Green HPTLC Method Validation in Pharma Analysis: A Sustainable Framework from Development to Compliance
This article provides a comprehensive guide for researchers and drug development professionals on validating High-Performance Thin-Layer Chromatography (HPTLC) methods aligned with green analytical chemistry (GAC) principles.
Green HPTLC Method Validation in Pharma Analysis: A Sustainable Framework from Development to Compliance
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on validating High-Performance Thin-Layer Chromatography (HPTLC) methods aligned with green analytical chemistry (GAC) principles. It covers the foundational rationale for adopting sustainable HPTLC, detailed methodologies for method development and application, strategies for troubleshooting and optimization, and a complete framework for validation as per ICH Q2(R1) guidelines. By integrating modern sustainability assessment tools like AGREE and GAPI, the content demonstrates how green HPTLC offers a robust, cost-effective, and environmentally friendly solution for pharmaceutical quality control, impurity profiling, and compliance with evolving global pharmacopeial standards.
The Rise of Green HPTLC: Principles, Drivers, and Regulatory Alignment
The fundamental goal of Green Analytical Chemistry (GAC) is to eliminate or significantly reduce the production of dangerous compounds throughout any chemical process, a principle first defined by Anastas [1]. In the field of analytical chemistry, this translates to redesigning equipment and procedures to make them more ecologically sustainable. The twelve principles of GAC provide a structured framework for achieving this, emphasizing waste reduction, safer solvents, and energy efficiency [1].
High-Performance Thin-Layer Chromatography (HPTLC) is increasingly recognized as an inherently green analytical technique due to its minimal environmental footprint. Its advantages for quantitative analysis include accurate sample application, quicker and efficient resolution of mixtures, significantly lower solvent consumption, and reduced sample size compared to other chromatographic methods [2]. A key differentiator from HPLC is HPTLC's ability to examine numerous samples of varying complexity simultaneously and in parallel, leading to a high sample throughput and low consumption of solvent and energy per sample [2]. Furthermore, HPTLC often requires little to no sample pre-treatments, such as liquid–liquid extraction or solid-phase extraction, which further conserves environmental resources [2]. The ongoing innovation in Green HPTLC focuses on replacing traditional organic solvents with greener alternatives in the mobile phase, thereby reducing environmental pollution without compromising analytical performance [2].
The Twelve Principles of Green Analytical Chemistry Applied to HPTLC
The twelve principles of GAC offer a direct pathway to sustainability in the laboratory. The table below outlines how each principle is specifically implemented in Green HPTLC practice.
Table 1: Application of GAC Principles in Green HPTLC
| GAC Principle | Application in Green HPTLC |
|---|---|
| 1. Direct Analytical Techniques | Minimizing or eliminating sample preparation to reduce solvent use and waste generation [1]. |
| 2. Reducing Sample Size | Using minimal sample volumes applied as narrow bands, typically in the microliter range [2]. |
| 3. In-line Measurements | Enabling multiple sample processing with a single mobile phase, reducing repetitive tasks [1]. |
| 4. Analytical Methodologies | Integrating derivatization or detection steps directly on the plate to simplify the process [1]. |
| 5. Automation & Miniaturization | Employing automated sample applicators and compact TLC plates to enhance precision and reduce reagent use [1]. |
| 6. Avoiding Derivatization | Using UV/VIS/FLD detection directly on the plate without derivative reagents where possible [1]. |
| 7. Energy Conservation | Operating at room temperature without energy-intensive pumps, unlike HPLC [1] [2]. |
| 8. Multi-analyte Determination | Simultaneously analyzing multiple samples and standards on a single plate [2]. |
| 9. Green Solvents & Reagents | Using ethanol, water, or acetone in the mobile phase instead of hazardous solvents like chloroform [1] [3] [4]. |
| 10. Waste Reduction | Generating significantly less solvent waste (a few mL per sample) compared to HPLC [1] [2]. |
| 11. Recycling | Reusing materials where feasible and implementing safe waste streams [1]. |
| 12. Safe Operator Handling | Using non-toxic and non-volatile solvents to minimize operator exposure to hazardous fumes [1]. |
Experimental Design for a Green HPTLC Method
Strategic Approach and Workflow
Developing a green HPTLC method requires a systematic approach that prioritizes environmental safety at every stage, from initial solvent selection to final greenness assessment. The protocol should be designed to comply with international guidelines for analytical techniques while maintaining high performance [2].
Materials and Reagents: The Green HPTLC Toolkit
The selection of appropriate materials and reagents is critical for developing an effective green HPTLC method. The following table details essential components and their environmentally conscious alternatives.
Table 2: Research Reagent Solutions for Green HPTLC
| Item | Function | Green Alternatives & Notes |
|---|---|---|
| Stationary Phase | Separation medium | Silica gel 60 F₂₅₄ or RP-18 F₂₅₄S HPTLC plates [4] [5]. |
| Mobile Phase | Sample elution | Binary mixtures of green solvents (e.g., ethanol-water, acetone-water) [3] [4]. |
| Sample Solvent | Dissolving analytes | Ethanol, water, or ethanol-water mixtures instead of acetonitrile or methanol [6]. |
| Derivatization Reagent | Compound visualization | Avoid where possible; use non-toxic reagents if necessary [1]. |
| Reference Standards | Method calibration | Prepare in green solvents at appropriate concentrations [7]. |
Detailed Protocol for Green HPTLC Method Development
Step 1: Green Mobile Phase Selection and Optimization Begin by testing binary mixtures of green solvents. For reversed-phase HPTLC, ethanol-water in ratios from 40:60 to 90:10 v/v has proven effective [4]. For normal-phase HPTLC, explore combinations like cyclohexane-ethyl acetate (90:10 v/v) [3]. The optimal mobile phase should produce a sharp, symmetrical peak with an Rf value between 0.2 and 0.8 [4] [5].
Step 2: Sample Preparation with Minimal Environmental Impact Where possible, employ direct analysis with simple dilution in a green solvent like ethanol or ethanol-water mixtures [1] [7]. If extraction is necessary, prioritize green approaches such as QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe), which use smaller amounts of solvent compared to traditional extraction methods [1].
Step 3: Chromatographic Separation
- Sample Application: Apply samples as bands (e.g., 6 mm width) using an automated applicator like a CAMAG ATS4 or Linomat V. A fixed application rate of 150 nL/s is typical [5].
- Plate Development: Pre-saturate the development chamber with mobile phase vapor for 15-30 minutes at room temperature (22°C) [8] [5]. Use an ascending development distance of 70-80 mm [8].
- Drying: Air-dry the developed plates at room temperature to avoid energy consumption from heating.
Step 4: Detection and Quantification Employ densitometric detection at the appropriate wavelength for the analyte. Scanning speeds of 20 mm/s with slit dimensions of 4 × 0.45 mm are standard [7] [5]. This non-destructive mode of detection allows for potential re-analysis or further derivatization if needed [2].
Validation of Green HPTLC Methods
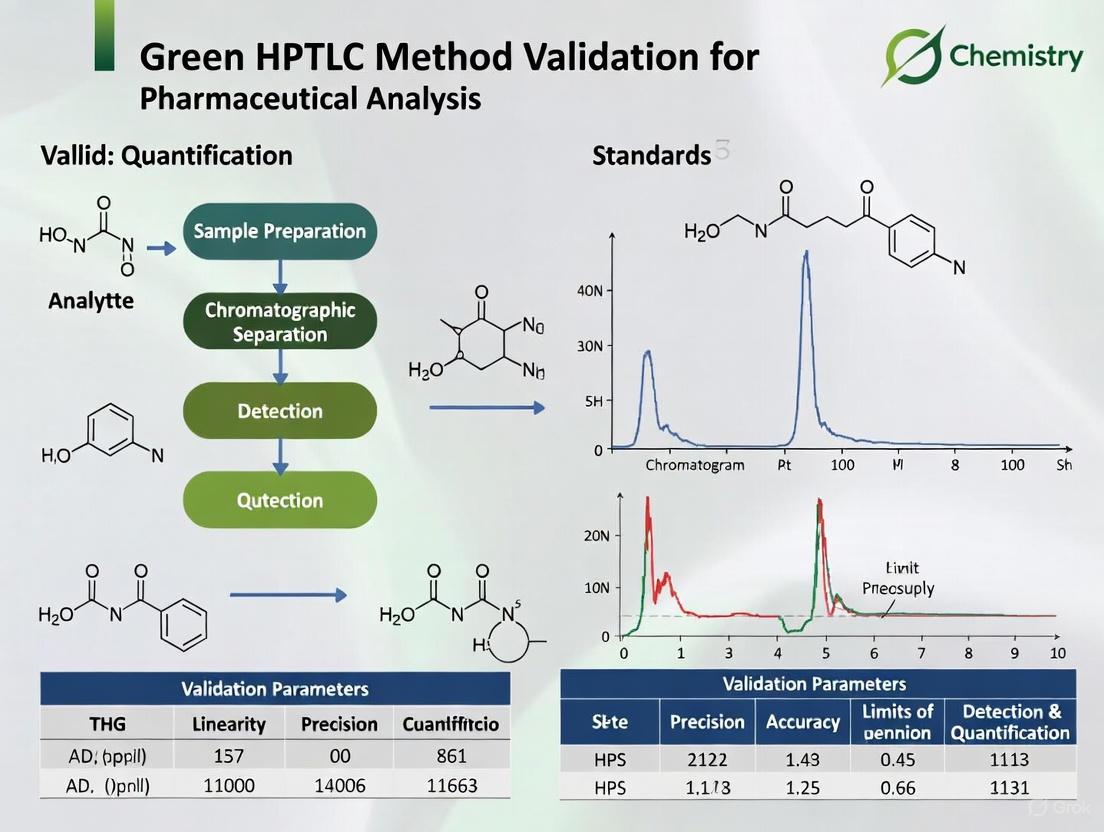
Validating a Green HPTLC method is crucial to ensure it produces reliable, accurate, and reproducible results while maintaining its environmentally friendly profile. Validation should be conducted in accordance with ICH Q2(R2) guidelines [5].
Table 3: Validation Parameters for Green HPTLC Methods
| Parameter | Protocol | Acceptance Criteria |
|---|---|---|
| Linearity | Analyze minimum 5 concentrations in triplicate [5]. | Correlation coefficient (R²) ≥ 0.995 [8] [7]. |
| Accuracy | Recovery studies via standard addition at 3 levels (50%, 100%, 150%) with n=6 [5]. | Recovery of 98-102% [7]. |
| Precision | Intra-day (n=6 on same day) and inter-day (n=6 over 3 days) [5]. | RSD ≤ 2% [7] [4]. |
| Robustness | Deliberate, small changes in mobile phase composition (±2%) [5]. | RSD of peak areas < 2% [4]. |
| Sensitivity (LOD/LOQ) | Based on standard deviation of response and slope of calibration curve [5]. | LOD as low as 0.03 µg/band; LOQ as low as 0.099 µg/band [9]. |
| Specificity | Analyze standard, sample, and forced degradation products [10]. | Baseline separation of analytes from degradants [8]. |
Greenness Assessment Tools for HPTLC Methods
Evaluating the environmental friendliness of an analytical method requires specialized metrics. Several tools have been developed to quantify and compare the greenness of HPTLC methods.
Table 4: Greenness Assessment Tools for HPTLC Methods
| Tool | Scoring System | Application Example in HPTLC |
|---|---|---|
| Analytical Eco-Scale | Ideal score = 100; score > 75 = excellent greenness [4]. | A method for Thymoquinone using ethanol-water (80:20) scored 89 [5]. |
| AGREE | 0 to 1 scale; closer to 1 = greener method [4]. | NP-HPTLC and RP-HPTLC for Thymoquinone scored 0.82 and 0.84, respectively [3]. |
| NEMI | Pictogram with 4 criteria: persistent, bioaccumulative, toxic, corrosive [4]. | RP-HPTLC for Ertugliflozin using ethanol-water scored better than NP-HPTLC with chloroform-methanol [4]. |
| GAPI | Pictogram with 5 pentagrams color-coded for environmental impact [8] [7]. | Applied in the evaluation of methods for Carvedilol and Remdesivir [8] [7]. |
| ChlorTox | Calculates total chlorinated solvent toxicity (g) [4]. | A method for Croconazole HCl scored 1.08 g [5]. |
Comparative Case Studies: Green vs. Conventional HPTLC
Ertugliflozin Analysis: NP-HPTLC vs. RP-HPTLC
A direct comparison between normal-phase and reversed-phase HPTLC for the analysis of Ertugliflozin (ERZ) demonstrates the environmental advantages of green solvent selection [4].
- NP-HPTLC Method: Utilized chloroform/methanol (85:15 v/v) as the mobile phase. Chloroform is classified as hazardous due to its potential health and environmental risks [4].
- RP-HPTLC Method: Employed ethanol-water (80:20 v/v) as the mobile phase. Ethanol is a renewable, biodegradable, and much less toxic solvent [4].
The greenness assessment using multiple tools (NEMI, AES, AGREE, ChlorTox) consistently demonstrated that the RP-HPTLC strategy was significantly greener than the NP-HPTLC approach. Furthermore, the RP-HPTLC method showed better analytical performance in terms of robustness, accuracy, precision, linearity, and sensitivity [4].
Representative Applications in Pharmaceutical Analysis
Green HPTLC methods have been successfully developed for a wide range of pharmaceutical compounds, demonstrating their versatility and robustness.
- Carvedilol: An eco-friendly, stability-indicating method was developed using toluene-isopropanol-ammonia, avoiding carcinogenic solvents. The method was validated and shown to be suitable for stability studies [8].
- Remdesivir, Linezolid, and Rivaroxaban: A single, green densitometric method was developed for simultaneous quantification in spiked human plasma using dichloromethane-acetone, demonstrating applicability to complex matrices [7].
- Thymoquinone: Both NP-HPTLC (cyclohexane-ethyl acetate) and RP-HPTLC (ethanol-water) methods were developed with excellent greenness profiles (AGREE scores of 0.82 and 0.84), highlighting the flexibility of green approaches [3].
- Sacubitril and Valsartan: A green HPLC-fluorescence method using ethanol-phosphate buffer was developed, showing that green principles can be effectively applied across different chromatographic techniques [6].
These case studies confirm that Green HPTLC methods are not only environmentally preferable but also meet rigorous analytical performance standards for pharmaceutical analysis, making them ideal for quality control and regulatory purposes.
Within pharmaceutical quality control and research laboratories, the selection of an analytical technique is increasingly guided by the principles of Green Analytical Chemistry (GAC), which emphasize the reduction of hazardous solvent use, energy consumption, and waste generation. High-performance liquid chromatography (HPLC) has long been the established gold standard for pharmaceutical analysis. However, high-performance thin-layer chromatography (HPTLC) is re-emerging as a robust, versatile, and eco-friendly platform. This application note provides a detailed comparative analysis of HPTLC versus HPLC, focusing on solvent consumption, operational cost, and analytical throughput, to guide scientists in making informed, sustainable choices for method validation and routine analysis.
The core differences between HPTLC and HPLC stem from their fundamental operational principles: HPTLC is an open-bed, parallel-processing system, whereas HPLC is a closed-column, sequential-processing system. This distinction has profound implications for their economic and environmental footprint.
Table 1: Core Characteristics and Environmental Impact at a Glance
| Feature | HPTLC | HPLC |
|---|---|---|
| System Operation | Open-bed, planar chromatography | Closed-column, pump-driven system |
| Sample Processing | Parallel (Multiple samples on one plate) | Sequential (One sample per injection) |
| Typical Solvent Consumption per Analysis | ~10-15 mL for an entire plate [8] [11] | ~500-1000 mL per day for a single set of analyses [12] |
| Analytical Throughput | High (Up to 19 samples/standard in one run) [13] | Lower (Dependent on run time, typically 10-30 min/sample) |
| Energy Consumption | Low (No high-pressure pumps) [14] | High (Constant operation of high-pressure pumps) |
| Instrumentation and Maintenance Costs | Lower (No expensive columns; lower solvent costs) [12] [15] | Higher (Costly columns; high solvent consumption and disposal) |
| Greenness Assessment (AGREE Score Example) | High score reported for anti-COVID-19 drug analysis [12] | Lower score in comparative studies [12] |
Detailed Comparative Analysis
Solvent Consumption and Environmental Impact
Solvent consumption is a primary differentiator and a key metric in greenness assessments.
- HPTLC: The technique is inherently minimalistic in solvent use. A single development chamber requires a small volume of mobile phase, typically 10-15 mL, to separate multiple samples on one plate. A study on carvedilol quantification emphasized its eco-friendly nature, using a minimal solvent volume to enhance sustainability [8]. The reduced solvent use directly translates to less waste generation and lower purchasing and disposal costs.
- HPLC: In contrast, HPLC operates on a continuous flow principle. The mobile phase is constantly pumped through the system at typical rates of 0.5-2.0 mL/min, leading to the consumption of hundreds of milliliters per day for a single instrument. This high consumption has a significant environmental impact and contributes substantially to operational costs.
Greenness assessment tools, such as the Analytical GREEnness (AGREE) metric, quantitatively highlight this advantage. A 2025 comparative study of anti-COVID-19 drug analysis demonstrated that the HPTLC method achieved a superior AGREE score compared to a reported HPLC-high-resolution mass spectrometry method, largely due to its reduced solvent requirements [12].
Cost Analysis
The cost-effectiveness of HPTLC extends beyond just solvent savings.
- Instrumentation and Consumables: HPTLC systems have a lower initial investment and maintenance cost. A significant saving comes from the absence of expensive analytical columns, which in HPLC have a limited lifespan and require replacement [12] [15]. HPTLC plates are far less costly per sample analyzed.
- Operational Costs: The "green" advantages of HPTLC directly reduce operational expenses. Lower solvent consumption cuts purchasing costs, and reduced waste volume decreases hazardous waste disposal fees. Furthermore, HPTLC's lower energy demand, as it does not require continuous high-pressure pumping, contributes to reduced utility costs [14].
Analytical Throughput and Efficiency
Throughput is critical in high-demand environments like quality control.
- HPTLC - Parallel Processing: The ability to run up to 19 or more samples and standards simultaneously on a single HPTLC plate drastically reduces total analysis time [13]. This parallel analysis makes HPTLC particularly suited for high-throughput applications such as stability studies, bioequivalence assessments, and routine quality control of multiple samples [13] [11].
- HPLC - Sequential Processing: HPLC analyzes samples one after another. Each sample must complete its run cycle (often 10-30 minutes) before the next injection, creating a bottleneck. While techniques like ultra-high-performance liquid chromatography (UHPLC) can shorten run times, they do not change this fundamental sequential nature.
Table 2: Quantitative Comparison of Analytical Performance
| Parameter | HPTLC | HPLC |
|---|---|---|
| Sample Processing Mode | Parallel | Sequential |
| Typical Analysis Time for 19 Samples | ~30-50 minutes (one plate) | ~285-475 minutes (at 15-25 min/sample) |
| Limit of Quantification (LOQ) | Demonstrated at 50 ng/band for Furosemide in plasma [13] | Generally offers very high sensitivity, often lower than HPTLC |
| Method Development | Flexible; mobile phase can be easily modified | More complex; requires column re-equilibration |
| Hyphenation Potential | High (compatible with MS, SERS, NIR) [11] | The industry standard (LC-MS is ubiquitous) |
The following workflow diagrams illustrate the procedural and efficiency differences between the two techniques.
HPTLC Parallel Analysis Workflow
HPLC Sequential Analysis Workflow
Detailed Experimental Protocols
This protocol outlines a general method for quantifying an active pharmaceutical ingredient (API) from tablets, showcasing the green and efficient nature of HPTLC.
I. Research Reagent Solutions
Table 3: Essential Materials for HPTLC Method Development
| Item | Function / Specification |
|---|---|
| HPTLC Plates | Silica gel 60 F₂₅₄, pre-coated, 10x10 cm or 20x10 cm [8] [16] |
| Mobile Phase Components | Toluene, Isopropanol, Ammonia solution (for normal-phase) [8] |
| Sample Solvent | Methanol or mixture (e.g., Acetonitrile:Methanol, 1:1) [13] |
| Standard Solution | High-purity reference standard of the target API |
| Microliter Syringe | 100 µL, for precise sample application [16] |
| Twin-Trough Chamber | For plate development with chamber saturation [16] |
| Densitometer Scanner | For quantitative measurement of band intensities [16] |
II. Procedure
- Plate Pre-washing and Activation: Pre-wash HPTLC plates with methanol. Activate in an oven at 110°C for 5 minutes [16].
- Standard and Sample Preparation:
- Standard Stock Solution: Accurately weigh 10 mg of API reference standard into a 10 mL volumetric flask. Dissolve and dilute to volume with methanol to obtain a 1000 µg/mL stock solution.
- Sample Solution: Finely powder and weigh tablet powder equivalent to 10 mg of API. Transfer to a 100 mL volumetric flask, add ~70 mL of methanol, sonicate for 15-20 minutes, dilute to volume, and filter.
- Prepare working solutions by appropriate dilution.
- Sample Application: Using an automatic applicator (e.g., CAMAG Linomat 5), apply bands of standard and sample solutions (e.g., 6 mm wide) onto the HPTLC plate. Maintain a constant application rate (e.g., 150 nL/s) [16].
- Chromatographic Development: Pour the mobile phase (e.g., Toluene:Isopropanol:Ammonia, 7.5:2.5:0.1 v/v/v [8]) into one trough of the twin-trough chamber. Allow saturation for 20 minutes at room temperature. Place the plate in the other trough and develop linearly to a distance of 75 mm.
- Densitometric Analysis: After development and drying, scan the plate using a TLC scanner (e.g., CAMAG TLC Scanner 3) at the optimal wavelength for the API (e.g., 224 nm for some compounds [16]). Generate a calibration curve from standard bands and quantify the API in the sample bands.
Protocol: Representative HPLC Method for Pharmaceutical Analysis
This protocol represents a standard isocratic HPLC method, provided for contrast.
I. Research Reagent Solutions
Table 4: Essential Materials for HPLC Method Development
| Item | Function / Specification |
|---|---|
| HPLC System | Binary or quaternary pump, autosampler, column oven, UV/Vis or DAD detector [17] |
| Analytical Column | C18, 150 mm x 4.6 mm, 5 µm (or sub-2 µm for UHPLC) [17] |
| Mobile Phase | Filtered and degassed mixture (e.g., Buffer:Acetonitrile, 60:40 v/v) |
| Standard Solution | High-purity reference standard of the target API |
| Sample Solvent | Often matching the initial mobile phase composition |
II. Procedure
- Mobile Phase Preparation: Prepare the mobile phase as per the method, typically a mixture of aqueous buffer and an organic solvent like acetonitrile or methanol. Filter through a 0.45 µm membrane and degas thoroughly.
- Standard and Sample Preparation: Prepare standard and sample solutions at known concentrations, similar to the HPTLC protocol, but often in a solvent compatible with the mobile phase.
- System Equilibration: Pump the mobile phase through the system at the method-specified flow rate (e.g., 1.0 mL/min) until a stable baseline is achieved. This can take 30-60 minutes.
- Chromatographic Run:
- Inject the standard and sample solutions sequentially via the autosampler.
- The pump delivers the mobile phase through the column, separating the components.
- The effluent passes through the detector, and the signal is recorded as a chromatogram.
- Each sample run may take 10-30 minutes, followed by a column re-equilibration time before the next injection.
- Data Analysis: Identify the API peak by its retention time compared to the standard. Use the peak area to calculate the concentration in the sample via external or internal standard calibration.
The choice between HPTLC and HPLC is not a matter of declaring one universally superior, but of selecting the right tool for the specific application. HPLC remains indispensable for applications demanding ultra-high sensitivity and is the cornerstone of LC-MS workflows. However, for a vast number of routine pharmaceutical analyses—including assay of dosage forms, dissolution testing, and stability-indicating studies—HPTLC presents a compelling, sustainable alternative.
HPTLC's minimal solvent consumption, lower operational costs, and superior throughput due to parallel processing align perfectly with the goals of Green Analytical Chemistry. The development of sophisticated hyphenated techniques like HPTLC-MS and HPTLC-SERS further expands its capabilities, solidifying its role as a powerful, modern analytical platform [11]. For drug development professionals and researchers aiming to enhance laboratory sustainability and efficiency without compromising data quality, integrating HPTLC into the analytical toolbox is a strategically sound decision.
High-Performance Thin-Layer Chromatography (HPTLC) has undergone a significant transformation from a simple qualitative tool to a sophisticated quantitative analytical platform, necessitating alignment with international regulatory standards. The convergence of ICH Q2(R1) validation guidelines, United States Pharmacopeia (USP) monographs, and emerging World Health Organization (WHO) standards creates a comprehensive framework for ensuring the reliability, reproducibility, and regulatory acceptance of HPTLC methods in pharmaceutical analysis. This evolution is particularly relevant in the context of green analytical chemistry, where HPTLC's minimal solvent consumption, low energy requirements, and reduced waste generation position it as an environmentally sustainable choice for modern quality control laboratories [18].
The regulatory landscape is dynamically adapting to incorporate advanced HPTLC technologies. Notably, the USP has published a draft chapter, PF 50(5), proposing Method III for the identification of fixed oils using HPTLC, which utilizes more environmentally friendly solvents and derivatization reagents while incorporating the latest generation of reverse-phase plates [19]. This regulatory development aligns with initiatives from the WHO and other pharmacopeial bodies to standardize HPTLC methodologies for complex matrices, including botanical materials and pharmaceutical formulations, within a structured validation framework [20].
Core Regulatory Guidelines and Their Applications
ICH Q2(R1) Validation for HPTLC Methods
The ICH Q2(R1) guideline, "Validation of Analytical Procedures: Text and Methodology," establishes fundamental validation parameters for analytical methods, with specific implementation considerations for HPTLC platforms. The following table summarizes the core validation parameters and their typical acceptance criteria for HPTLC methods in pharmaceutical analysis:
Table 1: ICH Q2(R1) Validation Parameters and Acceptance Criteria for HPTLC
| Validation Parameter | Experimental Requirement | Typical Acceptance Criteria |
|---|---|---|
| Linearity | Analysis of 5-7 concentration levels | Correlation coefficient (r²) ≥0.995 [7] [21] |
| Range | Established from Linearity study | Within the specified linear interval [22] |
| Accuracy (Recovery) | Spiked samples at 3 levels (80%, 100%, 120%) | Recovery 98-102% [7] [23] |
| Precision | ||
| - Repeatability | Multiple measurements of same sample | RSD ≤2% [7] |
| - Intermediate Precision | Different days/analysts/instruments | RSD ≤3% [20] |
| Detection Limit (LOD) | Signal-to-noise 3:1 or calculated | Visual or mathematical determination [22] |
| Quantitation Limit (LOQ) | Signal-to-noise 10:1 or calculated | Visual or mathematical determination [7] [22] |
| Specificity | Resolution from potential interferents | Baseline resolution (Rf difference ≥0.05) [24] |
| Robustness | Deliberate variations in parameters | No significant effect on results [22] [21] |
For qualitative HPTLC methods, particularly for botanical identification, specificity becomes the paramount validation parameter. The method must demonstrate capability to distinguish the target species from related species or potential adulterants, accounting for natural variability in plant materials [20]. The validation should include assessment against botanically defined reference materials and related species to establish discrimination capability [20].
