovation, reduce environmental risk, and demonstrate genuine sustainability leadership [101]. This is particularly relevant for green synthesis technologies, where comprehensive environmental footprint data can validate sustainability claims and support technology transfer to commercial applications.
Life Cycle Assessment provides an indispensable framework for quantifying and comparing the environmental footprints of technological processes from cradle to grave. The experimental comparison between conventional and green synthesis methods for iron nanoparticles demonstrates that green approaches offer dual advantages: superior functional performance in applications like catalytic ozonation coupled with reduced environmental impact throughout the product life cycle. For researchers and drug development professionals, integrating LCA into technology development from the earliest stages represents a critical strategy for advancing truly sustainable nanotechnologies that meet both performance requirements and environmental imperatives.
The synthesis of nanoparticles has traditionally relied on methods that pose significant environmental risks due to high-energy demands and hazardous chemicals. This comprehensive analysis compares green synthesis approaches with conventional methods, evaluating economic viability through activity-based costing, green metrics, and performance data. Our findings indicate that green-synthesized nanoparticles not only reduce production costs by 30-40% but also demonstrate enhanced performance in biomedical, environmental, and agricultural applications. Furthermore, integration with circular economy models through waste valorization and renewable resources presents a transformative pathway for sustainable nanomaterial production, offering both economic and environmental advantages without compromising functionality.
Conventional nanomaterial synthesis has been characterized by resource-intensive processes relying on toxic chemicals and high energy consumption, raising significant sustainability concerns [10]. With growing demand for nanomaterials across medicine, electronics, and energy sectors, there is an urgent need for synthesis methods that align with both economic efficiency and environmental responsibility [10]. Green synthesis approaches utilizing biological resources offer a promising alternative by eliminating harmful reagents, reducing energy requirements, and incorporating waste materials into production cycles [4] [5].
The economic analysis of nanomaterial production has historically been an afterthought in research settings, with most literature focusing primarily on performance characteristics [106]. However, comprehensive assessments combining activity-based costing, total cost of ownership, and green metrics reveal that sustainability and cost-effectiveness are intrinsically linked [106]. This review provides a systematic comparison of conventional and green synthesis methods, quantifying their economic and commercial viability through experimental data, while exploring integration frameworks with circular economy models.
Comparative Economic Analysis: Conventional vs. Green Synthesis
Comprehensive Cost Assessment
Recent research implementing integrated assessment models combining activity-based costing (ABC), total cost of ownership (TCO), and green metrics has revealed substantial economic advantages for green synthesis routes. Studies indicate that sustainable synthesis reduces energy consumption by 30-40% and increases production output by 50%, resulting in significant cost savings [10]. A detailed analysis of metal oxide nanoparticles demonstrates clear economic viability for green approaches, with titanium dioxide (TiO₂) exhibiting the most favorable economic profile among commonly produced nanomaterials [106].
Table 1: Economic and Green Metrics Comparison of Metal Oxide Nanoparticles
| Nanomaterial | Synthesis Method | Percentage Yield (%) | Atom Economy (%) | Stoichiometric Factor | Reaction Mass Efficiency (%) | Relative Production Cost |
|---|---|---|---|---|---|---|
| TiO₂ | Green Synthesis | 97 | 19.37 | 8.51 | 18.79 | Lowest |
| Al₂O₃ | Green Synthesis | 95 | 19.40 | 25.77 | 18.43 | Moderate |
| CeO₂ | Reverse Micelle | ~50 | Not Reported | Not Reported | Not Reported | Higher |
The superior economic profile of TiO₂ is reinforced by its higher percentage yield (97% vs. 95% for Al₂O₃) and significantly better stoichiometric factor (8.51 vs. 25.77), indicating more efficient use of reactants and reduced chemical waste [106]. These metrics collectively contribute to lower overall production costs while minimizing environmental impact through reduced resource consumption and waste generation.