USP Standards and Compendial HPTLC Methods
The United States Pharmacopeia has progressively incorporated HPTLC methods into its monographs, with recent emphasis on green chemistry principles and technological advancements. The newly proposed Method III in the revised USP General Chapter <202> "Identification of Fixed Oils by Thin-Layer Chromatography" represents a significant step toward environmentally sustainable analysis through:
- Implementation of more environmentally friendly solvents [19]
- Use of greener derivatization reagents [19]
- Adoption of latest generation HPTLC reverse-phase plates [19]
- Inclusion of additional reference standards for various fixed oils [19]
This compendial approach facilitates the standardization of HPTLC methodologies across laboratories and establishes a framework for method validation that aligns with both ICH Q2(R1) principles and green analytical chemistry objectives.
WHO and Global Standards for HPTLC
Global standardization efforts for HPTLC methodology are increasingly important for ensuring consistency in pharmaceutical analysis worldwide. The WHO's development of HPTLC standards, particularly for quality control of complex natural products, addresses several critical challenges:
- Natural variability in botanical materials requires method validation approaches that account for chemical profile fluctuations [20]
- Adulteration detection necessitates methods with sufficient specificity to identify wrong species in samples [20]
- Reference material qualification requires botanically defined reference standards with voucher specimens [20]
The validation of qualitative HPTLC methods for identification purposes must establish that the method can reliably determine whether a sample represents the same species as the botanical reference material, considering the inherent natural variability [20].
Experimental Protocols for Regulatory Compliance
HPTLC Method Development and Optimization Protocol
This protocol outlines a systematic approach to HPTLC method development incorporating Quality by Design (QbD) principles and green chemistry considerations.
Table 2: Essential Research Reagent Solutions for HPTLC Analysis
| Reagent/Material | Specification | Function/Application |
|---|---|---|
| HPTLC Plates | Silica gel 60 F254, aluminum-backed, 0.25 mm thickness | Stationary phase for separation [7] [23] |
| Mobile Phase | HPLC grade solvents; green alternatives preferred | Sample transport across stationary phase [19] [21] |
| Sample Solvent | Methanol, ethanol, acetonitrile, or aqueous buffers | Sample dissolution and application [7] |
| Derivatization Reagent | Anisaldehyde, vanillin, or greener alternatives [19] | Visualization of non-UV absorbing compounds |
| Reference Standards | USP, EP, or certified reference standards | Method calibration and qualification [19] [20] |
Procedure:
Stationary Phase Selection:
Mobile Phase Optimization:
- Utilize Hansen Solubility Parameters (HSP) and software tools (e.g., HSPiP) to predict solvent combinations based on solute-solvent interactions [21]
- Apply QbD principles with experimental designs (e.g., Box-Behnken, Central Composite Design) to optimize critical parameters [22] [21]
- Prioritize green solvent alternatives (e.g., ethanol, ethyl acetate, acetone) over traditional hazardous solvents [19] [21]
Sample Preparation:
Chromatographic Development:
Detection and Visualization:
Method Validation Protocol Per ICH Q2(R1)
This protocol provides a detailed procedure for validating HPTLC methods in compliance with ICH Q2(R1) requirements.
Linearity and Range:
- Prepare standard solutions at minimum 5 concentration levels across the expected range
- Apply each concentration in triplicate on HPTLC plates
- Develop plates and scan bands following optimized method
- Plot peak area versus concentration, calculate regression statistics
- Acceptable criteria: correlation coefficient (r²) ≥ 0.995 [7] [21]
Accuracy:
- Prepare samples spiked with known quantities of analyte at 80%, 100%, and 120% of target concentration
- Analyze using validated method (n=3 for each level)
- Calculate percentage recovery: (Measured Concentration/Spiked Concentration) × 100
- Acceptable criteria: mean recovery 98-102% with RSD ≤2% [7] [23]
Precision:
- Repeatability: Analyze six replicates of same sample preparation in one session
- Intermediate Precision: Analyze same samples on different days, by different analysts, or with different instruments
- Calculate relative standard deviation (RSD%) for peak areas and retention factors (Rf)
- Acceptable criteria: RSD ≤2% for repeatability, ≤3% for intermediate precision [7] [20]
Specificity:
- Analyze blank matrix, standard, and sample solutions
- Confirm baseline resolution of analyte from potential interferents (Rf difference ≥0.05) [24]
- For identification methods, demonstrate discrimination from closely related species [20]
Robustness:
- Deliberately vary method parameters (mobile phase composition ±2%, saturation time ±5 minutes, development distance ±5 mm)
- Evaluate impact on chromatographic results (Rf values, resolution, peak areas)
- Use experimental design approaches to study multiple factors simultaneously [22] [21]
LOD and LOQ:
- Visual Method: Serial dilution until detectable/quantifiable band observed
- Signal-to-Noise: LOD = 3:1 S/N, LOQ = 10:1 S/N
- Standard Deviation Method: LOD = 3.3σ/S, LOQ = 10σ/S (where σ = residual SD, S = slope) [7] [22]
Advanced Applications and Regulatory Case Studies
Pharmaceutical Quality Control Applications
HPTLC methods validated according to regulatory guidelines have been successfully applied across diverse pharmaceutical analysis scenarios:
Fixed-dose combination products: Simultaneous quantification of antidiabetic drugs linagliptin and dapagliflozin using QbD-optimized HPTLC method with well-resolved bands at Rf = 0.41 and 0.66, respectively, demonstrating linearity (r² = 0.9989 and 0.9505) across therapeutic ranges [21]
COVID-19 therapeutics: Green HPTLC method for simultaneous analysis of remdesivir with co-administered drugs linezolid and rivaroxaban in spiked human plasma, showing linearity ranges of 0.2-5.5 μg/band, 0.2-4.5 μg/band, and 0.1-3.0 μg/band respectively, with outstanding recoveries (98.3-101.2%) [7]
Veterinary drug residues: Eco-friendly HPTLC method for quantification of florfenicol and meloxicam in bovine tissues, validated according to ICH guidelines with linearity ranges of 0.50-9.00 μg/band and 0.03-3.00 μg/band, respectively, meeting regulatory requirements for monitoring veterinary drug residues [23]
Botanical Product Identification
For botanical materials, HPTLC identification methods require specialized validation approaches addressing natural variability:
The validation process for botanical identification methods must establish that the method can reliably distinguish the target species from related species and potential adulterants, accounting for natural variability in plant materials [20]. This requires testing against multiple botanical reference materials and establishing similarity criteria rather than exact matches [20].
Sustainability Assessment in Regulatory HPTLC
Greenness Evaluation Tools and Metrics
Modern regulatory HPTLC method development incorporates formal assessment of environmental impact using validated greenness metrics:
- AGREE Calculator: Comprehensive assessment across multiple environmental impact categories [7] [21]
- GAPI (Green Analytical Procedure Index): Pictorial representation of method greenness [7] [24]
- Analytical Eco-Scale: Semi-quantitative scoring system evaluating hazards of reagents and methodology [7]
- White Analytical Chemistry (WAC): Integrative assessment balancing analytical, ecological, and practical attributes [24]
HPTLC methods consistently demonstrate high greenness ratings due to inherently low solvent volumes (<10 mL per analysis), minimal energy requirements, elimination of derivatization in many cases, and capacity for parallel sample processing [18]. The "HPTLC+" multimodal platform is increasingly recognized as an environmentally friendly analysis tool aligned with Green Analytical Chemistry principles [18].
Integration of Green Principles in Regulatory Methods
The regulatory landscape is increasingly incorporating sustainability considerations:
- USP's proposed Method III for fixed oil identification explicitly incorporates environmentally friendly solvents and greener derivatization reagents [19]
- Method development approaches prioritize alternative green solvents identified through predictive tools like HSPiP [21]
- Waste minimization strategies including reduced plate dimensions and solvent recycling are implemented [18]
- Energy-efficient operations utilizing ambient temperature development rather than heated chambers [18]
The regulatory framework for HPTLC continues to evolve, with emerging trends focusing on the integration of advanced detection technologies (HPTLC-MS, HPTLC-SERS, HPTLC-NIR) within validated methodologies [18]. The application of convolutional neural networks (CNNs) for automated spot recognition represents a promising advancement for enhancing reproducibility and standardization in regulatory analysis [18]. Furthermore, the harmonization of HPTLC standards across ICH, USP, and WHO guidelines facilitates global acceptance of HPTLC methodologies while maintaining alignment with green analytical chemistry principles. As demonstrated through the case studies and protocols presented in this article, HPTLC methodologies developed within this comprehensive regulatory framework provide robust, sustainable solutions for modern pharmaceutical analysis across diverse applications from raw material identification to complex formulation analysis.
The adoption of Green High-Performance Thin-Layer Chromatography (HPTLC) in pharmaceutical analysis is being driven by a powerful convergence of sustainability demands, economic pressures, and the need for robust analytical performance. This paradigm shift represents a fundamental rethinking of analytical method development, moving from traditional "take-make-dispose" linear models toward circular, sustainable practices without compromising data quality [25]. The pharmaceutical industry's ambitious environmental targets, including AstraZeneca's goal of achieving carbon zero status for analytical laboratories by 2030, further accelerate this transition [26]. This application note examines the key drivers behind this adoption trend through specific case studies and quantitative data, providing researchers with validated protocols and frameworks for implementation.
Quantitative Assessment of Adoption Drivers
Sustainability Metrics Comparison of Analytical Techniques
Table 1: Greenness Assessment Scores of HPTLC Methods Versus Conventional HPLC
| Analytical Method | Analyzed Compound(s) | AGREE Score | NEMI Profile | Eco-Scale Assessment | GAPI Profile | Reference |
|---|---|---|---|---|---|---|
| Green HPTLC (NP) | Thymoquinone | 0.82 | Perfect | N/A | N/A | [3] |
| Green HPTLC (RP) | Thymoquinone | 0.84 | Perfect | N/A | N/A | [3] |
| Green HPTLC | Carvedilol | N/A | Perfect | High | Excellent | [8] |
| Green HPTLC | Suvorexant | 0.88 | N/A | 93 | N/A | [27] |
| HPTLC | Hydroxyzine, Ephedrine, Theophylline | Moderate | N/A | N/A | Moderate | [28] |
| Conventional HPLC | Various Pharmaceuticals | Typically <0.2 | Poor | Low | Poor | [25] |
AGREE Score Interpretation: 0.00-0.30 (Poor), 0.31-0.60 (Moderate), 0.61-0.80 (Good), 0.81-1.00 (Excellent Greenness)
Economic and Operational Efficiency Comparison
Table 2: Cost and Efficiency Analysis: HPTLC vs. Conventional Chromatography
| Parameter | Green HPTLC | Conventional HPLC/UHPLC | Reference |
|---|---|---|---|
| Solvent Consumption per Analysis | ~15-25 mL | ~500-1000 mL | [14] [26] |
| Energy Consumption | Low (minimal instrument operation time) | High (continuous pump operation) | [26] |
| Sample Throughput | High (parallel processing of 15-20 samples) | Low (sequential analysis) | [25] [14] |
| Analysis Time per Sample | ~2-5 minutes (parallel processing) | ~10-30 minutes (sequential) | [29] [28] |
| Equipment Cost | Low to Moderate | High | [14] |
| Column Consumption | None | Significant cost factor | [14] |
| Waste Generation | Minimal | Substantial | [26] |
| Operational Simplicity | High (minimal training required) | Moderate to High (specialized training) | [29] |
Detailed Experimental Protocols
Protocol 1: Green HPTLC Method for Simultaneous API Determination
Application: Simultaneous quantification of mirabegron and tamsulosin in pharmaceutical dosage forms [29]
Materials and Reagents:
- HPTLC plates: Silica gel 60 F₂₅₄ (20 × 20 cm, 0.25 mm thickness)
- Mobile phase: Methanol-ethyl acetate-ammonia (3:7:0.1, v/v/v)
- Standard solutions: 1 mg/mL in methanol
- Sample preparation: Methanol extraction with sonication
Instrumentation Conditions:
- Application device: CAMAG Linomat autosampler
- Development chamber: Twin-trough glass chamber (20 × 10 cm)
- Saturation time: 30 minutes at room temperature
- Development distance: 75 mm
- Development time: 15 minutes
- Detection wavelength: 270 nm
- Scanning speed: 20 mm/s
- Slit dimensions: 6.00 × 0.45 mm
Validation Parameters:
- Linearity range: 0.15–7.5 µg/band (mirabegron), 0.05–2.5 µg/band (tamsulosin)
- Precision: RSD ≤ 2%
- Accuracy: 99.98%-100.04% recovery
- Robustness: Deliberate variations in mobile phase composition (±0.1 mL)
- Specificity: Baseline separation of degradation products
Protocol 2: Stability-Indicating Method for Carvedilol
Application: Eco-friendly stability-indicating method for carvedilol in pharmaceutical dosage forms [8]
Materials and Reagents:
- Mobile phase: Toluene-isopropanol-ammonia (7.5:2.5:0.1, v/v/v)
- Standard solution: 20-120 ng/band in methanol
- Forced degradation: Acidic (0.1N HCl), basic (0.1N NaOH), oxidative (3% H₂O₂)
Chromatographic Conditions:
- Stationary phase: Silica gel 60F₂₅₄ TLC plates
- Development: Ascending development to 75 mm at room temperature
- Saturation time: 20 minutes
- Detection: Densitometric scanning at 284 nm
Method Validation:
- Linearity: R² = 0.995 (20-120 ng/band)
- Precision: Intra-day RSD < 1.5%, inter-day RSD < 2.0%
- Forced degradation: Significant degradation in acidic, alkaline, and oxidative conditions
- Greenness assessment: NEMI, AGREE, and White Analytical Chemistry metrics
Sustainability and Greenness Assessment Framework
The transition to green HPTLC aligns with the principles of Green Analytical Chemistry (GAC) and Circular Analytical Chemistry (CAC), addressing the limitations of traditional "take-make-dispose" analytical models [25]. The environmental impact of analytical methods can be quantified using multiple assessment tools:
Analytical Method Greenness Score (AMGS): This comprehensive metric evaluates environmental impact across dimensions including solvent energy consumption during production and disposal, safety/toxicity profiles, and instrument energy consumption [26].
Multi-Metric Assessment Approach:
- AGREE: Provides a comprehensive 0-1 score based on twelve green chemistry principles
- Analytical Eco-Scale: Penalty point system with higher scores indicating better greenness
- GAPI: Visual assessment using color-coded pentagrams
- NEMI: Simple pictogram indicating method environmental profile
Regulatory Context: Recent assessments of 174 standard methods from CEN, ISO, and Pharmacopoeias revealed that 67% scored below 0.2 on the AGREEprep scale, highlighting the urgent need for greener method adoption in regulated environments [25].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Green HPTLC Method Development
| Reagent/Material | Specification | Function | Green Alternatives |
|---|---|---|---|
| HPTLC Plates | Silica gel 60 F₂₅₄, 0.25 mm thickness | Stationary phase for separation | RP-18 for reversed-phase methods |
| Ethanol | HPLC grade | Green solvent for mobile phase | Can replace methanol or acetonitrile |
| Ethyl Acetate | HPLC grade | Medium-polarity solvent | Preferred over chlorinated solvents |
| Water | Purified (Milli-Q grade) | Aqueous component | Solvent with minimal environmental impact |
| Ammonia Solution | 25-33% | Modifier for peak symmetry | Volatile, easier to remove than buffers |
| Acetic Acid | Glacial, HPLC grade | Acidic modifier | Green alternative to TFA |
| Standard Compounds | Pharmacopoeial standards | Method development and validation | Certified reference materials |
Strategic Implementation Workflows
Diagram 1: Strategic Framework for Green HPTLC Adoption Decision-Making. This workflow illustrates the integrated assessment of sustainability, economic, and performance factors driving adoption decisions.
The adoption of green HPTLC methods represents a strategic imperative for pharmaceutical analysis, driven by the powerful convergence of environmental responsibility, economic efficiency, and analytical reliability. The quantitative data presented demonstrates that modern HPTLC methodologies can achieve excellent greenness profiles (AGREE scores >0.8) while maintaining rigorous analytical performance standards required for pharmaceutical quality control. The provided protocols and assessment frameworks offer researchers practical tools for implementation, supporting the pharmaceutical industry's transition toward more sustainable analytical practices without compromising data quality or regulatory compliance. As the field continues to evolve, the integration of green chemistry principles with robust method validation will remain essential for advancing both environmental stewardship and pharmaceutical quality assurance.
Developing and Applying Robust Green HPTLC Methods
The pharmaceutical sector is increasingly adopting green solvents as environmentally friendly substitutes for conventional solvents in response to rising ecological concerns and regulatory restrictions [30]. In High-Performance Thin-Layer Chromatography (HPTLC), mobile phase selection represents a significant opportunity to incorporate green chemistry principles by reducing toxic solvent consumption, minimizing waste generation, and improving operator safety [31]. Eco-friendly HPTLC methods align with the broader objectives of sustainable drug development by maintaining analytical performance while reducing environmental impact [30].
Green solvents for chromatographic applications are characterized by their low toxicity, biodegradability, and renewable origin [30]. The alignment of HPTLC with green analytical chemistry stems from its inherent advantages, including low solvent consumption per analysis, minimal sample preparation, and parallel processing of multiple samples, which significantly reduces environmental footprint compared to other chromatographic techniques [32] [33]. Modern green HPTLC methods have demonstrated successful application across various pharmaceutical analyses, including caffeine quantification in energy drinks [32], tenoxicam determination in commercial formulations [33], and simultaneous quantification of combination drugs [34].
Eco-Friendly Solvent Alternatives for Mobile Phases
Classification and Properties of Green Solvents
Green solvents for HPTLC mobile phases can be categorized into several classes based on their origin and properties. Bio-based solvents such as ethyl lactate, limonene, and ethanol derived from renewable biomass offer advantages of biodegradability with low volatile organic compound (VOC) emissions [30]. Water-based solvent systems modified with small percentages of ethanol or acetone provide non-flammable and non-toxic alternatives for polar compounds [30]. Deep eutectic solvents (DES) created by combining hydrogen bond donors and acceptors have unique properties suitable for extraction and chromatographic separation processes [30].
The greenness profile of HPTLC methods can be quantitatively evaluated using metrics such as the Analytical GREEnness (AGREE) scale, which assesses methods against all twelve principles of green analytical chemistry [32] [33]. Methods employing ethanol-water mobile phases have demonstrated high AGREE scores of 0.80 for caffeine analysis and 0.75 for tenoxicam determination, confirming their excellent environmental profile [32] [33].
Solvent Selection Guide
Table 1: Eco-Friendly Solvent Alternatives for HPTLC Mobile Phases
| Solvent Category | Specific Solvents | Polarity Index | Green Attributes | Common Applications |
|---|---|---|---|---|
| Alcohols | Ethanol, Isopropanol | 5.2 (EtOH), 4.3 (IPA) | Renewable, low toxicity, biodegradable | Primary modifier for medium-polarity compounds [32] [33] |
| Water | Deionized Water | 9.0 | Non-toxic, non-flammable, renewable | Base solvent for reverse-phase systems [32] |
| Esters | Ethyl Acetate | 4.3 | Biodegradable, low bioaccumulation | Medium-polarity modifier [33] |
| Ketones | Acetone | 5.4 | Low toxicity, recyclable | Alternative to acetonitrile [33] |
| Hydrocarbons | Cyclohexane | 0.2 | Reusable, low aquatic toxicity | Non-polar modifier [33] |
Table 2: Mobile Phase Systems for Specific Compound Classes
| Compound Class | Recommended Green Mobile Phase | Ratio (v/v/v) | Stationary Phase | Rf Range |
|---|---|---|---|---|
| Pseudo-alkaloids (Caffeine) | Ethanol-Water | 55:45 | RP-18 F254S | 0.2-0.8 [32] |
| NSAIDs (Tenoxicam) | Ethanol-Water-Ammonia | 50:45:5 | Silica Gel 60 F254 | 0.85 [33] |
| Flavonoids | Toluene-Ethyl Acetate-Formic Acid | 3:7:0.1 | Silica Gel 60 F254 | 0.3-0.7 [35] |
| Combination Drugs | Ethanol-Methylene Chloride-Ammonia | 7:2.5:0.5 | Silica Gel 60 F254 | 0.2-0.8 [34] |
Systematic Method Development Protocol
Initial Solvent System Screening
A systematic approach to green mobile phase optimization begins with screening eco-friendly solvents based on analyte characteristics. The PRISMA model (Polarity-Ratio-Index-Systematic-Mobile-phase-Addition) provides a structured framework for testing ternary solvent mixtures to identify optimal conditions [36]. Initial screening should evaluate binary and ternary combinations of green solvents such as ethanol-water, acetone-water, and ethanol-water-ammonia in varying proportions [33].
For normal-phase HPTLC, the eluotropic series of green solvents guides initial selection, with polarity increasing from cyclohexane through ethyl acetate to ethanol [36]. Target Rf values between 0.2-0.8 provide reliable identification and quantification, with optimal separations often achieved at Rf = 0.4 for the compound of interest [36]. Systematic optimization should test three different polarities with ternary solvent mixtures to balance separation efficiency and analysis time [36].
Optimization and Validation Procedures
After initial screening, method optimization focuses on improving resolution, spot morphology, and analysis time. Chamber saturation for a minimum of 20 minutes is critical for reproducible development, particularly for low-polarity mobile phases sensitive to humidity variations [36]. For problematic separations showing tailing or streaking, mobile phase modifiers such as 2-3 drops of triethylamine per 100 mL for basic compounds or acetic acid for acidic compounds can improve peak symmetry [36].
The optimized method must be validated according to ICH guidelines for linearity, accuracy, precision, robustness, and sensitivity [32] [33]. Validation should include stress testing under acidic, basic, thermal, and oxidative conditions to demonstrate specificity and stability-indicating properties [33]. The greenness profile should be quantitatively assessed using the AGREE metric or complementary green assessment tools [32] [23].
Experimental Protocols
Protocol 1: Green HPTLC for Caffeine Analysis
This protocol details the determination of caffeine in commercial energy drinks and pharmaceutical formulations using an ethanol-water mobile phase [32].
Materials and Reagents:
- Caffeine standard (potency: 98.8%)
- Absolute ethanol (HPLC-grade)
- Deionized water
- HPTLC plates: RP-18 F254S plates
- Commercial energy drinks and pharmaceutical formulations
Mobile Phase Preparation: Prepare ethanol-water in ratio 55:45 (v/v). Measure 55 mL of absolute ethanol and 45 mL of deionized water using graduated cylinders. Transfer to a mobile phase bottle and mix thoroughly by shaking. Degas by sonication for 5 minutes.
Standard Solution Preparation:
- Accurately weigh 10 mg of caffeine standard and transfer to 100 mL volumetric flask.
- Dissolve in and dilute to volume with ethanol-water (55:45) to obtain 100 μg/mL stock solution.
- Prepare working standards in concentration range of 50-800 ng/band by serial dilution.
Sample Preparation: For Energy Drinks:
- Degas commercial energy drinks using ultrasonic bath for 10 minutes.
- Lyophilize degassed samples for five days.
- Reconstitute dried samples in methanol-water (25:75 v/v).
- Perform liquid-liquid extraction with chloroform (3 × 50 mL).
- Combine chloroform fractions and evaporate under reduced pressure at 40°C.
- Reconstitute residue in ethanol-water (55:45) for analysis.
For Pharmaceutical Formulations:
- Compute average weight of ten tablets and powder thoroughly.
- Weigh powder equivalent to 10 mg caffeine and extract with chloroform (3 × 70 mL, 30 min each).
- Combine extracts, concentrate under vacuum.
- Reconstitute in ethanol-water (55:45) to final concentration.
Chromatographic Conditions:
- Stationary phase: RP-18 F254S plates
- Mobile phase: Ethanol-water (55:45 v/v)
- Application volume: 0.5-2.0 μL as 6 mm bands
- Development distance: 80 mm in ADC2 chamber
- * Chamber saturation*: 30 minutes at 22°C
- Detection: Densitometry at 275 nm
Validation Parameters:
- Linearity: 50-800 ng/band (R² > 0.995)
- Precision: RSD <5% for replicate analyses
- Accuracy: 95-105% recovery for spiked samples
- LOD/LOQ: <2.0 ng/band and <5.0 ng/band respectively
Protocol 2: Green HPTLC for Tenoxicam Analysis
This protocol describes the determination of tenoxicam in commercial tablets and capsules using ethanol-water-ammonia mobile phase [33].
Materials and Reagents:
- Tenoxicam standard
- Absolute ethanol (HPLC-grade)
- Deionized water
- Ammonia solution (30%)
- HPTLC plates: Silica Gel 60 F254
Mobile Phase Preparation: Prepare ethanol-water-ammonia in ratio 50:45:5 (v/v/v). Measure 50 mL absolute ethanol, 45 mL deionized water, and 5 mL ammonia solution (30%). Combine in mobile phase bottle and mix thoroughly. Degas by sonication for 5 minutes.
Standard Solution Preparation:
- Accurately weigh 10 mg tenoxicam standard and transfer to 100 mL volumetric flask.
- Dissolve in and dilute to volume with methanol to obtain 100 μg/mL stock solution.
- Prepare working standards in concentration range of 25-1400 ng/band by appropriate dilution.
Sample Preparation: For Tablets/Capsules:
- Weigh and powder twenty tablets/capsules.
- Accurately weigh powder equivalent to 10 mg tenoxicam.
- Transfer to 100 mL volumetric flask with 50 mL methanol.
- Sonicate for 30 minutes with occasional shaking.
- Dilute to volume with methanol and filter through 0.45 μm membrane.
- Dilute filtrate appropriately with mobile phase for analysis.
Chromatographic Conditions:
- Stationary phase: Silica Gel 60 F254 plates
- Mobile phase: Ethanol-water-ammonia (50:45:5 v/v/v)
- Application volume: 1.0-5.0 μL as 6 mm bands
- Development distance: 90 mm in saturated chamber
- Detection: Densitometry at 375 nm
Method Validation:
- Linearity: 25-1400 ng/band (R² > 0.995)
- Accuracy: 98.24-101.48% recovery
- Precision: RSD 0.87-1.02%
- Robustness: RSD 0.87-0.94%
- LOD/LOQ: 0.98 ng/band and 2.94 ng/band respectively
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Materials for Green HPTLC Analysis
| Item Category | Specific Products | Key Specifications | Application & Function |
|---|---|---|---|
| HPTLC Plates | Silica Gel 60 F254, RP-18 F254S | Layer thickness: 0.25 mm (analytical), Particle size: 5-12 μm | Stationary phase for separation [32] [33] |
| Green Solvents | Ethanol, Water, Ethyl Acetate, Acetone | HPLC-grade purity, low UV cutoff | Mobile phase components [32] [33] |
| Application System | CAMAG Automatic Sampler 4 (ATS4) | Application rate: 150 nL/s, Band length: 6 mm | Precise sample application [32] |
| Development Chamber | CAMAG Automatic Developing Chamber 2 (ADC2) | Saturation control, Development distance: 80-90 mm | Controlled mobile phase migration [32] |
| Detection System | CAMAG TLC Scanner 3 with winCATS | Wavelength range: 190-900 nm, Slit dimensions: 4 × 0.45 mm | Densitometric quantification [33] [35] |
| Validation Standards | Certified Reference Materials | Purity: >98%, Potency verification | Method calibration and validation [32] [33] |
Sustainability Assessment and Method Validation
Greenness Evaluation Metrics
The environmental performance of developed HPTLC methods should be quantitatively assessed using multiple metrics. The Analytical GREEnness (AGREE) calculator employs a circular diagram showing performance across all twelve principles of green analytical chemistry, providing a comprehensive score between 0-1 [32] [33]. Complementary assessment tools including GAPI, ECO-SCALE, and whiteness and blueness metrics provide additional perspectives on method sustainability [23] [34].
Methods employing ethanol-water mobile phases typically achieve AGREE scores >0.75, confirming their excellent greenness profile [32] [33]. The green credentials of these methods stem from several factors: ethanol is biodegradable, renewable, and poses minimal toxicity to operators and the environment [32]. Water is inherently non-toxic and non-flammable, enhancing method safety [30]. Additionally, HPTLC's minimal solvent consumption per analysis (typically 10-20 mL per development) significantly reduces waste generation compared to column chromatographic techniques [31].
Troubleshooting and Optimization Guidelines
Common issues in green HPTLC method development include spot tailing, streaking, and inadequate resolution. Spot tailing often results from acidic or basic functional groups interacting with active sites on the stationary phase; adding 2-3 drops of triethylamine per 100 mL mobile phase for basic compounds or acetic acid for acidic compounds can suppress ionization and improve spot morphology [36]. Streaking typically indicates sample overloading and requires reduction of application volume or sample concentration [36].
For inadequate resolution, systematic mobile phase adjustment is necessary. If compounds co-elute (Rf difference <0.15), decrease mobile phase polarity to increase retention differences. If compounds remain near the origin (Rf values <0.2), increase polarity to enhance migration [36]. Method robustness should be verified by deliberate variations in mobile phase composition (±2%), development distance (±5 mm), and chamber saturation time (±5 minutes) [32].
The strategic selection of eco-friendly solvent systems for HPTLC mobile phases represents a significant advancement toward sustainable pharmaceutical analysis. Methodologies employing ethanol-water and ethanol-water-ammonia systems have demonstrated excellent chromatographic performance while minimizing environmental impact [32] [33]. The successful application of these green methods to diverse pharmaceuticals confirms their reliability for routine analysis in quality control and research settings.
Future developments in green HPTLC will likely focus on hybrid solvent systems incorporating novel green solvents such as natural deep eutectic solvents (NADES) and improved bio-based solvents [30] [31]. The integration of computational methods for predicting solvent selectivity and retention behavior will further streamline method development [30]. Additionally, the coupling of HPTLC with renewable energy-powered instrumentation may provide further sustainability improvements. As regulatory emphasis on green chemistry continues to grow, the adoption of eco-friendly HPTLC methods will become increasingly essential for pharmaceutical laboratories committed to sustainable practices.
In High-Performance Thin-Layer Chromatography (HPTLC), the stationary phase forms the foundational component upon which separation science is built. Traditional plates, such as those pre-coated with silica gel 60 F₂₅₄, have long been the workhorse of pharmaceutical analysis, prized for their robustness and well-characterized normal-phase separation mechanisms [37] [38]. These conventional phases operate primarily through adsorption chromatography, where analytes interact with active silanol groups on the silica surface, enabling separation based on polarity differences [37]. The inherent simplicity, cost-effectiveness, and compatibility with a wide range of mobile phases make these layers particularly suitable for method development in regulated environments, where they meet the guidelines for validated analytical methods in current good laboratory practice (cGLP) and current good manufacturing practice (cGMP) [37].
Recent innovations have transformed this landscape through the introduction of advanced materials, particularly Metal-Organic Frameworks (MOFs). MOFs are crystalline porous materials consisting of metal ions or clusters coordinated with organic linkers, creating structures with exceptionally high surface areas and tunable pore geometries [11]. The integration of MOFs into HPTLC stationary phases represents a paradigm shift, enhancing selectivity through specific host-guest interactions, molecular sieving, and surface functionality that can be customized for target analyte classes [11]. This innovation addresses key analytical challenges in complex pharmaceutical matrices, where lipid interference and pigment overlap often obscure band resolution in traditional systems [11]. The evolution from conventional to MOF-modified plates aligns with the broader objectives of green analytical chemistry by potentially reducing solvent consumption through improved efficiency and enabling more sensitive detection at trace levels [11].
Conventional Stationary Phases in Pharmaceutical Analysis
Types and Properties
Conventional HPTLC stationary phases predominantly include silica gel, reversed-phase C18, and cellulose layers, each offering distinct separation mechanisms tailored to different analytical needs [37]. Silica gel plates remain the most widely used normal-phase stationary phase, characterized by their hydrophilic nature and surface silanol groups that facilitate hydrogen bonding and dipole-dipole interactions with analytes [37]. The standard silica gel 60 F₂₅₄ features a mean particle size of approximately 5-6 μm with a narrow distribution, significantly finer than traditional TLC, which enhances resolution, spot compactness, and separation efficiency [39]. The "F₂₅₄" designation indicates the incorporation of a fluorescent indicator, enabling UV detection at 254 nm through fluorescence quenching [37].
Reversed-phase plates, typically modified with C18 (octadecylsilane) or C8 chains, provide an alternative hydrophobic interaction mechanism where the stationary phase is less polar than the mobile phase [37] [4]. These phases are particularly valuable for separating non-polar to moderately polar compounds and are often employed with hydro-organic mobile phases containing methanol, acetonitrile, or tetrahydrofuran mixed with water [37]. The dual-layer plates (Multi-K, Whatman/GE Healthcare), which combine adjacent C18 and silica layers on a single plate, enable orthogonal separation in two-dimensional development, significantly improving resolution for complex mixtures [37].
Cellulose layers, derived from natural polymer sources, offer a different separation environment based on partition chromatography and are particularly suited to separating hydrophilic compounds, including amino acids, sugars, and inorganic ions [37]. The hydrophilic nature and chiral recognition properties of native cellulose make it valuable for certain enantioselective separations without additional chiral modifiers.
Table 1: Properties and Applications of Conventional HPTLC Stationary Phases
| Stationary Phase Type | Separation Mechanism | Common Applications | Typical Mobile Phases |
|---|---|---|---|
| Silica Gel 60 F₂₅₄ | Adsorption (normal-phase) | Pharmaceutical compounds, natural products, clinical samples [37] [39] | Toluene-acetone-methanol mixtures, chloroform-methanol [38] [16] |
| Reversed-Phase (C18) | Partition (reversed-phase) | Non-polar to moderately polar drugs, environmental contaminants [37] [4] | Methanol-water, acetonitrile-water, tetrahydrofuran-water [37] [4] |
| Cellulose | Partition (normal-phase) | Hydrophilic compounds, amino acids, sugars [37] | n-Butanol-acetic acid-water, salt solutions [37] |
Experimental Protocol: Method Development on Conventional Phases
Protocol Title: HPTLC Method Development and Validation for Pharmaceutical Compounds Using Conventional Silica Gel Plates
Principle: This protocol describes systematic method development for analyzing pharmaceutical compounds using silica gel 60 F₂₅₄ plates, with application to mycophenolate mofetil quantification as a representative model [38].
Materials and Reagents:
- HPTLC plates: Aluminum plates precoated with silica gel 60 F₂₅₄, 250 μm thickness (E. Merck, Darmstadt, Germany) [38]
- Sample applicator: Automated applicator (Camag Linomat V) equipped with 100 μL syringe [38]
- Development chamber: Twin trough glass chamber 10 × 10 cm (Camag) [38]
- Scanner: Densitometric scanner (Camag TLC Scanner 3) with WinCATS software [38]
- Mycophenolate mofetil standard (pharmaceutical grade) [38]
- Mobile phase components: Toluene, acetone, methanol (analytical grade) [38]
- Methanol for sample preparation (HPLC grade) [38]
Procedure:
- Plate Pre-washing and Activation: Pre-wash HPTLC plates with methanol. Activate at 110°C for 5 minutes before sample application [38].
- Standard Solution Preparation: Accurately weigh 10 mg of mycophenolate mofetil reference standard. Dissolve in methanol and transfer to a 10 mL volumetric flask. Dilute to volume with methanol to obtain a stock solution of 1 mg/mL. Prepare working standards in the concentration range of 100-500 ng/μL by appropriate dilution [38].
- Sample Application: Using the automated applicator, apply standards and samples as bands 6 mm wide and 10 mm apart, with application rate of 150 nL/s. Position bands 10 mm from the bottom and side edges of the plate [38].
- Chromatographic Development: Prepare mobile phase of toluene-acetone-methanol (6:2:2 v/v/v). Add to twin-trough chamber and saturate for 10 minutes. Develop plate in ascending mode to a distance of 8.5 cm at room temperature (approximately 25°C) [38].
- Post-Chromatography Processing: Dry developed plate in a current of air using an air-blowing drier [38].
- Detection and Quantification: Perform densitometric scanning at 254 nm using deuterium lamp. Set slit dimensions to 5 × 0.45 mm and scanning speed to 20 mm/s [38].
- Data Analysis: Construct calibration curve by plotting peak areas against corresponding amounts (100-500 ng/band). Determine unknown concentrations from the calibration curve [38].
Validation Parameters:
- Linearity: Verify correlation coefficient (r² ≥ 0.999) across the working range [38]
- Precision: Evaluate intra-day and inter-day precision (%RSD < 2%) [38]
- Accuracy: Perform recovery studies at 50%, 100%, and 150% levels (target recovery: 98-102%) [38]
- Specificity: Confirm absence of interference from excipients or degradation products [38]
Diagram 1: HPTLC Method Development Workflow for Conventional Stationary Phases. This flowchart illustrates the sequential steps involved in pharmaceutical analysis using conventional silica gel plates, from plate preparation through to data analysis [38].
MOF-Modified Stationary Phases
Principles and Advancements
Metal-Organic Frameworks represent a class of coordination polymers consisting of metal ions or clusters connected by organic linkers to form one-, two-, or three-dimensional porous structures [11]. Their integration into HPTLC stationary phases leverages several unique properties: exceptionally high surface areas (often exceeding 7000 m²/g), tunable pore sizes from micropores to mesopores, and customizable surface functionality that can be tailored to specific separation needs [11]. Unlike conventional stationary phases that rely on relatively non-specific adsorption or partition mechanisms, MOF-modified plates exhibit selective separation based on molecular sieving, host-guest interactions, and specific chemical affinities that can be precisely engineered at the molecular level [11].
The modular architecture of MOFs allows for strategic design to target specific analyte classes relevant to pharmaceutical analysis. By selecting appropriate metal clusters (e.g., Zn²⁺, Cu²⁺, Zr⁺) and functionalized organic linkers, MOF stationary phases can be engineered for specific applications such as chiral separations, isolation of polar compounds, or selective capture of trace contaminants in complex matrices [11]. This material-enabled enhancement significantly improves the detection of trace-level contaminants in complex food and pharmaceutical matrices, where conventional stationary phases often suffer from matrix interference effects [11]. The enhanced selectivity of MOF-modified plates can reduce the need for extensive sample cleanup and enable more precise quantification of low-abundance analytes.
Synthesis and Characterization Protocols
Protocol Title: Preparation and Evaluation of MOF-Modified HPTLC Plates
Principle: This protocol describes the in-situ growth of zeolitic imidazolate framework-8 (ZIF-8) on silica gel HPTLC plates to create MOF-modified stationary phases with enhanced separation capabilities for pharmaceutical compounds [11].
Materials and Reagents:
- Pre-coated HPTLC silica gel 60 F₂₅₄ plates (E. Merck, Germany) [11]
- Zinc nitrate hexahydrate (Zn(NO₃)₂·6H₂O), analytical grade [11]
- 2-Methylimidazole, 99% purity [11]
- Methanol, HPLC grade [11]
- N,N-dimethylformamide (DMF), analytical grade [11]
- Ultrasonic cleaning bath
- Vacuum oven
Procedure:
- Plate Pre-treatment: Cut HPTLC plates to desired size (typically 5 × 5 cm). Clean plates ultrasonically in methanol for 15 minutes to remove impurities. Dry at 80°C under vacuum for 1 hour [11].
- MOF Precursor Solution Preparation:
- Solution A: Dissolve 2.93 g Zn(NO₃)₂·6H₂O in 100 mL methanol
- Solution B: Dissolve 3.24 g 2-methylimidazole in 100 mL methanol [11]
- In-situ MOF Growth: Combine solutions A and B in a 1:1 ratio. Immediately immerse pre-treated HPTLC plates in the mixture. Allow reaction to proceed at room temperature for 6 hours with gentle agitation [11].
- Post-treatment: Remove plates from reaction mixture and rinse thoroughly with methanol to remove unreacted precursors. Dry plates at 100°C under vacuum for 12 hours to activate the MOF-modified stationary phase [11].
- Characterization:
- Morphological Analysis: Examine surface morphology using scanning electron microscopy (SEM) to verify uniform MOF crystal distribution and estimate crystal size [11].
- Chemical Characterization: Perform Fourier-transform infrared spectroscopy (FTIR) to confirm presence of characteristic ZIF-8 absorption bands [11].
- Porosity Assessment: Analyze nitrogen adsorption-desorption isotherms to determine specific surface area and pore size distribution [11].
- Chromatographic Performance Evaluation: Test separation efficiency using standard analyte mixtures and compare performance to unmodified silica gel plates [11].
Quality Control Parameters:
- MOF coating uniformity: SEM should show homogeneous crystal distribution without aggregation [11]
- Stability: MOF layer should remain intact after multiple development cycles with common mobile phases [11]
- Enhancement factor: Compare separation efficiency and selectivity to unmodified plates using challenging analyte pairs [11]
Table 2: Comparison of Conventional Silica Gel and MOF-Modified Stationary Phases
| Parameter | Conventional Silica Gel | MOF-Modified Plates |
|---|---|---|
| Surface Area | Moderate (300-500 m²/g) | Very high (700-5000 m²/g) [11] |
| Separation Mechanism | Adsorption (polar interactions) | Molecular sieving, host-guest interactions, specific affinity [11] |
| Selectivity Tunability | Limited (mobile phase dependent) | Highly tunable (metal cluster and linker selection) [11] |
| Matrix Tolerance | Moderate (susceptible to interference) | High (enhanced selectivity reduces matrix effects) [11] |
| Analysis Cost | Low | Moderate to high (synthesis dependent) |
| Method Development Complexity | Low to moderate | High (requires optimization of MOF chemistry) |
Diagram 2: MOF-Modified HPTLC Plate Fabrication Process. This workflow illustrates the sequential steps for synthesizing and characterizing metal-organic framework modified stationary phases, from precursor preparation to final performance evaluation [11].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Reagents for HPTLC Stationary Phase Research
| Item | Function/Application | Representative Examples |
|---|---|---|
| Conventional HPTLC Plates | Normal-phase separation backbone | Silica gel 60 F₂₅₄ plates (E. Merck) [38] [16] |
| Reversed-Phase HPTLC Plates | Hydrophobic interaction separations | RP-18F₂₅₄S plates (Merck) [4] |
| MOF Precursors | Stationary phase functionalization | Zinc nitrate, 2-methylimidazole, copper acetate, terephthalic acid [11] |
| Mobile Phase Components | Chromatographic development | Toluene, ethyl acetate, chloroform, methanol, ammonia solutions [38] [16] [4] |
| Detection Reagents | Zone visualization and derivatization | Iodine, ninhydrin, sulfuric acid (charring), fluorescence inducers [37] |
| Reference Standards | Method development and validation | Pharmaceutical grade active ingredients (e.g., mycophenolate mofetil, dapagliflozin, bisoprolol fumarate) [38] [16] |
Green Analytical Chemistry and Method Validation
The evolution of stationary phases aligns with the growing emphasis on Green Analytical Chemistry (GAC) principles in pharmaceutical analysis. HPTLC inherently supports several GAC principles through minimal solvent consumption (typically <10 mL per analysis), capacity for parallel sample processing, and reduced energy requirements compared to column chromatography techniques [11]. Modern greenness assessment tools, including the Modified Green Analytical Procedure Index (MoGAPI), Analytical GREEnness (AGREE), and Analytical Eco-Scale, provide quantitative metrics to evaluate the environmental impact of analytical methods [16] [4] [11].
The integration of MOF-modified stationary phases further enhances the greenness profile of HPTLC methods through improved separation efficiency, which can reduce the need for solvent-intensive mobile phases or extensive sample cleanup procedures [11]. The ability to selectively capture target analytes from complex matrices minimizes waste generation and reduces overall chemical consumption throughout the analytical workflow [11]. When combined with green solvent systems (such as ethanol-water mixtures in reversed-phase applications), MOF-modified plates contribute to more sustainable pharmaceutical analysis while maintaining the high-throughput capabilities essential for modern drug development [4].
Method validation for both conventional and MOF-modified HPTLC methods follows ICH Q2(R2) guidelines, encompassing parameters such as linearity, precision, accuracy, specificity, and robustness [38] [16]. The stability-indicating capability of these methods is particularly crucial for pharmaceutical applications, requiring demonstration of specificity in the presence of degradation products formed under various stress conditions [16]. The complementary selectivity offered by different stationary phases enhances the reliability of pharmaceutical analysis, with conventional silica gel providing robust normal-phase separations and MOF-modified plates offering tailored selectivity for challenging separation problems [37] [11].
Stationary phase innovation in HPTLC represents a dynamic field bridging fundamental separation science with practical pharmaceutical applications. Conventional silica gel plates continue to offer reliable, cost-effective solutions for routine analysis, while MOF-modified stationary phases provide unprecedented opportunities for selective separations through engineered materials design. The integration of these advanced stationary phases within the framework of green analytical chemistry principles supports the development of sustainable, robust methods for pharmaceutical quality control and drug development. As stationary phase technology continues to evolve, the synergy between material science and separation fundamentals will undoubtedly yield further innovations, enhancing the capabilities of HPTLC as a versatile analytical platform for modern pharmaceutical analysis.
In the development of green High-Performance Thin-Layer Chromatography (HPTLC) methods for pharmaceutical analysis, achieving reproducibility is paramount for method validation and regulatory acceptance. Reproducibility ensures that analytical results remain consistent across different laboratories, analysts, instruments, and time periods, establishing reliability for quality control applications. The critical operational phases where reproducibility must be carefully controlled are sample application and chromatographic development. Even in eco-friendly methods that utilize safer solvents, without strict control of these parameters, method validation fails, compromising the entire analytical workflow. This protocol details the optimized procedures and controlled variables essential for achieving reproducible results in green HPTLC pharmaceutical analysis.
Critical Factors Influencing Reproducibility
Fundamental HPTLC Parameters
The reproducibility of HPTLC analysis depends on numerous interconnected factors throughout the analytical process. Studies have demonstrated that uncontrolled or poorly defined variables significantly impact the developed plate, leading to inconsistent retardation factor (Rf) values and band resolution [40]. The fundamental parameters requiring standardization include:
- Plate characteristics: Stationary phase consistency, pre-elution treatments, and layer uniformity
- Application parameters: Spotting technique, band dimensions, and application volume
- Development conditions: Chamber saturation, mobile phase composition, and development distance
- Environmental factors: Temperature, humidity, and solvent stability [40]
When these variables are properly defined and controlled, HPTLC transitions from a merely comparative technique to a reproducible quantitative analytical tool suitable for pharmaceutical quality control and regulatory compliance [40] [41].
The Reproducibility Challenge
The limited use of investigative HPTLC analysis in some scientific fields stems primarily from the range of uncontrolled or poorly defined variables that affect the reproducibility of the developed plate [40]. Research involving over fifty HPTLC plates and a six-component test dye mixture has demonstrated that through systematic control of critical parameters, laboratories can achieve reproducible data suitable for building reference libraries and making significant analytical comparisons [40]. This systematic approach is equally applicable to pharmaceutical analysis, where method validation requires consistent performance across different batches and analysts.
Sample Application Protocol
Instrumentation and Materials
Table 1: Essential Research Reagent Solutions for Sample Application
| Item | Specification | Function | Example Sources |
|---|---|---|---|
| HPTLC Plates | Silica gel 60 F254, 20×10 cm or 10×10 cm, layer thickness 0.2-0.25 mm | Stationary phase for separation | Merck, Germany [42] [43] |
| Application Syringe | CAMAG Hamilton syringe, 100 μL capacity | Precise sample deposition | Bonaduz, Switzerland [42] |
| Sample Applicator | CAMAG Linomat IV/V semiautomatic applicator | Automated band application for reproducibility | Muttenz, Switzerland [42] [23] |
| Methanol (HPLC Grade) | ≥99.9% purity | Primary solvent for standard/sample preparation | Sigma-Aldrich Chemicals [42] |
Standard Solution Preparation
For pharmaceutical analysis, prepare standard solutions according to the following optimized protocol:
Primary Stock Solution: Precisely weigh 10 mg of reference standard and transfer to a 10 mL volumetric flask. Dissolve and make to volume with methanol to achieve 1000 μg/mL concentration [42]. For compounds with poor solubility in methanol, brief sonication for 15-20 minutes may be employed [43].
Working Standard Solutions: Prepare serial dilutions from stock solution using methanol to cover the validated linearity range. For simultaneous estimation of multiple compounds, mixed working standards may be prepared, such as combining 1 mL of dapagliflozin stock with 10 mg of vildagliptin in a 10 mL volumetric flask [42].
Sample Solution Preparation: For tablet formulations, weigh and powder twenty tablets. Transfer powder equivalent to single dosage unit to volumetric flask, add solvent, and sonicate for 15-20 minutes. Filter through Whatman filter paper No. 41, with thorough washing of residue [43]. Make filtrate to volume and further dilute if necessary.
Sample Application Technique
The sample application process significantly impacts separation efficiency and reproducibility:
Plate Pre-conditioning: Pre-wash plates with methanol if necessary and activate at 110°C for 15-20 minutes before application [43]. Store in desiccator until use.
Application Parameters:
Application Volume: For quantitative analysis, apply volumes between 1-10 μL per band, depending on concentration and detection limits. Volumes below 1 μL may challenge precision, while volumes above 10 μL can cause band broadening [43].
Figure 1: Sample Application Workflow for Reproducible HPTLC Analysis
Chromatographic Development Protocol
Mobile Phase Preparation
Mobile phase selection must balance separation efficiency with green chemistry principles, avoiding carcinogenic solvents like benzene in favor of safer alternatives [42]:
Solvent Selection: Choose solvents that provide adequate resolution while minimizing toxicity. Recent methods successfully utilize combinations like:
Preparation Technique: Measure components volumetrically in separate cylinders before combining in the development chamber. Mix thoroughly and degas by sonication if necessary.
Development Chamber Conditions
Table 2: Chromatographic Development Parameters for Reproducibility
| Parameter | Optimal Condition | Impact on Reproducibility |
|---|---|---|
| Chamber Type | Twin-trough glass chamber | Ensures even solvent distribution [42] |
| Chamber Saturation | 15-30 minutes prior to development [42] [23] | Critical for consistent RF values and band shape [40] |
| Saturation Method | Mobile phase in one trough, plate in dry trough initially | Prevents pre-elution before development |
| Development Distance | 70-80 mm from application point [43] | Must be consistent across analyses [40] |
| Development Temperature | Room temperature (25±2°C) | Minimizes solvent evaporation rate variations |
| Development Mode | Ascending, one-dimensional | Standard for most pharmaceutical applications |
Development Procedure
Chamber Preparation: Pour prepared mobile phase into one trough of twin-trough chamber. For 20×10 cm plates, approximately 20-25 mL mobile phase is sufficient. Ensure chamber is level on a stable surface.
Chamber Saturation: Place filter paper lining on three sides of chamber to enhance saturation. Allow chamber to saturate with mobile phase vapor for 20-30 minutes with lid closed [42] [23]. This saturation period is critical for achieving consistent Rf values [40].
Plate Development: After saturation, place pre-spotted plate in the mobile phase-containing trough. Seal chamber immediately and allow development to proceed until solvent front reaches pre-marked distance (typically 70-80 mm from origin).
Plate Removal and Drying: Immediately remove developed plate from chamber, mark solvent front, and air-dry for 10-15 minutes in a fume hood. For complete removal of residual solvents, use oven drying at 60°C for 2-5 minutes if analytes are thermally stable.