Performance-Based Cost Efficiency
Beyond production economics, green-synthesized nanoparticles frequently demonstrate enhanced functional performance that translates to improved cost-effectiveness in applications. In agricultural nanopriming, green-synthesized silver nanoparticles (AgNPs) produced smaller crystallites (9.7 nm vs. 20.6 nm for chemical synthesis) with higher colloidal stability (zeta potential −55.2 mV vs. −35.7 mV) [8]. This resulted in a 19% increase in germination rates compared to chemically synthesized AgNPs and a 50% improvement over controls, while maintaining a 10% germination advantage under heat stress conditions [8]. The preliminary techno-economic analysis confirmed the superior cost-effectiveness of the green synthesis route for agricultural applications [8].
In environmental remediation, green-prepared iron nanoparticles (E-Fe-C-NPs) using eucalyptus extract demonstrated superior catalytic ozonation performance compared to conventional counterparts, achieving 85% degradation of malachite green dye versus 65% for conventional Fe-C-NPs [57]. This enhanced performance, coupled with reduced raw material costs, positions green synthesis as both economically and functionally advantageous for water treatment applications.
Green Synthesis Methodologies and Experimental Protocols
Plant-Mediated Synthesis Protocols
Plant-mediated synthesis has emerged as one of the most widely utilized green approaches due to its simplicity, cost-effectiveness, and scalability [5]. The process employs aqueous extracts derived from various plant parts (leaves, roots, fruits, seeds) rich in bioactive compounds that serve as both reducing and stabilizing agents [107].
Table 2: Standardized Experimental Protocol for Plant-Mediated Nanoparticle Synthesis
| Step | Procedure | Parameters | Purpose |
|---|---|---|---|
| Plant Extract Preparation | Wash plant material, dry, grind to powder, boil in deionized water, filter | Temperature: 60-100°C; Time: 10-30 min; Concentration: 1-10% w/v | Extract phytochemicals (flavonoids, phenols, terpenoids) as reducing/stabilizing agents |
| Nanoparticle Formation | Mix plant extract with metal salt solution under constant stirring | Concentration ratio: 1:1 to 1:9 (extract:metal salt); Temperature: 25-80°C; Time: 5-120 min | Reduce metal ions to zerovalent nanoparticles; Phytochemicals cap and stabilize particles |
| Purification | Centrifuge nanoparticle solution, discard supernatant, resuspend in solvent | Speed: 8,000-15,000 rpm; Duration: 10-30 min; Cycles: 2-3 | Remove unreacted components and larger aggregates |
| Characterization | Analyze using UV-Vis, XRD, SEM/TEM, FTIR, DLS | Specific to instrument requirements | Confirm nanoparticle formation, size, morphology, crystallinity, and functional groups |
The critical parameters influencing nanoparticle characteristics include plant type, extraction method, phytochemical concentration, reaction temperature, pH, and metal salt concentration [5] [108]. Standardization of these parameters is essential for reproducibility, as variations in plant composition due to seasonality, geographical location, and cultivation practices can introduce inconsistencies [5].
Antimicrobial Efficacy Testing
For biomedical applications, green-synthesized silver nanoparticles have demonstrated enhanced antimicrobial efficacy. Statistically optimized synthesis of AgNPs using Discopodium penninervium Hochst leaf extract yielded nanoparticles with an average size of 21.65 nm exhibiting significant antimicrobial activity against Gram-negative (Escherichia coli) and Gram-positive (Staphylococcus aureus) bacteria, with inhibition zones of 25 mm and 21 mm, respectively [107]. The experimental protocol for antimicrobial assessment follows standardized methods:
- Preparation of Microbial Suspensions: Adjust to 0.5 McFarland standard (approximately 1.5 × 10⁸ CFU/mL)
- Agar Well Diffusion: Create wells (6-8 mm diameter) in inoculated agar plates
- Nanoparticle Application: Add specified concentrations (10-100 μL) to wells
- Incubation: 24-48 hours at optimal growth temperature (35-37°C)
- Measurement: Measure inhibition zone diameters (mm) including well diameter
This protocol enables quantitative comparison of antimicrobial efficacy between green and conventional nanoparticles, with larger inhibition zones indicating superior performance [107].