Figure 2: Chromatographic Development Workflow for Reproducible HPTLC
Method Validation Parameters
To confirm reproducibility, the developed method must be validated according to International Council for Harmonisation (ICH) guidelines. Key parameters include:
- Linearity: Typically demonstrated across 80-120% of target concentration with correlation coefficient (R²) ≥0.995 [42] [44]
- Precision: Expressed as %RSD for intra-day (repeatability) and inter-day (intermediate precision) measurements, with acceptance criteria ≤2% [43]
- Robustness: Deliberate, small variations in development conditions should not significantly affect results [43]
- Specificity: Ability to assess analyte unequivocally in presence of components expected to be present [44]
Reproducible HPTLC analysis in pharmaceutical quality control depends on meticulous attention to both sample application and chromatographic development parameters. By standardizing these critical steps—including controlled sample application using semi-automated instruments, mobile phase composition optimized for both separation and environmental safety, standardized chamber saturation protocols, and consistent development distances—researchers can achieve the reproducibility required for valid analytical results. The protocols outlined herein provide a framework for developing green HPTLC methods that deliver consistent, reliable data suitable for pharmaceutical analysis and regulatory submission.
High-Performance Thin-Layer Chromatography (HPTLC) has evolved significantly beyond simple visual analysis through the integration of advanced detection technologies. These technologies provide unparalleled capabilities for the identification, characterization, and quantification of compounds in complex pharmaceutical matrices. The combination of HPTLC with sophisticated detection methods represents a powerful approach that aligns with green analytical chemistry principles by minimizing solvent consumption and waste generation while maximizing analytical information. Modern HPTLC detection systems now include densitometry for precise quantification, Photo-Diode Array (PDA) detection for spectral characterization, and hyphenation with Mass Spectrometry (MS) for structural elucidation [45] [46]. These integrated approaches provide complementary data that ensures comprehensive analysis of pharmaceutical compounds, making them invaluable for quality control, method validation, and regulatory compliance in drug development.
The fundamental advantage of these detection systems lies in their ability to non-destructively analyze compounds on the chromatographic plate, allowing for sequential application of multiple detection techniques to the same separation. This multi-dimensional analytical approach is particularly valuable for the analysis of complex botanical drugs and multi-component pharmaceutical formulations where multiple markers must be monitored simultaneously [10] [46]. Furthermore, the hyphenation of HPTLC with MS creates a robust analytical platform that combines the separation efficiency of planar chromatography with the identification power of mass spectrometry, enabling researchers to obtain both qualitative and quantitative data from a single analysis.
Densitometry in HPTLC
Principles and Technical Specifications
Densitometry serves as the fundamental quantitative detection method in HPTLC, operating on the principle of measuring the absorbance or fluorescence of analyte bands directly on the chromatographic plate. This technique involves scanning the developed HPTLC plate with a light source in the UV or visible range and precisely measuring the reflected or transmitted light using a sensitive detector. The measurement occurs in reflectance mode, where the light reflected from the plate surface is captured, or in transmission mode, where light passing through the plate is measured [42] [47]. Modern densitometers are equipped with deuterium (190-400 nm) and tungsten halogen (400-800 nm) lamps, enabling scanning across a broad wavelength spectrum, with monochromators that provide bandwidth selection typically between 1-20 nm for optimal resolution and sensitivity.
The quantification process involves scanning the entire track or predefined zones of the HPTLC plate and generating a chromatographic profile where the peak areas or heights correlate with analyte concentrations. Contemporary instruments like the CAMAG TLC Scanner 4 offer automatic wavelength programming, allowing for different wavelengths to be applied to various zones on the plate, which is particularly beneficial for analyzing compounds with different absorption maxima in a single development [42] [46]. The data acquisition and processing are managed through sophisticated software such as WinCATS or visionCATS, which provide comprehensive tools for peak integration, baseline correction, and calibration curve generation.
Application Protocols and Validation
Standard Densitometry Protocol for Pharmaceutical Analysis:
- Plate Preparation: Activate pre-coated HPTLC plates (silica gel 60 F254) by heating at 105-110°C for 15-20 minutes prior to sample application [47].
- Sample Application: Apply samples as bands using an automated applicator (e.g., CAMAG Linomat 5) with nitrogen flow, maintaining band lengths of 6-8 mm and application rate of 150 nL/s [42] [47].
- Chromatographic Development: Develop the plate in a twin-trough chamber pre-saturated with mobile phase for 20 minutes at room temperature. Development distance is typically 70-80 mm from the point of application [42] [46].
- Plate Drying: Air-dry the developed plate for 5-10 minutes to completely evaporate the mobile phase.
- Densitometric Scanning: Scan the plate at the appropriate wavelength with a slit dimension of 6.00 × 0.45 mm and scanning speed of 10-20 mm/s [42] [47].
- Data Analysis: Process the chromatograms using integrated software to determine peak areas and generate calibration curves.
Method Validation Parameters: Densitometric methods must be rigorously validated according to ICH guidelines. The table below summarizes typical validation parameters for HPTLC-densitometry methods:
Table 1: Validation Parameters for HPTLC-Densitometry Methods
| Validation Parameter | Experimental Design | Acceptance Criteria | Application Example |
|---|---|---|---|
| Linearity | Analysis of 5-7 concentration levels in triplicate | Correlation coefficient (r²) > 0.995 | Dapagliflozin: 0.6-1.4 µg/band (r²=0.997) [42] |
| Precision | Intra-day (n=3) and inter-day (n=3) analysis at three concentration levels | RSD ≤ 2% | Tenoxicam: RSD < 5% at 100-400 ng/spot [47] |
| Accuracy | Recovery studies at 50%, 100%, 150% of target concentration | Recovery 98-102% | T. cordifolia markers: 98.06-101.56% recovery [46] |
| LOD/LOQ | Signal-to-noise ratio of 3:1 for LOD, 10:1 for LOQ | LOD: 25-50 ng/spot, LOQ: 50-100 ng/spot | Tenoxicam: LOD 25 ng/spot, LOQ 50 ng/spot [47] |
| Robustness | Deliberate variations in mobile phase, development time, etc. | RSD ≤ 2% for altered parameters | Robustness RSD < 2% for T. cordifolia markers [46] |
| Specificity | Analysis in presence of excipients and related compounds | No interference at analyte Rf | Baseline separation of dapagliflozin (Rf=0.57) and vildagliptin (Rf=0.26) [42] |
Green Analytical Chemistry Considerations
Densitometry aligns exceptionally well with green analytical chemistry principles due to its minimal solvent consumption and energy requirements. Unlike HPLC methods that continuously consume mobile phase throughout analysis, HPTLC-densitometry requires only minimal solvent volumes for plate development. A typical HPTLC analysis consumes approximately 10-20 mL of mobile phase compared to 500-1000 mL for comparable HPLC methods [4]. This represents a 95% reduction in solvent consumption, significantly reducing environmental impact and waste disposal requirements. Furthermore, the ability to analyze multiple samples simultaneously on a single plate dramatically improves energy efficiency compared to sequential injection-based techniques.
The greenness of HPTLC-densitometry methods can be quantitatively assessed using tools such as the Analytical GREENness (AGREE) metric, which provides a comprehensive evaluation of environmental impact [4]. Methods utilizing greener solvents like ethanol-water mixtures typically achieve higher AGREE scores (closer to 1.0) compared to those employing chlorinated solvents. For instance, reversed-phase HPTLC methods using ethanol-water mobile phases demonstrate superior environmental profiles compared to normal-phase methods utilizing chloroform-based mobile phases [4].
Diagram 1: HPTLC-Densitometry Workflow (76 characters)
Photo-Diode Array (PDA) Detection
Technical Fundamentals and Spectral Analysis
Photo-Diode Array detection represents a significant advancement in HPTLC detection capabilities by enabling simultaneous multi-wavelength scanning and acquisition of complete UV-Vis spectra directly from the chromatographic plate. Unlike conventional single-wavelength densitometry, PDA detectors employ an array of hundreds of photodiodes that capture absorbance data across a spectral range, typically 190-800 nm, in a single scan [46]. This technology generates three-dimensional data (Rf, absorbance, wavelength) that provides comprehensive spectral information for each compound separated on the HPTLC plate. The fundamental advantage of PDA detection lies in its ability to record complete UV-Vis spectra of individual bands without the need for multiple separations or scanning procedures.
The spectral information acquired through PDA detection serves multiple critical functions in pharmaceutical analysis. First, it enables peak purity assessment by comparing spectra across different regions of a chromatographic band, detecting potential co-elution that might be missed with single-wavelength detection [46]. Second, it facilitates compound identification through spectral matching with reference standards, with modern software calculating match factors to confirm identity. Third, optimal quantification wavelengths can be selected post-separation by reviewing the full spectral data, ensuring maximum sensitivity and minimal interference. For compounds with specific chromophores, PDA detection can target their characteristic absorption maxima, such as 210 nm for dapagliflozin and vildagliptin analysis or 247 nm for 20-β-hydroxyecdysone in Tinospora cordifolia [42] [46].
Application Protocols and Method Optimization
PDA Method Development Protocol:
- Preliminary Spectral Scanning: Apply reference standards and record spectra from 190-800 nm to identify λmax for target analytes.
- Wavelength Optimization: Test multiple wavelengths around the determined λmax to select the optimal quantification wavelength that balances sensitivity and selectivity.
- Spectral Library Development: Create a library of reference spectra for all target compounds under the specific chromatographic conditions.
- Peak Purity Assessment: Configure software parameters for automated peak purity checking, typically using threshold match factors of ≥0.999 for pure peaks.
- Multi-Wavelength Method Establishment: For multi-component analysis, establish wavelength programs that switch wavelengths at specific migration distances to optimize detection for each compound.
Pharmaceutical Applications: PDA detection has been successfully implemented in numerous pharmaceutical analyses. In the analysis of Tinospora cordifolia, a traditional immunomodulatory botanical, PDA detection enabled the quantification of multiple markers including cordifolioside A (UV λmax 221 nm), 20-β-hydroxyecdysone (UV λmax 247 nm), and columbin (UV λmax 210 nm) from a single chromatographic development [46]. The method demonstrated excellent linearity (r² > 0.99) across concentration ranges of 675-2250 ng/band for these markers. Similarly, for the simultaneous analysis of the anti-diabetic drugs dapagliflozin and vildagliptin, detection at 210 nm provided optimal sensitivity for both compounds, with linear ranges of 0.6-1.4 µg/band and 6.0-14 µg/band respectively [42].
Table 2: PDA Detection Parameters for Pharmaceutical Compounds
| Compound/Analyte | Matrix | λmax (nm) | Linearity Range | Correlation (r²) | Reference |
|---|---|---|---|---|---|
| Dapagliflozin | Pharmaceutical formulation | 210 | 0.6-1.4 µg/band | 0.997 | [42] |
| Vildagliptin | Pharmaceutical formulation | 210 | 6.0-14 µg/band | 0.998 | [42] |
| Cordifolioside A | T. cordifolia extract | 221 | 750-2250 ng/band | >0.99 | [46] |
| 20-β-hydroxyecdysone | T. cordifolia extract | 247 | 750-2250 ng/band | >0.99 | [46] |
| Columbin | T. cordifolia extract | 210 | 675-1875 ng/band | >0.99 | [46] |
| Tenoxicam | Microemulsion gels | 379 | 100-400 ng/spot | Linear | [47] |
| Florfenicol | Bovine tissue | 230 | 0.50-9.00 µg/band | Linear | [23] |
| Meloxicam | Bovine tissue | 230 | 0.03-3.00 µg/band | Linear | [23] |
Greenness Assessment of PDA Methods
The environmental impact of HPTLC-PDA methods can be systematically evaluated using multiple greenness assessment tools. The National Environmental Method Index (NEMI) provides a simple pictogram indicating whether a method incorporates hazardous chemicals [4]. Methods utilizing green solvents like ethanol-water mixtures typically achieve full NEMI compliance. The Analytical Eco-Scale assigns penalty points to non-green parameters, with scores above 75 representing excellent greenness [4]. HPTLC-PDA methods frequently achieve high Eco-Scale scores due to minimal reagent consumption and energy usage. The AGREE metric provides a comprehensive 0-1 score based on all twelve principles of green analytical chemistry, with HPTLC methods typically outperforming HPLC alternatives [4].
For instance, a reversed-phase HPTLC method for ertugliflozin utilizing ethanol-water (80:20 v/v) as mobile phase demonstrated superior greenness profiles compared to normal-phase methods using chloroform-methanol mixtures [4]. The AGREE scores were 0.81 for the reversed-phase method versus 0.65 for the normal-phase approach, highlighting the significant environmental advantages of greener solvent systems. This alignment with green analytical chemistry principles makes HPTLC-PDA an environmentally responsible choice for pharmaceutical analysis while maintaining rigorous analytical performance.
HPTLC-MS Hyphenation
Technical Configuration and Interface Design
HPTLC-MS hyphenation represents the most advanced detection approach, combining the separation power of planar chromatography with the structural elucidation capabilities of mass spectrometry. The technical configuration involves specialized interfaces that enable direct extraction and transfer of analytes from the HPTLC plate to the mass spectrometer. Two primary interface designs dominate current applications: the TLC-MS Interface 2 from CAMAG, which uses a fixed elution head that seals against the plate surface, and open-port sampling interfaces that employ liquid extraction surfaces [46]. Both systems enable solvent-based extraction of individual zones with subsequent transfer to the MS ion source, typically using flow rates of 0.1-0.3 mL/min and extraction times of 30-120 seconds per zone.
The mass spectrometers coupled with HPTLC systems range from single quadrupole instruments for simple confirmation to tandem triple quadrupole and Q-TOF systems for sophisticated structural characterization and method development. Electrospray Ionization (ESI) is the most prevalent ionization technique due to its compatibility with the liquid extracts and ability to handle a broad range of compound polarities [46]. Atmospheric Pressure Chemical Ionization (APCI) and Atmospheric Pressure Photoionization (APPI) serve as complementary techniques for less polar compounds. The HPTLC-MS interface must maintain robust connection between the planar chromatography separation and the MS detection while addressing challenges related to matrix effects from the stationary phase and the need for MS-compatible mobile phases that avoid non-volatile salts and buffers.
Experimental Protocols and Applications
HPTLC-ESI-MS/MS Protocol for Botanical Marker Analysis:
- Chromatographic Separation: Develop HPTLC plates using MS-compatible mobile phases (avoiding non-volatile buffers) and document under UV light at 254 and 366 nm [46].
- Zone Selection: Mark zones of interest based on reference standards and derivatization responses (if applicable).
- Interface Configuration: Set elution solvent composition (typically methanol or acetonitrile with 0.1% formic acid), flow rate (0.2 mL/min), and extraction time (60 seconds).
- MS Parameter Optimization: Optimize ESI parameters (capillary voltage, cone voltage, desolvation temperature) using reference standards.
- Data-Dependent Acquisition: Program MS to switch from survey scan to product ion scan when target ions exceed intensity thresholds.
- Data Interpretation: Correlate MS spectra with chromatographic Rf values and UV spectra for comprehensive compound identification.
Representative Applications: HPTLC-MS has been successfully applied to the analysis of complex botanical extracts and pharmaceutical formulations. In the characterization of Tinospora cordifolia, HPTLC-ESI-MS/MS enabled the identification and confirmation of cordifolioside A (m/z = 527 [M + Na]+), 20-β-hydroxyecdysone (m/z = 481.30 [M + H]+), and columbin (m/z = 359 [M + H]+) directly from the stem extracts [46]. The method provided both qualitative identification through mass spectral data and quantitative analysis through densitometric measurement, demonstrating the power of this hyphenated approach. Similarly, HPTLC-MS methods have been developed for pharmaceutical compounds including anti-diabetics, antibiotics, and anti-inflammatory drugs, allowing for both active pharmaceutical ingredient quantification and impurity profiling [45] [23].
Table 3: HPTLC-MS Applications in Pharmaceutical Analysis
| Application Area | Analytes | Interface Type | Ionization Mode | Key Ions (m/z) | Reference |
|---|---|---|---|---|---|
| Botanical Drug Standardization | Cordifolioside A, 20-β-hydroxyecdysone, Columbin | ESI-MS/MS | Positive | 527 [M+Na]+, 481 [M+H]+, 359 [M+H]+ | [46] |
| Veterinary Drug Residues | Florfenicol, Meloxicam | ESI-MS (hyphenated) | Not specified | Not specified | [23] |
| Anti-diabetic Drugs | Dapagliflozin, Vildagliptin | Not specified | Not specified | Not specified | [42] |
| Pharmaceutical Impurities | Degradation products | Various | ESI/APCI | Compound-dependent | [45] |
| Metabolite Profiling | Plant metabolites | HPTLC-ESI-MS/MS | Positive/Negative | Variable | [46] |
Greenness and Sustainability of HPTLC-MS Methods
HPTLC-MS methods demonstrate exceptional environmental performance when evaluated using modern greenness assessment tools. The ChlorTox tool, which assesses both chlorine content and toxicity of method components, typically shows significantly better scores for HPTLC-MS compared to conventional HPLC-MS methods due to substantially lower solvent consumption [4]. A comprehensive greenness assessment using multiple metrics (NEMI, AES, ChlorTox, AGREE) demonstrated that reversed-phase HPTLC methods utilizing ethanol-water mobile phases outperform normal-phase methods and most HPLC-based approaches [4]. The ability to analyze multiple samples on a single plate drastically reduces solvent waste, with typical solvent consumption of 10-15 mL for 10-15 samples compared to 500-1000 mL for equivalent HPLC analyses.
The hyphenation of HPTLC with MS further enhances the greenness profile by enabling comprehensive analysis without multiple separate techniques. The non-destructive nature of initial HPTLC analysis allows for sequential application of different detection methods (visualization, densitometry, PDA) followed by MS analysis of specific zones of interest, minimizing overall resource consumption [45] [46]. This multi-modal approach on a single chromatographic separation represents a fundamentally greener paradigm compared to techniques requiring separate injections for different detection methods. When combined with green solvent choices such as ethanol-water or ethyl acetate-methanol mixtures, HPTLC-MS emerges as a sustainable yet powerful analytical platform for pharmaceutical quality control and research.
Diagram 2: HPTLC-MS Hyphenation Workflow (79 characters)
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of advanced HPTLC detection methods requires careful selection of reagents, materials, and instrumentation. The following table comprehensively details the essential components for establishing these methodologies in pharmaceutical analysis.
Table 4: Essential Research Reagents and Materials for Advanced HPTLC Detection
| Category | Specific Items | Technical Specifications | Function/Purpose | Greenness Considerations |
|---|---|---|---|---|
| Stationary Phases | Silica gel 60 F254 (normal phase) | Aluminium-backed, 20×10 cm, layer thickness 0.25 mm [42] [47] | Separation matrix for compounds | Reusable with cleaning (limited) |
| RP-18 F254S (reversed phase) | Glass or aluminium-backed, 20×10 cm [4] | Separation of polar compounds | ||
| Mobile Phase Solvents | Toluene, Ethyl acetate, Methanol | HPLC/AR grade, filtered and degassed [42] | Normal-phase separation | Replace benzene with toluene [42] |
| Chloroform, Methanol | HPLC grade [4] | Normal-phase for neutral compounds | High environmental impact [4] | |
| Ethanol, Water | HPLC grade [4] | Reversed-phase green alternative | Preferred green solvent [4] | |
| Detection Reagents | Anisaldehyde-sulfuric acid | Freshly prepared 0.5% in methanol [46] | Derivatization for non-UV absorbing compounds | Corrosive, handle with care |
| UV-active compounds | Native fluorescence or UV absorption | Non-destructive detection | No additional reagents needed | |
| Reference Standards | Pharmaceutical standards | USP/EP grade, purity >98% [42] [23] | Method calibration and identification | Minimal quantities required |
| Botanical markers | Isolated compounds, purity >95% [46] | Herbal drug standardization | ||
| MS-Specific Materials | Volatile buffers | Ammonium formate, ammonium acetate (10-20 mM) | MS-compatible mobile phase additive | Low residue after evaporation |
| Elution solvents | Methanol, acetonitrile with 0.1% formic acid | HPTLC-MS interface elution | Volatile, MS-compatible |
Comparative Analysis and Future Perspectives
The integration of densitometry, PDA, and MS detection with HPTLC creates a powerful analytical platform that addresses the comprehensive needs of modern pharmaceutical analysis. Densitometry provides robust quantification, PDA enables spectral characterization and peak purity assessment, and MS delivers definitive structural identification. When employed in a complementary workflow, these techniques offer unparalleled capability for pharmaceutical quality control, method validation, and regulatory compliance.
The environmental advantages of HPTLC with advanced detection continue to drive its adoption in alignment with green analytical chemistry principles. The minimal solvent consumption, energy efficiency, and ability to analyze multiple samples simultaneously position HPTLC as a sustainable alternative to column chromatographic techniques [4]. Future developments will likely focus on improved interfaces for HPTLC-MS coupling, enhanced software integration for data management, and further refinement of green solvent systems that maintain analytical performance while reducing environmental impact. As regulatory requirements for pharmaceutical analysis continue to evolve, the comprehensive data generated by these advanced HPTLC detection techniques will play an increasingly vital role in ensuring drug quality, safety, and efficacy.
High-Performance Thin-Layer Chromatography (HPTLC) has evolved from a simple qualitative tool into a sophisticated quantitative analytical platform that aligns with the principles of Green Analytical Chemistry (GAC). This evolution addresses the pharmaceutical industry's critical need for sustainable methods that minimize environmental impact while maintaining analytical precision. Green HPTLC techniques significantly reduce organic solvent consumption, energy requirements, and hazardous waste generation compared to conventional HPLC methods, without compromising data quality [48] [11]. The inherent advantages of HPTLC include parallel sample processing, minimal sample preparation, and the renewable nature of its stationary phase, making it particularly suitable for high-throughput pharmaceutical analysis in quality control and research settings [48]. This application note details specific protocols and case studies demonstrating the practical implementation of green HPTLC for pharmaceutical quantification, impurity profiling, and herbal drug analysis within a rigorous method validation framework.
Application Note 1: API Quantification in Bulk and Formulated Drugs
Quantitative Analysis of Dasatinib Monohydrate
Background: Dasatinib is a tyrosine kinase inhibitor used in treating chronic myelogenous leukemia. The development of green HPTLC methods for its quantification addresses the need for sustainable therapeutic drug monitoring [49].
Table 1: Green HPTLC Methods for Dasatinib Quantification
| Parameter | Reverse-Phase HPTLC | Normal-Phase HPTLC |
|---|---|---|
| Mobile Phase | 2-propanol:water:glacial acetic acid (60:40:0.2, v/v/v) | methanol:n-butyl acetate:glacial acetic acid (50:50:0.2, v/v/v) |
| Rf Value | 0.31 ± 0.2 | 0.39 ± 0.2 |
| Detection Wavelength | 323 nm | 323 nm |
| Linear Range | 30–500 ng/spot | 200–1200 ng/spot |
| R² Value | 0.9998 | 0.9995 |
| AGREE Greenness Score | 0.90 | 0.88 |
Experimental Protocol:
Chromatographic Conditions:
- Use HPTLC silica gel 60 F254 plates (10 × 20 cm)
- Employ a CAMAG Linomat 5 autosampler with 100 µL syringe
- Set application rate to 150 nL/s as 6 mm bands
- Develop in CAMAG automated developing chamber (ADC2) saturated for 30 minutes with mobile phase vapor
- Maintain development distance of 80 mm at room temperature
Sample Preparation:
- Prepare stock solution of Dasatinib standard (1 mg/mL) in methanol
- Prepare serial dilutions covering the linear range: 3-50 µg/mL for RP-HPTLC and 20-120 µg/mL for NP-HPTLC
- For tablet formulation, powder and extract equivalent to 10 mg Dasatinib in 10 mL methanol with 15 minutes of sonication
Detection and Quantification:
- Scan developed plates using CAMAG TLC scanner 3 in reflectance-absorbance mode at 323 nm
- Generate calibration curve by plotting peak area against concentration
- Calculate sample concentrations using regression equation
Greenness Assessment:
- Evaluate method using AGREE software (12 GAC principles)
- Record scores of 0.90 (RP-HPTLC) and 0.88 (NP-HPTLC), indicating excellent green profiles [49]
Analysis of Carvedilol in Tablet Formulations
Background: An eco-friendly stability-indicating HPTLC method was developed for carvedilol (Coreg tablets) estimation, demonstrating the application of green principles to cardiovascular drugs [8].
Table 2: HPTLC Method for Carvedilol Quantification
| Parameter | Specification |
|---|---|
| Mobile Phase | toluene:isopropanol:ammonia (7.5:2.5:0.1, v/v/v) |
| Stationary Phase | Silica gel 60 F254 TLC plates |
| Rf Value | 0.44 ± 0.02 |
| Linear Range | 20–120 ng/band |
| Detection Wavelength | 284 nm |
| R² Value | 0.995 |
| Application | Carvedilol tablet analysis (99-101% of label claim) |
Experimental Protocol:
Chromatographic Conditions:
- Use silica gel 60 F254 TLC plates
- Prepare mobile phase: toluene:isopropanol:ammonia (7.5:2.5:0.1, v/v/v)
- Saturate developing chamber for 20 minutes
- Develop plates to 75 mm at room temperature
Sample Preparation:
- Prepare carvedilol standard solution (1 mg/mL) in methanol
- Prepare sample solution by powdering 20 tablets, weighing equivalent to 10 mg carvedilol, and extracting with 10 mL methanol with sonication
Forced Degradation Studies:
- Perform acid degradation (0.1M HCl), base degradation (0.1M NaOH), oxidative degradation (3% H₂O₂)
- Expose to thermal (105°C) and photolytic (UV light) stress conditions
- Demonstrate method specificity by separating degradants from parent compound
Validation Parameters:
- Establish linearity (20-120 ng/band), precision (RSD < 2%), accuracy (98-102%)
- Determine LOD and LOQ values
- Assess robustness by deliberate variations in mobile phase composition and development distance [8]
Application Note 2: Impurity Profiling of Pharmaceutical Compounds
Simultaneous Determination and Impurity Profiling of Ebastine and Phenylephrine
Background: This method demonstrates the capability of HPTLC for impurity profiling of co-formulated drugs, addressing the critical need for purity assessment in pharmaceutical quality control [48].
Table 3: Impurity Profiling of Ebastine and Phenylephrine
| Compound | Rf Value | Impurity | Rf Value | Linear Range | Application |
|---|---|---|---|---|---|
| Ebastine (EBS) | 0.72 | Benzhydrol (BEN) | 0.52 | 0.10–1.60 µg/band | Ebast DC tablets |
| Phenylephrine HCl (PHE) | 0.33 | Norphenylephrine (NOR) | 0.18 | 0.05–0.80 µg/band | Ebast DC tablets |
Experimental Protocol:
Chromatographic Conditions:
- Stationary phase: HPTLC plates silica gel 60 F254 (E. Merck)
- Mobile phase: ethyl acetate:methanol:glacial acetic acid (70:25:5, v/v/v)
- Chamber saturation: 30 minutes at room temperature
- Development distance: 80 mm
- Detection: Densitometric scanning at 220 nm
Standard Solution Preparation:
- Prepare stock solutions of EBS, PHE, BEN, and NOR (1 mg/mL each in methanol)
- Prepare mixed standard solutions containing drugs and their impurities at different concentration levels
Sample Preparation:
- Weight and powder 20 tablets
- Transfer powder equivalent to 10 mg EBS and 0.25 mg PHE to 100 mL volumetric flask
- Add 70 mL methanol, sonicate for 20 minutes, dilute to volume with methanol
- Filter through 0.45 µm membrane filter
Validation Parameters:
- Specificity: Resolution between drugs and their impurities > 2.0
- Linearity: R² > 0.998 for all compounds
- Accuracy: 98.5–101.2% recovery for all components
- Precision: RSD < 2% for intra-day and inter-day variations
Greenness Assessment:
- Compare with reported HPLC method showing 80% reduction in solvent consumption
- Calculate analytical Eco-scale score of 85 (excellent greenness) [48]
Analysis of Mupirocin with Co-formulated Drugs and Impurities
Background: This method demonstrates the application of green HPTLC for complex mixtures containing multiple active ingredients and their impurities, essential for comprehensive pharmaceutical quality assessment [50].
Experimental Protocol:
Chromatographic Conditions for Mixture 1 (Mupirocin + Fluticasone propionate + Impurities):
- Stationary phase: HPTLC plates silica gel 60 F254
- Mobile phase: toluene:chloroform:ethanol (5:4:2, by volume)
- Detection: 220 nm for Mupirocin and Pseudomonic acid D, 254 nm for Fluticasone propionate and Fluticasone impurity C
- Chamber saturation: 20 minutes at room temperature
Chromatographic Conditions for Mixture 2 (Mupirocin + Mometasone furoate + Impurity):
- Use same stationary phase and detection conditions
- Mobile phase: toluene:ethyl acetate:methanol:glacial acetic acid (45:35:15:0.1, v/v/v/v)
Sample Preparation:
- Prepare standard solutions of all compounds (1 mg/mL in methanol)
- For ointment formulations, weigh equivalent to 10 mg Mupirocin, transfer to 100 mL volumetric flask, add 70 mL methanol, sonicate for 30 minutes, dilute to volume and filter
Method Validation:
- Demonstrate specificity by baseline separation of all components
- Establish linearity over concentration ranges of 0.1-2.0 µg/band for different components
- Verify accuracy through recovery studies (98.0-101.5%)
- Assess method robustness by varying mobile phase composition ±0.5%
Greenness Evaluation:
- Assess using National Environmental Method Index (NEMI), Analytical Eco-scale, GAPI, and AGREE metrics
- Show superior green profile compared to reported HPLC methods [50]
Application Note 3: Herbal Drug Analysis
Estimation of Ascorbic Acid in Plant Extracts
Background: This application demonstrates the suitability of green HPTLC for analyzing bioactive compounds in herbal matrices, providing a sustainable approach for standardization of plant-based medicines [51].
Table 4: Analysis of Ascorbic Acid in Plant Extracts
| Parameter | Specification |
|---|---|
| Mobile Phase | Water:ethanol (70:30, v/v) |
| Stationary Phase | RP silica gel 60 F254S plates |
| Detection Wavelength | 265 nm |
| Linear Range | 25–1200 ng/band |
| Rf Value | 0.58 ± 0.02 |
| AGREE Score | 0.88 |
| Plants Analyzed | Phyllanthus emblica, Psidium guajava, Capsicum annuum |
Experimental Protocol:
Extraction Procedures:
- Solvent Extraction (SE): Homogenize 1 g plant material with 10 mL water, centrifuge at 5000 rpm for 15 minutes, collect supernatant
- Ultrasound-Assisted Extraction (UAE): Homogenize 1 g plant material with 10 mL water, sonicate at 35 kHz for 30 minutes at 40°C, centrifuge at 5000 rpm for 15 minutes
Chromatographic Conditions:
- Use RP silica gel 60 F254S HPTLC plates (10 × 20 cm)
- Prepare mobile phase: water:ethanol (70:30, v/v)
- Saturate ADC2 chamber for 30 minutes at 22°C
- Apply samples as 6 mm bands using CAMAG ATS4
- Develop to distance of 80 mm in linear ascending mode
Calibration Curve:
- Prepare ascorbic acid stock solution (100 µg/mL) in water
- Prepare dilutions in range of 25-1200 ng/band
- Plot peak area against concentration to generate calibration curve
Method Validation:
- Linearity: R² = 0.9992 in range 25-1200 ng/band
- Precision: RSD < 1.5% for intra-day and inter-day
- Accuracy: 98.5-101.3% recovery
- LOD and LOQ: 8 ng/band and 25 ng/band respectively
Quantification in Plant Samples:
- Apply suitable dilutions of plant extracts alongside calibration standards
- Calculate ascorbic acid content using regression equation
- Report results as mg/100 g plant material [51]
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 5: Essential Materials for Green HPTLC Analysis
| Item | Specification | Application/Function |
|---|---|---|
| HPTLC Plates | Silica gel 60 F254, 10×20 cm, 0.2 mm thickness (Merck) | Standard stationary phase for normal-phase separation |
| HPTLC RP Plates | RP-18 silica gel 60 F254S, 10×20 cm | Stationary phase for reverse-phase separation |
| Sample Applicator | CAMAG Linomat 5 with 100 µL syringe | Precise application of samples as narrow bands |
| Developing Chamber | CAMAG ADC2 automated developing chamber | Controlled mobile phase development with chamber saturation |
| Densitometer | CAMAG TLC Scanner 3 with winCATS software | Quantitative densitometric measurement at selected wavelengths |
| Green Solvents | Ethanol, 2-propanol, water, ethyl acetate, n-butyl acetate | Environmentally benign mobile phase components |
| Reference Standards | USP/EP certified reference standards | Method validation and quantitative accuracy |
| Documentation System | CAMAG TLC Visualizer | Digital documentation under UV/visible light |
Workflow Diagrams
Green HPTLC Method Development Workflow
Greenness Assessment Protocol
Green HPTLC methodologies provide robust, sustainable alternatives to conventional chromatographic techniques for pharmaceutical analysis. The application notes presented demonstrate successful implementation for API quantification, impurity profiling, and herbal drug analysis while maintaining compliance with ICH validation guidelines. The integrated greenness assessment using tools such as AGREE, NEMI, and Analytical Eco-Scale provides quantitative metrics for environmental impact evaluation, aligning with the growing emphasis on sustainable analytical practices. The versatility, cost-effectiveness, and minimal environmental footprint of green HPTLC position it as a valuable technique for modern pharmaceutical analysis that balances analytical excellence with ecological responsibility.
Solving Common HPTLC Challenges and Enhancing Method Performance
In the development and validation of green High-Performance Thin-Layer Chromatography (HPTLC) methods for pharmaceutical analysis, achieving optimal separation with sharp, symmetrical peaks is a common challenge. Poor resolution and peak tailing directly impact the accuracy, sensitivity, and reliability of quantitative analysis. These issues can obscure low-concentration analytes and complicate integration, threatening the validity of an entire analytical procedure [52]. Within the framework of green analytical chemistry, optimizing the mobile phase and buffer systems provides a powerful strategy to overcome these challenges without resorting to excessive solvent consumption or toxic additives, aligning sustainability with analytical excellence [8] [53]. This application note details evidence-based protocols for mobile phase and buffer optimization to mitigate poor resolution and tailing in pharmaceutical HPTLC.
Theoretical Foundations: Causes of Poor Resolution and Tailing
Peak Tailing in chromatography, particularly for basic compounds under reversed-phase conditions, is often attributed to undesirable secondary interactions with the stationary phase. On silica-based phases, acidic silanol groups (-Si-OH) can lose protons at higher pH, creating negatively charged sites that strongly interact with basic analytes, causing delayed elution and tailing [52]. Another proposed theory is "mutual repulsion," where initial adsorption of charged analyte molecules creates a zone that repels similarly charged molecules, leading to band broadening and tailing, a phenomenon observed for both cationic and anionic analytes [52].
Poor Resolution occurs when two or more compounds in a mixture are not sufficiently separated. This is a function of the selectivity (α) of the system for the analyte pair and the efficiency (N) of the chromatographic band. A poorly chosen mobile phase that fails to create differential migration, or one that leads to excessive band broadening (including from tailing), will result in inadequate resolution.
Mobile Phase & Buffer Optimization Strategies
Optimizing the mobile phase is the most direct way to address tailing and poor resolution. The following strategies, supported by recent research, are recommended.
Strategic Mobile Phase Modification
The composition of the mobile phase is the primary tool for controlling retention, selectivity, and peak shape.
- pH Control: For ionizable compounds, controlling the mobile phase pH is critical. Working at a low pH (e.g., 2-3) can suppress the ionization of acidic silanols on the silica surface, thereby reducing their undesirable interactions with basic analytes and minimizing tailing [52]. A recent method for phenylephrine and doxylamine used a phosphate buffer at pH 5.0 to achieve good separation with a reversed-phase C18 column [54].
- Surfactant Addition: The use of surfactants like Sodium Dodecyl Sulphate (SDS) in the mobile phase is a advanced strategy. Surfactants can adsorb onto the stationary phase, modifying its surface and blocking specific interaction sites that cause tailing. Research has demonstrated that varying the concentration of SDS in an acetonitrile-buffer mobile phase can significantly improve the band shape (as measured by tailing and asymmetry factors close to 1.0) for a range of neuropsychiatric drugs [55].
- Green Solvent Selection: Aligning with green chemistry principles, replacing hazardous solvents with eco-friendly alternatives is a key trend. Methods have been successfully developed using solvents like ethanol, isopropanol, and acetone [8] [54] [56]. For instance, a green HPTLC method for β-sitosterol was optimized using a Box-Behnken design to find the ideal balance of formic acid content, saturation time, and migration distance [53].
Table 1: Exemplary Mobile Phase Compositions from Recent HPTLC Research
| Analyte(s) | Mobile Phase Composition (v/v/v) | Stationary Phase | Key Outcome | Source |
|---|---|---|---|---|
| Carvedilol | Toluene : Isopropanol : Ammonia (7.5 : 2.5 : 0.1) | Silica Gel 60 F₂₅₄ | Sharp, symmetric peaks with minimal tailing. | [8] |
| Phenylephrine & Doxylamine | Ethanol : Methylene Chloride : Ammonia 30% (7 : 2.5 : 0.5) | Silica Gel 60 F₂₅₄ | Successful separation from degradation products. | [54] |
| Sorafenib (NP-HPTLC) | n-Butanol : Ethyl Acetate | Silica Gel 60 F₂₅₄ | Compact spots, Rf 0.7; AGREE score 0.82. | [57] |
| Neurodegenerative Drugs | Acetonitrile : Buffer with SDS | RP-18 W | Improved band shape; tailing factor close to 1.0 for most compounds. | [55] |
| Emtricitabine (Green RP-HPTLC) | Acetone : Water (70 : 30) | RP-18 W | Well-separated, compact peak at Rf 0.79; superior greenness. | [56] |
Buffer System Optimization
Buffers are essential for maintaining a stable pH, which is crucial for reproducible retention times and consistent peak shapes for ionizable compounds.
- Buffer Type and Concentration: The choice of buffer and its concentration can influence the ionic strength of the mobile phase, which in turn can affect the efficiency of the separation and the sharpness of the peaks. A common choice in recent methods is a 0.01 M phosphate buffer [54].
- Ammonia / Amine Modifiers: Small amounts of ammonia or organic amines like triethylamine (TEA) have historically been used as mobile phase additives. They act by competing with the basic analyte for the acidic silanol sites, effectively "blocking" them and reducing tailing. However, the use of TEA is declining with the advent of high-purity silica and new column technologies and is generally incompatible with mass spectrometric detection [52]. Its use should be considered carefully.
Table 2: Troubleshooting Guide for Resolution and Tailing Issues
| Symptom | Potential Cause | Corrective Action |
|---|---|---|
| Severe Tailing of Basic Compounds | Interaction with acidic silanols on stationary phase. | 1. Lower mobile phase pH (if column permits).2. Add a surfactant (e.g., SDS) to the mobile phase [55].3. Use a stationary phase designed for basic compounds. |
| Poor Resolution Between Two Peaks | Insufficient selectivity or efficiency. | 1. Adjust the ratio of organic modifiers in the mobile phase.2. Change the type of organic modifier (e.g., switch from methanol to acetonitrile).3. For ionizable compounds, fine-tune the buffer pH to alter ionization states. |
| Broad or Diffuse Peaks | Overloading, poor mass transfer, or secondary interactions. | 1. Reduce sample concentration/volume.2. Ensure mobile phase buffer concentration is adequate.3. Optimize chamber saturation time [53] [58]. |
| Inconsistent Rf Values | Unstable mobile phase pH or inadequate chamber saturation. | 1. Use a fresh, properly prepared buffer.2. Standardize and extend the chamber saturation time before plate development [58]. |
Experimental Protocols
Protocol 1: Optimizing a Green HPTLC Method for a Single API
This protocol outlines the development of a simple, eco-friendly method for a single active pharmaceutical ingredient (API), such as the one described for emtricitabine [56].
- Stationary Phase Selection: Select a standard HPTLC plate (e.g., Silica Gel 60 F₂₅₄).
- Sample Solution Preparation: Dissolve the API in a suitable green solvent like methanol or ethanol to prepare a stock solution of 1 mg/mL. Further dilute as needed.
- Initial Mobile Phase Screening: Test different ratios of green solvent pairs, such as:
- Application and Development:
- Apply the sample as bands (e.g., 6 mm width) to the plate using an autosampler.
- Develop the plate in a twin-trough chamber pre-saturated with the mobile phase vapor for 20 minutes at room temperature.
- The development distance is typically 70-80 mm.
- Detection and Analysis:
- Dry the plate thoroughly after development.
- Scan the plate densitometrically at the analyte's λmax.
- Evaluation: Assess the chromatogram for a compact, symmetrical band (asymmetry factor close to 1.0) and a reasonable Rf value (e.g., 0.2-0.8). The acetone/water (70:30, v/v) system, for example, yielded a compact peak for emtricitabine at Rf 0.79 [56].
Protocol 2: Developing a Stability-Indicating Method for a Drug Combination
This protocol is based on methods developed for complex mixtures, such as phenylephrine with doxylamine or hydroxyzine with ephedrine and theophylline [54] [58].
- Forced Degradation: Subject the drug substance to stress conditions (acidic, basic, oxidative, thermal, photolytic) to generate degradation products.
- Mobile Phase Optimization for Selectivity:
- The primary goal is to achieve baseline separation between all active ingredients and their degradants.
- Experiment with multi-component mobile phases. A mixture of ethanol, methylene chloride, and ammonia (7:2.5:0.5, v/v/v) successfully separated phenylephrine, doxylamine, and its oxidative degradant on a silica gel plate [54].
- For difficult separations, a buffer component may be necessary. A chloroform-ammonium acetate buffer (pH 6.5) system was used to resolve a three-drug mixture based on their differing pKa values [58].
- Validation: Validate the final method as per ICH guidelines for specificity, linearity, accuracy, precision, and robustness, proving its ability to accurately quantify the APIs in the presence of degradants [54].
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for HPTLC Optimization
| Reagent / Material | Function in Optimization | Exemplary Use Case |
|---|---|---|
| Ammonia Solution (e.g., 30-33%) | A volatile modifier used to control pH in normal-phase separations, often to deactivate silanols and reduce tailing of basic compounds. | Used in mobile phase for carvedilol and phenylephrine/doxylamine to improve peak shape [8] [54]. |
| Ammonium Acetate Buffer | Provides a volatile buffering system suitable for methods that may interface with mass spectrometry; used to control pH for ionizable compounds. | Utilized in a TLC-densitometric method for hydroxyzine, ephedrine, and theophylline at pH 6.5 [58]. |
| Phosphate Buffer | A common aqueous buffer for reversed-phase systems; effective for controlling pH in the low to mid range. | Used at 0.01 M, pH 5.0, in an HPLC method for phenylephrine and doxylamine [54]. |
| Sodium Dodecyl Sulphate (SDS) | An ionic surfactant used to modify the stationary phase, effectively blocking sites that cause tailing and improving band shape. | Added to mobile phase to improve separation and peak shape of neuropsychiatric drugs on RP-18 W plates [55]. |
| Green Solvents (Ethanol, Isopropanol, Acetone) | To replace more hazardous solvents (e.g., chloroform, acetonitrile) in the mobile phase, aligning with Green Analytical Chemistry principles. | Ethanol used in mobile phase for phenylephrine/doxylamine [54]; Acetone/water used for emtricitabine [56]. |
Workflow for Systematic Optimization
The following diagram illustrates a logical, step-by-step workflow for diagnosing and addressing resolution and tailing problems in HPTLC method development.
Matrix effects pose a significant challenge in the quantitative analysis of pharmaceuticals in complex samples, often leading to ion suppression or enhancement, reduced sensitivity, and compromised analytical accuracy. High-Performance Thin-Layer Chromatography (HPTLC) has emerged as a powerful platform for addressing these challenges due to its unique separation characteristics and compatibility with green analytical principles [11]. The evolution of HPTLC from a simple qualitative tool to a sophisticated versatile analytical platform enables researchers to implement effective cleanup and separation strategies that manage matrix effects while adhering to the principles of Green Analytical Chemistry (GAC) [11] [8].
Within pharmaceutical quality control, matrix effects originate from various sample components, including lipids, pigments, proteins, and excipients, which can co-elute with target analytes and interfere with detection [11]. These interfering substances can obscure band resolution, destabilize ionization during mass spectrometric detection, and ultimately limit method dynamic range and reproducibility [11]. This application note delineates integrated cleanup and separation strategies to mitigate these effects, with a specific focus on sustainable HPTLC methodologies validated under ICH Q2(R2) guidelines for pharmaceutical analysis [27].
Cleanup Strategies for Sample Preparation
Effective sample preparation is crucial for minimizing matrix effects prior to HPTLC analysis. Well-designed cleanup protocols reduce interfering substances, concentrate target analytes, and enhance overall method performance.
Solid Phase Extraction (SPE)
SPE provides a robust, high-throughput approach for sample cleanup, particularly valuable when analyzing complex biological matrices or pharmaceutical formulations with interfering excipients.
- Mechanism: SPE utilizes selective sorbents to retain target analytes while allowing matrix components to pass through. The retained analytes are subsequently eluted with an appropriate solvent [60].
- Sorbent Selection: The choice of sorbent is critical for optimal recovery. Hydrophilic-Lipophilic Balanced (HLB) sorbents offer broad-spectrum retention for analytes with diverse physicochemical properties, while mixed-mode sorbents (e.g., PSA + C18) provide additional selectivity for removing specific interferences like fatty acids or sugars [60].
- High-Throughput Applications: Transferring SPE protocols to 96-well plate formats significantly enhances throughput, making it suitable for exposome-wide association studies (ExWAS) and routine quality control in pharmaceutical laboratories. This approach can improve processing efficiency approximately 10-fold compared to traditional protein precipitation methods [60].
Table 1: Performance Metrics of SPE Sorbents for Cleanup
| Sorbent Type | Chemical Characteristics | Optimal For Analytes | Typical Recovery Range | Key Advantages |
|---|---|---|---|---|
| HLB (Hydrophilic-Lipophilic Balanced) | Macroporous copolymer of divinylbenzene and N-vinylpyrrolidone | Broad-spectrum; log P range: -0.7 to 6.8 [60] | 60-140% for >70% of analytes [60] | Balanced retention of polar and non-polar compounds; high capacity |
| Mixed-Mode (PSA + C18) | Primary Secondary Amine and C18 silica mixed | Polar and non-polar analytes; effective for pigment removal [60] | Varies by analyte polarity | Selective removal of fatty acids, sugars, and organic acids |
Green Solvent-Based Extraction
Leveraging environmentally friendly solvents aligns with GAC principles while effectively extracting target analytes.
- QuEChERS (Quick, Easy, Cheap, Effective, Rugged, Safe): While not detailed in the search results, this approach is widely used in conjunction with planar chromatography and aligns with green principles.
- Solvent Selection: Methods prioritizing solvents like ethanol, isopropanol, and water reduce environmental impact and toxicity. For instance, a green HPTLC method for Carvedilol uses a mobile phase of toluene:isopropanol:ammonia (7.5:2.5:0.1, v/v/v) to minimize hazardous solvent use [8]. Similarly, a method for Suvorexant employs ethanol:water (75:25, v/v) as the developing system, showcasing excellent greenness scores [27].
Separation Strategies via HPTLC
HPTLC's planar separation format offers inherent advantages for managing matrix effects through spatial resolution, enabling multiple sample processing and various detection schemes on a single plate.
Stationary Phase Engineering
The selection and modification of the stationary phase are pivotal for achieving high-resolution separation of target analytes from matrix components.
- Standard Phases: Silica gel 60 F254 is the most widely used adsorbent for normal-phase HPTLC. For reversed-phase applications, RP-18F254S plates provide a hydrophobic surface suitable for separating more lipophilic compounds [8] [27].
- Advanced Material Modifications: Incorporating Metal-Organic Frameworks (MOFs) onto HPTLC plates creates a tailored stationary phase with enhanced selectivity and enrichment capabilities for trace-level contaminants [11]. These materials offer high surface areas and modular architectures, allowing for selective interactions with target analytes [11].
Mobile Phase Optimization
Optimizing the mobile phase is critical for resolving the analyte of interest from matrix-derived bands.
- Green Solvent Systems: Replace traditional hazardous solvents like chlorinated hydrocarbons with greener alternatives. For example, a combination of methanol:toluene (2:6, v/v) has been used for the simultaneous determination of Quinfamide and Mebendazole [61].
- Additives for Peak Sharpening: Small amounts of additives, such as ammonia (0.1% v/v), can be incorporated into the mobile phase to improve band symmetry and reduce tailing, as demonstrated in the Carvedilol method [8].
Table 2: HPTLC Experimental Parameters for Pharmaceutical Compounds
| Pharmaceutical Compound | Stationary Phase | Mobile Phase Composition (v/v) | Retardation Factor (Rf) | Detection Wavelength |
|---|---|---|---|---|
| Carvedilol [8] | Silica gel 60 F254 | Toluene : Isopropanol : Ammonia (7.5 : 2.5 : 0.1) | 0.44 ± 0.02 | 255 nm |
| Suvorexant [27] | RP-18F254S | Ethanol : Water (75 : 25) | Not specified | 255 nm |
| Quinfamide & Mebendazole [61] | Silica gel HPTLC F254 | Methanol : Toluene (2 : 6) | Not specified | 254 nm |
Integrated Workflow and Advanced Detection
Coupling effective cleanup with high-resolution HPTLC separation and multimodal detection creates a comprehensive strategy for overcoming matrix effects.
Workflow Diagram
The following diagram visualizes the integrated workflow from sample preparation to final analysis, incorporating the "HPTLC+" concept for advanced detection.
Multimodal Detection Platforms (HPTLC+)
The "HPTLC+" platform integrates spectroscopic and spectrometric techniques directly with the HPTLC separation to confirm identity and quantify analytes in complex matrices.
- HPTLC-MS: This coupling leverages the separation simplicity of HPTLC with the high molecular specificity of Mass Spectrometry. It reduces ion suppression effects by physically separating the target analyte from interfering substances before in-situ ionization and transfer to the MS [11].
- HPTLC-SERS: Integrating Surface-Enhanced Raman Spectroscopy provides molecular-level specificity through fingerprinting. The HPTLC separation mitigates fluorescence interference often encountered in SERS, and the metallic nanoparticles required for signal enhancement can be applied directly to the analyte band on the plate [11].
- HPTLC-NIR: Near-Infrared Spectroscopy offers a non-destructive method for compositional profiling directly on the plate, preserving the sample for further analysis [11].
- HPTLC-Bioautography: This function-directed screening technique is valuable for detecting bioactive compounds in complex herbal or food matrices that might be missed by chemical fingerprinting alone. It involves developing the HPTLC plate and then exposing it to a biological agent (e.g., enzyme, microorganism) to reveal active compounds [11].
Detailed Experimental Protocol
This section provides a step-by-step protocol for a green, stability-indicating HPTLC method, adaptable for various pharmaceuticals, based on validated procedures for compounds like Carvedilol and Suvorexant [8] [27].
Materials and Reagents
- HPTLC System: CAMAG or equivalent system with Automatic TLC Sampler (ATS4), Automated Developing Chamber (ADC2), and TLC Scanner with winCATS software [27].
- Stationary Phase: Pre-coated silica gel 60 F254 or RP-18F254S HPTLC plates (10 x 20 or 20 x 10 cm) [8] [27].
- Reference Standard: Pharmaceutical-grade analyte reference standard (e.g., Carvedilol, Suvorexant).
- Solvents: LC or analytical grade methanol, ethanol, toluene, isopropanol, ammonia solution, and purified water.
- Sample: Pharmaceutical dosage forms (tablets) or prepared biological samples.
Sample Preparation Protocol
- For Tablet Analysis: Accurately weigh and triturate not less than twenty tablets. Transfer an amount of powder equivalent to the weight of one tablet into a volumetric flask.
- Extraction: Add a sufficient volume of the chosen green solvent system (e.g., ethanol:water 75:25 v/v or methanol) to the flask. Sonicate for 15-30 minutes to ensure complete dissolution and extraction of the analyte [27].
- Cleanup (if required): Pass the extract through a 0.45 μm membrane filter. For highly complex matrices, employ a pre-conditioned SPE cartridge (e.g., HLB). Load the sample, wash with an appropriate solvent (e.g., water or 2% methanol in water), and elute the analyte with a strong solvent like pure methanol [60]. Evaporate the eluent under a gentle stream of nitrogen and reconstitute in the HPTLC mobile phase.
- Standard Solution: Prepare a stock solution of the reference standard at a concentration of 100 μg/mL in the same solvent used for sample extraction. Dilute serially to obtain working standards for the calibration curve (e.g., 10-1200 ng/band) [27].
HPTLC Analysis and Validation Protocol
- Application: Using the automatic sampler, apply the standard and sample solutions as 6 mm bands onto the HPTLC plate. Maintain an application rate of 150 nL/s and leave appropriate margins between tracks [27].