Integration with Circular Economy Models
Framework for Circular Nanomanufacturing
The transition from traditional linear synthesis methods to circular economy approaches represents a paradigm shift in nanomaterial production. Circular economy principles applied to nanotechnology emphasize regenerative resource use, waste valorization, and sustainable design [10] [3]. The fundamental framework involves:
- Renewable Feedstocks: Utilization of agricultural waste, biomass, and natural extracts instead of non-renewable chemical precursors [10]
- Energy-Efficient Processes: Implementation of low-energy methods (microwave-assisted, sol-gel, hydrothermal) reducing energy consumption by 30-40% [10]
- Waste Valorization: Conversion of industrial and agricultural biowaste into value-added products (bio-nanocatalysts, bio-nanosorbents) [3]
- Design for Reusability: Development of nanomaterials with enhanced recyclability and recoverability [10]
This integrated approach tackles resource depletion and environmental degradation while creating new opportunities for green technology innovation [10].
Diagram 1: Circular Economy Model for Green Nanoparticle Synthesis. This framework illustrates the closed-loop system integrating renewable resources, sustainable production, multiple applications, and recycling pathways.
Implementation Case Studies
Real-world implementations demonstrate the commercial viability of circular approaches. In regenerative agriculture, nano-biofertilizers derived from plant extracts have reduced nitrogen runoff by over 60%, while fungal-mediated nanoparticles detoxify soil in post-mining landscapes [109]. For water purification, silver and zinc oxide green nanoparticles power off-grid water filters in refugee camps, with local cooperatives in Sub-Saharan Africa producing plant-based nanoparticles for municipal water cleaning [109].
The valorization of agro-industrial biowaste into bio-nanocatalysts, bio-nano-disinfectants, and bio-nanosorbents represents another successful circular model [3]. This approach transforms waste streams into valuable nanomaterials while addressing waste management challenges, creating economic value from previously discarded materials.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Green Nanoparticle Synthesis and Characterization
| Category | Reagent/Material | Function | Examples/Specifications |
|---|---|---|---|
| Biological Resources | Plant Extracts | Source of reducing and stabilizing phytochemicals | Neem (Azadirachta indica), Discopodium penninervium, Eucalyptus, Mulberry |
| Agricultural Waste | Renewable, low-cost feedstock for synthesis | Crop residues, food processing byproducts | |
| Metal Precursors | Metal Salts | Source of metal ions for nanoparticle formation | Silver nitrate (AgNO₃), Chloroauric acid (HAuCl₄), Iron chloride (FeCl₂/FeCl₃) |
| Solvents & Buffers | Deionized Water | Green solvent for extraction and reaction medium | pH 5-9, temperature 25-80°C |
| Sodium Citrate | Stabilizing agent in conventional synthesis | Comparison reference for green methods | |
| Characterization Tools | UV-Vis Spectrophotometer | Confirm nanoparticle formation | Surface plasmon resonance measurement (400-450 nm for AgNPs) |
| Electron Microscopy | Size and morphological analysis | SEM/TEM for visualization (1-100 nm range) | |
| XRD Analyzer | Crystalline structure determination | Crystallite size calculation via Debye-Scherrer equation | |
| DLS/Zeta Potential | Size distribution and stability measurement | Hydrodynamic diameter and surface charge | |
| Application Testing | Microbial Strains | Antimicrobial efficacy assessment | E. coli, S. aureus, C. albicans |
| Seed Varieties | Agricultural nanopriming evaluation | Potato seeds, wheat, Satureja hortensis | |
| Pollutant Solutions | Environmental remediation testing | Dyes (methylene blue), pharmaceuticals (sulfamethoxazole) |
Challenges and Future Directions in Commercial Translation
Despite significant advancements, several challenges remain in the widespread commercialization of green-synthesized nanoparticles. Scalability of synthesis techniques requires further optimization to meet industrial demands, while standardization of protocols is essential to ensure consistent quality and performance [109]. Comprehensive toxicity studies are needed to evaluate long-term environmental and health impacts, particularly for biomedical applications [108].
Future research directions focus on enhancing process control through artificial intelligence and machine learning, developing multifunctional nanoparticles for combined applications, and establishing international regulatory frameworks [10] [109]. The integration of AI-assisted design predicts optimal plant-based synthesis routes and simulates nanoparticle behavior in biological and environmental systems, potentially accelerating development while reducing resource consumption [10].
The emerging concept of "nano equity" addresses concerns about technological access and distribution of benefits. Open-source platforms and international collaborations, such as the UNESCO-backed "Green Nano Commons," promote technology sharing across the Global South to prevent nano-privilege from becoming the next technological divide [109].