- Chromatographic Development: Develop the plate in the ADC2 pre-saturated with mobile phase vapor for 20-30 minutes. Use an ascending linear development mode to a distance of 75-80 mm at room temperature (20-25°C) with the optimized mobile phase [8] [27].
- Drying and Visualization: After development, air-dry the plate completely in a fume hood. Examine the plate under a UV cabinet at 254 nm for initial visualization of bands.
- Densitometric Scanning: Scan the developed plate with the TLC scanner in absorbance mode at the selected wavelength (e.g., 255 nm for Suvorexant and Carvedilol). Set the slit dimensions to 4.00 x 0.45 mm and a scan speed of 20 mm/s [27].
- Method Validation:
- Linearity: Apply standard solutions in the range of 10-1200 ng/band (n=6). Plot peak area vs. concentration to determine the correlation coefficient (R²), which should be ≥0.995 [27].
- Accuracy (Recovery): Perform a standard addition method at three levels (e.g., 50%, 100%, 150%). Calculate % recovery, which should be in the range of 98-102% [27].
- Precision: Assess repeatability (intra-day) and intermediate precision (inter-day) by analyzing samples multiple times. The % CV should be ≤2% [8].
- Robustness: Deliberately introduce small changes in mobile phase composition (±2%), development distance (±5 mm), and saturation time (±5 min) to evaluate the method's resilience [8].
- Forced Degradation: Subject the analyte solution to stress conditions (acid, base, oxidation, heat, and light) to demonstrate the stability-indicating nature of the method by effectively separating the analyte from its degradants [8].
Greenness Assessment Protocol
- Tool Selection: Use available software or calculators for the Analytical GREEnness (AGREE) metric, Analytic Eco-Scale, and NEMI to evaluate the method's environmental impact [8] [27].
- Data Input: Input parameters related to the method, including solvent types and volumes, energy consumption, waste generation, and reagent toxicity.
- Interpretation: An AGREE score >0.8, an Analytic Eco-Scale score >75, and a full quadrant of green in the NEMI pictogram indicate an excellent green profile [8] [27].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagents and Materials for HPTLC Method Development
| Item Name | Function / Purpose | Example Specifications / Notes |
|---|---|---|
| HPTLC Plates (Silica gel 60 F254) | Stationary phase for normal-phase chromatographic separation. | Pre-coated glass plates, particle size ~5 μm; F254 indicates fluorescent indicator for UV detection at 254 nm [8]. |
| HPTLC Plates (RP-18 F254S) | Stationary phase for reversed-phase chromatographic separation. | Silica gel modified with octadecylsilyl groups; suitable for more lipophilic compounds [27]. |
| Hydrophilic-Lipophilic Balanced (HLB) SPE Sorbent | Sample cleanup to remove matrix interferences and pre-concentrate analytes. | Macroporous copolymer; provides balanced retention for acidic, basic, and neutral compounds [60]. |
| Green Solvents (e.g., Ethanol, Isopropanol) | Extraction and mobile phase components. | Lower toxicity and environmental impact compared to traditional solvents like acetonitrile or chlorinated hydrocarbons [8] [27]. |
| Metal-Organic Frameworks (MOFs) | Stationary phase modification for enhanced selectivity and enrichment. | Nanomaterials with high surface area and tunable porosity; can be incorporated into HPTLC plates [11]. |
| Mass Spectrometry (MS) Interface | Coupling for hyphenated detection (HPTLC-MS) for structural confirmation. | Enables in-situ elution and ionization of analyte bands from the HPTLC plate for transfer to the MS [11]. |
| SERS-Active Nanoparticles | Enables HPTLC-SERS for molecular fingerprinting. | Colloidal suspensions of gold or silver nanoparticles applied directly to separated bands on the plate [11]. |
Effectively managing matrix effects in complex pharmaceutical samples requires a holistic strategy that integrates efficient cleanup procedures with advanced HPTLC separation and detection. The protocols outlined herein, emphasizing green chemistry principles and multimodal HPTLC platforms, provide a robust framework for developing sustainable, accurate, and precise analytical methods. The adoption of these strategies, validated per ICH guidelines, ensures reliable quality control and stability testing of pharmaceuticals, contributing to safer and more efficacious drug products.
In the development of green High-Performance Thin-Layer Chromatography (HPTLC) methods for pharmaceutical analysis, reproducibility stands as a critical pillar for regulatory acceptance and reliable quality control. Achieving consistent results demands precise control over environmental conditions and application parameters, which directly influence chromatographic behavior and quantitative accuracy. This protocol outlines a standardized approach to manage these variables, ensuring that eco-friendly methods not only minimize environmental impact through reduced solvent use and safer chemicals but also deliver robust, trustworthy data for drug development professionals [8] [42].
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key reagents and materials essential for executing a reproducible green HPTLC method, emphasizing their function within an eco-friendly framework.
Table 1: Key Research Reagent Solutions and Materials for Green HPTLC Analysis
| Item | Function & Importance in Green HPTLC |
|---|---|
| Pre-coated HPTLC plates (Silica gel 60 F₂₅₄) | Standardized stationary phase for separation. The F₂₅₄ indicator allows for UV visualization. Using pre-coated plates ensures layer uniformity, a prerequisite for reproducibility [42] [16]. |
| Methanol, Ethyl Acetate, Toluene | Common mobile phase components in greener methods. Safer than class 1 solvents (e.g., benzene, chloroform). Volumes are measured precisely for volumetric preparation to ensure mobile phase consistency [42] [62]. |
| Ammonia Solution | Modifier used in the mobile phase to control pH and improve peak shape of ionizable compounds, reducing tailing and enhancing separation efficiency [8] [16]. |
| CAMAG Linomat Automatic Applicator | An automated sample applicator critical for applying samples as bands of precise length and volume. This eliminates the variability associated with manual spotting and is fundamental for quantitative reproducibility [63] [16]. |
| Twin-Trough Development Chamber | A glass chamber used for plate development. Its twin-trough design allows for controlled chamber saturation, a key environmental factor affecting RF values and separation [42] [62]. |
| CAMAG TLC Scanner with winCATS Software | Densitometer for in-situ quantitative analysis of the developed chromatogram. It converts band intensity into a digital signal, and the software is used for data processing, calibration, and validation [62] [16]. |
Core Experimental Protocol for a Reproducible Green HPTLC Method
This section provides a detailed, step-by-step protocol for the simultaneous estimation of a model drug combination—Dapagliflozin (DAP) and Vildagliptin (VIL)—using a green-optimized mobile phase, as established in recent literature [42].
Materials and Preparation
- Chemicals: Toluene, methanol, and ethyl acetate (all of Analytical Reagent grade).
- Standard Solutions: Precisely weigh 10 mg of DAP and 10 mg of VIL pure substances. Transfer each into separate 10 mL volumetric flasks, dissolve, and make up to volume with methanol to obtain stock solutions of 1000 µg/mL. Prepare a combined working standard from these stocks [42].
- Sample Solution: For tablet analysis, weigh and powder tablets. Dissolve a quantity of powder equivalent to the label claim of each drug in methanol, sonicate, and filter to obtain a test solution [16].
- HPTLC Plates: Use aluminum-backed pre-coated silica gel 60 F₂₅₄ plates (20x10 cm).
Critical Step 1: Plate Pre-treatment and Sample Application
- Pre-washing: Pre-wash the HPTLC plates with methanol via ascending development in a twin-trough chamber to remove impurities. Dry the plates in an oven at 110°C for 5-10 minutes [16].
- Activation: After drying, allow the plates to equilibrate with the laboratory atmosphere (controlled at 25 ± 2°C and 60 ± 5% relative humidity) in a dust-free environment for 15-30 minutes. This standardizes the activity of the silica gel layer [63].
- Automated Sample Application:
- Use a CAMAG Linomat automatic applicator equipped with a 100 µL syringe.
- Program the instrument to apply bands 8.0 mm in length, 10.0 mm from the bottom edge of the plate, and 11.4 mm apart.
- Set a constant application rate of 150 nL/s.
- Apply standard and sample bands in the volume range of 2-5 µL to deliver the required mass per band (e.g., 0.6–1.4 µg/band for DAP and 6–14 µg/band for VIL) [42].
Critical Step 2: Chromatographic Development under Controlled Conditions
- Mobile Phase Preparation: Precisely measure toluene, methanol, and ethyl acetate in a volumetric ratio of 5:3:2 (v/v/v). Mix thoroughly in a stoppered vessel. This specific ratio avoids toxic solvents like benzene, aligning with green principles [42].
- Chamber Saturation: Pour approximately 10-15 mL of the mobile phase into one trough of a twin-trough glass chamber. Place the prepared plate in the empty trough. Seal the chamber and allow it to saturate for 20 minutes at room temperature to achieve a stable vapor environment [42].
- Plate Development: After saturation, transfer the plate to the trough containing the mobile phase. Develop the plate by linear ascending technique to a migration distance of 70 mm.
- Drying: Remove the plate and air-dry it in a fume hood for 10 minutes to completely evaporate the mobile phase.
Analysis and Validation
- Densitometric Scanning: Scan the developed, dried plate using a CAMAG TLC scanner IV in absorbance mode at 210 nm. Use a deuterium lamp, slit dimensions of 6.00 x 0.45 mm, and a scanning speed of 10 mm/s [42].
- Data Analysis: Use visionCATS or similar software to record peak areas. Generate calibration curves by plotting the peak area against the concentration for each drug.
- Method Validation: Validate the reproducible method as per ICH Q2(R2) guidelines, assessing linearity, accuracy, precision, specificity, LOD, LOQ, and robustness [64] [16].
Quantitative Data for Reproducibility Assessment
The following table compiles key validation parameters from recent green HPTLC studies, demonstrating the level of reproducibility achievable when environmental and application parameters are strictly controlled.
Table 2: Reproducibility and Validation Parameters from Recent Green HPTLC Studies
| Analytical Method (Drugs) | Retention Factor (Rf) ± SD | Linearity (ng/band) & Correlation (R²) | Precision (% RSD) | Reference |
|---|---|---|---|---|
| Dapagliflozin & Vildagliptin | DAP: 0.57 ± 0.02VIL: 0.26 ± 0.02 | DAP: 600-1400 (R²=0.997)VIL: 6000-14000 (R²=0.998) | Intra-day & Inter-day < 2% | [42] |
| Dapagliflozin & Bisoprolol | DAPA: 0.22 ± 0.003BSF: 0.63 ± 0.006 | DAPA: 200-1200 (R²=0.9995)BSF: 100-600 (R²=0.9991) | Intra-day & Inter-day < 2% | [16] |
| Ivabradine & Metoprolol (UV Mode) | IVA: 0.45 ± 0.05MET: 0.89 ± 0.01 | IVA: 50-600 (R² >0.999)MET: 50-900 (R² >0.999) | Intra-day & Inter-day < 2% | [62] |
| Carvedilol (Stability-Indicating) | 0.44 ± 0.02 | 20-120 (R²=0.995) | N/R | [8] |
N/R: Not Reported; SD: Standard Deviation; RSD: Relative Standard Deviation
Workflow and Parameter Interrelationships
The diagram below illustrates the critical controlled parameters and their interactions throughout the HPTLC process, highlighting how they collectively ensure analytical reproducibility.
The rigorous control of environmental factors and application parameters is not merely a procedural requirement but the foundation of reproducibility in green HPTLC. By adhering to standardized protocols for plate pretreatment, automated sample application, chamber saturation, and development, scientists can ensure that their eco-friendly methods are both sustainable and scientifically robust. This approach, validated under ICH Q2(R2) guidelines, provides the drug development industry with reliable analytical tools that meet the dual demands of environmental responsibility and pharmaceutical quality control.
Leveraging Chemometrics and Experimental Design (DoE) for Method Robustness
In the field of pharmaceutical analysis, the validation of robust, precise, and environmentally sustainable analytical methods is paramount. High-performance thin-layer chromatography (HPTLC) has emerged as a powerful technique that aligns with the principles of Green Analytical Chemistry (GAC) and White Analytical Chemistry (WAC) due to its minimal solvent consumption, low energy requirements, and high throughput capabilities [14] [8]. The integration of chemometrics and Design of Experiments (DoE) represents a transformative approach to enhancing method robustness during the validation process. This protocol details the application of these advanced statistical tools within a green HPTLC framework, providing a structured pathway for developing methods that are not only analytically sound but also environmentally responsible.
The synergy of HPTLC with chemometrics and DoE addresses two critical challenges in modern pharmaceutical analysis: the need for methods that can reliably quantify multiple analytes simultaneously—including active pharmaceutical ingredients and their mutagenic impurities—and the growing imperative to reduce the environmental impact of analytical procedures [14]. By implementing strategic experimental designs and sophisticated data processing algorithms, researchers can systematically optimize analytical parameters, validate method performance, and ensure regulatory compliance while adhering to sustainability goals.
Theoretical Background and Key Concepts
Chemometrics in Analytical Science
Chemometrics applies mathematical and statistical methods to chemical data to maximize the information obtained. In HPTLC method development, two chemometric approaches are particularly valuable: multivariate calibration for spectral analysis and algorithm-driven optimization for parameter selection.
The Firefly Algorithm (FA), inspired by natural swarm intelligence, has shown exceptional utility in multivariate calibration for HPTLC-densitometry and spectrophotometry. This algorithm strategically identifies the most influential variables in partial least squares (PLS) modeling, effectively transforming traditional approaches into refined, precise analytical tools [14]. By mimicking the flashing patterns of fireflies, the algorithm efficiently navigates complex variable spaces to find optimal combinations that enhance predictive accuracy while reducing model complexity.
For validation set construction, the Hammersley Sequence Sampling (HSS) technique provides a sophisticated alternative to random data partitioning. This advanced statistical method systematically constructs representative validation sets by dividing modeled variables into equally probable levels, ensuring comprehensive sample space coverage and minimizing sampling bias that often plagues conventional approaches [14]. The integration of HSS with a 52 mixture experimental design for calibration has demonstrated significant enhancements in model robustness and predictive capability in pharmaceutical applications.
Design of Experiments (DoE) Fundamentals
DoE represents a systematic approach to understanding the relationship between factors affecting a process and its output. Instead of the traditional one-factor-at-a-time approach, DoE enables simultaneous evaluation of multiple parameters, revealing interaction effects that might otherwise go undetected.
In HPTLC method validation, Fractional Factorial Design (FFD) has emerged as particularly valuable for robustness testing. This design reduces the number of experiments needed by considering only a fraction of all possible combinations while still providing sufficient information about the system [65]. FFD allows researchers to identify the most critical factors affecting method robustness, detect potential interactions between parameters, and optimize analytical conditions with minimal experimental runs—aligning perfectly with green chemistry principles by reducing solvent consumption and waste generation.
Experimental Protocols
Protocol 1: Firefly Algorithm-Optimized PLS for HPTLC-Densitometry
Objective
To develop a chemometric model for the simultaneous quantification of multiple cardiovascular drugs (bisoprolol fumarate and amlodipine besylate) and their mutagenic impurity (4-hydroxybenzaldehyde) using FA-PLS optimization [14].
Materials and Equipment
Table 1: Research Reagent Solutions for FA-PLS HPTLC Protocol
| Item | Specification | Function |
|---|---|---|
| HPTLC Plates | Silica gel 60 F₂₅₄ (20 × 20 cm, 0.2 mm thickness) | Stationary phase for chromatographic separation |
| Mobile Phase | Ethyl acetate–ethanol (7:3, v/v) | Eco-friendly solvent system for compound separation |
| Standard Solutions | Bisoprolol fumarate, amlodipine besylate, 4-hydroxybenzaldehyde | Analytical targets for method development |
| CAMAG ADC2 Chamber | Automated development chamber | Controlled mobile phase development |
| CAMAG TLC Scanner 3 | Densitometer with deuterium/tungsten lamps | Quantification of separated compounds |
| MATLAB Software | R2013a with PLS Toolbox v2.0 | Chemometric modeling and FA-PLS implementation |
Step-by-Step Procedure
Sample Preparation and Application
- Prepare standard solutions of bisoprolol fumarate (BIP), amlodipine besylate (AML), and 4-hydroxybenzaldehyde (HBZ) in methanol at concentrations spanning the expected analytical range.
- Apply samples as 8 mm bands onto silica gel 60 F₂₅₄ HPTLC plates using an automated applicator (Camag Linomat 5) with 10 mm intervals between bands.
Chromatographic Development
- Condition the HPTLC plates in a Camag ADC2 automated development chamber pre-saturated for 25 minutes with the mobile phase (ethyl acetate-ethanol, 7:3 v/v) at controlled environmental conditions (25 ± 0.5°C, 40 ± 2% relative humidity).
- Develop the chromatogram using ascending mode to a distance of 75 mm from the origin.
Densitometric Analysis
- Scan the developed HPTLC plate using a Camag TLC Scanner 3 operated in reflectance-absorbance mode with 8 × 0.1 mm slit dimension and scanning speed of 100 nm/s.
- Record absorbance spectra for each compound across the wavelength range of 200-400 nm.
Data Preprocessing
- Export the densitometric data to MATLAB format.
- Perform baseline correction and normalize spectral data to correct for plate-to-plate variations.
- Arrange data into a matrix format suitable for chemometric analysis.
Firefly Algorithm Implementation
- Initialize FA parameters: number of fireflies (20-50), maximum iterations (100-500), absorption coefficient (0.5-1.0), and randomization parameter (0.1-0.5).
- Define the objective function as minimization of the root mean square error of cross-validation (RMSECV).
- Execute the FA to identify the most informative wavelengths for PLS modeling.
- Validate the selected variables through iterative cross-validation.
PLS Model Development and Validation
- Construct the PLS model using the wavelength regions identified by the FA.
- Employ Hammersley Sequence Sampling to divide the data into representative calibration and validation sets.
- Validate model performance using external validation samples not included in the model development.
- Calculate figures of merit: root mean square error of prediction (RMSEP), relative standard error of prediction (RSEP), and correlation coefficients.
Figure 1: FA-PLS HPTLC Protocol Workflow. This diagram illustrates the sequential steps for implementing the Firefly Algorithm-optimized Partial Least Squares protocol for HPTLC-densitometry.
Protocol 2: Fractional Factorial Design for Robustness Testing
Objective
To systematically evaluate the robustness of an HPTLC method for simultaneous analysis of levodropropizine and chlorpheniramine maleate in syrup formulation using a 2^(4-1) fractional factorial design [65].
Materials and Equipment
Table 2: Research Reagent Solutions for DoE Robustness Protocol
| Item | Specification | Function |
|---|---|---|
| HPTLC Plates | TLC Silica Gel 60F254 aluminum sheets (20 × 20 cm) | Stationary phase for separation |
| Mobile Phase | Triethylamine:Toluene:Methanol (0.5:3:16 v/v/v) | Solvent system for compound elution |
| Standard Solutions | Levodropropizine (1500-7500 ng/band), Chlorpheniramine maleate (100-500 ng/band) | Analytical targets for robustness study |
| CAMAG TLC Scanner 3 | With winCATS software and deuterium lamp | Densitometric quantification at 270 nm |
| Statistical Software | Package capable of factorial design analysis (e.g., Minitab, R, Design-Expert) | Experimental design and data analysis |
Step-by-Step Procedure
Critical Parameter Identification
- Select four factors for investigation based on preliminary risk assessment: chamber saturation time, solvent front distance, detection wavelength, and methanol volume in mobile phase.
- Define the high (+1) and low (-1) levels for each factor based on the nominal method conditions:
- Chamber saturation time: ±5 minutes from nominal
- Solvent front distance: ±5 mm from nominal
- Detection wavelength: ±5 nm from nominal
- Methanol volume: ±2% from nominal composition
Experimental Design Setup
- Construct a 2^(4-1) fractional factorial design with resolution IV, requiring 8 experimental runs instead of the full 16 runs.
- Randomize the run order to minimize systematic error.
- Include center points (3-5 replicates) to estimate pure error and check for curvature.
Sample Preparation and Analysis
- Prepare standard solutions of levodropropizine (3000 ng/band) and chlorpheniramine maleate (300 ng/band) as test samples.
- Execute the experimental runs according to the designed sequence.
- For each run, apply samples to HPTLC plates, develop in the twin-trough chamber with specified saturation time and solvent front.
- Scan the developed plates at the specified wavelength and record the peak areas and Rf values.
Data Analysis
- Calculate the response variables: peak area and Rf value for each compound.
- Perform analysis of variance (ANOVA) to identify significant factors and interactions.
- Construct main effects and interaction plots to visualize factor influences.
- Calculate factor effects and determine the statistical significance (p < 0.05).
Method Robustness Assessment
- Evaluate the impact of each factor variation on the analytical responses.
- Establish system suitability criteria based on the observed robustness ranges.
- Define control strategies for critical method parameters that significantly affect method performance.
Figure 2: DoE Robustness Assessment Workflow. This diagram illustrates the systematic approach for evaluating method robustness using Fractional Factorial Design.
Applications and Case Studies
Case Study 1: Green HPTLC with Chemometrics for Cardiovascular Drugs
A recent study demonstrated the successful application of FA-PLS chemometrics for the simultaneous quantification of bisoprolol fumarate, amlodipine besylate, and 4-hydroxybenzaldehyde (a mutagenic impurity) using HPTLC-densitometry [14]. The method employed an eco-friendly mobile phase of ethyl acetate-ethanol (7:3, v/v), achieving baseline separation with Rf values of 0.29 ± 0.02 (HBZ), 0.72 ± 0.01 (AML), and 0.83 ± 0.01 (BIP).
The FA-PLS model incorporated Hammersley Sequence Sampling for validation set construction, ensuring uniform concentration space coverage and eliminating sampling bias inherent in conventional random approaches. This innovation, combined with a 52 mixture experimental design for calibration (25 mixtures), significantly enhanced model robustness and predictive capability [14]. Both methods demonstrated superior analytical performance with detection limits of 3.56–20.52 ng/band (HPTLC) and 0.011–0.120 μg/mL (FA-PLS), correlation coefficients ≥ 0.9995, and precision (RSD) ≤ 2%.
The sustainability assessment using multiple evaluation tools revealed exceptional environmental profiles: perfect NEMI, AGREE, and ComplexGAPI scores, high GEMAM indices (7.015 and 7.487), minimal carbon footprints (0.037 and 0.021 kg CO₂/sample), and outstanding BAGI (87.50 and 90.00), VIGI (75.00 and 80.00), and RGBfast scores (81.00 and 85.00) for HPTLC and FA-PLS, respectively [14].
Case Study 2: DoE for Stability-Indicating HPTLC Method
In the development of a stability-indicating HPTLC method for levodropropizine and chlorpheniramine maleate in syrup formulation, a 2^(4-1) fractional factorial design was implemented for robustness studies [65]. The design examined four factors (chamber saturation time, solvent front, wavelength, and methanol volume in mobile phase) at both high level (+1) and low level (-1).
The study revealed that methanol volume in mobile phase, chamber saturation time, and wavelength had minor effects on the response of Rf value, confirming method robustness against minor variations in these parameters [65]. This systematic approach allowed for efficient investigation of multiple factors with reduced experimental runs, aligning with green chemistry principles by minimizing solvent consumption and waste generation.
The forced degradation studies conducted as part of method validation demonstrated effective separation of parent compounds from degradants, with the method proving capable of detecting degradation under various stress conditions while maintaining robustness within the defined parameter ranges [65].
Data Analysis and Interpretation
Statistical Treatment of Chemometric Data
For FA-PLS models, evaluate performance using the following statistical measures:
- Root Mean Square Error of Calibration (RMSEC): Should be minimized while avoiding overfitting
- Root Mean Square Error of Prediction (RMSEP): Should be comparable to RMSEC for a robust model
- Coefficient of Determination (R²): Should approach 1.0 for both calibration and validation sets
- Relative Standard Error of Prediction (RSEP): Should be below 5% for acceptable model performance
For the Firefly Algorithm, monitor convergence behavior to ensure optimal variable selection. The algorithm should show progressive improvement in objective function value with iterations, stabilizing at the optimum solution.
Interpretation of DoE Results
In fractional factorial designs for robustness testing, focus on the following aspects:
- Main Effects: Factors with large absolute effect sizes significantly influence method performance
- Interaction Effects: Significant two-factor interactions indicate interdependent parameter effects
- Normal Probability Plots: Help distinguish significant effects from random noise
- Analysis of Variance (ANOVA): Provides statistical significance of factor effects
Table 3: Quantitative Performance Metrics from Chemometric HPTLC Applications
| Application | Analytical Technique | Chemometric Approach | Linearity (R²) | LOD | LOQ | Greenness Score |
|---|---|---|---|---|---|---|
| Cardiovascular Drugs & Impurity [14] | HPTLC-Densitometry | FA-PLS with HSS | ≥0.9995 | 3.56-20.52 ng/band | 11.8-67.8 ng/band | AGREE: 0.95 |
| Antiviral Agents [12] | NP-HPTLC vs RP-HPTLC | Comparative DoE | >0.9998 | 1.2-3.5 ng/band | 3.6-10.5 ng/band | RGB12: 82-85 |
| Anti-asthmatic Drugs [28] | TLC-Densitometry | Mobile Phase Optimization | >0.9990 | 4.8-7.3 ng/band | 14.5-22.1 ng/band | AGREE: 0.78 |
| Salivary Caffeine [66] | HPTLC | Method Robustness Testing | >0.9900 | 2.42 ng/band | 7.34 ng/band | N/A |
Establish acceptable robustness ranges for each critical method parameter based on the experimental results. Parameters showing insignificant effects (p > 0.05) can tolerate wider variations, while significant parameters require tighter control strategies in the final method protocol.
Troubleshooting and Optimization Guidelines
Common Issues in Chemometric Modeling
- Overfitting: If the model shows excellent calibration but poor prediction performance, reduce the number of latent variables in PLS or adjust FA parameters to favor simpler models.
- Wavelength Selection Bias: When the Firefly Algorithm consistently selects narrow wavelength regions, verify that these regions correspond to compound-specific spectral features, not background noise.
- Sample Representation: If Hammersley Sequence Sampling produces validation sets with poor predictive performance, increase the number of calibration samples or adjust the sequence parameters to ensure better space coverage.
DoE Implementation Challenges
- Factor Range Definition: If no significant factors are identified in robustness testing, consider widening the factor ranges to detect potential effects, while ensuring the variations remain within practically relevant limits.
- Aliasing in Fractional Factorial Designs: Be aware that in resolution IV designs, two-factor interactions are aliased with each other. If significant interactions are detected, consider augmenting the design to resolve the aliases.