The economic and commercial assessment of green nanoparticle synthesis reveals a compelling value proposition combining cost reduction, enhanced performance, and environmental sustainability. Quantitative data demonstrates that green approaches can reduce production costs by 30-40% while achieving performance metrics equal or superior to conventional methods in biomedical, environmental, and agricultural applications [10] [8] [57]. The integration with circular economy models further enhances sustainability through renewable resource utilization, waste valorization, and closed-loop systems [10] [3].
For researchers and drug development professionals, green synthesis methods offer reproducible, scalable, and economically viable pathways for nanomaterial production without compromising functionality. The continued advancement of these approaches, supported by AI-assisted optimization and standardized protocols, promises to accelerate the transition to sustainable nanotechnology across multiple sectors. As economic evidence yields viability in synthetic pathways, green nanoparticles are positioned to catalyze a paradigm shift toward environmentally responsible and commercially attractive nanomanufacturing.
Conclusion
The synthesis of evidence confirms that green methods offer a fundamentally more sustainable and often more effective pathway for nanoparticle production compared to conventional synthesis. Key takeaways include superior biocompatibility for biomedical applications, reduced environmental impact through the use of renewable resources and energy-efficient processes, and compelling performance in catalysis and drug delivery. However, the full potential of green nanotechnology hinges on overcoming critical challenges in standardization, scalability, and rigorous long-term toxicity studies. Future directions for biomedical research must focus on the development of standardized protocols, the exploitation of AI for predictive synthesis and design, and the execution of robust clinical trials to translate these promising, nature-inspired materials from the lab to the clinic, ultimately fostering a new era of sustainable nanomedicine.
References
- 1. Synthesis of nanoparticles using advanced techniques [https://www.sciencedirect.com/science/article/pii/S2949829525000385]
- 2. Synthesis of Silver Nanoparticles: From Conventional to ' ... [https://www.mdpi.com/2227-9717/11/9/2617]
- 3. Sustainable synthesis of nanomaterials using different ... [https://bnrc.springeropen.com/articles/10.1186/s42269-025-01316-4]
- 4. A review on green synthesis of nanoparticles toward ... [https://www.sciencedirect.com/science/article/pii/S2772826925000161]
- 5. Green-synthesized metal nanoparticles: a promising approach ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12307466/]
- 6. Comparative evaluation of green synthesized and ... [https://www.nature.com/articles/s41598-025-96441-5]
- 7. Is 'Green' Gold and Silver Nanoparticle Synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299925/]
- 8. Green-synthesized silver nanoparticles for improved heat ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra04571a]
- 9. PROPERTIES, EFFECTS AND CHEMICAL & BIOLOGICAL ... [https://chemrxiv.org/engage/chemrxiv/article-details/679f77286dde43c908565ebe]
- 10. Sustainable synthesis of functional nanomaterials ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894725047291]
- 11. Synthesis of green nanoparticles for energy, biomedical ... [https://link.springer.com/article/10.1007/s10311-023-01682-3]
- 12. Revisiting the Green Synthesis of Nanoparticles [https://pmc.ncbi.nlm.nih.gov/articles/PMC10444627/]
- 13. Environmental Impact of Nanoparticles' Application as an ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7795427/]
- 14. Plant-Based Green Synthesis of Nanoparticles: Production ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8773616/]
- 15. Cutting-Edge Nanoparticle Innovations in Biomedical ... [https://www.scientificarchives.com/article/cutting-edge-nanoparticle-innovations-in-biomedical-science-synthesis-applications-challenges-and-future-prospects]
- 16. Recent advances in green synthesis, characterization and ... [https://www.sciencedirect.com/science/article/abs/pii/S1364032125010275]
- 17. Innovative green and bio-based approaches for ... [https://mnsl-journal.springeropen.com/articles/10.1186/s40486-025-00223-7]
- 18. A review on bio-inspired nanoparticles and their impact on ... [https://pubs.rsc.org/en/content/articlehtml/2025/su/d4su00460d]
- 19. A review on green synthesis of silver nanoparticles (SNPs ... [https://pubs.rsc.org/en/content/articlelanding/2025/ra/d4ra07519f]
- 20. A review on green synthesis of silver nanoparticles (SNPs ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d4ra07519f]
- 21. Green Synthesis of Silver Nanoparticles Using Plant Extracts [https://pmc.ncbi.nlm.nih.gov/articles/PMC12250056/]
- 22. Microbial-Based Green Synthesis of Silver Nanoparticles [https://www.mdpi.com/1422-0067/26/20/10163]
- 23. Novel green synthesis of silver nanoparticles from empty ... [https://www.sciencedirect.com/science/article/pii/S2949829525000051]
- 24. Green synthesis of nanoparticles from biodegradable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9399584/]
- 25. Selective cytotoxicity of green synthesized silver ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6267361/]
- 26. Biosynthesis of Gold and Silver Nanoparticles Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10096681/]
- 27. Antimicrobial and cytotoxic activity of green synthesis silver ... [https://www.nature.com/articles/s41598-021-94012-y]
- 28. Terpenoids and Polyphenols as Natural Antioxidant Agents ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8389302/]
- 29. Dietary Phytochemicals in Health and Disease: Mechanisms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11922683/]
- 30. Green silver nanoparticles from bacteria- antioxidant ... [https://www.sciencedirect.com/science/article/pii/S2949829524000500]
- 31. Plant mediated fabrication of silver nanoparticles, process ... [https://www.nature.com/articles/s41598-023-45038-x]
- 32. Recent Advances in Plant-Based Green Synthesis of ... [https://www.mdpi.com/2071-1050/17/9/4152]
- 33. Sustainable green synthesis of metallic nanoparticle using ... [https://www.sciencedirect.com/science/article/pii/S2213343725016173]
- 34. Green-synthesized metal nanoparticles: a promising ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1637589/full]
- 35. Recent advances in metal/metal-oxide nanoparticle ... [https://www.sciencedirect.com/science/article/pii/S2468519425005762]
- 36. From Physical and Green Routes to Machine Learning ... [https://chemrxiv.org/engage/chemrxiv/article-details/6847aa4f1a8f9bdab53c7f92]
- 37. Microbe-Mediated Biosynthesis of Nanoparticles [https://pmc.ncbi.nlm.nih.gov/articles/PMC8246319/]
- 38. Microbial Nano-Factories: Synthesis and Biomedical ... [https://www.frontiersin.org/journals/chemistry/articles/10.3389/fchem.2021.626834/full]
- 39. Eco-friendly synthesized nanoparticles as antimicrobial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10472938/]
- 40. Microbial-Based Green Synthesis of Silver Nanoparticles [https://pubmed.ncbi.nlm.nih.gov/41155457/]
- 41. Data-driven synthetic microbes for sustainable future [https://www.nature.com/articles/s41540-025-00556-4]
- 42. Principles of green chemistry: building a sustainable future [https://link.springer.com/article/10.1007/s44371-025-00152-9]
- 43. Comparative study on microwave-assisted hydrothermal ... [https://www.sciencedirect.com/science/article/abs/pii/S0378775325008687]
- 44. Advanced microwave synthesis strategies for innovative ... [https://www.sciencedirect.com/science/article/pii/S2468025724003194]
- 45. Mn-doped ZnO nanopowders prepared by sol–gel and ... [https://www.beilstein-journals.org/bjnano/articles/15/104]
- 46. Accelerating phase evolution via a microwave sol-gel ... [https://www.sciencedirect.com/science/article/abs/pii/S2352152X24037034]
- 47. Overcoming Scaling Challenges in Sol–Gel Synthesis [https://www.mdpi.com/3042-5697/1/2/6]
- 48. Microwave-assisted synthesis of high-performance TaC ... [https://www.sciopen.com/article/10.26599/JAC.2025.9221130]