- Center Point Curvature: If significant curvature is detected at center points, the method may have nonlinear responses within the studied range, requiring more complex modeling approaches.
Sustainability Assessment in Method Validation
The integration of chemometrics and DoE inherently supports green chemistry principles by reducing the number of experimental runs, minimizing solvent consumption, and decreasing waste generation. To quantitatively assess the environmental impact of the developed methods, employ modern greenness assessment tools:
- AGREE Metric: Evaluates method greenness based on the 12 principles of GAC, providing a score between 0-1 [28] [12]
- BAGI Tool: Assesses method practicality and applicability, complementing greenness evaluation [12]
- NEMI Scale: Provides a simple pictogram representing method environmental performance [8]
- RGB12 Model: Implements a trichromatic approach evaluating greenness, blueness, and whiteness simultaneously [12]
Document the sustainability profile of the developed methods as an integral part of the validation protocol, aligning with the United Nations Sustainable Development Goals, particularly SDG 3 (Good Health and Well-being), SDG 9 (Industry, Innovation and Infrastructure), and SDG 12 (Responsible Consumption and Production) [14].
Validating Method Reliability and Assessing Greenness
The validation of analytical methods is a critical, mandatory process in pharmaceutical development and quality control, ensuring that analytical procedures yield results that are reliable, consistent, and suitable for their intended purpose. The International Council for Harmonisation (ICH) Q2(R1) guideline, titled "Validation of Analytical Procedures: Text and Methodology," provides the internationally accepted framework for this validation [67] [68]. For researchers focused on Green High-Performance Thin-Layer Chromatography (HPTLC), validation demonstrates that a method is not only environmentally friendly but also scientifically sound, ensuring the identity, purity, potency, and performance of drug substances and products [8] [69]. The objective of validation is to demonstrate that the analytical procedure is suitable for its intended purpose, a principle that must guide the selection and execution of every validation test [70].
This protocol details the experimental workflow for validating a green HPTLC method, covering the core parameters defined by ICH Q2(R1): Specificity, LOD/LOQ, Linearity, Accuracy, Precision, and Robustness.
Core Validation Parameters & Experimental Protocols
Specificity
Definition and Purpose: Specificity is the ability to assess unequivocally the analyte in the presence of components that may be expected to be present, such as impurities, degradants, or excipients [67]. For a stability-indicating method, it is paramount to demonstrate that the method can separate and accurately quantify the analyte from its degradation products.
Experimental Protocol:
- Preparation of Solutions:
- Prepare a standard solution of the pure analyte.
- Prepare a sample solution from the formulated product (e.g., powdered tablet) including all excipients.
- Subject the analyte to forced degradation under stress conditions (acid, base, oxidative, thermal, and photolytic) as per ICH stability testing guidelines [69].
- Chromatographic Procedure:
- Apply the standard, sample, and degraded solutions onto the HPTLC plate (e.g., silica gel 60 F254) [42] [71].
- Develop the plate using the optimized, green mobile phase (e.g., toluene:ethyl acetate:methanol or similar, avoiding carcinogenic solvents like benzene) [42] [8].
- Scan the developed plate with a densitometer at the selected wavelength.
- Data Analysis and Acceptance Criteria:
- The analyte peak from the sample solution should be identified by comparing its Rf value with the standard.
- The chromatogram should show a sharp, symmetric peak for the analyte with baseline separation from any other peak arising from excipients or degradation products [8].
- The peak purity of the analyte in the sample and stressed samples should be confirmed by comparing spectra at peak start, apex, and peak end.
Limit of Detection (LOD) and Limit of Quantification (LOQ)
Definition and Purpose: The LOD is the lowest amount of analyte in a sample that can be detected, but not necessarily quantified. The LOQ is the lowest amount of analyte that can be quantitatively determined with suitable precision and accuracy [67] [72].
Experimental Protocols:
- Based on Signal-to-Noise Ratio (S/N):
- This approach is commonly used in chromatographic methods [72].
- Prepare and analyze samples with decreasing concentrations of the analyte.
- The LOD is the concentration that yields a signal-to-noise ratio of 3:1.
- The LOQ is the concentration that yields a signal-to-noise ratio of 10:1.
- Based on Standard Deviation of the Response and Slope:
- This method is based on the standard deviation of the response (σ) and the slope (S) of the calibration curve.
- Measure the magnitude of analytical background response by analyzing blank samples.
- Calculate the standard deviation (σ) of this response.
- Determine the slope (S) of the linearity calibration curve (see Section 2.3).
- Calculate LOD and LOQ using the formulas:
- ( LOD = \frac{3.3 \times \sigma}{S} )
- ( LOQ = \frac{10 \times \sigma}{S} )
Table 1: Exemplary LOD and LOQ Data from a Green HPTLC Method [42]
| Analyte | Linear Range (µg/band) | LOD (µg/band) | LOQ (µg/band) |
|---|---|---|---|
| Dapagliflozin | 0.6 - 1.4 | 0.02 | 0.07 |
| Vildagliptin | 6.0 - 14.0 | 0.19 | 0.58 |
Linearity
Definition and Purpose: Linearity is the ability of the method to obtain test results that are directly proportional to the concentration of the analyte within a given range [67] [73]. It is crucial to demonstrate that the method provides accurate quantification across the intended working range.
Experimental Protocol:
- Preparation of Solutions:
- Prepare a stock standard solution of the analyte.
- Prepare a series of at least five different concentrations of the analyte from the stock solution, covering the specified range (e.g., 80%, 90%, 100%, 110%, 120% of the target concentration).
- Chromatographic Procedure:
- Spot each concentration in triplicate on the HPTLC plate.
- Develop and scan the plate using the validated method.
- Data Analysis and Acceptance Criteria:
- Plot the average peak area (or height) against the corresponding concentration for each level.
- Calculate the regression line using the least-squares method (y = mx + c).
- The coefficient of determination (R²) is typically used to assess linearity. For accurate quantification, an R² value ≥ 0.995 is generally expected [42].
- Newer approaches using double logarithm function linear fitting are also being explored to more rigorously assess the proportionality of results [73].
Table 2: Exemplary Linearity Data from a Green HPTLC Method [42] [69]
| Analyte | Linear Range | Correlation Coefficient (R²) | Regression Equation |
|---|---|---|---|
| Dapagliflozin | 0.6 - 1.4 µg/band | 0.997 | Not Specified |
| Vildagliptin | 6.0 - 14.0 µg/band | 0.998 | Not Specified |
| Mirabegron | 0.15 - 7.5 µg/band | >0.995 | Not Specified |
| Tamsulosin | 0.05 - 2.5 µg/band | >0.995 | Not Specified |
Accuracy
Definition and Purpose: Accuracy expresses the closeness of agreement between the value found and the value accepted as a true or reference value. It is typically established by spiking known amounts of analyte into the sample matrix and measuring recovery [67] [70].
Experimental Protocol (Recovery Study):
- Preparation of Samples:
- Select a batch of placebo (excipients without active ingredient) or a pre-analyzed sample.
- Prepare samples spiked with the analyte at three concentration levels (e.g., 80%, 100%, 120% of the target concentration), each in triplicate.
- Also prepare standard solutions corresponding to 100% level for comparison.
- Chromatographic Procedure:
- Analyze all prepared samples and standards using the developed HPTLC method.
- Data Analysis and Acceptance Criteria:
- Calculate the percentage recovery for each spike level using the formula:
- ( \% Recovery = \frac{\text{Found Concentration}}{\text{Theoretical Concentration}} \times 100 )
- Calculate the mean recovery and the relative standard deviation (%RSD) for each level.
- The mean recovery should be within 98.0% - 102.0%, and the %RSD should typically be ≤ 2.0% [69]. Confidence intervals should be reported for a more robust assessment of trueness [70].
- Calculate the percentage recovery for each spike level using the formula:
Precision
Definition and Purpose: Precision expresses the closeness of agreement between a series of measurements obtained from multiple sampling of the same homogeneous sample under the prescribed conditions. It is investigated at three levels: repeatability, intermediate precision, and reproducibility [67].
Experimental Protocol:
- Repeatability:
- Intermediate Precision:
- Demonstrate the impact of random, within-laboratory variations, such as different days, different analysts, or different equipment.
- Repeat the repeatability experiment on a different day, with a different analyst using the same homogeneous sample.
- The combined %RSD from both sets of results should meet the acceptance criteria (e.g., %RSD ≤ 2.0%). It is critical that the data set for precision studies is generated from independently worked up samples, individually applied on the plate, to be a true representation of method variability [70].
Robustness
Definition and Purpose: Robustness is a measure of the method's capacity to remain unaffected by small, deliberate variations in method parameters. It indicates the reliability of a method during normal usage.
Experimental Protocol:
- Identification of Parameters:
- Identify critical method parameters that could vary, such as mobile phase composition (± 0.5-1% for each component), development distance (± 5 mm), chamber saturation time (± 5 minutes), or detection wavelength (± 2-3 nm) [71].
- Experimental Design:
- Using a standard solution at 100% concentration, perform the analysis by deliberately introducing small changes to one parameter at a time while keeping others constant.
- Each variation should be performed in duplicate or triplicate.
- Data Analysis and Acceptance Criteria:
- Evaluate the impact of each variation on the results, such as Rf value, peak area, tailing factor, and resolution from the nearest peak.
- The method is considered robust if the %RSD of the results across all variations is within the pre-defined precision limit (e.g., ≤ 2.0%) and the system suitability criteria are met in all instances.
The Green HPTLC Validation Workflow
The following diagram illustrates the logical sequence and key decision points in the validation of a green HPTLC method, integrating core ICH Q2(R1) parameters with green chemistry principles.
Diagram 1: Green HPTLC Method Validation Workflow. The validation process is sequential, with each parameter building upon the previous. The final step before deployment is a formal greenness assessment.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Green HPTLC Validation
| Item | Function & Rationale in Validation | Green Consideration |
|---|---|---|
| Pre-coated Silica gel 60 F₂₅₄ plates | The stationary phase. F₂₅₄ indicates the fluorescent indicator for UV visualization. Standardized plates are critical for reproducibility of Rf values [42] [71]. | - |
| Green Mobile Phase Components (e.g., Ethyl Acetate, Ethanol, Methanol) | Solvents for the mobile phase. Selected for their lower toxicity and environmental impact compared to class 1 solvents (e.g., benzene) [42] [8]. | Prefer less hazardous, biodegradable solvents. |
| Ammonia Solution | Used in small proportions in the mobile phase to modify pH, improve peak shape, and achieve separation without tailing [8] [69]. | Use minimal required concentration. |
| Pure Drug Standards | Used to prepare stock and working solutions for constructing calibration curves and determining accuracy, linearity, LOD, and LOQ [42] [71]. | - |
| Pharmaceutical Placebo | A mixture of all formulation excipients without the active ingredient. Essential for proving specificity and for conducting accurate recovery studies [69]. | - |
| CAMAG or Equivalent HPTLC System (Linomat, Chamber, Scanner) | Automated sample applicator, development chamber, and densitometric scanner. Essential for obtaining precise, quantitative, and reproducible results [42] [71]. | - |
This protocol provides a detailed roadmap for the complete validation of a green HPTLC method according to the ICH Q2(R1) guideline. By systematically assessing specificity, LOD/LOQ, linearity, accuracy, precision, and robustness, researchers can generate robust scientific evidence that their method is fit-for-purpose. Integrating this rigorous validation with a conscious selection of green chemicals and processes, assessed by tools like AGREE and GAPI, ensures the development of sustainable and high-quality analytical methods for modern pharmaceutical analysis [8] [69]. The final proof of any method's validity lies in its ability to reliably achieve its intended purpose, which for drug analysis is often the accurate quantification of the active ingredient within a defined confidence interval, such as ±2.5% of the labeled claim at a 95% significance level [70].
The pharmaceutical industry is increasingly embracing the principles of Green Analytical Chemistry (GAC), which aims to minimize the environmental impact of analytical methods while maintaining their efficacy and reliability. This paradigm shift is particularly evident in the field of pharmaceutical analysis, where techniques like High-Performance Thin-Layer Chromatography (HPTLC) are being developed with sustainability as a core objective [8]. The drive toward eco-friendly methodologies has necessitated the development of robust, standardized tools to quantitatively assess and validate the greenness of these analytical procedures.
Within this context, three assessment tools have emerged as cornerstones for evaluating the sustainability of analytical methods: AGREE (Analytical GREEnness), GAPI (Green Analytical Procedure Index), and BAGI (Blue Applicability Grade Index). These complementary tools provide researchers, scientists, and drug development professionals with a comprehensive framework for designing, optimizing, and validating analytical methods that align with green chemistry principles [74] [75]. Their application is particularly crucial in the development of green HPTLC methods, which offer advantages such as reduced solvent consumption, minimal sample preparation, and the ability for simultaneous analysis of multiple samples [4].
This guide provides a detailed examination of these three assessment tools, complete with structured protocols for their implementation in pharmaceutical research, specifically focusing on HPTLC method validation. By integrating these metrics into routine analytical practice, researchers can systematically quantify and enhance the sustainability profile of their methodologies while ensuring they remain fit for purpose in pharmaceutical quality control and stability studies.
Comparative Analysis of AGREE, GAPI, and BAGI
The three primary assessment tools—AGREE, GAPI, and BAGI—each offer unique perspectives on method evaluation, addressing different aspects of sustainability and practicality.
Table 1: Comparison of Key Green Assessment Tools
| Tool Name | Assessment Focus | Output Format | Score Interpretation | Key Principles Covered |
|---|---|---|---|---|
| AGREE | Comprehensive greenness based on GAC principles | Pictogram with overall score (0-1) | Closer to 1 indicates greener method | All 12 principles of GAC |
| GAPI | Environmental impact across method lifecycle | Pictogram with colored segments | More green segments indicate lower environmental impact | Sample collection, preparation, analysis, waste disposal |
| BAGI | Practical applicability and method robustness | Numerical score (0-100) | Higher scores indicate better practicality | Cost, efficiency, safety, operational simplicity, energy consumption |
AGREE provides the most holistic assessment of greenness by evaluating all 12 principles of Green Analytical Chemistry, resulting in a circular pictogram divided into 12 sections, each corresponding to one GAC principle [75]. The tool generates an overall score between 0 and 1, with scores closer to 1 indicating superior greenness. For example, a recently developed eco-friendly HPTLC method for Trifluridine and Tipiracil determination achieved an AGREE score of 0.81, confirming its environmental sustainability [74].
GAPI employs a multi-criteria evaluation system that covers the entire analytical procedure through a pictogram with five pentagrams, each color-coded to represent the environmental impact of different method stages [4]. This tool is particularly valuable for identifying specific areas where environmental improvements can be made throughout the analytical workflow.
BAGI complements these greenness assessments by focusing on the practical applicability of analytical methods, evaluating factors such as cost, efficiency, safety, operational simplicity, and energy consumption [75]. It generates a score from 0 to 100, with higher scores indicating better practicality and robustness. For instance, a green HPTLC method for thioctic acid and biotin analysis achieved a BAGI score of 82.5, demonstrating excellent applicability for routine quality control [76].
The AGREE (Analytical GREEnness) Assessment Tool
Theoretical Foundation and Calculation Methodology
The AGREE assessment tool is built upon the 12 fundamental principles of Green Analytical Chemistry, providing a comprehensive framework for evaluating method sustainability [75]. Each principle is assigned a score based on how thoroughly the analytical method addresses it, and these scores are integrated into an overall assessment using a specialized software calculator available at https://mostwiedzy.pl/AGREE [76].
The AGREE pictogram consists of 12 segmented sections arranged in a circle, with each segment corresponding to one GAC principle. The color of each segment ranges from red (poor compliance) to green (excellent compliance), while the center of the pictogram displays a numerical score between 0 and 1, where higher values indicate superior greenness. This visual representation allows researchers to quickly identify both the strengths and weaknesses of a method's environmental profile.
Experimental Protocol for AGREE Implementation
Protocol: Conducting AGREE Assessment for HPTLC Methods
Step 1: Data Collection - Compile all relevant information about the HPTLC method, including: sample preparation requirements (number of steps, reagents used, energy consumption), sample size, analysis time, cartridge/plate type, mobile phase composition, waste generation per analysis, and safety data for all chemicals [4] [27].
Step 2: Software Input - Launch the AGREE calculator software and input the collected data, responding to all prompts regarding the 12 GAC principles. The software is freely available and user-friendly, requiring no specialized computational skills [76].
Step 3: Score Interpretation - Analyze the generated pictogram and numerical score. Methods scoring above 0.75 are generally considered to have excellent greenness characteristics. For example, a reverse-phase HPTLC method for suvorexant analysis achieved an outstanding AGREE score of 0.88, indicating superior environmental performance [27].
Step 4: Method Optimization - Use the AGREE output to identify areas for improvement. If specific segments display yellow or red coloring, focus method optimization efforts on those aspects to enhance overall greenness.
Application Example in HPTLC Method Development
A green stability-indicating HPTLC method for the estimation of Carvedilol in pharmaceutical dosage forms was successfully evaluated using the AGREE tool alongside other metrics [8]. The method employed a mobile phase of toluene, isopropanol, and ammonia (7.5:2.5:0.1, v/v/v), specifically optimized to avoid carcinogenic solvents, thus addressing multiple GAC principles related to waste reduction and safer chemical usage.
The GAPI (Green Analytical Procedure Index) Assessment Tool
Structural Framework and Assessment Criteria
GAPI provides a comprehensive visual assessment of the environmental impact of analytical methods throughout their entire lifecycle [4]. The tool employs a pentagram-shaped pictogram divided into five sections that represent key stages of the analytical process: sample collection, preservation, transportation, storage, and sample preparation; and the final three sections dedicated to the analysis method itself, including instrumentation, reagents, and solvents used.
Each section of the GAPI pictogram is color-coded (green, yellow, red) to indicate the environmental impact level, with green representing low impact, yellow moderate impact, and red high impact. This detailed visualization helps researchers quickly identify which specific aspects of their method contribute most significantly to its environmental footprint, enabling targeted optimization efforts.
Experimental Protocol for GAPI Implementation
Protocol: Applying GAPI to HPTLC Method Validation
Step 1: Method Deconstruction - Break down the HPTLC method into its constituent steps, from sample collection through final analysis. For pharmaceutical analysis using HPTLC, this typically includes: sample collection and preservation, sample transport and storage, sample preparation (extraction, filtration, derivation), application to HPTLC plates, mobile phase preparation, chromatographic development, and detection/visualization [7].
Step 2: Impact Assessment - For each step identified, evaluate its environmental impact based on established GAPI criteria. Consider factors such as energy requirements, reagent toxicity, waste generation, and safety hazards. Recent studies have introduced Modified GAPI (MoGAPI) which provides enhanced differentiation between impact levels [76].
Step 3: Pictogram Generation - Complete the GAPI pictogram by coloring each segment according to the impact assessment. For HPTLC methods, the sample preparation and mobile phase composition segments often present opportunities for greenness improvement.
Step 4: Comparative Analysis - Use the completed GAPI pictogram to compare alternative method configurations or to benchmark against existing methods. This visual comparison facilitates decision-making during method development and optimization.
Application Example in Pharmaceutical HPTLC
In the development of an HPTLC method for simultaneous determination of thioctic acid and biotin, researchers employed MoGAPI (Modified GAPI) to evaluate the method's greenness profile [76]. The assessment revealed that the use of ethanol-water mobile phases in reversed-phase HPTLC significantly improved the greenness score compared to normal-phase methods utilizing chloroform-methanol mixtures, directly influencing the solvent selection during method optimization.
The BAGI (Blue Applicability Grade Index) Assessment Tool
Practicality Assessment Framework
While AGREE and GAPI focus primarily on environmental impact, BAGI addresses the equally important aspect of method practicality and applicability in routine analytical settings [75]. This tool evaluates ten key criteria related to the practical implementation of analytical methods, including: total analysis time, cost per analysis, operational simplicity, health hazard safety, throughput, efficiency, waste management, energy consumption, alignment with green chemistry principles, and automation potential.
BAGI generates a numerical score from 0 to 100, with higher scores indicating better practicality and suitability for routine use. This quantitative output complements the greenness assessments provided by AGREE and GAPI, ensuring that methods are not only environmentally friendly but also operationally viable for pharmaceutical quality control laboratories.
Experimental Protocol for BAGI Implementation
Protocol: Assessing Method Practicality Using BAGI
Step 1: Practicality Data Collection - Gather comprehensive data on the practical aspects of the HPTLC method, including: total analysis time (from sample preparation to result), estimated cost per analysis (reagents, plates, instrumentation), required operator skill level, safety considerations, sample throughput capacity, and energy requirements [75].
Step 2: Online Assessment - Access the BAGI online calculator at https://bagi-index.anvil.app/ and input the collected data. The platform features an intuitive interface that guides users through the assessment process [76].
Step 3: Score Analysis - Interpret the resulting BAGI score according to established benchmarks: scores above 75 indicate excellent applicability, scores between 60-75 represent good practicality, while scores below 60 suggest significant practical limitations for routine implementation.
Step 4: Optimization Strategy - Based on the BAGI output, identify practical limitations and implement improvements. For HPTLC methods, this might involve automating sample application, optimizing development time, or simplifying mobile phase preparation to enhance throughput.
Application Example in Veterinary Pharmaceutical Analysis
A recent study developed a cost-effective HPTLC densitometric method for simultaneous quantification of meloxicam and florfenicol in pharmaceutical formulations and spiked bovine muscle samples [9]. The method was subjected to BAGI assessment alongside other greenness metrics, demonstrating that it offered an optimal balance between analytical performance, environmental sustainability, and practical applicability for regulatory and surveillance purposes in veterinary drug residue monitoring.
Integrated Workflow for Comprehensive Method Assessment
Combined Implementation Strategy
For a thorough evaluation of analytical methods, researchers should implement AGREE, GAPI, and BAGI in a complementary fashion, as each tool provides unique insights that collectively inform a complete sustainability and practicality profile.
The diagram above illustrates a systematic workflow for integrating all three assessment tools throughout the method development process. This approach ensures that environmental considerations and practicality are embedded from the initial stages rather than being evaluated as an afterthought.
Case Study: Trifaceted Sustainability Assessment
A recent study on thioctic acid and biotin analysis provides an excellent example of this integrated approach [76]. The researchers employed a tri-faceted assessment strategy incorporating:
- Greenness metrics: Analytical Eco-Scale (score: 80), AGREE (score: 0.72), and MoGAPI
- Blueness assessment: BAGI (score: 82.5)
- Whiteness evaluation: RGB12 algorithm (score: 92.2%)
This comprehensive evaluation demonstrated that the developed HPTLC method successfully balanced environmental sustainability with excellent practical applicability, making it suitable for routine quality control laboratories, particularly those with limited resources in developing countries.
Essential Research Reagent Solutions
Table 2: Key Research Reagents and Materials for Green HPTLC Analysis
| Reagent/Material | Function in HPTLC Analysis | Green Alternatives | Sustainability Considerations |
|---|---|---|---|
| Silica Gel Plates | Stationary phase for separation | Biodegradable plates | Choose F254S for UV detection to avoid derivatization |
| Ethanol-Water Systems | Mobile phase components | Replace with methanol-acetonitrile | Less toxic, biodegradable, renewable source |
| Ethyl Acetate | Mobile phase modifier | Replace hexane/chloroform | Lower toxicity, biodegradable |
| Ammonia Solution | pH modifier in mobile phase | Dilute concentrations | Minimize concentration to reduce environmental impact |
| Densitometer Scanner | Quantitative analysis | - | Enables precise quantification at low concentrations |
| Automated Sample Applicator | Sample application | - | Reduces solvent consumption and improves reproducibility |
The selection of appropriate reagents and materials is crucial for developing sustainable HPTLC methods. As demonstrated in multiple studies, the choice of mobile phase constituents significantly influences the greenness profile of analytical methods [4] [27]. For instance, replacing traditional normal-phase solvents like chloroform with greener alternatives such as ethanol-water mixtures in reversed-phase HPTLC can dramatically improve AGREE, GAPI, and BAGI scores [4].
The AGREE, GAPI, and BAGI assessment tools collectively provide researchers with a comprehensive framework for developing, evaluating, and optimizing sustainable analytical methods in pharmaceutical analysis. When implemented as part of an integrated workflow, these tools enable the creation of HPTLC methods that successfully balance environmental responsibility with analytical performance and practical applicability.
As the pharmaceutical industry continues to embrace green chemistry principles, the systematic application of these assessment metrics will play an increasingly vital role in method validation and harmonization. By adopting these tools, researchers and drug development professionals can contribute to the advancement of sustainable analytical practices while maintaining the high standards required for pharmaceutical quality control and stability studies.
The development of environmentally conscious analytical methods has become a paramount objective in modern pharmaceutical analysis. This case study, framed within a broader thesis on green analytical technique validation, details the development and validation of a stability-indicating High-Performance Thin-Layer Chromatography (HPTLC) method for the simultaneous quantification of Sacubitril and Valsartan, a critical cardiovascular drug combination used in heart failure management. The method was designed to align with the twelve principles of green analytical chemistry (GAC), prioritizing the reduction of hazardous solvent use and enhancing sustainability without compromising analytical performance [8] [6]. This protocol serves as a comprehensive guide for researchers and drug development professionals seeking to implement green, cost-effective, and robust quality control methods.
Experimental Design and Workflow
The method was validated for the simultaneous analysis of Sacubitril and Valsartan in pharmaceutical dosage forms. The workflow below outlines the key stages of the analytical process.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 1: Key Research Reagent Solutions and Materials
| Item | Function/Description | Specification/Example |
|---|---|---|
| HPTLC Plates | Stationary phase for chromatographic separation | Silica gel 60 F254 plates (e.g., Merck), 0.20-0.25 mm thickness [27] [16] |
| Green Solvents | Mobile phase components; sample dissolution | Ethanol, Water, Ethyl Acetate. Chosen for lower toxicity [6] [27] [69] |
| Standard Substances | Method development and calibration | High-purity Sacubitril (≥98.8%) and Valsartan (≥98.8%) [6] |
| HPTLC Instrumentation | Automated sample application, development, and detection | CAMAG system (e.g., Linomat autosampler, ADC2 chamber, TLC Scanner 3) [27] |
| Densitometer | Quantification of separated analyte bands | CAMAG TLC Scanner with UV/Vis detector and WinCATS software [77] [27] |
| Forced Degradation Reagents | For stability-indicating studies | HCl, NaOH, H2O2 for acid, base, and oxidative stress [8] [69] |
Detailed Experimental Protocols
Protocol 1: Preparation of Standard and Sample Solutions
Objective: To prepare accurate and precise stock, working standard, and sample solutions for analysis.