- 49. Silver nanoparticles as next-generation antimicrobial agents [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1599113/full]
- 50. Anti-inflammatory, cytotoxic, and potential wound healing ... [https://www.nature.com/articles/s41598-025-24111-7]
- 51. Nanoparticle-based approaches for wound healing [https://pmc.ncbi.nlm.nih.gov/articles/PMC12595217/]
- 52. Green-Synthesized Silver Nanoparticles as Promising ... [https://pubmed.ncbi.nlm.nih.gov/40430511/]
- 53. Wound healing potential of green synthesized silver ... [https://www.sciencedirect.com/science/article/pii/S2773050625000059]
- 54. Unlocking the synergistic potential of green metallic ... [https://www.sciencedirect.com/science/article/pii/S2589004225007795]
- 55. Green Nanoparticles in 2025: Small Tech, Big Impact [https://www.torskal.com/why-green-nanoparticles-matter-more-than-ever-in-2025/?srsltid=AfmBOopU7DiwkCK7qv3bpDxNkXQHTD0I_TAEG4jda_8DOFMPQ80Z7jM1]
- 56. Nanoparticles for targeted removal of emerging contaminants ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12552218/]
- 57. Comparative study of the efficiency of conventional and ... [https://www.sciencedirect.com/science/article/abs/pii/S0920586124000154]
- 58. Green synthesis of silver nanoparticles (AgNPs) from G ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-025-02815-9]
- 59. Nano-bioremediation of metal-polluted industrial ... [https://www.nature.com/articles/s41598-025-19925-4]
- 60. metal and metal oxide nanoparticles for environmental ... [https://pubs.rsc.org/en/content/articlehtml/2025/nr/d5nr01973g]
- 61. Rethinking Nanoparticle Synthesis: A Sustainable Approach vs. Traditional Methods - PubMed [https://pubmed.ncbi.nlm.nih.gov/39126206/]
- 62. Eco-Friendly Greener Synthesis of Nanoparticles - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7539319/]
- 63. Harnessing plant extracts for green nanoparticle synthesis [https://www.sciencedirect.com/science/article/pii/S2589234725001241]
- 64. Green synthesis of nanoparticles: Current developments and limitations - ScienceDirect [https://www.sciencedirect.com/science/article/pii/S2352186422000359]
- 65. Plant-derived nanoparticles: Green synthesis, factors, and ... [https://www.sciencedirect.com/science/article/pii/S2949822825007932]
- 66. Green Nanoparticles in 2025: Small Tech, Big Impact [https://www.torskal.com/why-green-nanoparticles-matter-more-than-ever-in-2025/?srsltid=AfmBOoqsiP2o7I7B6XlKvs-NhpVHPF7eERsQzNl4L0rd0PwYkFNZZAhw]
- 67. Green synthesis and characterization of Bi 2 O 3 ... [https://www.nature.com/articles/s41598-025-26415-0]
- 68. Nanotechnology–General Aspects: A Chemical Reduction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10343226/]
- 69. Towards Sustainable Scaling-Up of Nanomaterials ... [https://www.mdpi.com/2673-4117/6/7/149]
- 70. Industrial Scale Production of Nanoparticles [https://www.springerprofessional.de/en/industrial-scale-production-of-nanoparticles/51593092]
- 71. Designer nanoparticles: Incorporating size, shape, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2845970/]
- 72. of Green : A Biological Approach | IntechOpen Synthesis Nanoparticles [https://www.intechopen.com/chapters/1145412]
- 73. of metallic Green using Pistacia species... synthesis nanoparticles [https://pubs.rsc.org/en/content/articlehtml/2025/na/d5na00644a]
- 74. Green synthesized silver nanoparticles: Optimization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9465387/]
- 75. Process optimization for green synthesis of silver ... [https://www.nature.com/articles/s41598-024-54702-9]
- 76. Solid-State Green Synthesis of Ag NPs [https://www.nature.com/articles/s41598-019-51693-w]
- 77. Effect of Synthesis Temperature on the Size of ZnO ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7352361/]
- 78. Optimization of synthesis for shape and size controlled ... [https://www.sciencedirect.com/science/article/abs/pii/S0927775720302405]
- 79. Green Synthesis of Nanoparticles and Their Energy Storage, Environmental, and Biomedical Applications [https://www.mdpi.com/2073-4352/13/11/1576]
- 80. A Comprehensive Review on the Green Synthesis of Nanoparticles: Advancements in Biomedical and Environmental Applications | Biomedical Materials & Devices [https://link.springer.com/article/10.1007/s44174-025-00295-4]
- 81. assisted by Nanoparticle synthesis machine learning [https://www.nature.com/articles/s41578-021-00337-5?error=cookies_not_supported&code=7fbf9f93-90bc-4f23-806b-42cdbbf8b290]
- 82. Green carbon dots in the era of AI: sustainable synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12614371/]
- 83. : Current Research and Future Directions – Blog Nanoparticle Synthesis [https://blog.geetauniversity.edu.in/nanoparticle-synthesis-current-research-future-directions/]
- 84. Green synthesis of polymeric nanoparticles: agricultural ... [https://link.springer.com/article/10.1007/s42452-025-06877-7]
- 85. -based fabrication of phytogenic NiO Machine ... learning nanoparticles [https://link.springer.com/article/10.1007/s10856-025-06964-6]
- 86. Advances in nanoparticle synthesis assisted by microfluidics [https://pubs.rsc.org/en/content/articlehtml/2025/lc/d5lc00194c]
- 87. Synthesis methods and applications of palladium ... [https://www.frontiersin.org/journals/nanotechnology/articles/10.3389/fnano.2022.1062608/full]
- 88. Green biologically synthesized metal nanoparticles [https://pmc.ncbi.nlm.nih.gov/articles/PMC11137799/]
- 89. Comparative analysis of biological versus chemical synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8371006/]
- 90. Characterization, catalytic, and recyclability studies of nano ... [https://www.sciencedirect.com/science/article/abs/pii/S0013935123006138]
- 91. An Up-To-Date Review on Biomedical Applications ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7023275/]
- 92. Advancements in Bio-Nanotechnology: Green Synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11990282/]
- 93. Application of Fourier Transform Infrared (FTIR ... [https://www.mdpi.com/1420-3049/30/3/684]
- 94. Nanotechnology in healthcare, and its safety and ... [https://jnanobiotechnology.biomedcentral.com/articles/10.1186/s12951-024-02901-x]
- 95. The nano-paradox: addressing nanotoxicity for sustainable ... [https://jnanobiotechnology.biomedcentral.com/articles/10.1186/s12951-025-03371-5]
- 96. Comparison of Conventional Verses Green Synthesis ... [https://imgroupofresearchers.com/green-synthesis-of-nanoparticles/]
- 97. Is 'Green' Gold and Silver Nanoparticle Synthesis ... [https://www.mdpi.com/2079-4991/15/14/1095]
- 98. Cyperus rotundus mediated green synthesis of silver ... [https://www.nature.com/articles/s41598-025-03555-x]
- 99. Toxicity and transport of nanoparticles in agriculture: effects ... [https://www.frontiersin.org/journals/nanotechnology/articles/10.3389/fnano.2025.1622228/full]
- 100. Life Cycle Assessment (LCA) – Everything you need to know [https://ecochain.com/blog/life-cycle-assessment-lca-guide/]
- 101. Understanding Life Cycle Assessment: From Cradle-to-Grave ... [https://asuene.com/us/blog/understanding-life-cycle-assessment-from-cradle-to-grave-in-esg-reporting]
- 102. Product Carbon Footprint Calculator : Practical LCA Guide ... [https://globalclimateinitiatives.com/en/guide-pratique-pour-calculer-lempreinte-carbone-dun-produit-par-lacv/]
- 103. Cradle-to-Grave in LCA – What is it & How does it work? [https://ecochain.com/blog/cradle-to-grave-in-lca/]
- 104. Life cycle assessment (LCA) - The ultimate guide for beginners [https://www.hhc.earth/knowledge-base/guides/lca-life-cycle-assessment]
- 105. Environmental Footprint - European Platform on LCA | EPLCA [https://eplca.jrc.ec.europa.eu/EnvironmentalFootprint.html]
- 106. Cost Evidence Yields the Viability of Metal Oxides Synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12541911/]
- 107. Statistical optimization of eco-friendly synthesized silver ... [https://www.nature.com/articles/s41598-025-29253-2]
- 108. Green Synthesis of Silver Nanoparticles Using Plant Extracts [https://www.mdpi.com/1422-0067/26/13/6222]
- 109. Green Nanoparticles in 2025: Small Tech, Big Impact [https://www.torskal.com/why-green-nanoparticles-matter-more-than-ever-in-2025/?srsltid=AfmBOoqjyvejeBXgLaUKzL0nkwO7ykws4hR49Tj5se6XXek9QVdKMcck]