Materials: Sacubitril and Valsartan reference standards (high purity), ethanol, water, volumetric flasks (10 mL, 100 mL), analytical balance, ultrasonic bath.
Steps:
- Stock Standard Solutions (1 mg/mL): Accurately weigh 10 mg of each Sacubitril and Valsartan into separate 10 mL volumetric flasks. Dissolve in and dilute to volume with a mixture of ethanol and water (75:25, v/v) to obtain final concentrations of 1 mg/mL [6] [27].
- Working Standard Solutions: Perform appropriate serial dilutions of the stock solutions using the ethanol-water mobile phase to prepare working standards covering the calibration range (e.g., 10–1200 ng/band for Sacubitril) [27].
- Pharmaceutical Formulation (Tablet) Solution:
- Weigh and finely powder not less than 20 tablets.
- Accurately weigh a portion of the powder equivalent to about 10 mg of Sacubitril (or the target analyte) into a 100 mL volumetric flask.
- Add approximately 70 mL of the ethanol-water mixture, sonicate for 30 minutes with intermittent shaking, and cool.
- Dilute to volume with the same solvent and mix well.
- Filter the solution through a 0.45 µm membrane filter, discarding the first few mL of the filtrate [27] [16].
Protocol 2: Chromatographic Separation and Densitometric Analysis
Objective: To achieve baseline separation of the target analytes and perform their quantification.
Materials: Pre-coated HPTLC plates (Silica gel 60 F254), mobile phase (Ethanol:Water, 75:25 v/v), twin-trough developing chamber, CAMAG HPTLC system or equivalent.
Steps:
- Plate Pre-washing & Pre-conditioning: Pre-wash the HPTLC plates with methanol and activate at 110 °C for 5 minutes before sample application [16].
- Sample Application: Using an automatic sample applicator (e.g., CAMAG Linomat 5), apply the standard and sample solutions as 6 mm bands, 10 mm from the bottom and with a minimum of 4 mm distance between bands. The application rate should be set at 150 nL/s [27].
- Chromatographic Development: Pour the mobile phase into one trough of the twin-trough chamber. Saturate the chamber for 30 minutes at room temperature (22 ± 2 °C). Develop the plate in the other trough in a linear ascending mode to a distance of 80 mm. Dry the developed plate at room temperature for 2-5 minutes [27] [69].
- Densitometric Scanning: Scan the dried plate using a TLC scanner equipped with a deuterium lamp. Set the scanning wavelength to 255 nm, with a scanning speed of 20 mm/s and slit dimensions of 4.00 x 0.45 mm [27]. Record the chromatograms and measure the peak areas.
Protocol 3: Forced Degradation (Stability-Indicating) Studies
Objective: To demonstrate the method's specificity and its ability to quantify the analytes in the presence of degradation products.
Materials: Standard drug solutions, 0.1 M HCl, 0.1 M NaOH, 3% H2O2, thermal oven, UV light chamber.
Steps:
- Acidic & Alkaline Hydrolysis: Treat 1 mL of standard stock solution (1 mg/mL) separately with 1 mL of 0.1 M HCl or 0.1 M NaOH. Heat these solutions at 60 °C for 1-2 hours. Neutralize before application on the HPTLC plate [8] [69].
- Oxidative Degradation: Treat 1 mL of standard stock solution with 1 mL of 3% hydrogen peroxide. Allow the solution to stand at room temperature for 30-60 minutes before analysis [8] [16].
- Dry Heat Degradation: Expose the pure drug powder to a dry heat of 60 °C in an oven for 24 hours. Prepare the sample solution from the exposed powder [8].
- Photolytic Degradation: Expose the solid drug and drug product to direct sunlight for 24-48 hours or to UV light (e.g., 254 nm) in a chamber for a specified duration [8].
- Analysis: After degradation, prepare solutions of the stressed samples and subject them to analysis using the developed HPTLC method. Assess the chromatograms for the appearance of additional peaks (degradants) and the resolution from the main analyte peak [8] [16].
Method Validation and Data Analysis
The developed method was validated as per the International Council for Harmonisation (ICH) Q2(R2) guideline for the following parameters [27] [16].
Table 2: Summary of Validation Parameters and Results
| Validation Parameter | Results for Sacubitril | Results for Valsartan | Acceptance Criteria |
|---|---|---|---|
| Linearity Range | 10–1200 ng/band [27] | 100–600 ng/band [16] | — |
| Correlation Coefficient (r²) | >0.999 [27] | >0.999 [16] | r² ≥ 0.995 |
| Accuracy (% Recovery) | 98.18 – 99.30% [27] | 99.19 – 100.15% [16] | 98–102% |
| Precision (% RSD) | ≤ 2% [16] | ≤ 2% [16] | RSD ≤ 2% |
| Repeatability (n=6) | RSD = 0.78–0.94% [27] | — | RSD ≤ 2% |
| Robustness | Deliberate variations accepted [8] | Deliberate variations accepted [8] | RSD of peak area < 2% |
| LOD | 3.32 ng/band [27] | — | — |
| LOQ | 9.98 ng/band [27] | — | — |
| Specificity | Resolved from degradation products [8] [16] | Resolved from degradation products [8] [16] | No interference |
The signaling pathway below illustrates the logical sequence and decision points in the method validation process, which is integral to confirming the method's suitability for its intended purpose.
Greenness Assessment
The environmental impact of the analytical method was evaluated using multiple greenness assessment tools, a critical step in modern method validation [8] [27].
- Analytical Eco-Scale: The method scored 93 (out of 100), indicating an excellent green profile. A score above 75 is considered excellent green analysis. This score is calculated by penalizing the method for hazardous reagents, energy consumption, and waste [27].
- AGREE Metric: The method achieved a high score of 0.88, reflecting strong compliance with the 12 principles of GAC. The AGREE tool provides a comprehensive pictogram for easy visualization of a method's greenness [8] [27].
- GAPI/MoGAPI: These multi-criteria assessment tools visually confirmed the method's environmental friendliness, particularly highlighting the use of ethanol-water as a green mobile phase compared to traditional acetonitrile or n-hexane [69] [16].
This case study successfully demonstrates the development and comprehensive validation of a green, stability-indicating HPTLC method for the analysis of a cardiovascular drug combination. The method is linear, precise, accurate, robust, and specific, successfully separating the active pharmaceuticals from their forced degradation products. The greenness assessment, conducted using the Analytical Eco-Scale, AGREE, and GAPI tools, confirms its minimal environmental impact. This protocol provides a reliable, cost-effective, and sustainable solution for the routine quality control and stability monitoring of this vital drug combination in pharmaceutical formulations, aligning with the evolving paradigm of Green Analytical Chemistry.
High-Performance Thin-Layer Chromatography (HPTLC) is a well-established analytical technique in pharmaceutical analysis, valued for its efficiency, minimal sample preparation requirements, and capacity for parallel sample processing. In recent years, a paradigm shift toward Green Analytical Chemistry (GAC) principles has prompted the development of eco-friendly HPTLC methods that reduce environmental impact while maintaining analytical performance [8] [33] [31]. This application note provides a comparative analysis of green versus traditional HPTLC approaches, offering validated protocols and performance metrics to guide researchers in implementing sustainable pharmaceutical analysis methods. The transition to greener methods aligns with global sustainability initiatives while offering unexpected analytical advantages in sensitivity, precision, and cost-effectiveness [78] [4].
Comparative Performance Data
The table below summarizes quantitative validation parameters and greenness metrics for green and traditional HPTLC methods from recent studies, demonstrating the comparative performance of each approach.
Table 1: Comparative Validation Parameters of Traditional vs. Green HPTLC Methods
| Analyte | Method Type | Linear Range (ng/band) | LOD/LOQ (ng/band) | Precision (% RSD) | Greenness Score (AGREE) | Reference |
|---|---|---|---|---|---|---|
| Colchicine | Traditional NP-HPTLC | 100-600 | Not specified | Not specified | 0.46 | [78] |
| Colchicine | Greener RP-HPTLC | 25-1200 | Not specified | Better precision | 0.75 | [78] |
| Ertugliflozin | Traditional NP-HPTLC | 50-600 | Not specified | Not specified | 0.46 | [4] |
| Ertugliflozin | Greener RP-HPTLC | 25-1200 | Not specified | Better precision | 0.75 | [4] |
| Tenoxicam | Greener HPTLC | 25-1400 | LOD: 0.98, LOQ: 2.94 | 0.87-1.02 | 0.75 | [33] |
| Trifluridine/Tipiracil | Greener HPTLC | Not specified | LOD: 0.0011-0.0022 µg/mL | <0.74 (intra-day) | 0.81 | [74] |
| Thioctic Acid/Biotin | Greener HPTLC | 2.5-30 (TH), 2.5-20 (BO) | LOD: 0.58 (TH), 0.33 (BO) | ≤2.0 | 0.72 | [79] |
Table 2: Mobile Phase Composition Comparison
| Method Type | Typical Mobile Phase Composition | Environmental Impact | Waste Generation |
|---|---|---|---|
| Traditional HPTLC | Chloroform/methanol (85:15 v/v) [4] | High toxicity, environmental concerns | Significant |
| Green HPTLC | Ethanol/water (80:20 v/v) [4] | Low toxicity, biodegradable | Minimal |
| Green HPTLC | Ethanol/water/ammonia (50:45:5 v/v/v) [33] | Low toxicity, safer alternatives | Reduced |
Experimental Protocols
Protocol 1: Green HPTLC Method Development for Pharmaceutical Compounds
This protocol outlines the development and validation of a greener reversed-phase HPTLC method for the analysis of pharmaceutical compounds, adapting approaches validated for tenoxicam [33] and ertugliflozin [4].
Materials and Reagents:
- Pre-coated HPTLC plates: RP-18 F254S or silica gel 60 F254
- Green solvents: Ethanol, water, ethyl acetate, isopropanol
- Ammonia solution (for pH adjustment)
- Standard pharmaceutical compounds
- Pharmaceutical formulations (tablets, capsules)
Instrumentation:
- HPTLC system with sample applicator (e.g., CAMAG Linomat)
- Development chamber with saturation capability
- Densitometer with UV/Vis detector
- Software for data analysis (e.g., WinCATS)
Procedure:
- Mobile Phase Optimization:
- Test binary and ternary mixtures of green solvents (ethanol/water, ethanol/water/ammonia)
- Evaluate different proportions (e.g., from 40:60 to 90:10 v/v for ethanol/water)
- Select optimal composition based on peak symmetry, theoretical plates, and retardation factor
Sample Preparation:
- Accurately weigh standard compounds and prepare stock solutions in methanol or ethanol
- For formulations, extract powdered tablets/capsules with green solvents using ultrasonication
- Filter solutions through 0.45 µm membrane before application
Chromatographic Conditions:
- Apply samples as 6-8 mm bands using automated applicator
- Develop plates in twin-trough chamber pre-saturated with mobile phase for 15-20 minutes
- Development distance: 70-80 mm at room temperature
- Scanning wavelength: Compound-specific (e.g., 375 nm for tenoxicam, 199 nm for ertugliflozin)
Method Validation:
- Linearity: Analyze minimum of six concentrations in triplicate
- Precision: Repeat intra-day (n=3) and inter-day (n=3) analyses
- Accuracy: Perform recovery studies at 80%, 100%, 120% levels
- Robustness: Deliberately vary parameters (mobile phase composition, development distance)
Greenness Assessment:
- Calculate AGREE score using available software
- Evaluate with complementary metrics (Eco-Scale, GAPI)
Protocol 2: Forced Degradation Studies for Stability-Indicating Methods
This protocol describes forced degradation studies to establish the stability-indicating properties of green HPTLC methods, based on approaches for thioctic acid/biotin [79] and tenoxicam [33].
Materials:
- Acidic degradant: 0.1-2M HCl
- Basic degradant: 0.1-3M NaOH
- Oxidative degradant: 3-30% H₂O₂
- Thermal degradation: Oven at 60-80°C
- Photolytic degradation: UV chamber
Procedure:
- Acidic/Basic Degradation:
- Expose drug solution to 2.5M HCl or 3M NaOH at room temperature for 30 minutes
- Neutralize after exposure and prepare for HPTLC analysis
Oxidative Degradation:
- Treat drug solution with 3-15% H₂O₂ at room temperature for 30 minutes
- Dilute and apply directly to HPTLC plates
Thermal Degradation:
- Heat solid drug substance at 80°C for 24 hours
- Prepare solution in green solvent for analysis
Photolytic Degradation:
- Expose drug solution to UV light (254 nm) for 24 hours
- Analyze alongside protected control samples
Degradation Kinetics (Optional):
- Withdraw samples at regular intervals
- Plot log % remaining vs. time to determine degradation rate constants
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Green HPTLC
| Item | Function/Application | Green Characteristics |
|---|---|---|
| Ethanol (Pharmaceutical Grade) | Primary green solvent for mobile phases [33] [4] | Biodegradable, low toxicity, renewable source |
| Water (HPLC Grade) | Eco-friendly solvent component [33] [4] | Nontoxic, readily available |
| Ethyl Acetate | Green organic solvent alternative [74] [14] | Lower toxicity than chlorinated solvents |
| Isopropanol | Modifier for mobile phases [8] | Less hazardous than acetonitrile |
| Ammonia Solution | pH adjustment in mobile phases [33] [79] | Volatile, minimal residue |
| Silica Gel 60 F₂₅₄ HPTLC Plates | Stationary phase for normal-phase separations [80] [79] | Standard HPTLC substrate |
| RP-18 F₂₅₄ HPTLC Plates | Stationary phase for reversed-phase separations [4] | Enables water-rich mobile phases |
| Ultrasound Bath | Enhanced extraction from formulations [78] [79] | Reduces solvent consumption, improves efficiency |
Workflow and Decision Pathway
The following diagram illustrates the systematic workflow for developing and validating green HPTLC methods, incorporating method selection, optimization, and greenness assessment.
Discussion
Analytical Performance Comparison
The comparative data demonstrates that green HPTLC methods consistently match or exceed the analytical performance of traditional approaches. For colchicine analysis, the greener reversed-phase HPTLC method showed superior sensitivity with a wider linear range (25-1200 ng/band) compared to the traditional normal-phase method (100-600 ng/band) [78]. Similarly, for ertugliflozin, the green RP-HPTLC method exhibited enhanced sensitivity and better precision compared to its traditional counterpart [4]. These findings challenge the conventional assumption that greener methods require analytical performance compromises.
Greenness Metrics and Environmental Impact
The AGREE scoring system provides a comprehensive assessment of method greenness based on all 12 principles of Green Analytical Chemistry. Traditional HPTLC methods typically score approximately 0.46, while greener alternatives achieve scores of 0.72-0.81 [78] [74] [79]. The environmental advantages of green HPTLC include:
- Reduced solvent toxicity: Replacement of chlorinated solvents and acetonitrile with ethanol, water, and ethyl acetate [4] [31]
- Minimized waste generation: Lower solvent consumption and biodegradable mobile phases [33] [74]
- Enhanced safety: Reduced exposure to hazardous chemicals for laboratory personnel [8] [31]
Implementation Challenges and Solutions
While green HPTLC offers significant advantages, implementation may present challenges including method transfer from existing traditional methods, initial method development time, and analyst training. These challenges can be mitigated through:
- Systematic method development following Quality by Design principles [74]
- Phased implementation alongside existing methods
- Comprehensive training on green chemistry principles and alternative solvents
Green HPTLC methodologies represent a significant advancement in sustainable pharmaceutical analysis, offering comparable or superior analytical performance while reducing environmental impact. The protocols and data presented herein provide researchers with practical guidance for implementing these methods in quality control and drug development settings. The continued adoption of green HPTLC approaches will contribute to more environmentally responsible pharmaceutical analysis while maintaining the rigorous analytical standards required for regulatory compliance.
Conclusion
Green HPTLC method validation represents a significant advancement in sustainable pharmaceutical analysis, successfully merging analytical rigor with environmental responsibility. The framework outlined demonstrates that HPTLC is not merely an alternative but often a superior choice for routine quality control, offering proven reliability, compliance with ICH guidelines, and a drastically reduced ecological footprint. Future directions point toward deeper integration with advanced detection platforms like HPTLC-MS and HPTLC-SERS, increased use of artificial intelligence for method optimization and data analysis, and broader acceptance driven by global harmonization of pharmacopeial standards. This evolution will further solidify the role of green HPTLC as an indispensable tool for ensuring drug quality and safety in an eco-conscious world.
References
- 1. Recent application of green : eco-friendly... analytical chemistry [https://fjps.springeropen.com/articles/10.1186/s43094-024-00658-6]
- 2. - Green - High : A Step... Performance Thin Layer Chromatography [https://link.springer.com/article/10.1007/s10337-025-04397-5]
- 3. Green NP-HPTLC and green RP-HPTLC methods for the ... [https://pubmed.ncbi.nlm.nih.gov/34227167/]
- 4. Comparing the Greenness and Validation Metrics of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11137692/]
- 5. Green stability-indicating RP-HPTLC technique for determi... [https://www.degruyterbrill.com/document/doi/10.1515/gps-2024-0107/html?srsltid=AfmBOoqDpQCK6bn_DQ_pUMfrkTB2WfSsI-c6fQrFdouhRlr09v5DrKTZ]
- 6. A sensitive green HPLC-fluorescence method for ... [https://www.nature.com/articles/s41598-025-23181-x]
- 7. An eco-friendly and cost-effective HPTLC method for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11063142/]
- 8. Eco-friendly and stability-indicating HPTLC method for the ... [https://www.sciencedirect.com/science/article/pii/S2772577425000333]
- 9. FDA validated ecofriendly HPTLC method for quantification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12436624/]
- 10. Method Validation in High-Performance Thin Layer ... [https://www.eurofinsus.com/food-testing/resources/method-validation-in-hptlc-methods-for-food-and-supplements/]
- 11. Versatile platforms based on HPTLC: Multimodal and ... [https://www.sciencedirect.com/science/article/abs/pii/S0924224425004467]
- 12. Comparative study of Normal-phase versus ... - BMC Chemistry [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-025-01439-9]
- 13. A High-Throughput and Green Planar Chromatographic ... [https://www.ajgreenchem.com/article_221633.html]
- 14. Dual-platform integration of HPTLC and firefly algorithm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12345108/]
- 15. Sustainable and smart multi-analyte HPTLC determination ... [https://www.sciencedirect.com/science/article/pii/S2666831925000189]
- 16. Greenness assessment and stability-indicating HPTLC ... [https://link.springer.com/article/10.1007/s44371-025-00348-z]
- 17. New HPLC, MS, and CDS Products from 2024–2025 [https://www.chromatographyonline.com/view/new-hplc-ms-and-cds-products-from-2024-2025-a-brief-review]
- 18. Versatile Platforms based on HPTLC: Multimodal and ... [https://www.sciencedirect.com/science/article/pii/S0924224425004467?dgcid=rss_sd_all]
- 19. USP Draft Chapter on HPTLC Identification of Fixed Oils ... [https://www.gmp-compliance.org/gmp-news/usp-draft-chapter-on-hptlc-identification-of-fixed-oils-published-for-comments]
- 20. Validation of High-Performance Thin-Layer Chromatographic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2662610/]
- 21. HSPiP and quality by design-assisted optimized analytical ... [https://jast-journal.springeropen.com/articles/10.1186/s40543-025-00508-x]
- 22. Development and validation of an HPTLC method for the ... [https://www.sciencedirect.com/science/article/pii/S165836551500179X]
- 23. FDA validated ecofriendly HPTLC method for quantification ... [https://www.nature.com/articles/s41598-025-18548-z]
- 24. Independent concentration extraction as a novel green ... [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-024-01260-w]
- 25. HPLC 2025 Preview: The Road To Sustainable Analytical ... [https://www.chromatographyonline.com/view/hplc-2025-preview-the-road-to-sustainable-analytical-chemistry]
- 26. Utilisation of Analytical Method Greenness Score to drive ... [https://pubs.rsc.org/pt-br/content/articlehtml/2025/gc/d5gc01574j]
- 27. Green stability-indicating RP-HPTLC approach for ... [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-025-01431-3]
- 28. Assessment of the Method's Greenness and Blueness [https://www.mdpi.com/1424-8247/17/8/1002]
- 29. Development of Green HPTLC method for simultaneous ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10541718/]
- 30. Eco-friendly alternatives to conventional solvents [https://www.sciencedirect.com/science/article/pii/S2772826925000859]
- 31. Recent Advances In Green Chromatography Techniques ... [https://www.sciencedirect.com/science/article/pii/S2772391725000830]
- 32. A Green High-Performance Thin-Layer Chromatography ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9103461/]
- 33. Eco-Friendly High-Performance Thin-Layer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10601416/]
- 34. Two sustainable chromatographic approaches for ... [https://pubmed.ncbi.nlm.nih.gov/41131091/]
- 35. High-performance Thin-layer Chromatography Method ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5004512/]
- 36. Complete Guide to Thin Layer Chromatography Sample ... [https://www.organomation.com/complete-guide-to-thin-layer-chromatography-sample-preparation]
- 37. Stationary Phases for Modern Thin-Layer Chromatography [https://www.chromatographyonline.com/view/stationary-phases-modern-thin-layer-chromatography]
- 38. Development and validation of HPTLC method for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3658090/]
- 39. High-performance thin layer chromatography: A powerful analytical technique in pharmaceutical drug discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3658041/]
- 40. Reproducibility of high-performance thin-layer ... [https://www.sciencedirect.com/science/article/abs/pii/S2468170917301352]
- 41. Key Components for Ensuring Accurate HPTLC Analysis [https://www.eurofinsus.com/food-testing/resources/key-components-for-ensuring-accurate-hptlc-analysis/]
- 42. Development and Validation of High-Performance Thin ... - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12628033/]
- 43. HPTLC METHOD DEVELOPMENT AND VALIDATION OF ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3255440/]
- 44. Development and Validation of a Stability-Indicating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388184/]
- 45. Hyphenated analytical techniques for validation of herbal ... [https://www.sciencedirect.com/science/article/abs/pii/B978032385542600038X]
- 46. A validated HPTLC method for quantification of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8356894/]
- 47. Development and Validation of HPTLC Method for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3507343/]
- 48. profiling of ebastine and phenylephrine hydrochloride [https://www.sciencedirect.com/science/article/abs/pii/S0026265X18318071]
- 49. and NP-HPTLC methods in API and formulation with ... [https://www.sciencedirect.com/science/article/pii/S2352554125000646]
- 50. Greenness assessment of two chromatographic methods ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10612294/]
- 51. A Simple, Cost-Effective, and Green HPTLC Method for the ... [https://www.mdpi.com/2073-4395/12/5/1016]
- 52. The Evolution of LC Troubleshooting [https://www.chromatographyonline.com/view/the-evolution-of-lc-troubleshooting-strategies-for-improving-peak-tailing]
- 53. Development and validation of a green - high - performance ... thin layer [https://link.springer.com/article/10.1007/s00764-025-00380-w]
- 54. Two sustainable chromatographic approaches for ... [https://www.nature.com/articles/s41598-025-22295-6]
- 55. High performance thin layer chromatography with eluents ... [https://www.nature.com/articles/s41598-025-99617-1]
- 56. Quantitative Analysis of Emtricitabine in Dosage Forms Using Green ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7774264/]
- 57. Ecofriendly densitometric (RP and NP) HPTLC for... methods [https://pubs.rsc.org/en/content/articlelanding/2025/ay/d5ay01031d]
- 58. High Performance Thin Layer Chromatography (HPTLC ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11357029/]
- 59. Development and validation of high-performance thin layer ... [https://www.sciencedirect.com/science/article/pii/S0026265X25000815]
- 60. High-Throughput Solid Phase Extraction for Targeted and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11948174/]
- 61. Development and Validation of HPTLC and Green HPLC ... [https://pubmed.ncbi.nlm.nih.gov/31879753/]
- 62. Comparative HPTLC study for simultaneous determination of ... [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-023-01025-x]
- 63. [ high ] | PPT performance thin layer chromatography HPTLC [https://www.slideshare.net/slideshow/high-performance-thin-layer-chromatography-hptlc-207726429/207726429]
- 64. ICH Guidelines for Analytical Method Validation Explained [https://amsbiopharma.com/ich-guidelines-analytical-method/]
- 65. Stability indicating HPTLC method development and ... [https://link.springer.com/article/10.1007/s44371-025-00124-z]
- 66. Development of a Validated High-Performance Thin-Layer ... [https://www.mdpi.com/1420-3049/30/19/3859]
- 67. Q2(R1) Validation of Analytical Procedures: Text and ... [https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q2r1-validation-analytical-procedures-text-and-methodology-guidance-industry]
- 68. ICH Q2(R1) Validation of Analytical Procedures [https://www.gmp-compliance.org/guidelines/gmp-guideline/ich-q2r1-validation-of-analytical-procedures-text-and-methodology]
- 69. Development of Green HPTLC method for simultaneous ... [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-023-01043-9]
- 70. Editorial | JPC – Journal of Planar Chromatography [https://link.springer.com/article/10.1007/s00764-022-00210-3]
- 71. ICH and US-FDA validated HPTLC methods with ... [https://link.springer.com/article/10.1007/s00764-022-00201-4]
- 72. The Limit of Detection [https://www.chromatographyonline.com/view/limit-detection]
- 73. A linear validation method of analytical procedures based ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267024004963]
- 74. Eco-friendly HPTLC method for Trifluridine and Tipiracil ... [https://www.sciencedirect.com/science/article/pii/S2772577425000308]
- 75. Green and white analytical approach for parallel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12185713/]
- 76. Hydrolytic stress degradation study and concomitant HPTLC ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12574119/]
- 77. An eco-friendly and cost-effective HPTLC method for ... [https://www.nature.com/articles/s41598-024-56923-4]
- 78. Determination of Colchicine in Pharmaceutical Formulations... [https://pubmed.ncbi.nlm.nih.gov/35807719/]
- 79. Hydrolytic stress degradation study and concomitant HPTLC estimation... [https://bmcchem.biomedcentral.com/articles/10.1186/s13065-025-01637-5]
- 80. A validated HPTLC method for quantitative analysis of ... [https://www.sciencedirect.com/science/article/pii/S1674638420300253]